Simultaneous Selective Chlorination and Carbothermic Reduction of High-Iron Manganese Ore for the Recovery of Manganese Chloride and Metallic Iron
Abstract
:1. Introduction
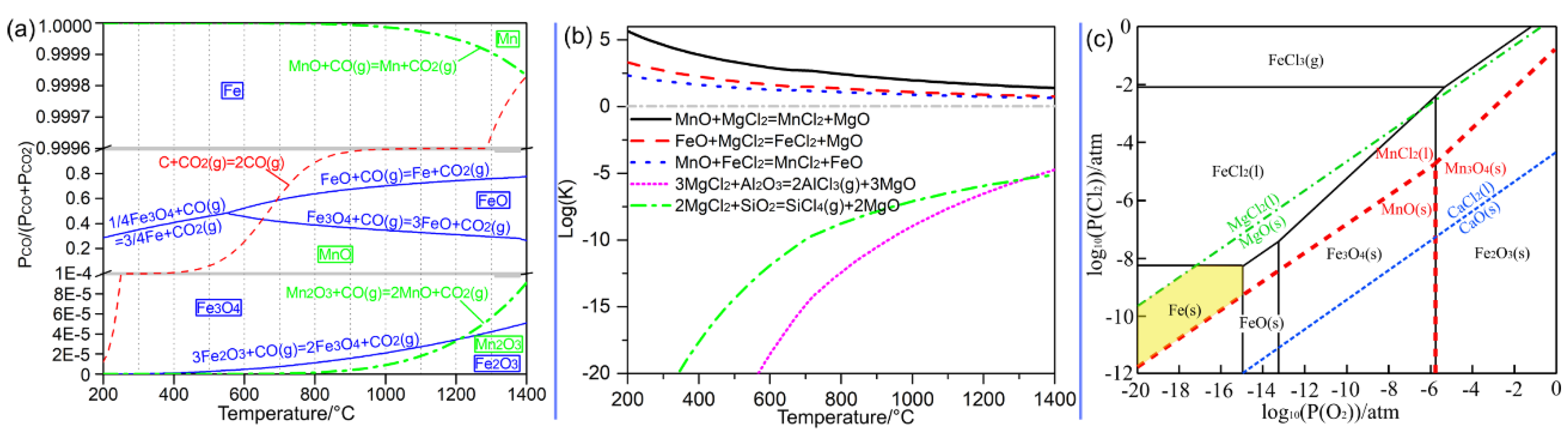
Thermodynamic Considerations
2. Materials and Methods
2.1. Materials
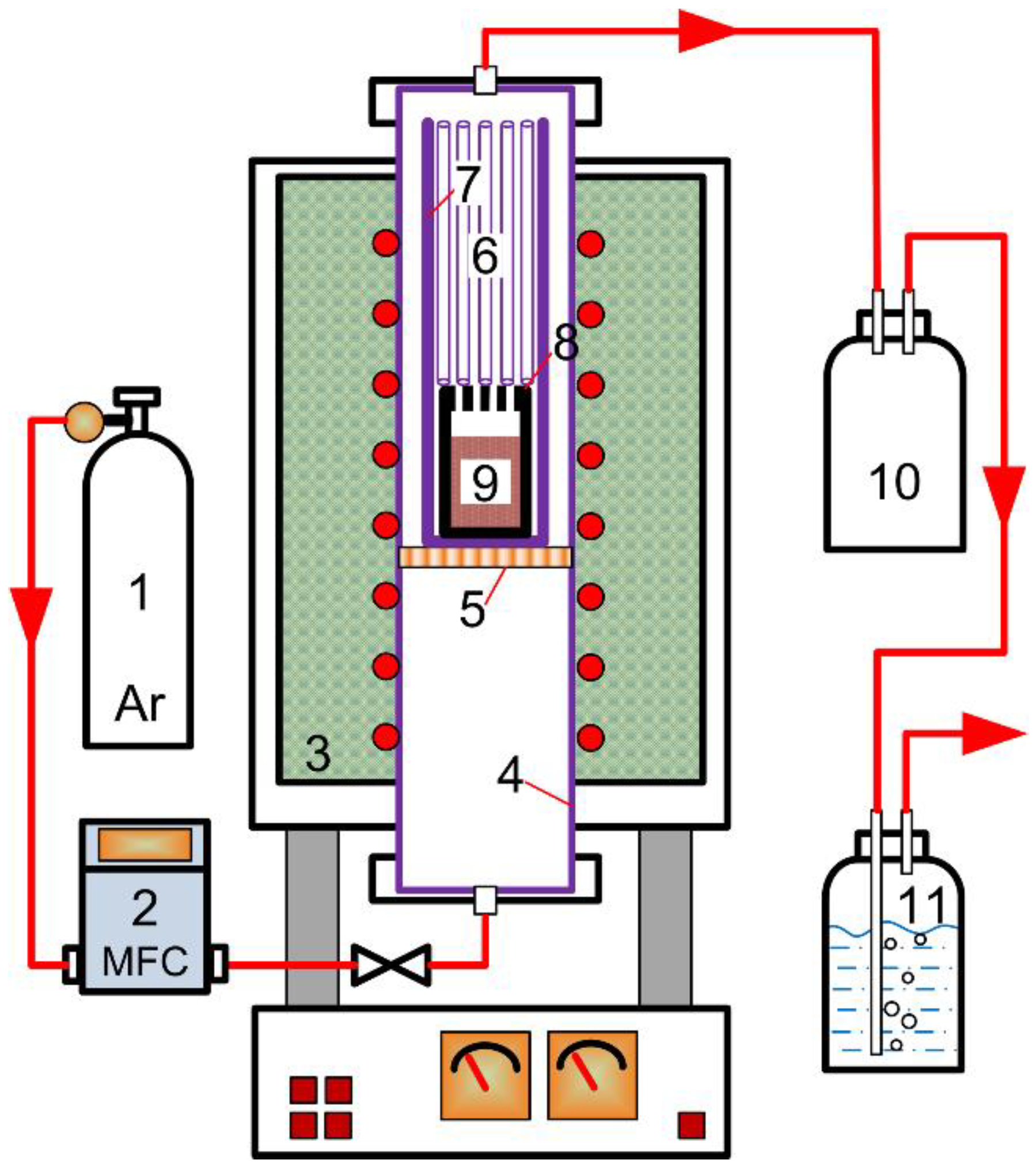
2.2. Experimental Procedure
2.3. Analytical Methods
3. Results and Discussion
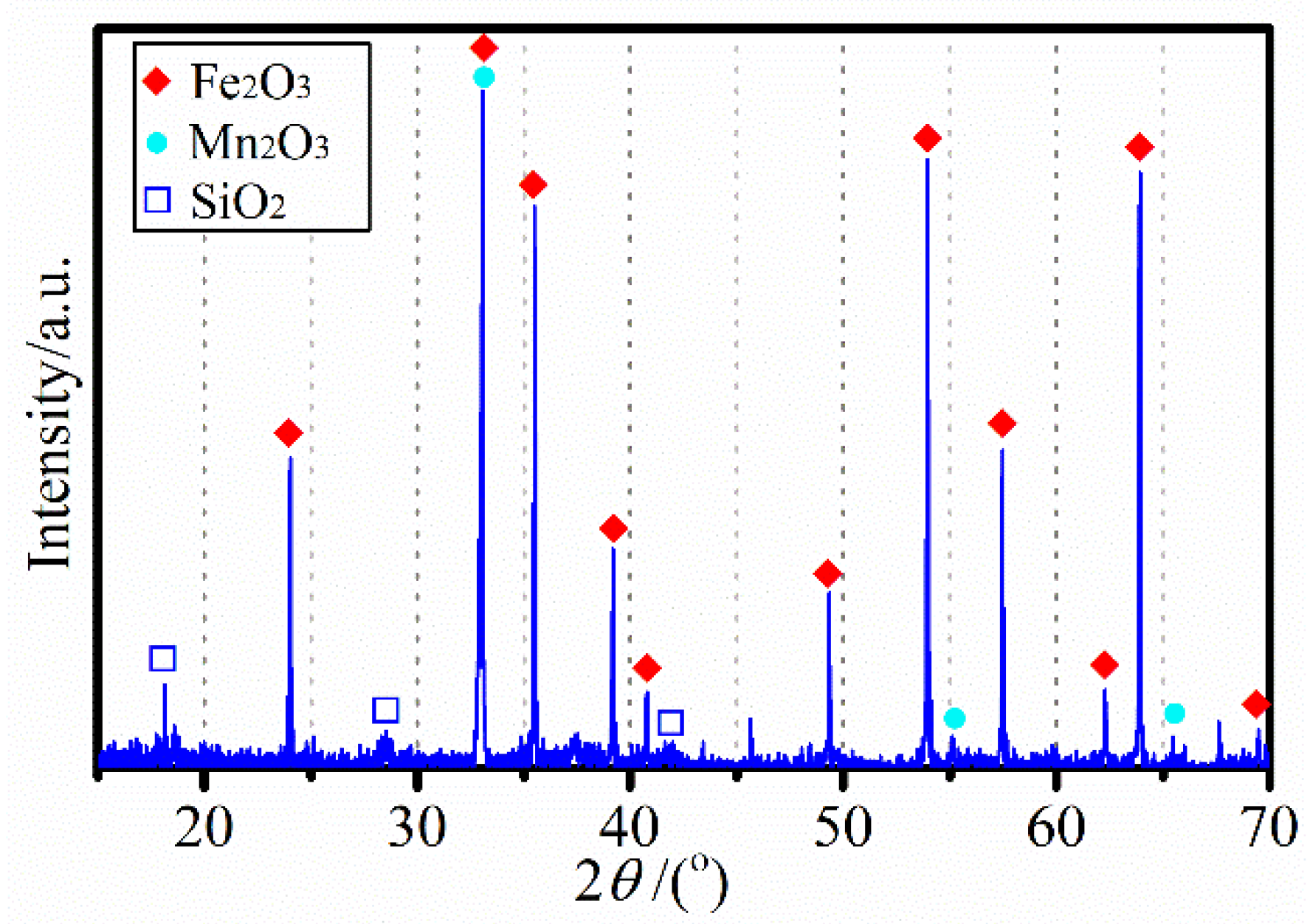
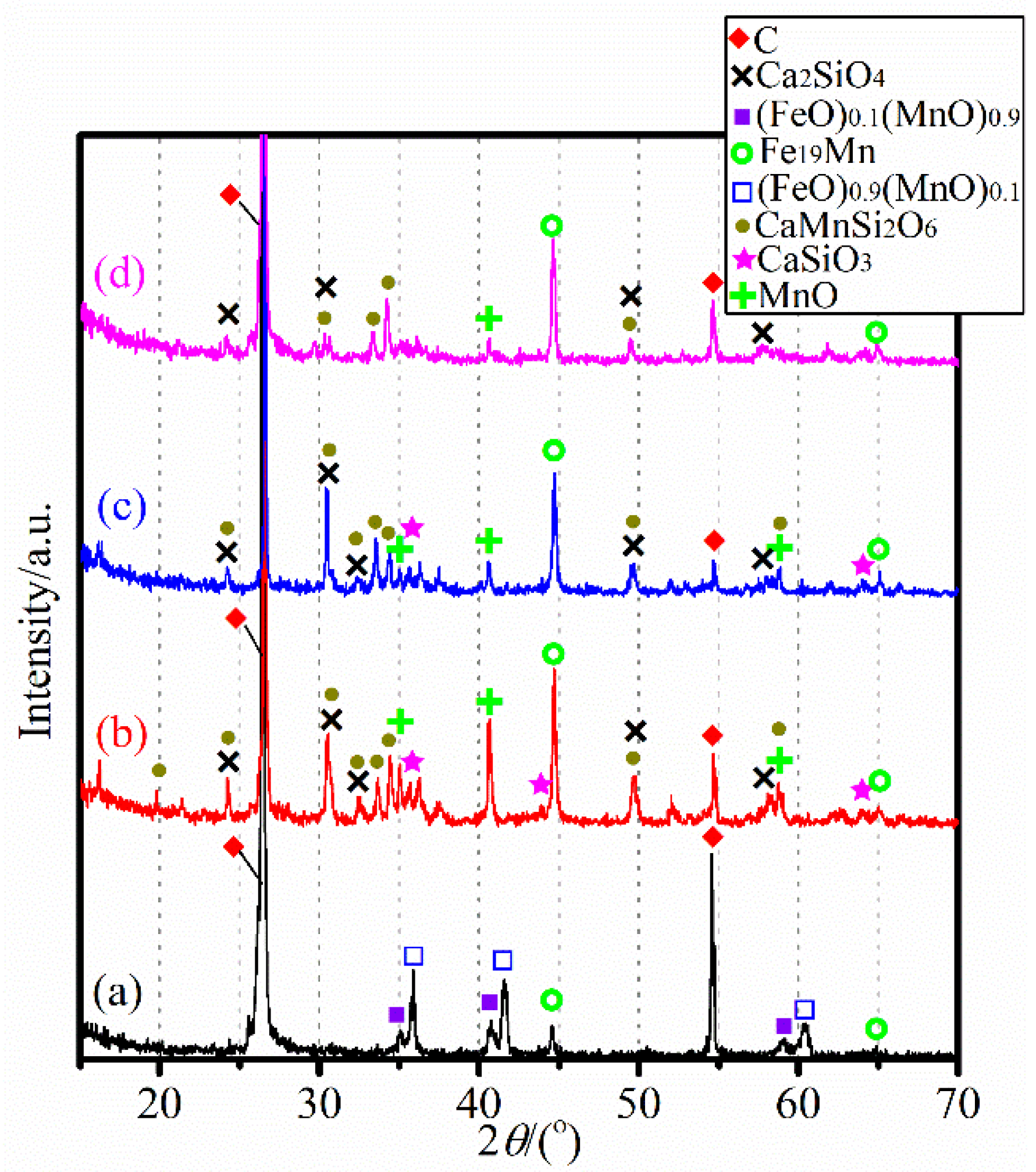
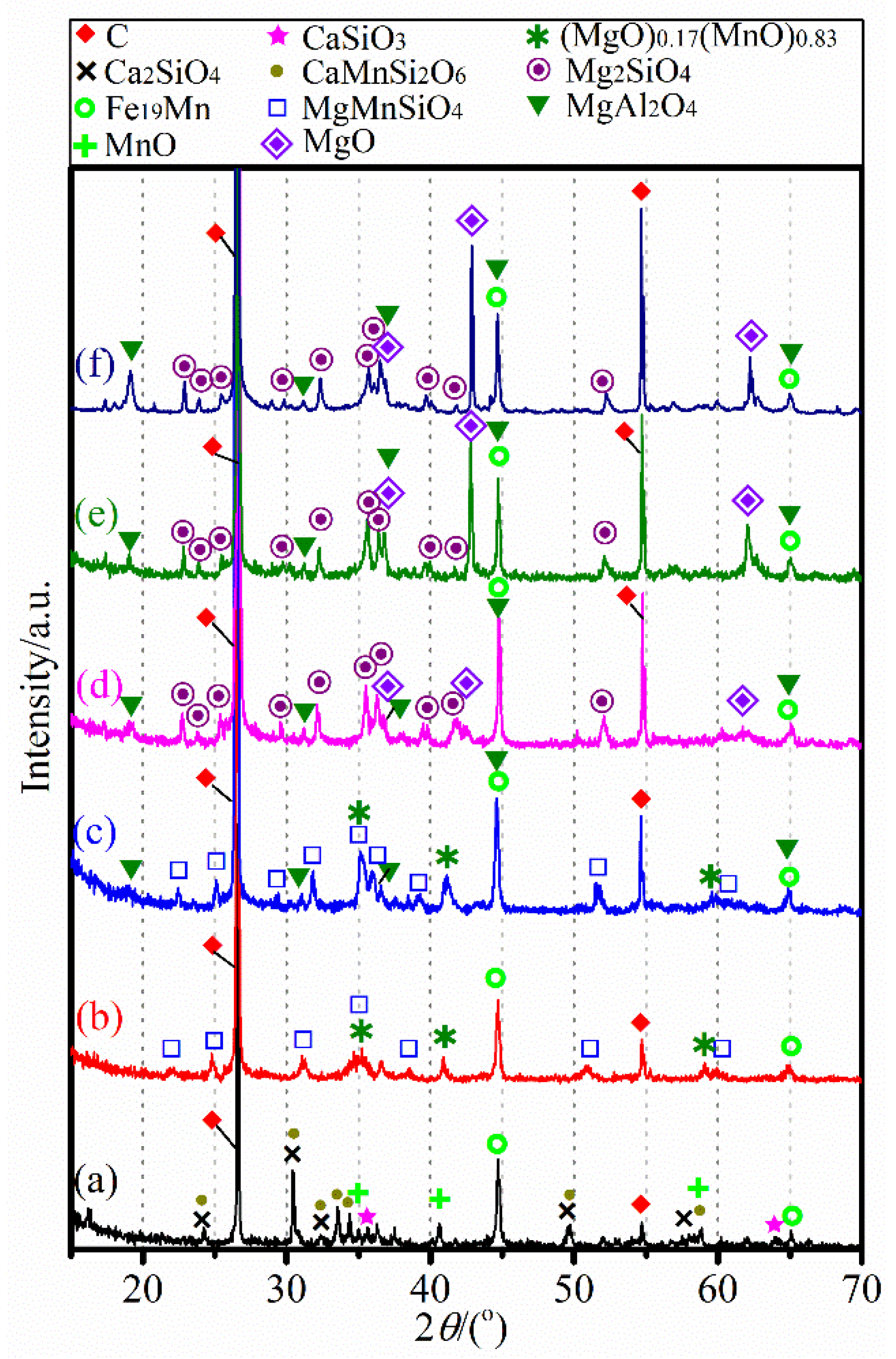
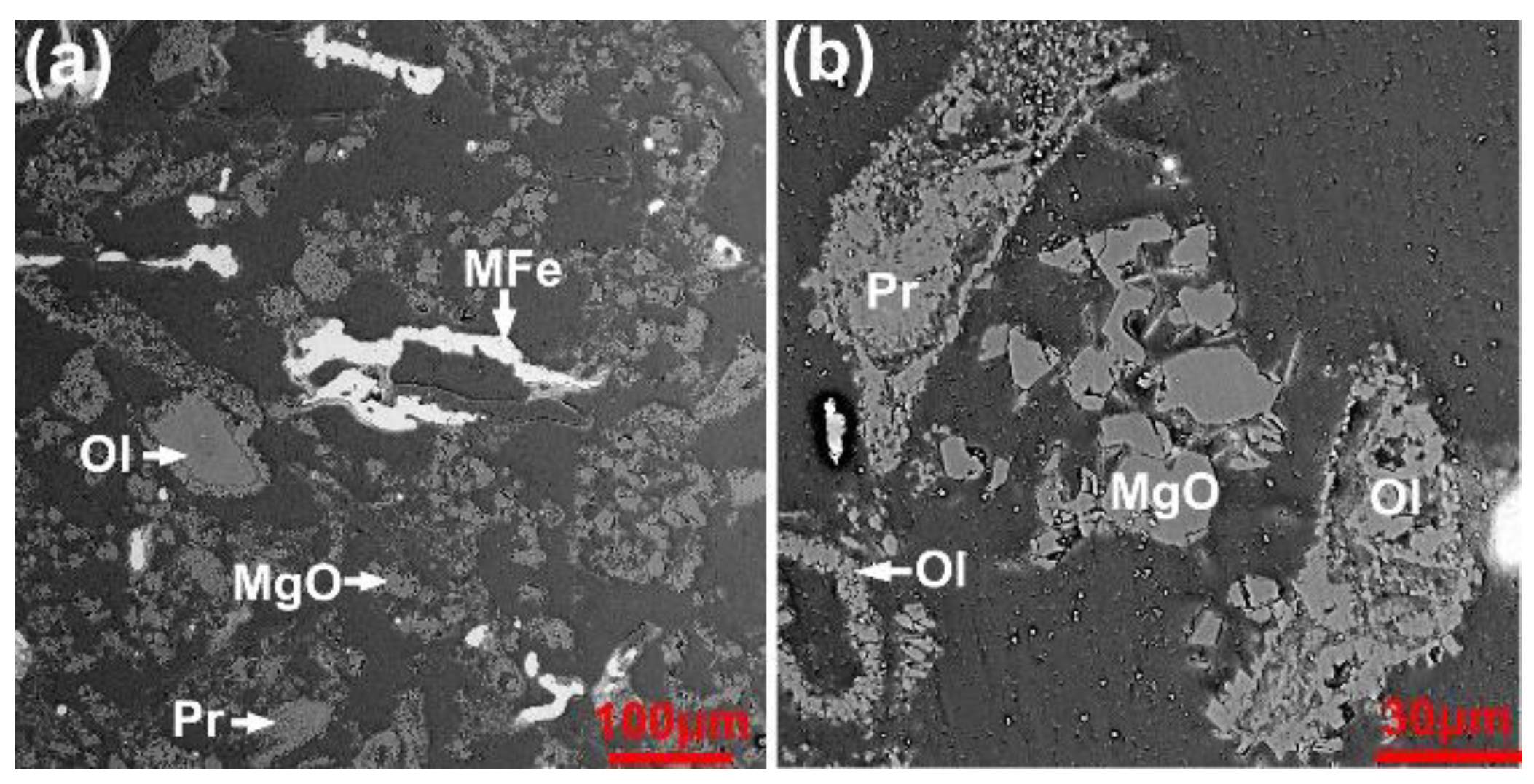
3.1. Characterizaiton of the Mn Ore
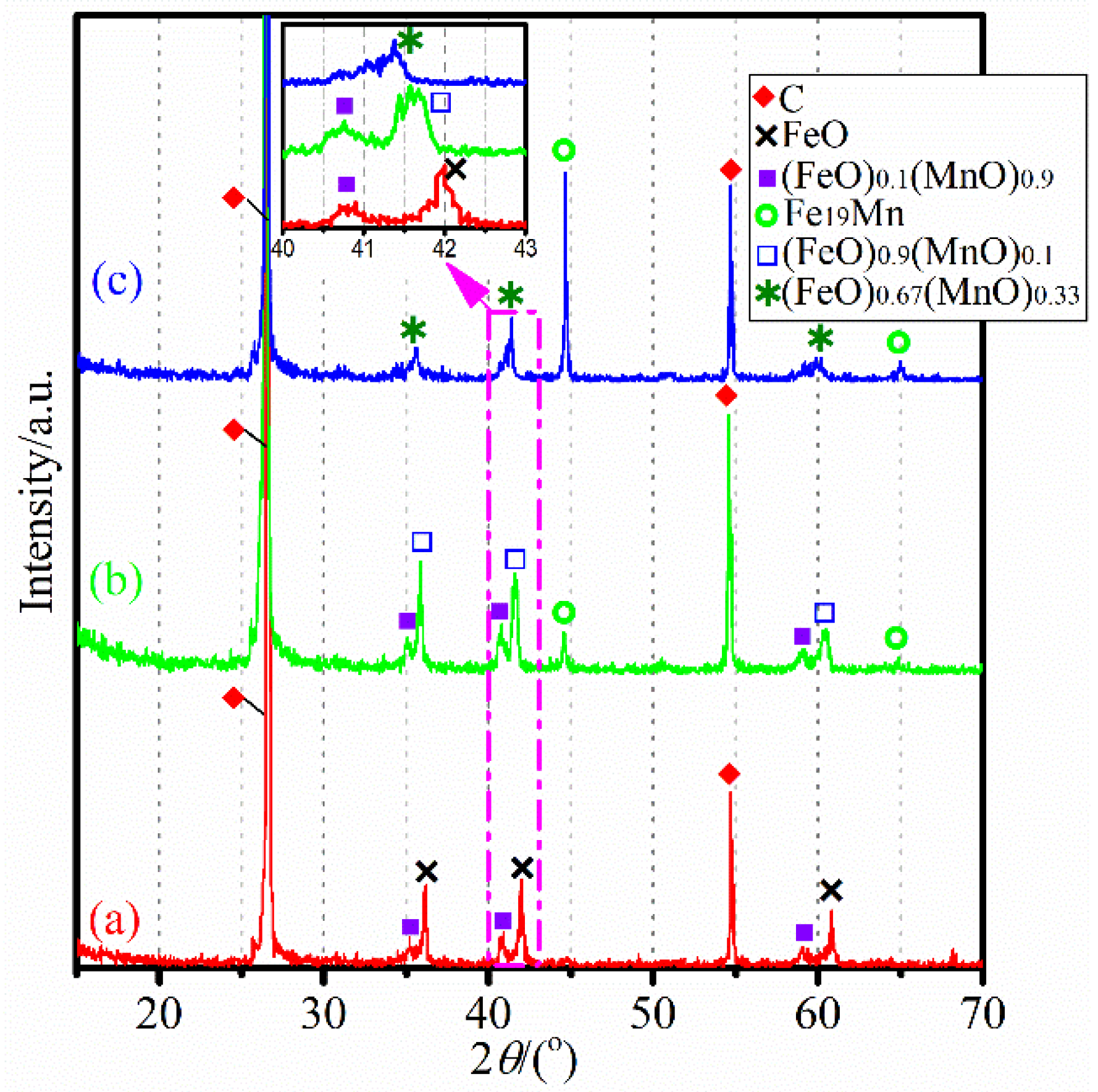
3.2. Carbothermic Reduction without Additives
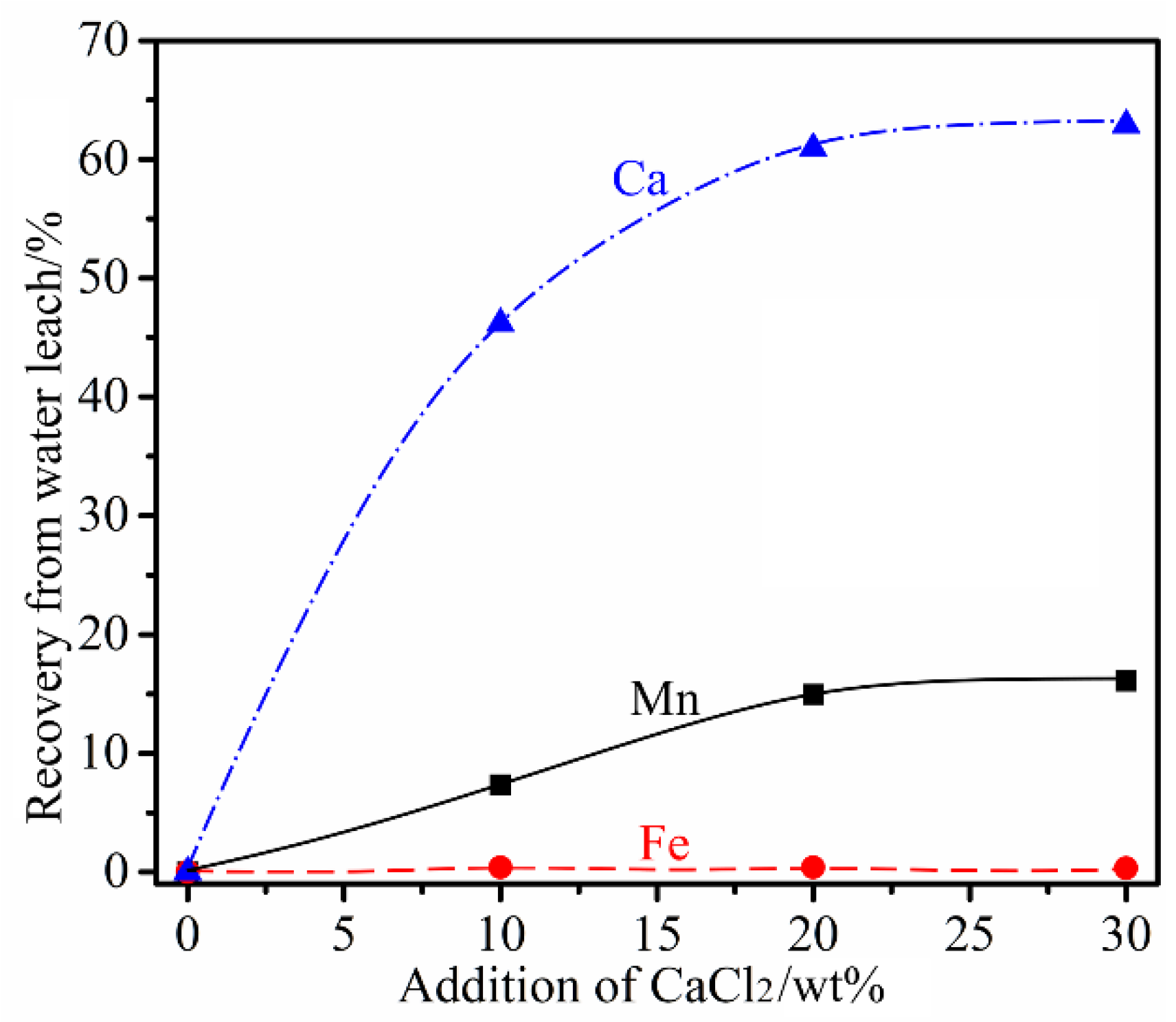
3.3. Influence of CaCl2 Addition
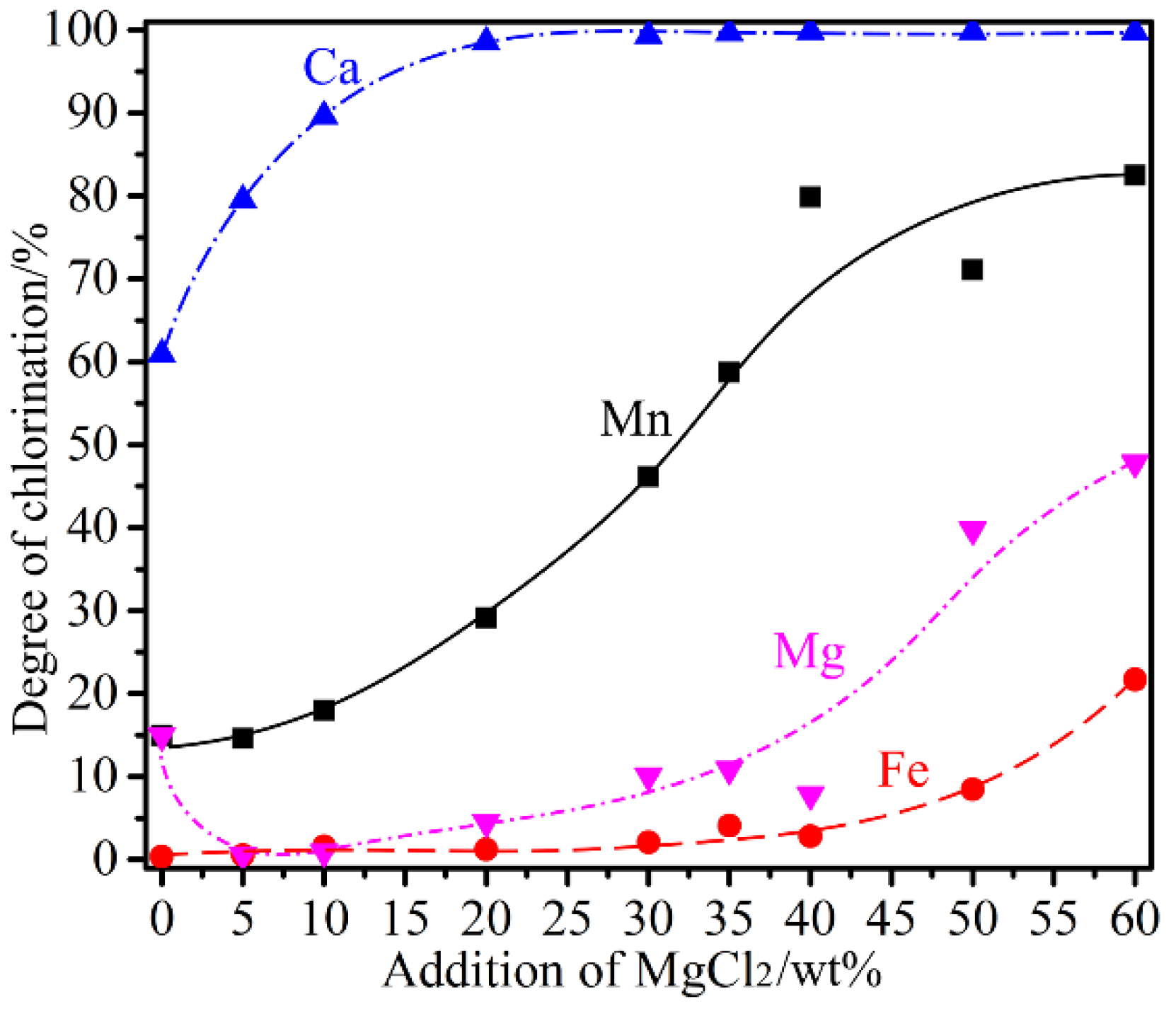
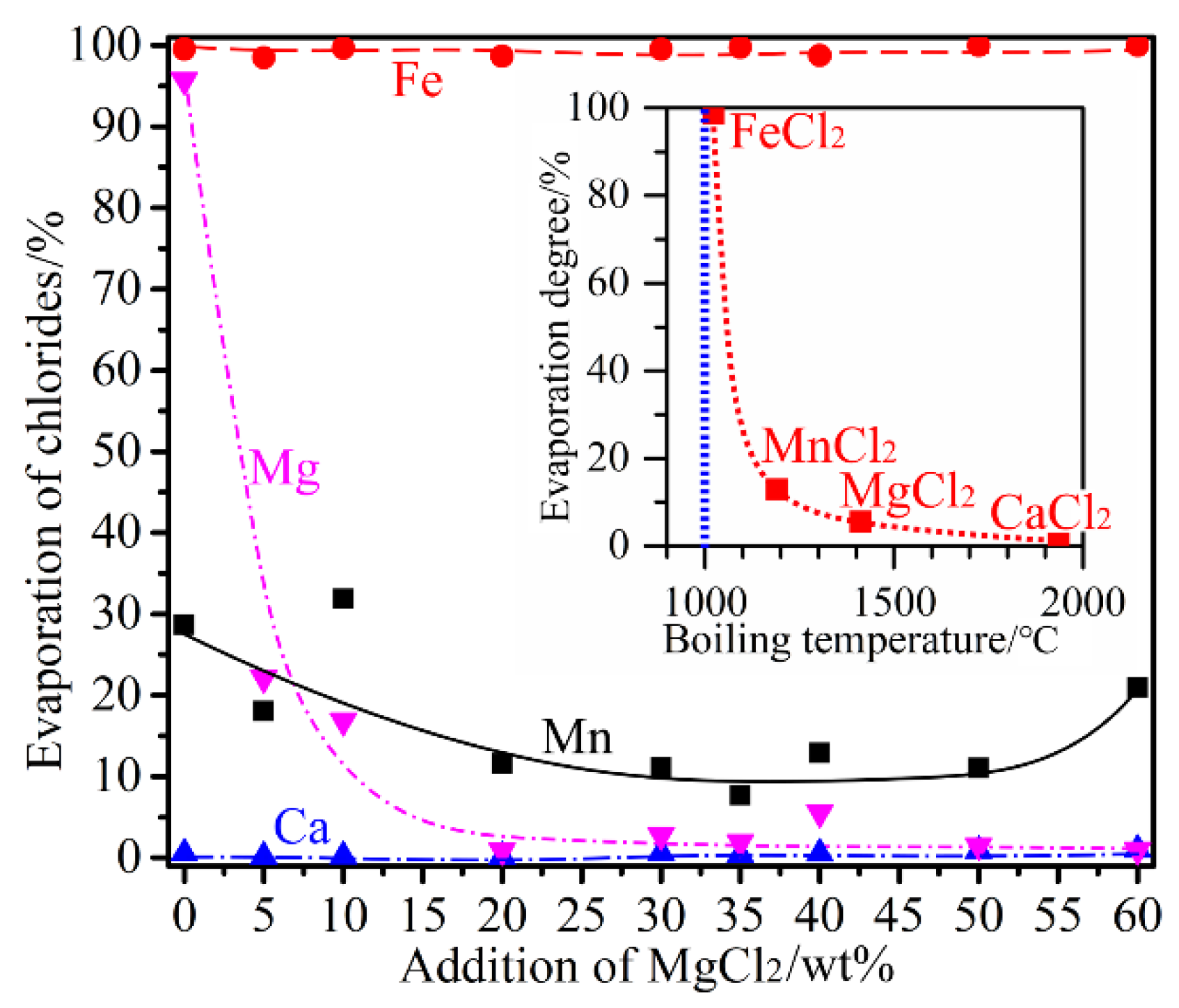
3.4. Influence of MgCl2 Addition
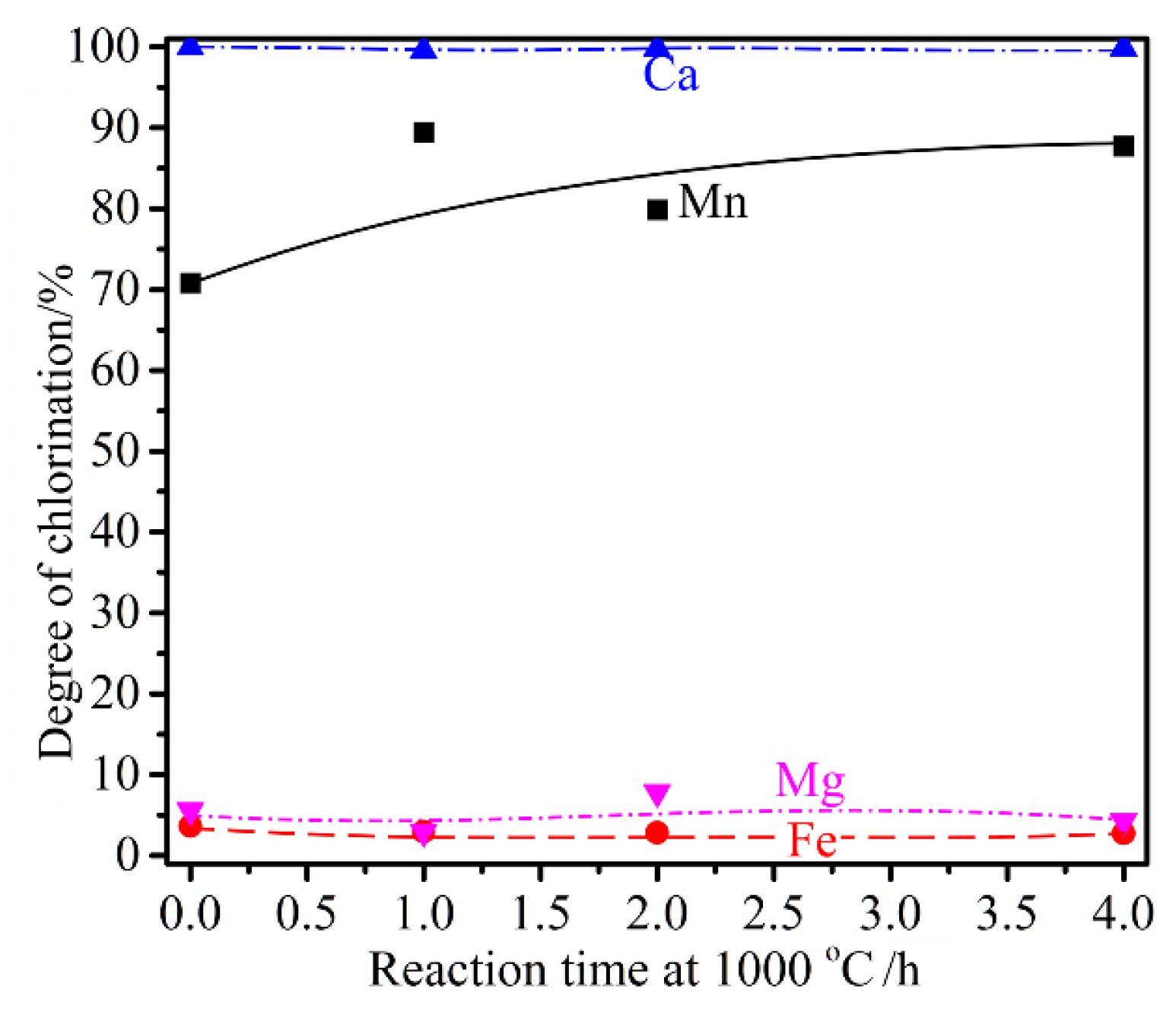
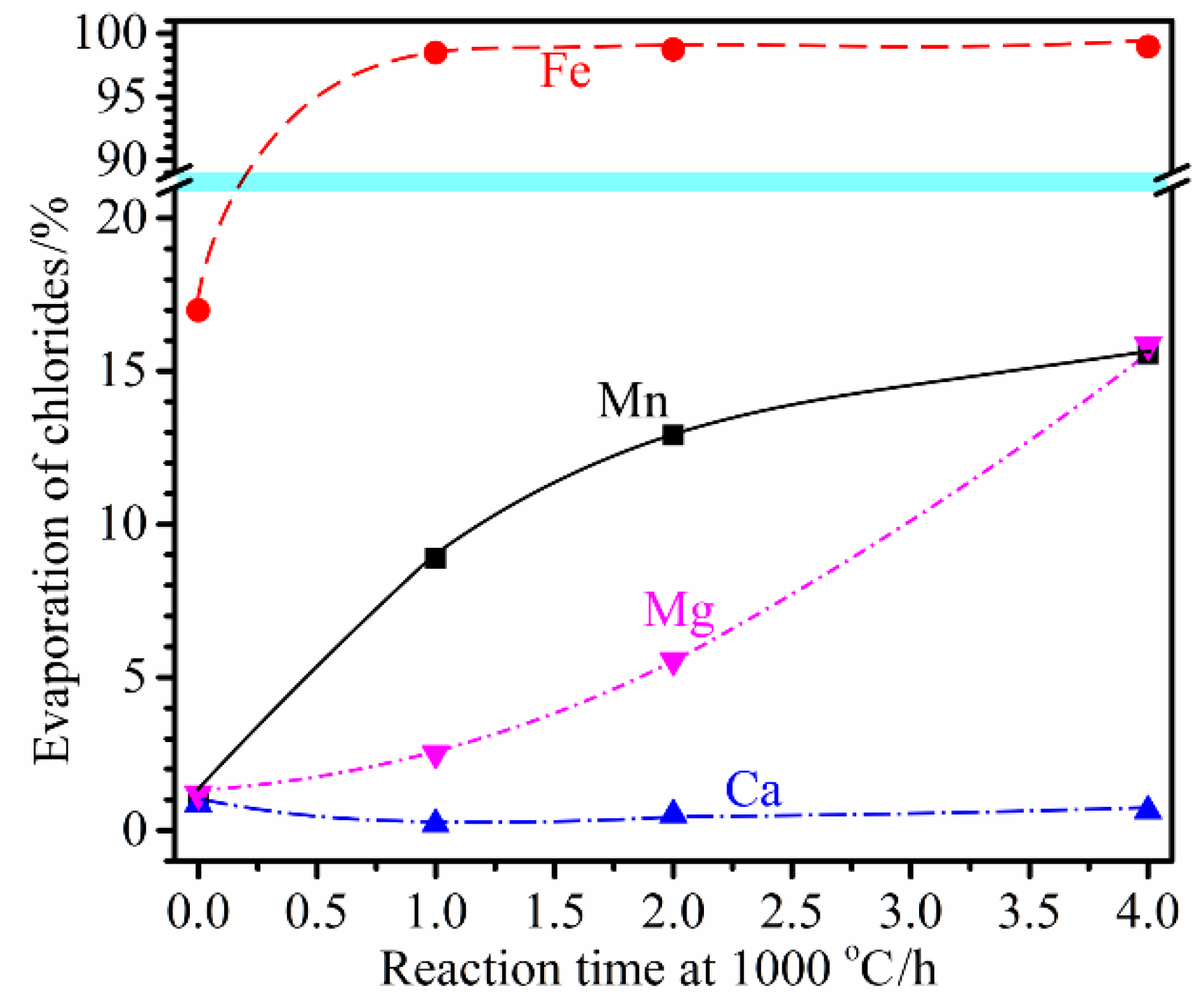
3.5. Influence of Reaction Time
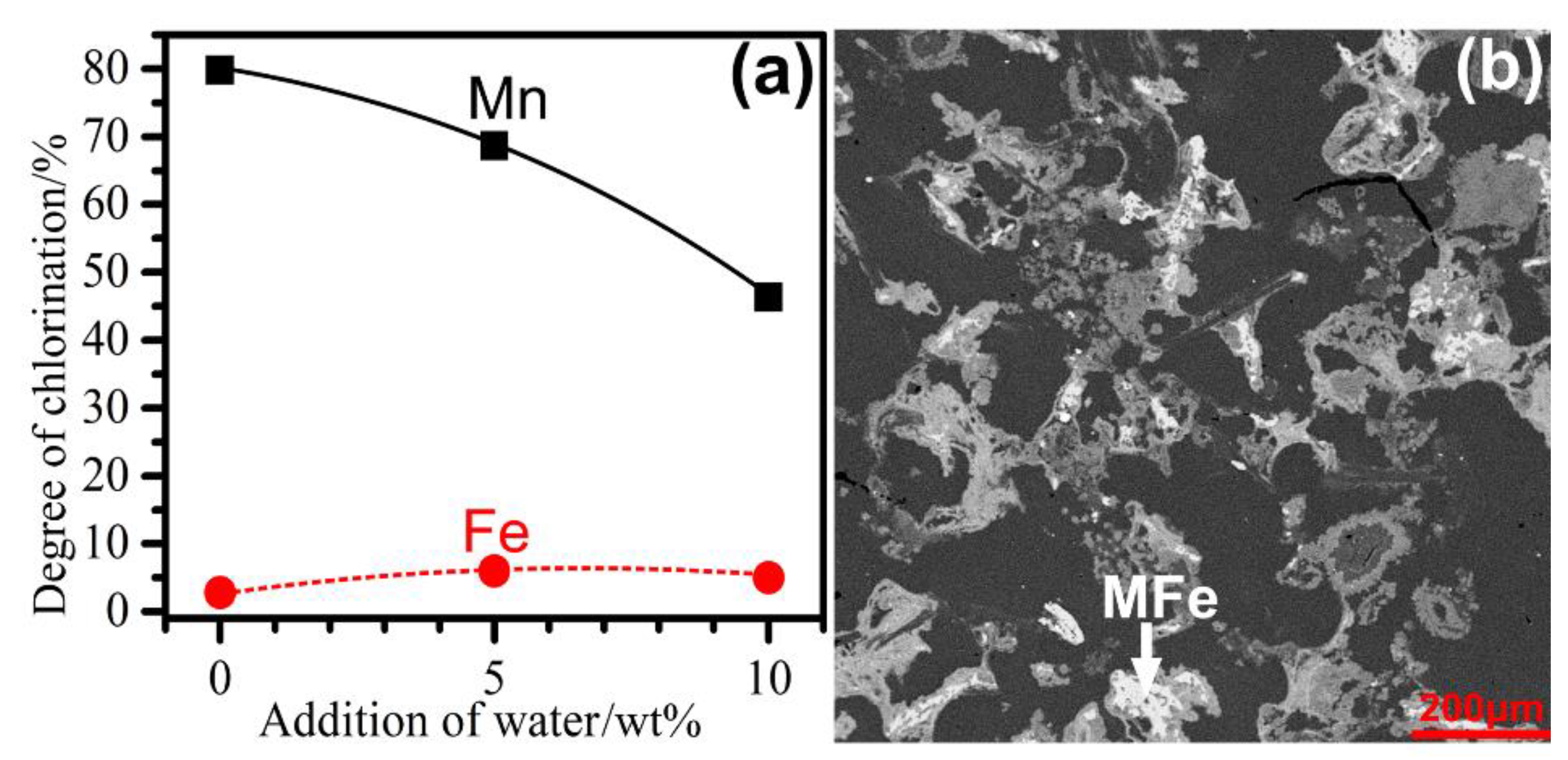
3.6. Influence of the Presence of Water
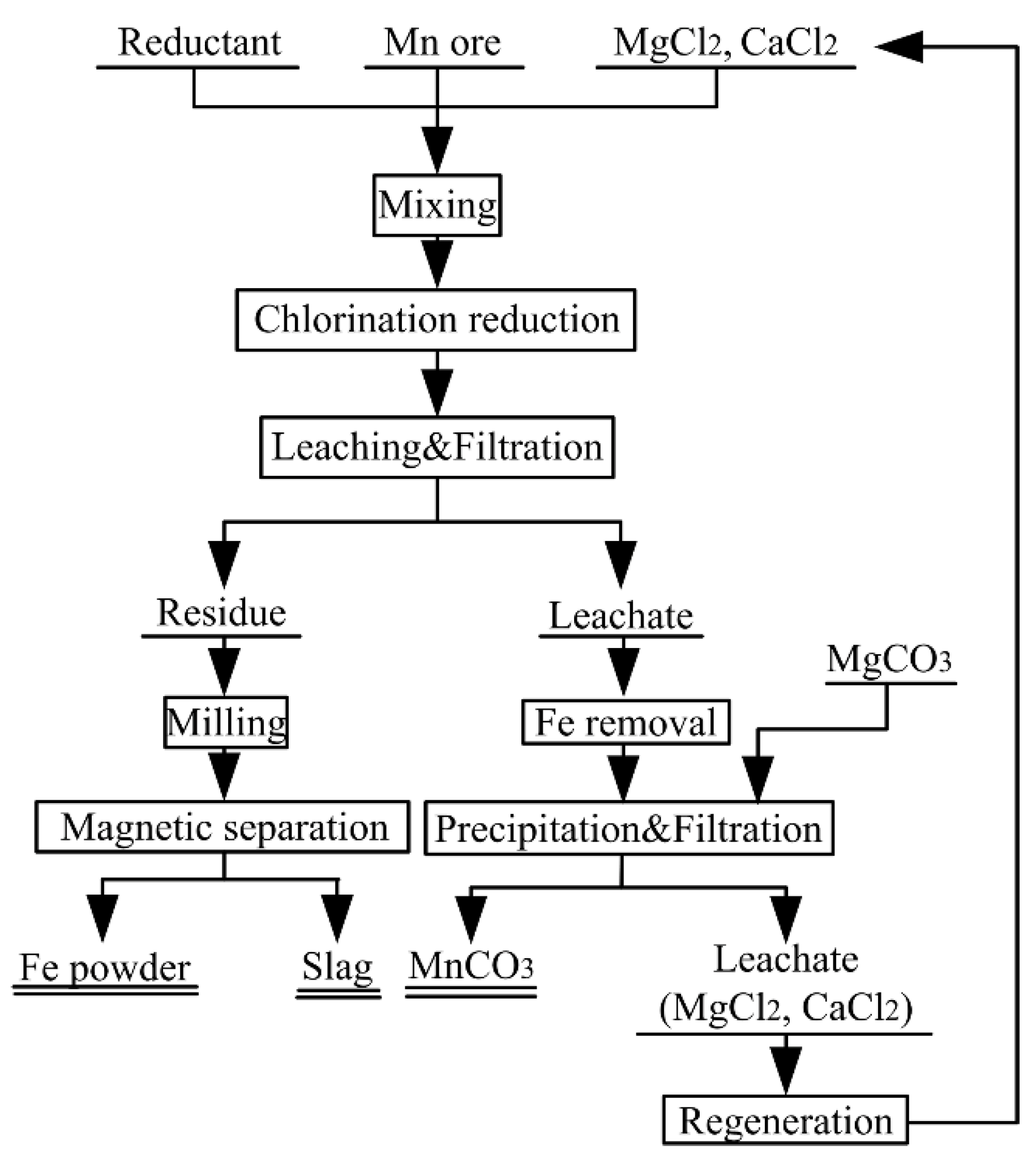
3.7. Proposed Flow Sheet
4. Conclusions
- (1)
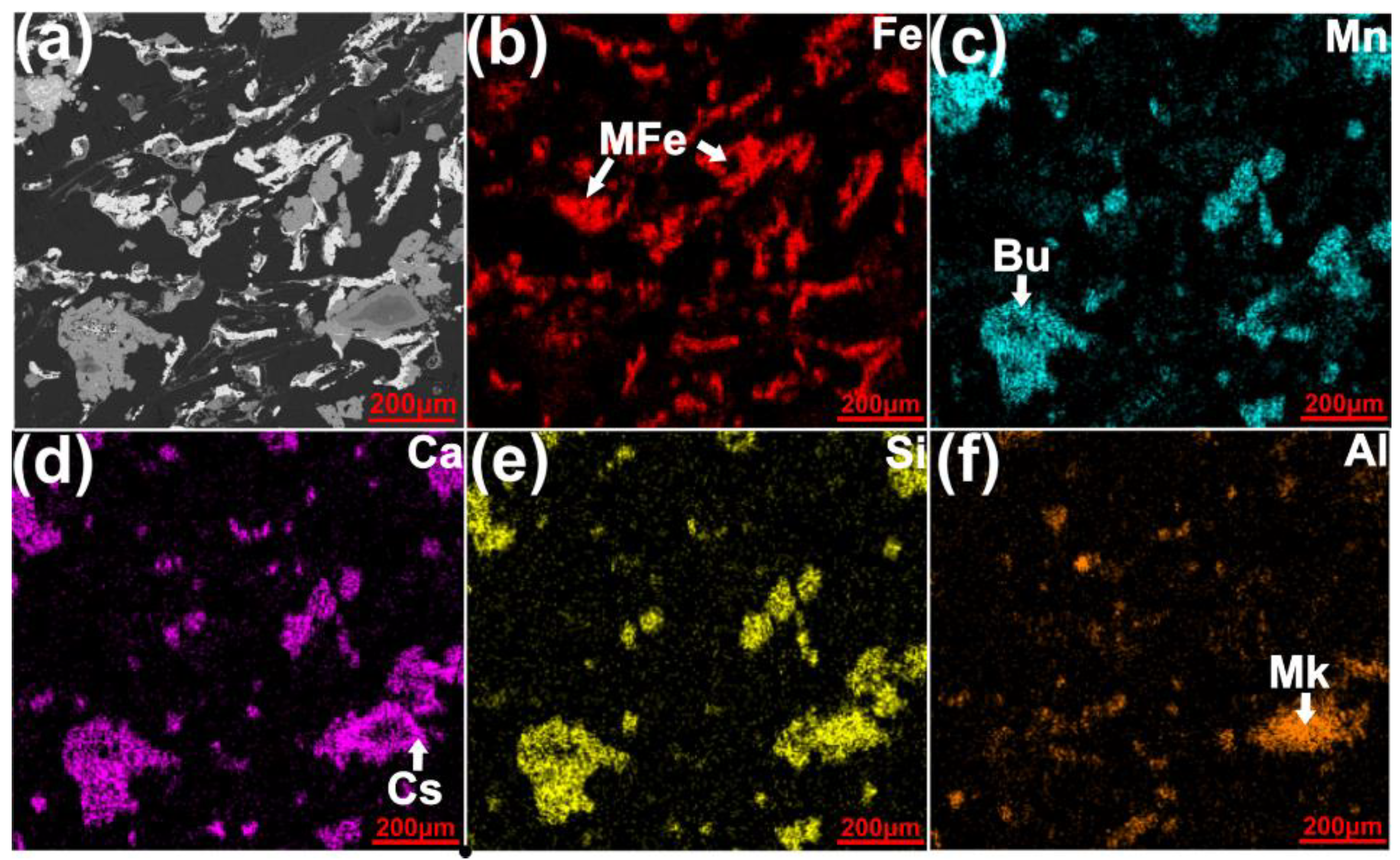
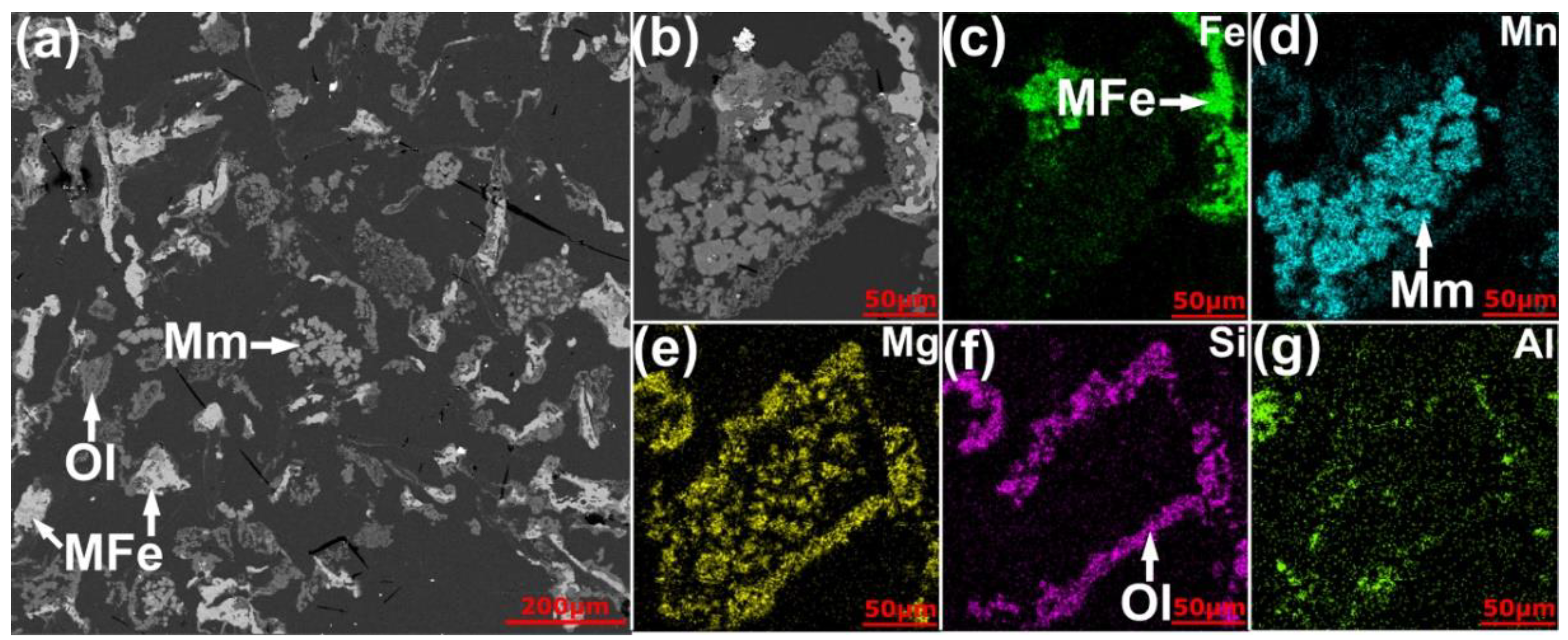
- Simultaneous chlorination of Mn and metallization of Fe from the high-Fe manganese ore is feasible. With the addition of 18 wt% graphite as the carbonaceous reductant, 20 wt% CaCl2 and 40 wt% MgCl2 as the chlorinating agent, heating the manganese ore at 1000 °C for 1 h resulted in the selective chlorination of Mn reaching as much as 89.4%, with the Fe chlorination of only 3.0%. Most Fe oxides in the ore were reduced to the metallic form, with the particle sizes generally in the range of 100–200 µm, allowing subsequent easier separation and upgrading of the metallic Fe particles.
- (2)
- The presence of CaCl2 greatly accelerated the selective reduction of Fe oxides to their metallic form. In addition, it promoted the formation of relatively large metallic Fe particles, facilitating their subsequent separation. In the absence of MgCl2, CaCl2 alone could selectively chlorinate Mn only to a limited degree. The chlorination involved reacting with quartz which was present in the Mn ore as gangue, forming calcium silicates and MnCl2 (Equations (10)–(12)).
- (3)
- MgCl2 was a stronger chlorinating agent comparing to CaCl2. Selective chlorination of Mn to a high degree was possible in the presence of MgCl2, in which case the presence of CaCl2 did not contribute to the chlorination. However, addition of excessive amounts of MgCl2 would lead to gradual increase in the chlorination of Fe without further improving the Mn chlorination degree. Chlorination using MgCl2 would preferably involve reacting with quartz forming magnesium silicates and MnCl2 Equations (13) and (14). Once the quartz was consumed completely, direct chlorination of MnO by MgCl2 would take place (Equation (15)).
- (4)
- The presence of water was detrimental to the selective chlorination of Mn from the ore, likely due to the presence of HCl-generating reaction (Equation (17)) competing for MgCl2.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Singh, V.; Biswas, A. Physicochemical processing of low grade ferruginous manganese ores. Int. J. Miner. Process. 2017, 158, 35–44. [Google Scholar] [CrossRef]
- Pagnanelli, F.; Garavini, M.; Vegliò, F.; Toro, L. Preliminary screening of purification processes of liquor leach solutions obtained from reductive leaching of low-grade manganese ores. Hydrometallurgy 2004, 71, 319–327. [Google Scholar] [CrossRef]
- Hagelstein, K. Globally sustainable manganese metal production and use. J. Environ. Manag. 2009, 90, 3736–3740. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Huang, Z.; Jiang, T.; Zhao, P.; Zhong, R.; Liang, Z. Carbothermic Reduction of Ferruginous Manganese Ore for Mn/Fe Beneficiation: Morphology Evolution and Separation Characteristic. Minerals 2017, 7, 167. [Google Scholar] [CrossRef]
- Corathers, L.A. USGS 2015 Minerals Yearbook-Manganese; U.S. Geological Survey: Reston, VA, USA, 2019.
- Kononov, R.; Ostrovski, O.; Ganguly, S. Carbothermal Solid State Reduction of Manganese Ores: 1. Manganese Ore Characterisation. ISIJ Int. 2009, 49, 1099–1106. [Google Scholar] [CrossRef]
- Gao, Y.B.; Kim, H.G.; Sohn, H.Y. Kinetics of pre-reduction of manganese ore by CO. Min. Process. Extr. Met. 2012, 121, 109–116. [Google Scholar] [CrossRef]
- Akdogan, G.; Eric, R.H. Kinetics of the solid-state carbothermic reduction of wessel manganese ores. Met. Mater. Trans. B 1995, 26, 13–24. [Google Scholar] [CrossRef]
- Abdel Halim, K.S.; Bahgat, M.; Morsi, M.B.; El-Barawy, K. Pre-reduction of manganese ores for ferromanganese industry. Ironmak. Steelmak. 2011, 38, 279–284. [Google Scholar] [CrossRef]
- Braga, R.S.; Takano, C.; Mourão, M.B. Prereduction of self-reducing pellets of manganese ore. Ironmak. Steelmak. 2007, 34, 279–284. [Google Scholar] [CrossRef]
- Zhang, W.; Cheng, C.Y. Manganese metallurgy review. Part I: Leaching of ores/secondary materials and recovery of electrolytic/chemical manganese dioxide. Hydrometallurgy 2007, 89, 137–159. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, S.; Cao, Z.F.; Zhong, H. Recovery of manganese from manganese oxide ores in the EDTA solution. Met. Res. Technol. 2018, 115, 306. [Google Scholar] [CrossRef]
- Acharya, C.; Kar, R.N.; Sukla, L.B. Studies on reaction mechanism of bioleaching of manganese ore. Min. Eng. 2003, 16, 1027–1030. [Google Scholar] [CrossRef]
- Mpho, M.; Samson, B.; Ayo, A. Evaluation of reduction roasting and magnetic separation for upgrading Mn/Fe ratio of fine ferromanganese. Int. J. Min. Sci. Technol. 2013, 23, 537–541. [Google Scholar] [CrossRef]
- You, Z.; Li, G.; Zhang, Y.; Peng, Z.; Jiang, T. Extraction of manganese from iron rich MnO2 ores via selective sulfation roasting with SO2 followed by water leaching. Hydrometallurgy 2015, 156, 225–231. [Google Scholar] [CrossRef]
- Paixdo, J.M.M.; Amaral, J.C.; Memória, L.E.; Freitas, L.R. Sulphation of Carajás manganese ore. Hydrometallurgy 1995, 39, 215–222. [Google Scholar] [CrossRef]
- Zhang, Y.; You, Z.; Li, G.; Jiang, T. Manganese extraction by sulfur-based reduction roasting–acid leaching from low-grade manganese oxide ores. Hydrometallurgy 2013, 133, 126–132. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Zhao, Y.; You, Z.X.; Duan, D.X.; Li, G.H.; Jiang, T. Manganese extraction from high-iron-content manganese oxide ores by selective reduction roasting-acid leaching process using black charcoal as reductant. J. Cent. South Univ. 2015, 22, 2515–2520. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, G.; Cheng, Z. Thermal analysis and kinetic modeling of manganese oxide ore reduction using biomass straw as reductant. Hydrometallurgy 2010, 105, 96–102. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, G.; Yan, H.; Li, T.; Feng, X. Thermogravimetric Analysis and Kinetics on Reducing Low-Grade Manganese Dioxide Ore by Biomass. Met. Mater. Trans. B 2013, 44, 878–888. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, G.; Yan, H.; Zhao, Y.; Li, T.; Feng, X. Reduction of low-grade manganese dioxide ore pellets by biomass wheat stalk. Acta Met. Sin. (Engl. Lett.) 2013, 26, 167–172. [Google Scholar] [CrossRef]
- Long, Y.F.; Su, J.; Ye, X.J.; Su, H.F.; Wen, Y.X. Reduction-Roast Leaching of Low-Grade Pyrolusite Using Bagasse as a Reducing Agent. Adv. Mater. Res. 2013, 699, 28–33. [Google Scholar] [CrossRef]
- Cheng, Z.; Zhu, G.; Zhao, Y. Study in reduction-roast leaching manganese from low-grade manganese dioxide ores using cornstalk as reductant. Hydrometallurgy 2009, 96, 176–179. [Google Scholar] [CrossRef]
- Sharma, T. Physico-chemical processing of low grade manganese ore. Int. J. Miner. Process. 1992, 35, 191–203. [Google Scholar] [CrossRef]
- Sinha, M.K.; Purcell, W. Reducing agents in the leaching of manganese ores: A comprehensive review. Hydrometallurgy 2019, 187, 168–186. [Google Scholar] [CrossRef]
- Gao, Y.; Olivas-Martinez, M.; Sohn, H.Y.; Kim, H.G.; Kim, C.W. Upgrading of Low-Grade Manganese Ore by Selective Reduction of Iron Oxide and Magnetic Separation. Met. Mater. Trans. B 2012, 43, 1465–1475. [Google Scholar] [CrossRef]
- Wu, Y.; Shi, B.; Ge, W.; Yan, C.J.; Yang, X. Magnetic Separation and Magnetic Properties of Low-Grade Manganese Carbonate Ore. JOM 2015, 67, 361–368. [Google Scholar] [CrossRef]
- Yu, D.; Paktunc, D. Calcium Chloride-Assisted Segregation Reduction of Chromite: Influence of Reductant Type and the Mechanism. Minerals 2018, 8, 45. [Google Scholar] [CrossRef]
- Yu, D.; Paktunc, D. Direct Production of Ferrochrome by Segregation Reduction of Chromite in the Presence of Calcium Chloride. Metals 2018, 8, 69. [Google Scholar] [CrossRef]
- Roine, A. HSC Chemistry; Outokumpu Research Oy: Pori, Finland, 2007. [Google Scholar]
- Boudouard, M.O. Recherches sur les equilibres chimiques. Ann. Chim. Phys. 1901, 24, 1–85. [Google Scholar]
- Yu, D.; Zhu, M.; Utigard, T.A.; Barati, M. TG/DTA study on the carbon monoxide and graphite thermal reduction of a high-grade iron nickel oxide residue with the presence of siliceous gangue. Thermochim. Acta 2014, 575, 1–11. [Google Scholar] [CrossRef]
- Lide, D.R. CRC Handbook of Chemistry and Physics, Internet Version 2005; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Zhang, W.; Cheng, C.Y. Manganese metallurgy review. Part II: Manganese separation and recovery from solution. Hydrometallurgy 2007, 89, 160–177. [Google Scholar] [CrossRef]


| Total Fe | Total Mn | SiO2 | Al2O3 | CaO | MgO | BaO | K2O | Na2O | PbO | LOI * |
|---|---|---|---|---|---|---|---|---|---|---|
| 40.54 | 13.86 | 7.14 | 2.57 | 0.18 | 0.18 | 1.17 | 0.30 | 0.04 | 0.02 | 3.41 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, D.; Cui, F.; Cong, Y.; Zhang, C.; Tian, Q.; Guo, X. Simultaneous Selective Chlorination and Carbothermic Reduction of High-Iron Manganese Ore for the Recovery of Manganese Chloride and Metallic Iron. Metals 2019, 9, 1124. https://doi.org/10.3390/met9101124
Yu D, Cui F, Cong Y, Zhang C, Tian Q, Guo X. Simultaneous Selective Chlorination and Carbothermic Reduction of High-Iron Manganese Ore for the Recovery of Manganese Chloride and Metallic Iron. Metals. 2019; 9(10):1124. https://doi.org/10.3390/met9101124
Chicago/Turabian StyleYu, Dawei, Fuhui Cui, Yunxiang Cong, Chunxi Zhang, Qinghua Tian, and Xueyi Guo. 2019. "Simultaneous Selective Chlorination and Carbothermic Reduction of High-Iron Manganese Ore for the Recovery of Manganese Chloride and Metallic Iron" Metals 9, no. 10: 1124. https://doi.org/10.3390/met9101124
APA StyleYu, D., Cui, F., Cong, Y., Zhang, C., Tian, Q., & Guo, X. (2019). Simultaneous Selective Chlorination and Carbothermic Reduction of High-Iron Manganese Ore for the Recovery of Manganese Chloride and Metallic Iron. Metals, 9(10), 1124. https://doi.org/10.3390/met9101124





