Kinetics of Iron Extraction from Coal Fly Ash by Hydrochloric Acid Leaching
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
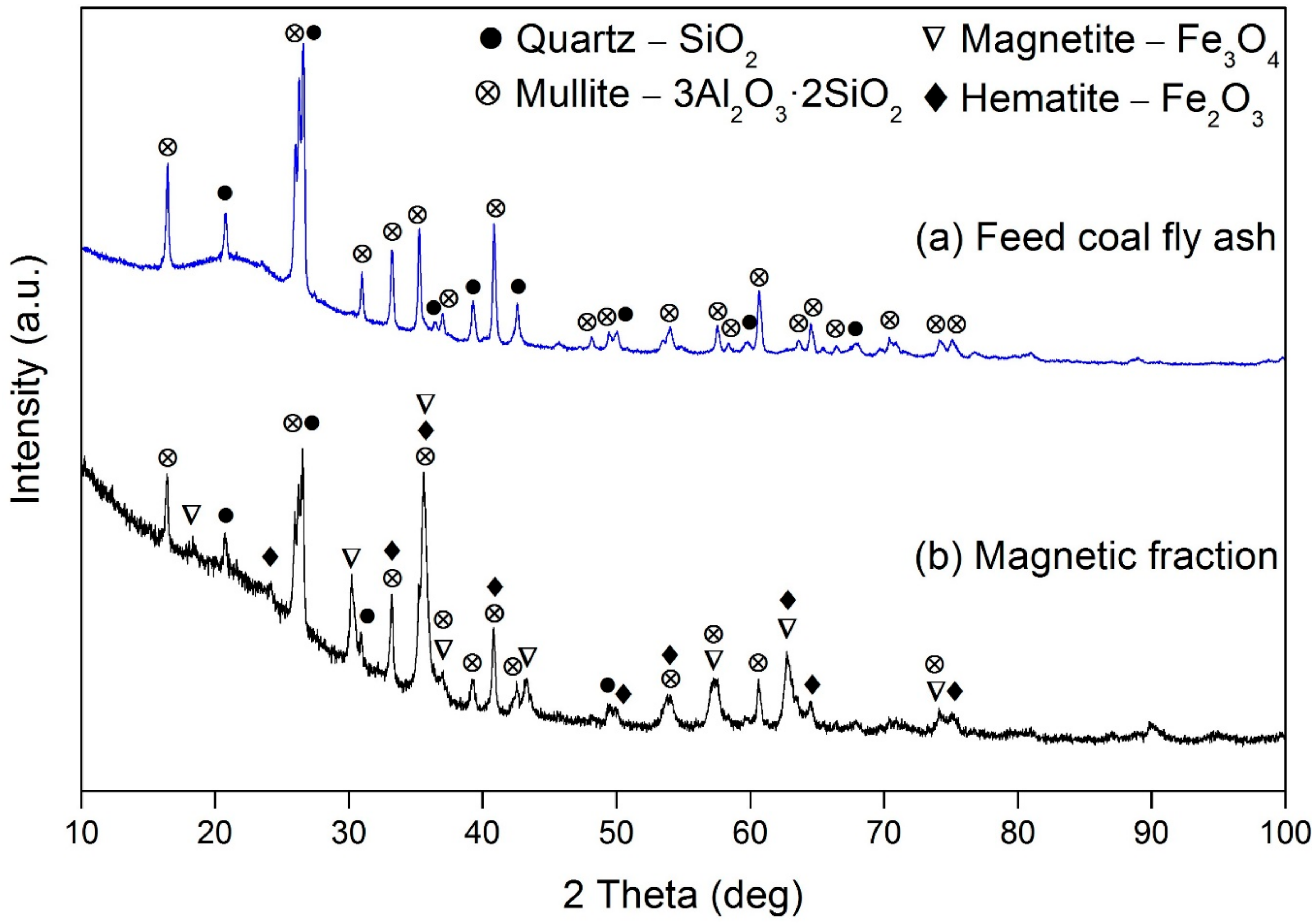
3.1. Characterization of the Coal Fly Ash
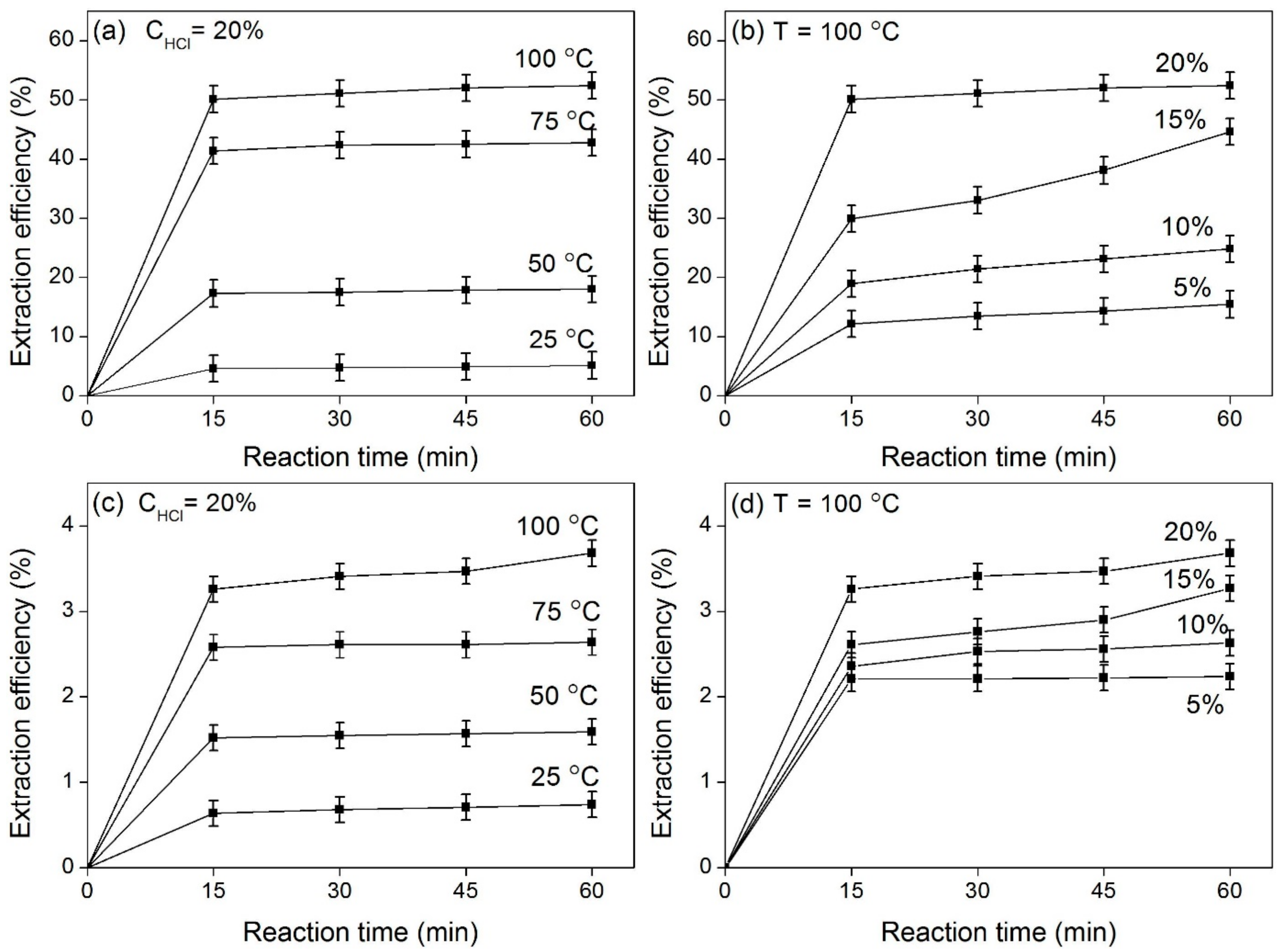
3.2. CFA Leaching by Hydrochloric Acid
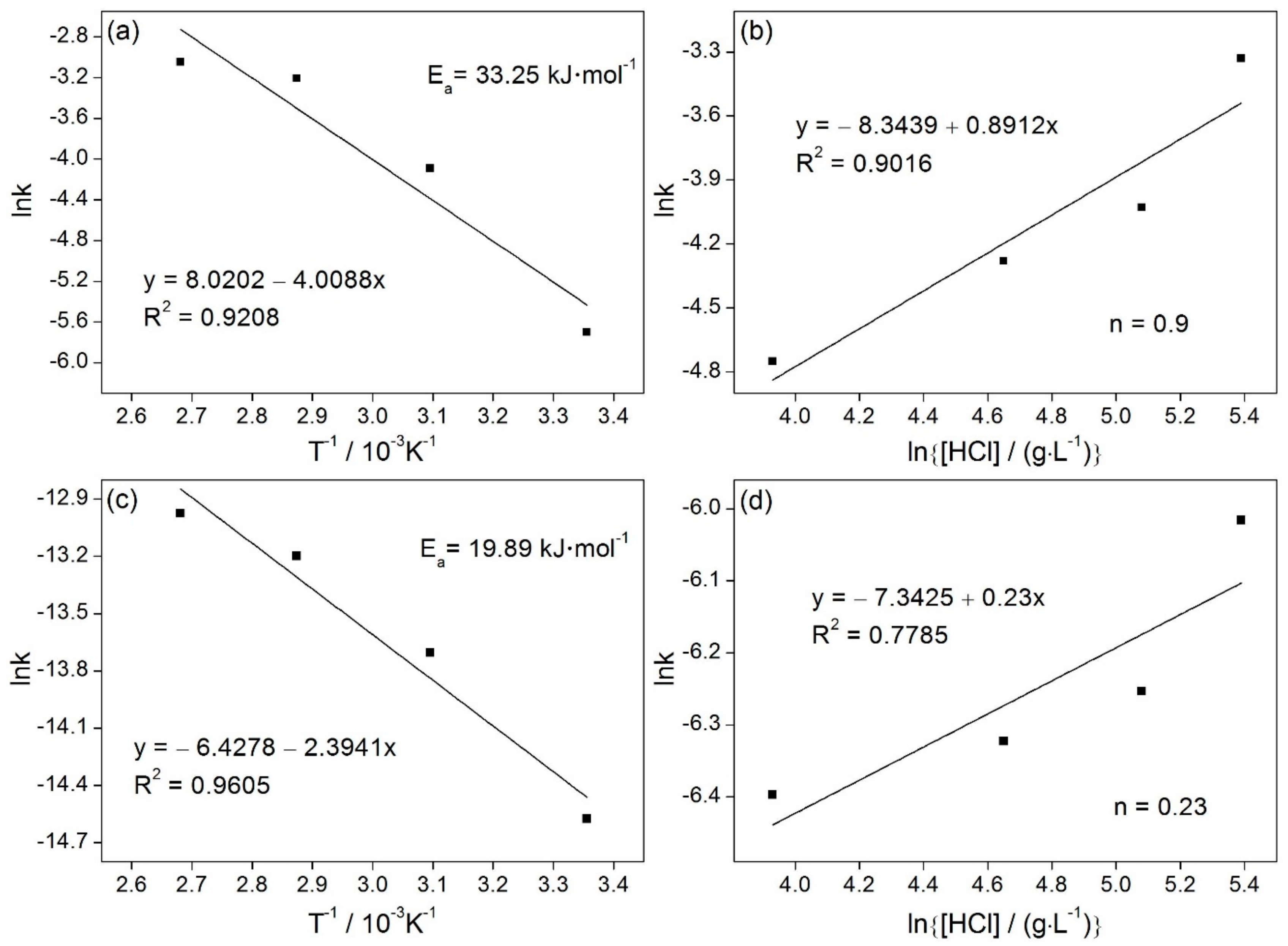
3.3. Kinetic Study of Fe and Al Leaching in HCl Solutions
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- World Energy Council (WEC). World Energy Resources; World Energy Council: London, UK, 2016; p. 1028. [Google Scholar]
- Ryabov, Y.V.; Delitsyn, L.M.; Ezhova, N.N.; Lavrinenko, A.A. PAV-2 conditioning agent application efficiency in flotation of unburned carbon from coal-fired power plants fly ash. Obogashchenie Rud 2018, 43–49. [Google Scholar] [CrossRef]
- Shemi, A.; Mpana, R.N.; Ndlovu, S.; van Dyk, L.D.; Sibanda, V.; Seepe, L. Alternative techniques for extracting alumina from coal fly ash. Miner. Eng. 2012, 34, 30–37. [Google Scholar] [CrossRef]
- Yao, Z.T.; Xia, M.S.; Sarker, P.K.; Chen, T. A review of the alumina recovery from coal fly ash, with a focus in China. Fuel 2014, 120, 74–85. [Google Scholar] [CrossRef]
- Ding, J.; Ma, S.; Shen, S.; Xie, Z.; Zheng, S.; Zhang, Y. Research and industrialization progress of recovering alumina from fly ash: A concise review. Waste Manag. 2017, 60, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Luo, K.; Fan, J.; Lu, H. Experimental study of extracting alumina from coal fly ash using fluidized beds at high temperature. Fuel 2017, 199, 22–27. [Google Scholar] [CrossRef]
- Lu, M.; Chang, Y.; Chen, L. Kinetics of desilification pretreatment from high-aluminium coal fly ash in alkaline medium under pressure. Chem. Res. Chin. Univ. 2017, 33, 282–286. [Google Scholar] [CrossRef]
- Jiang, Z.Q.; Yang, J.; Ma, H.W.; Wang, L.; Ma, X. Reaction behaviour of Al2O3 SiO2 in high alumina coal fly ash during alkali hydrothermal process. Trans. Nonferr. Met. Soc. China 2015, 25, 2065–2072. [Google Scholar] [CrossRef]
- Wang, R.C.; Zhai, Y.C.; Ning, Z.Q.; Ma, P.H. Kinetics of SiO2 leaching from Al2O3 extracted slag of fly ash with sodium hydroxide solution. Trans. Nonferr. Met. Soc. China 2014, 24, 1928–1936. [Google Scholar] [CrossRef]
- Sangita, S.; Nayak, N.; Panda, C.R. Extraction of aluminium as aluminium sulphate from thermal power plant fly ashes. Trans. Nonferr. Met. Soc. China 2017, 27, 2082–2089. [Google Scholar] [CrossRef]
- Seidel, A.; Zimmels, Y. Mechanism and kinetics of aluminum and iron leaching from coal fly ash by sulfuric acid. Chem. Eng. Sci. 1998, 53, 3835–3852. [Google Scholar] [CrossRef]
- Wu, C.Y.; Yu, H.F.; Zhang, H.F. Extraction of aluminum by pressure acid-leaching method from coal fly ash. Trans. Nonferr. Met. Soc. China 2012, 22, 2282–2288. [Google Scholar] [CrossRef]
- Xu, D.; Li, H.; Bao, W.; Wang, C. A new process of extracting alumina from high-alumina coal fly ash in NH4HSO4 + H2SO4 mixed solution. Hydrometallurgy 2016, 165, 336–344. [Google Scholar] [CrossRef]
- Seidel, A.; Sluszny, A.; Shelef, G.; Zimmels, Y. Self inhibition of aluminum leaching from coal fly ash by sulfuric acid. Chem. Eng. J. 1999, 72, 195–207. [Google Scholar] [CrossRef]
- Tripathy, A.K.; Sarangi, C.K.; Tripathy, B.C.; Sanjay, K.; Bhattacharya, I.N.; Mahapatra, B.K.; Behera, P.K.; Satpathy, B.K. Aluminium recovery from NALCO fly ash by acid digestion in the presence of fluoride ion. Int. J. Miner. Process. 2015, 138, 44–48. [Google Scholar] [CrossRef]
- Cheng, F.; Cui, L.; Miller, J.D.; Wang, X. Aluminum leaching from calcined coal waste using hydrochloric acid solution. Miner. Process. Extr. Metall. Rev. 2012, 33, 391–403. [Google Scholar] [CrossRef]
- Smirnov, A.; Kibartas, D.; Senyuta, A.; Panov, A. Miniplant Tests of HCl Technology of Alumina Production; Springer: Berlin/Heidelberg, Germany, 2018; pp. 3–8. [Google Scholar]
- Cui, L.; Cheng, F.; Zhou, J. Preparation of high purity AlCl3·6H2O crystals from coal mining waste based on iron(III) removal using undiluted ionic liquids. Sep. Purif. Technol. 2016, 167, 45–54. [Google Scholar] [CrossRef]
- Guo, Y.; Lv, H.; Yang, X.; Cheng, F. AlCl3·6H2O recovery from the acid leaching liquor of coal gangue by using concentrated hydrochloric inpouring. Sep. Purif. Technol. 2015, 151, 117–183. [Google Scholar] [CrossRef]
- Zhang, N.; Yang, Y.; Wang, Z.; Shi, Z.; Gao, B.; Hu, X.; Tao, W.; Liu, F.; Yu, J. Study on the thermal decomposition of aluminium chloride hexahydrate. Can. Metall. Q. 2018, 57, 235–244. [Google Scholar] [CrossRef]
- Al-Othman, A.; Demopoulos, G.P. Gypsum crystallization and hydrochloric acid regeneration by reaction of calcium chloride solution with sulfuric acid. Hydrometallurgy 2009, 96, 95–102. [Google Scholar] [CrossRef]
- Singh, R.; Singh, L.; Singh, S.V. Beneficiation of iron and aluminium oxides from fly ash at lab scale. Int. J. Miner. Process. 2015, 145, 32–37. [Google Scholar] [CrossRef]
- Haider, U.; Bittnar, Z.; Kopecky, L.; Šmilauer, V.; Pokorny, J.; Zaleska, M.; Prošek, Z.; Hrbek, V. Determining the role of individual fly ash particles in influencing the variation in the overall physical, morphological, and chemical properties of fly ash. Acta Polytech. 2016, 56, 265–282. [Google Scholar] [CrossRef]
- Reddy, B.; Mishra, S.; Banerjee, G. Kinetics of leaching of a gibbsitic bauxite with hydrochloric acid. Hydrometallurgy 1999, 51, 131–138. [Google Scholar] [CrossRef]
- Zhao, A.C.; Liu, Y.; Zhang, T.A.; Lü, G.Z.; Dou, Z.H. Thermodynamics study on leaching process of gibbsitic bauxite by hydrochloric acid. Trans. Nonferr. Met. Soc. China 2013, 23, 266–270. [Google Scholar] [CrossRef]
- Zhao, A.; Zhang, T.A.; Lv, G.; Tian, W. Kinetics of the Leaching Process of an Australian Gibbsitic Bauxite by Hydrochloric Acid. Adv. Mater. Sci. Eng. 2016, 2016, 5813542. [Google Scholar] [CrossRef]
- Zhu, P.-W.; Zeng, W.-Q.; Xu, X.-L.; Cheng, L.-M.; Jiang, X.; Shi, Z.-L. Influence of acid leaching and calcination on iron removal of coal kaolin. Int. J. Miner. Metall. Mater. 2014, 21, 317–325. [Google Scholar] [CrossRef]
- Li, Z.; Cao, Y.; Jiang, Y.; Han, G.; Fan, G.; Chang, L. Removal of Potassium and Iron in Low Grade Bauxite by a Calcination-Acid Leaching Process. Minerals 2018, 8, 125. [Google Scholar] [CrossRef]
- Han, H.J.; Chen, Y.G.; Lu, J.; Xu, T.T.; Jiang, Y.H.; Bai, J.L. Investigation of removing iron from fly ash. Adv. Mater. Res. 2013, 807–809, 1194–1197. [Google Scholar] [CrossRef]
- Li, S.; Qin, S.; Kang, L.; Liu, J.; Wang, J.; Li, Y. An Efficient Approach for Lithium and Aluminum Recovery from Coal Fly Ash by Pre-Desilication and Intensified Acid Leaching Processes. Metals 2017, 7, 272. [Google Scholar] [CrossRef]
- Kastrinaki, G.; Lorentzou, S.; Karagiannakis, G.; Rattenbury, M.; Woodhead, J.; Konstandopoulos, A.G. Parametric synthesis study of iron based nanoparticles via aerosol spray pyrolysis route. J. Aerosol Sci. 2018, 115, 96–107. [Google Scholar] [CrossRef]
- Zheng, J.; Liu, K.; Cai, W.; Qiao, L.; Ying, Y.; Li, W.; Yu, J.; Lin, M.; Che, S. Effect of chloride ion on crystalline phase transition of iron oxide produced by ultrasonic spray pyrolysis. Adv. Powder Technol. 2018. [Google Scholar] [CrossRef]
- Gurmen, S.; Ebin, B. Production and characterization of the nanostructured hollow iron oxide spheres and nanoparticles by aerosol route. J. Alloys Compd. 2010, 492, 585–589. [Google Scholar] [CrossRef]
- Koo, H.Y.; Kim, J.H.; Hong, S.K.; Han, J.M.; Ko, Y.N.; Kang, Y.C.; Kang, S.H.; Cho, S.B. Characteristics of Fe powders prepared by spray pyrolysis from various types of Fe precursors as a heat pellet material. Met. Mater. Int. 2010, 16, 941–946. [Google Scholar] [CrossRef]
- Lee, M.-S.; Ahn, J.-G.; Oh, Y.-J. Chemical Model of the FeCl3-HCl-H2O Solutions at 25 °C. Mater. Trans. 1997, 5639, 1117–1133. [Google Scholar] [CrossRef]
- Jamett, N.E.; Hernández, P.C.; Casas, J.M.; Taboada, M.E. Speciation in the Fe(III)-Cl(I)-H2O System at 298.15 K, 313.15 K, and 333.15 K (25 °C, 40 °C, and 60 °C). Metall. Mater. Trans. B 2018, 49, 451–459. [Google Scholar] [CrossRef]
- Livenspiel, O. Chemical Reaction Engineering, 3rd ed.; John Wiley & Sons, Inc.: New York, NY, USA, 1999; ISBN 047125424X. [Google Scholar]
- Tian, L.; Liu, Y.; Zhang, T.A.; Lv, G.Z.; Zhou, S.; Zhang, G.Q. Kinetics of indium dissolution from marmatite with high indium content in pressure acid leaching. Rare Met. 2017, 36, 69–76. [Google Scholar] [CrossRef]


| Composition | SiO2 | Al2O3 | Fe2O3 | TiO2 | K2O | CaO | MgO | MnO | Na2O | P2O5 | Loss on Ignition |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Concentration, wt % | 57.1 | 31.3 | 5.31 | 1.30 | 0.41 | 1.78 | 0.65 | 0.09 | 0.42 | 0.33 | 1.31 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valeev, D.; Mikhailova, A.; Atmadzhidi, A. Kinetics of Iron Extraction from Coal Fly Ash by Hydrochloric Acid Leaching. Metals 2018, 8, 533. https://doi.org/10.3390/met8070533
Valeev D, Mikhailova A, Atmadzhidi A. Kinetics of Iron Extraction from Coal Fly Ash by Hydrochloric Acid Leaching. Metals. 2018; 8(7):533. https://doi.org/10.3390/met8070533
Chicago/Turabian StyleValeev, Dmitry, Alexandra Mikhailova, and Alexandra Atmadzhidi. 2018. "Kinetics of Iron Extraction from Coal Fly Ash by Hydrochloric Acid Leaching" Metals 8, no. 7: 533. https://doi.org/10.3390/met8070533
APA StyleValeev, D., Mikhailova, A., & Atmadzhidi, A. (2018). Kinetics of Iron Extraction from Coal Fly Ash by Hydrochloric Acid Leaching. Metals, 8(7), 533. https://doi.org/10.3390/met8070533






