Calculation Model for Activity of FeO in Quaternary Slag System SiO2-CaO-Al2O3-FeO
Abstract
:1. Introduction
2. Calculation Model of Activity
3. Results and Discussion
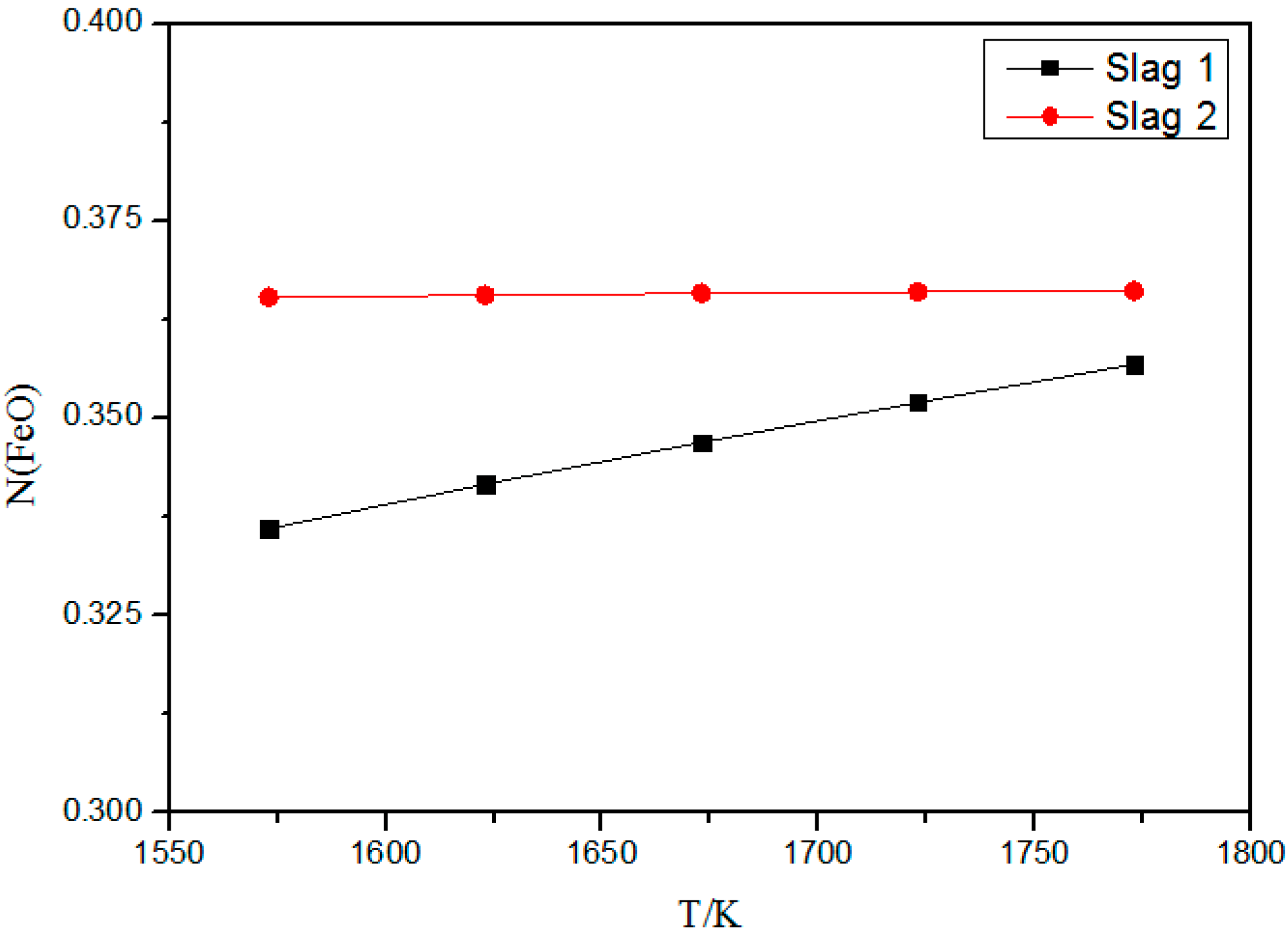
3.1. Effect of Temperature on Activity of FeO
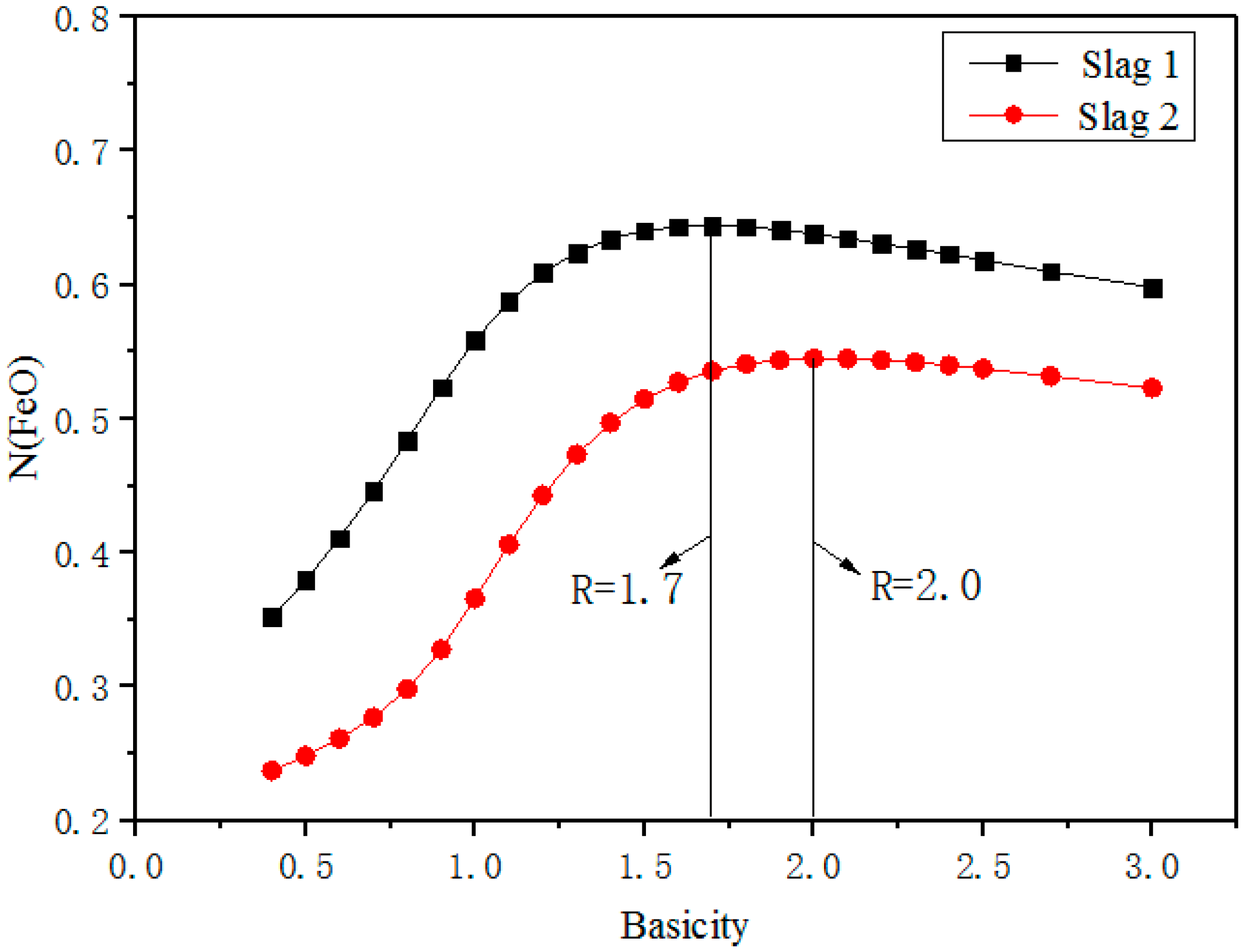
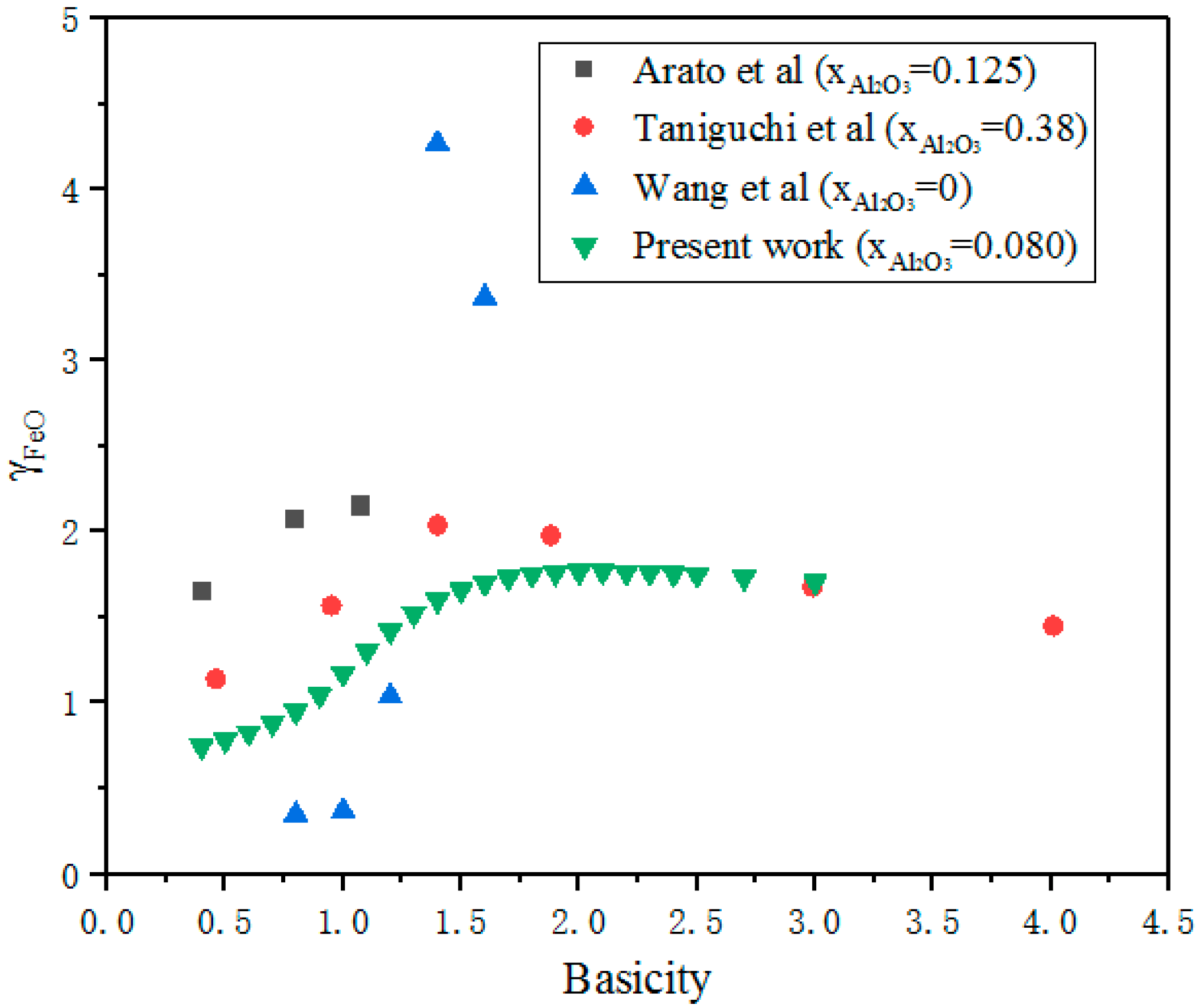
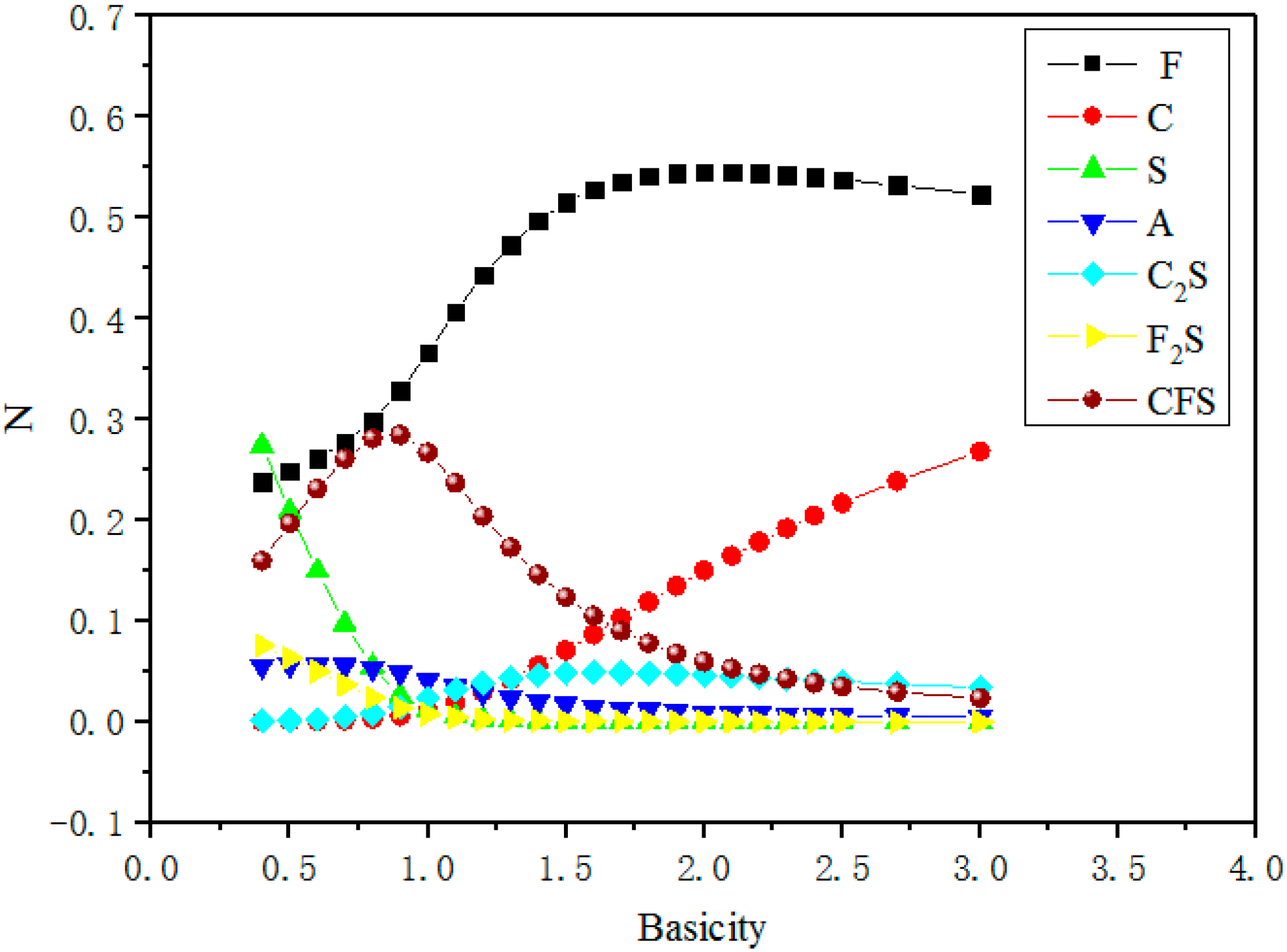
3.2. Effect of Basicity on Activity of FeO
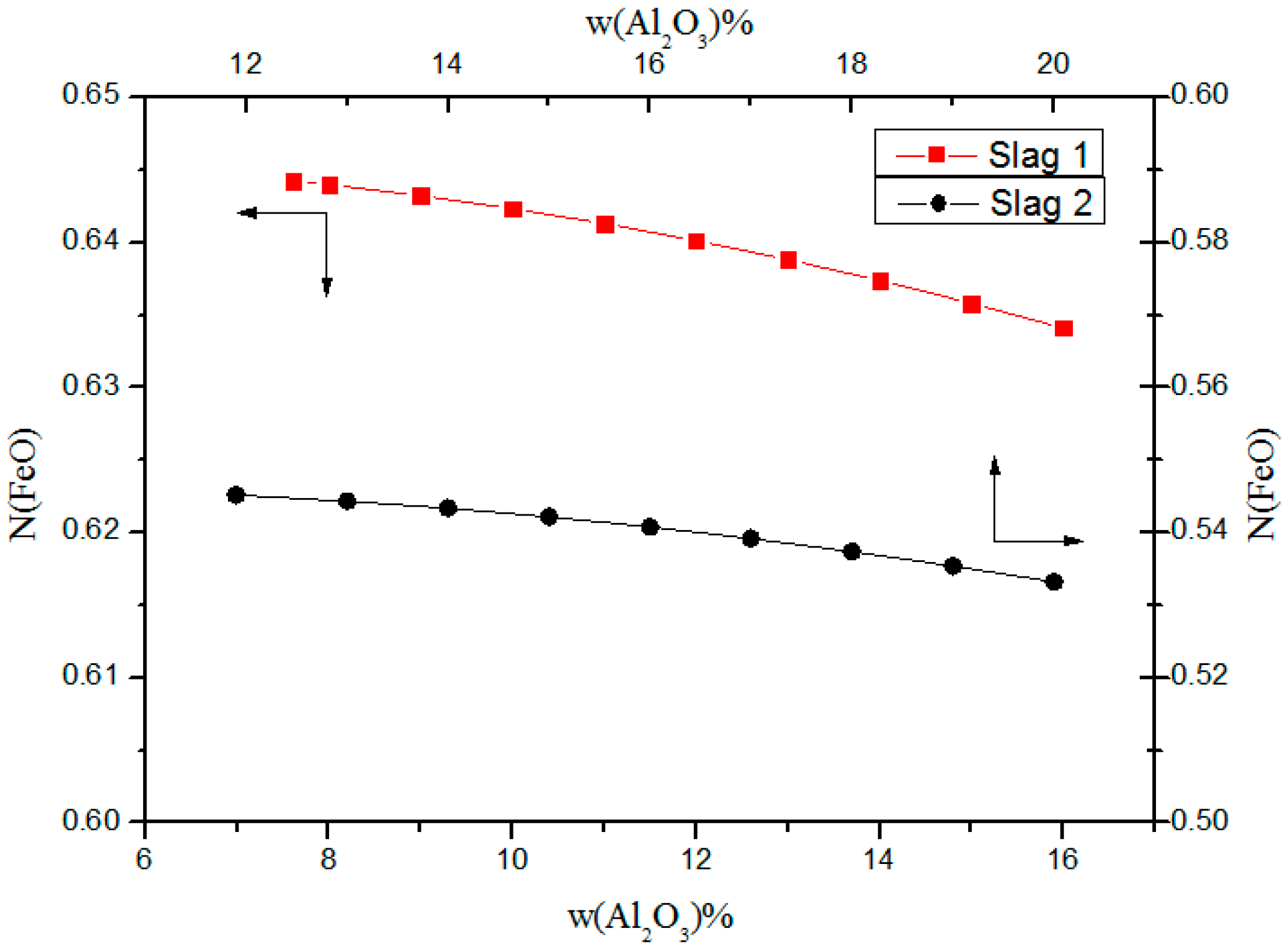
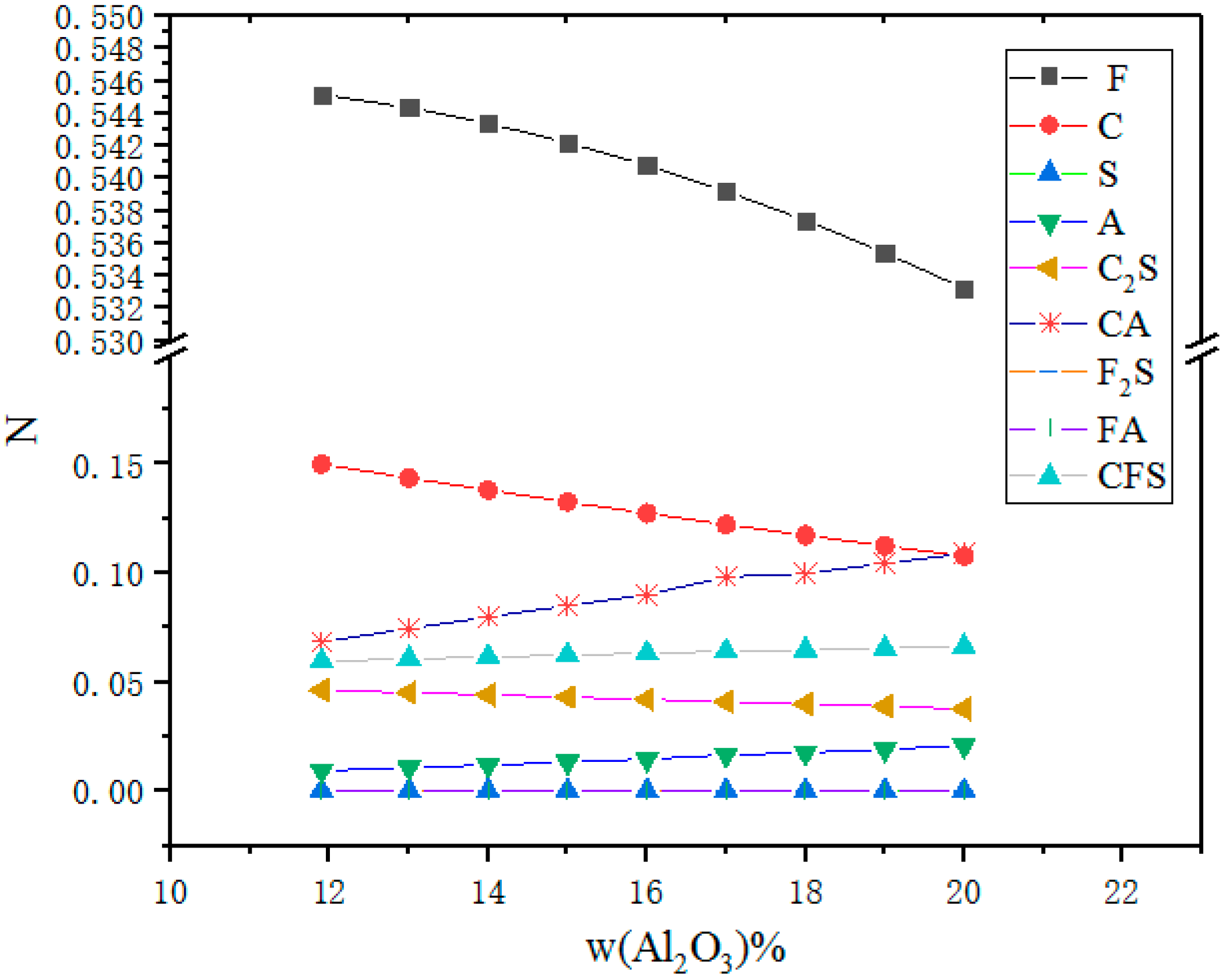
3.3. Effect of Al2O3 on Activity of FeO
4. Conclusions
- The calculation model can predict the activity of components generated in the quaternary slag system SiO2-CaO-Al2O3-FeO with different temperatures, basicity, and Al2O3 content. Temperature influences the activity of NFeO slightly at 1573–1773 K.
- Basicity is the major factor affecting the activity of FeO. With basicity ranging from 0.4 to 0.8, NFeO increased slightly due to the formation of CaO·FeO·SiO2. When basicity reaches near 2.0, there is maximum NFeO, as CaO can react with SiO2 to form the most stable and simplest structure, (Si/O = 1/4). When basicity is higher than 2.0, CaO not only reacts with SiO2, but also combines with FeO to form calcium ferrate compounds to decrease NFeO.
- The value of NFeO slightly decreases with increased Al2O3 content, as CaO reacts with Al2O3, which limits the free FeO generated from 2FeO·SiO2.
Author Contributions
Funding
Conflicts of Interest
References
- The International Copper Study Group (ICSG). The World Copper Factbook 2016. Available online: http://www.icsg.org/index.php?option=com_content&task=view&id=22&itemid=26 (accessed on 5 May 2018).
- Gorai, B.; Jana, R.K.; Premchand. Characteristics and utilization of copper slag—A review. Resour. Conserv. Recycl. 2003, 39, 299–313. [Google Scholar] [CrossRef]
- Gyurov, S.; Rabadjieva, D.; Kovacheva, D.; Kostova, Y. Kinetics of copper slag oxidation under nonisothermal conditions. J. Therm. Anal. Calorim. 2014, 116, 945–953. [Google Scholar] [CrossRef]
- Sarfo, P.; Young, J.; Ma, G.J.; Young, C. Characterization and recovery of valuables from waste copper smelting slag. In Advances in Molten Slags, Fluxes, and Salts, Proceedings of the 10th International Conference on Molten Slags, Fluxes and Salts, Seattle, WA, USA, 22–25 May 2016; Springer International: Cham, Switzerland, 2016; pp. 889–898. [Google Scholar]
- Yang, Z.H.; Lin, Q.; Lu, S.C.; He, Y.; Liao, G.D.; Ke, Y. Effect of CaO/SiO2 ratio on the preparation and crystallization of glass-ceramics from copper slag. Ceram. Int. 2014, 40, 7297–7305. [Google Scholar] [CrossRef]
- Yang, Z.H.; Lin, Q.; Xia, J.X.; He, Y.; Liao, G.D.; Ke, Y. Preparation and crystallization of glass-ceramics derived from iron-rich copper slag. J. Alloys Compd. 2013, 547, 354–360. [Google Scholar] [CrossRef]
- Sarfo, P.; Wyss, G.; Ma, G.J.; Das, A.; Young, C. Carbothermal reduction of copper smelter slag for recycling into pig iron and glass. Miner. Eng. 2017, 107, 8–19. [Google Scholar] [CrossRef]
- Zhang, J.; Dai, X.T.; Yan, D.L.; Qi, Y.H. Carbothermic reduction for collocation use between circulating steelmaking slag and copper slag. Steels 2015, 50, 114–118. [Google Scholar]
- Zhao, K.; Gong, X.R.; Li, J.; Liu, W.X.; Xing, H.W. Thermodynamic of recovering iron, copper, zinc in copper slag by direct reduction method. J. Environ. Eng. 2016, 10, 2638–2646. [Google Scholar]
- Tang, W.D.; Zhu, W.W.; Jiang, P.G.; Jing, Q.X. Kinetics of CuO chlorination process. Nonferr. Met. Sci. Eng. 2017, 8, 46–50. [Google Scholar]
- Cornelis, G.; Johnson, C.A.; Gerven, T.V.; Vandecasteele, C. Leaching mechanisms of oxyanionic metalloid and metal species in alkaline solid wastes: A review. Appl. Geochem. 2008, 23, 955–976. [Google Scholar] [CrossRef]
- Dixit, S.; Hering, J.G. Comparison of arsenic(V) and arsenic(III) sorption onto iron oxide minerals: Implications for arsenic mobility. Environ. Sci. Technol. 2003, 37, 4182–4189. [Google Scholar] [CrossRef]
- O’Neill, H.S.C.; Eggins, S.M. The effect of melt composition on trace element partitioning: An experimental investigation of the activity coefficients of FeO, NiO, CoO, MoO2 and MoO3 in silicate melts. Chem. Geol. 2002, 186, 151–181. [Google Scholar] [CrossRef]
- Taniguchi, Y.; Morita, K.; Sano, N. Activities of FeO in CaO-Al2O3-SiO2-FeO and CaO-Al2O3-CaF2-FeO Slags. ISIJ Int. 1997, 37, 956–961. [Google Scholar] [CrossRef]
- Arato, T.; Tokuda, M.; Ohtani, M. Activity measurement of FeTO in molten blast furnace type slags by EMF method. Trans. ISIJ 1982, 68, 2263–2270. [Google Scholar]
- Zhang, J. Computational Thermodynamics of Metallurgical Metals and Solutions, 1st ed.; Metallurgical Industry Press: Beijing, China, 2007; pp. 307–313. ISBN 9787502441647. [Google Scholar]
- Timucin, M.; Morris, A.E. Phase equilibria and thermodynamic studies in the system CaO-FeO-Fe2O3-SiO2. Metall. Trans. 1970, 1, 3193–3201. [Google Scholar]
- Wang, H.; Li, L. Applied Basic Research on the Effective Recovery of Valuable Metals from Copper Slags, 1st ed.; Science Press: Beijing, China, 2013; pp. 34–36. ISBN 9787030360250. [Google Scholar]
- Chuiko, N.M. Chemical bonds and the theory of slag structure. Metallurgist 1971, 19, 2–22. [Google Scholar]
- Wang, J.L.; Zhang, C.F.; Tong, C.R.; Zhang, W.H. Action concentration calculation model for slag system of CaO-FeO-Fe2O3-SiO2-Cu2O. J. Cent. South. Univ. Technol. 2009, 40, 282–287. [Google Scholar]
- Ma, X.C.; Yu, C.M.; Cheng, G.G.; Zhang, J. Calculation model for the concentration of MnO-FeO-SiO2-Al2O3 slag system. J. Iron. Steel Res. Int. 2011, 23, 4–7. [Google Scholar]
- Eisenhüttenleute, V.D.; Neuschütz, D. Slag Atlas, 2nd ed.; Verlag Stahleisen GmbH: Düsseldorf, Germany, 1995; pp. 39–154. ISBN 3514004579. [Google Scholar]
- Fu, C.S. The Principle of Non-Ferrous Metallurgy, 2nd ed.; Metallurgical Industry Press: Beijing, China, 1993; pp. 35–36. ISBN 9787502411503. [Google Scholar]
- Xue, Z.L.; Zou, F.; Xiong, R.; Li, J.L.; Zhu, H.Y. Activity model of FeO in refining slag. J. Chongqing Univ. 2015, 5, 98–103. [Google Scholar]
| Simple Ions | Ca2+, Fe2+, O2− |
| Complex Molecules | CaO·SiO2, 2CaO·SiO2, 3CaO·SiO2, 3CaO·2SiO2, 3Al2O3·2SiO2, 3CaO·Al2O3, 12CaO·7Al2O3, CaO·2Al2O3, CaO·6Al2O3, CaO·Al2O3, 2FeO·SiO2, FeO·Al2O3, CaO·Al2O3·2SiO2, 2CaO·Al2O3·SiO2, CaO·FeO·SiO2, Al2O3, SiO2 |
| (Ca2+ + O2−) + SiO2 = CaO·SiO2 | = −81416 − 10.498T | N5 = k1N2N3 |
| 2(Ca2+ + O2−) + SiO2 = 2CaO·SiO2 | = −160431 + 4.160T | N6 = k2N3 |
| 3(Ca2+ + O2−) + SiO2 = 3CaO·SiO2 | = −93366 − 23.03T | N7 = k3N3 |
| 3(Ca2+ + O2−) + 2SiO2 = 3CaO·2SiO2 | = −236973 + 9.63T | N8 = k4 |
| 3Al2O3 + 2SiO2 = 3Al2O3·2SiO2 | = 8589.9 − 17.39T | N9 = k5 |
| 3(Ca2+ + O2−) + Al2O3 = 3CaO·Al2O3 | = −17000 − 32.0 T | N10 = k6N4 |
| 12(Ca2+ + O2−) + 7Al2O3 = 12CaO·7Al2O3 | = −86100 − 205.1T | N11 = k7 |
| (Ca2+ + O2−) + 2Al2O3 = CaO·2Al2O3 | = −16400 − 26.8T | N12 = k8N2 |
| (Ca2+ + O2−) + 6Al2O3 = CaO·6Al2O3 | = −17430 − 37.2T | N13 = k9N2 |
| (Ca2+ + O2−) + Al2O3 = CaO·Al2O3 | = −18120 − 18.62T | N14 = k10N2N4 |
| 2(Fe2+ + O2−) + SiO2 = 2FeO·SiO2 | = −28596 + 3.349T | N15 = k11N3 |
| (Fe2+ + O2−) + Al2O3 = FeO·Al2O3 | = −33272.8 + 6.1028T | N16 = k12N1N4 |
| (Ca2+ + O2−) + Al2O3 + 2SiO2 = CaO·Al2O3·2SiO2 | = 28006 − 74.795T | N17 = k13N2N4 |
| 2(Ca2+ + O2−) + Al2O3 + SiO2 = 2CaO·Al2O3·SiO2 | = −17092 + 8.778T | N18 = k14N3N4 |
| (Ca2+ + O2−) + (Fe2+ + O2−) + SiO2 = CaO·FeO·SiO2 | = −72996.8 − 29.3169T | N19 = k15N1N2N3 |
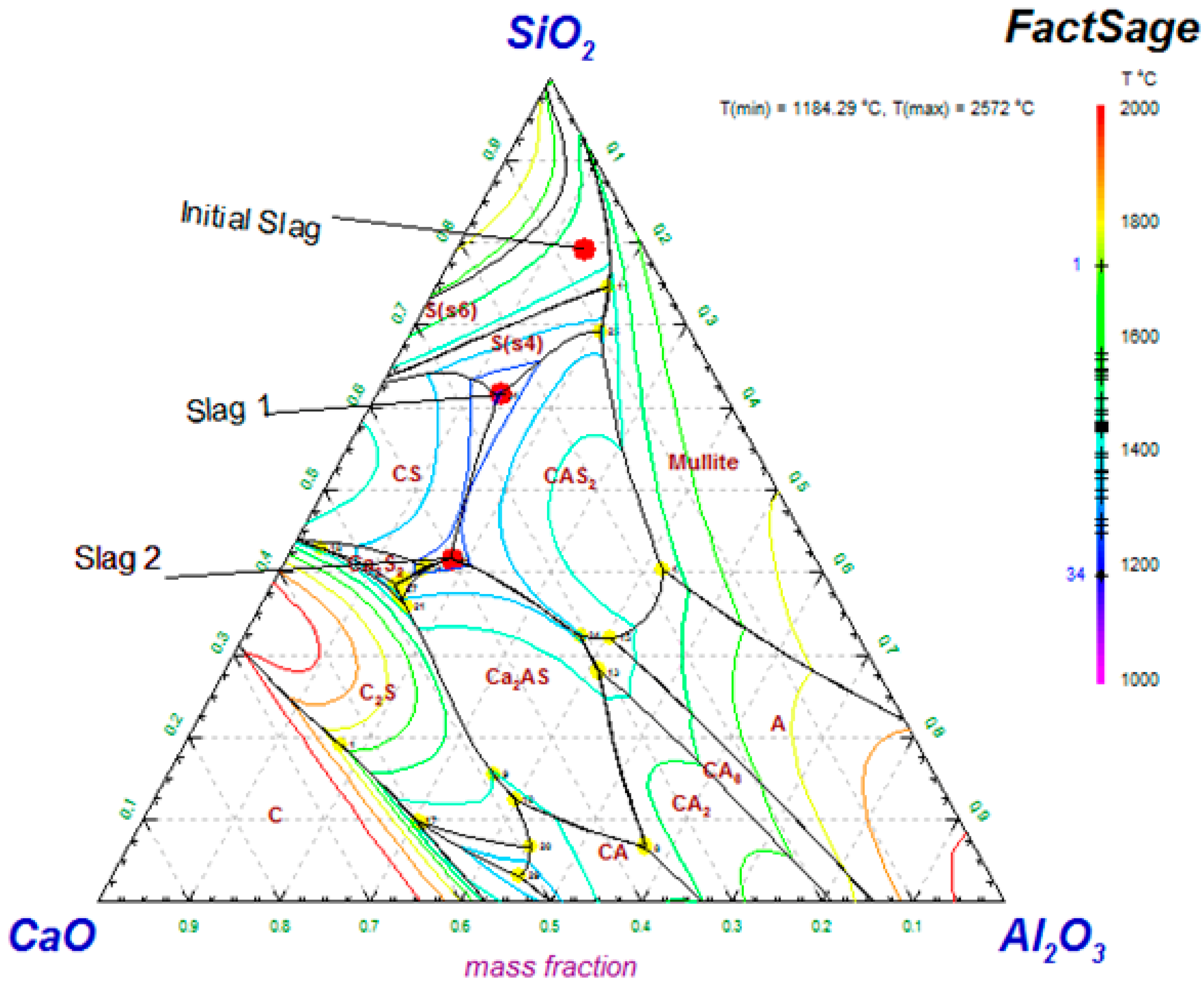
| Slag Type | SiO2 | Al2O3 | CaO | FeO | R |
|---|---|---|---|---|---|
| Initial | 39.96 | 7.21 | 3.35 | 49.47 | 0.08 |
| 1 | 34.91 | 7.61 | 14.27 | 43.22 | 0.4 |
| 2 | 27.50 | 11.90 | 26.55 | 34.05 | 0.95 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Ma, G.; Liu, M.; Zou, J. Calculation Model for Activity of FeO in Quaternary Slag System SiO2-CaO-Al2O3-FeO. Metals 2018, 8, 714. https://doi.org/10.3390/met8090714
Li Z, Ma G, Liu M, Zou J. Calculation Model for Activity of FeO in Quaternary Slag System SiO2-CaO-Al2O3-FeO. Metals. 2018; 8(9):714. https://doi.org/10.3390/met8090714
Chicago/Turabian StyleLi, Zhi, Guojun Ma, Mengke Liu, and Jingjing Zou. 2018. "Calculation Model for Activity of FeO in Quaternary Slag System SiO2-CaO-Al2O3-FeO" Metals 8, no. 9: 714. https://doi.org/10.3390/met8090714
APA StyleLi, Z., Ma, G., Liu, M., & Zou, J. (2018). Calculation Model for Activity of FeO in Quaternary Slag System SiO2-CaO-Al2O3-FeO. Metals, 8(9), 714. https://doi.org/10.3390/met8090714





