Impurity Effects in Electroplated-Copper Solder Joints
Abstract
1. Introduction
2. Electroplating of Cu
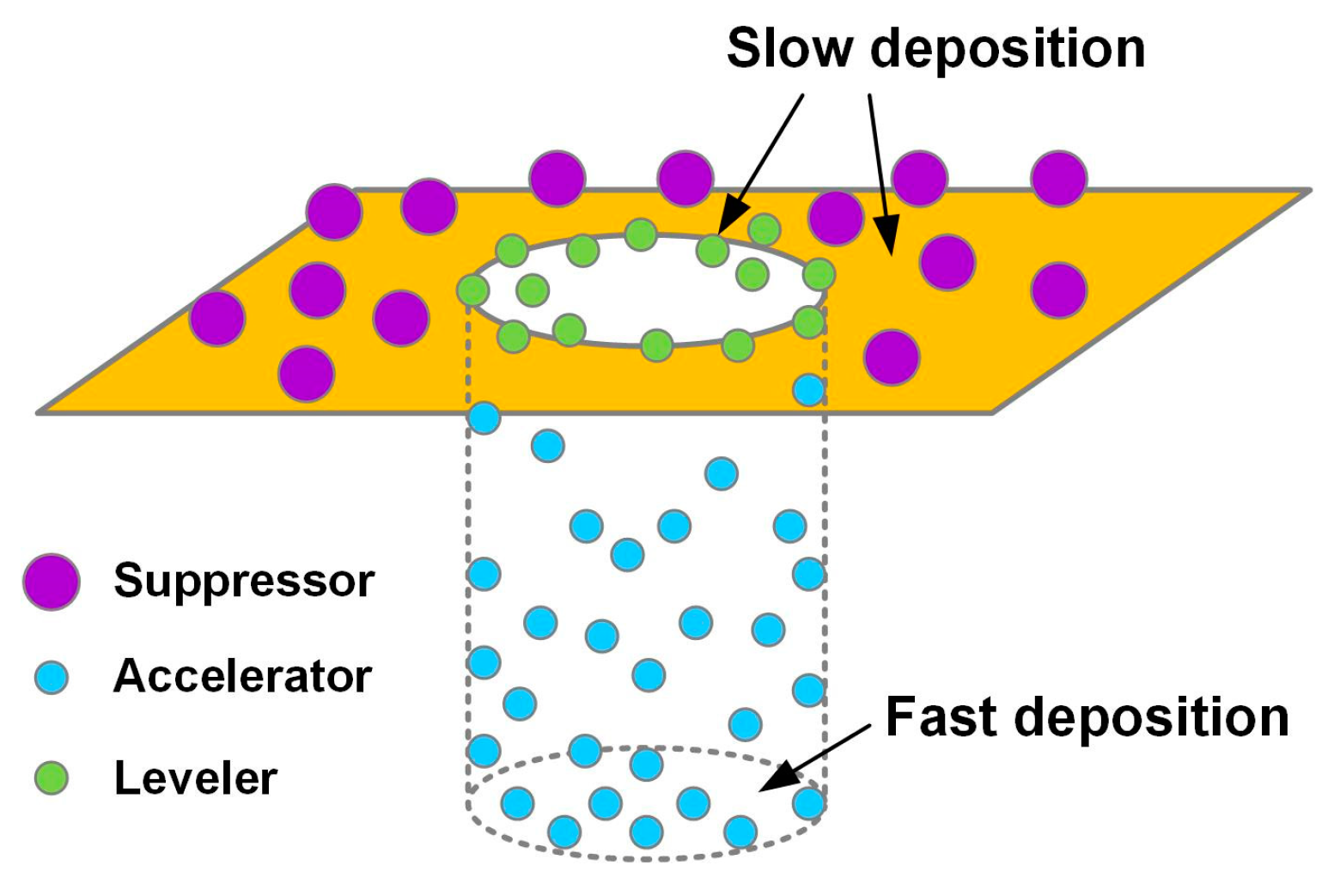
2.1. Functional Additives Used for Electrodeposition of Cu
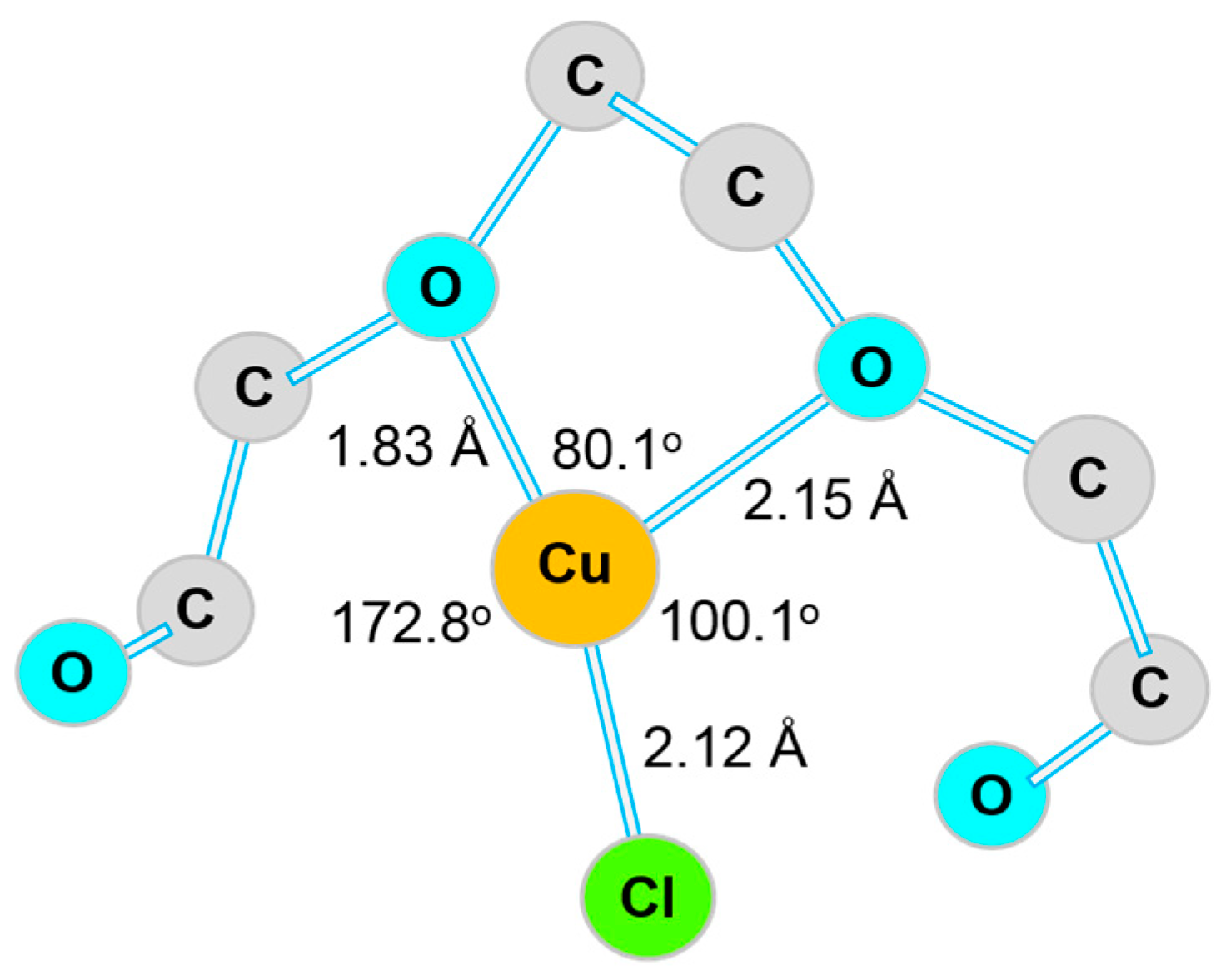
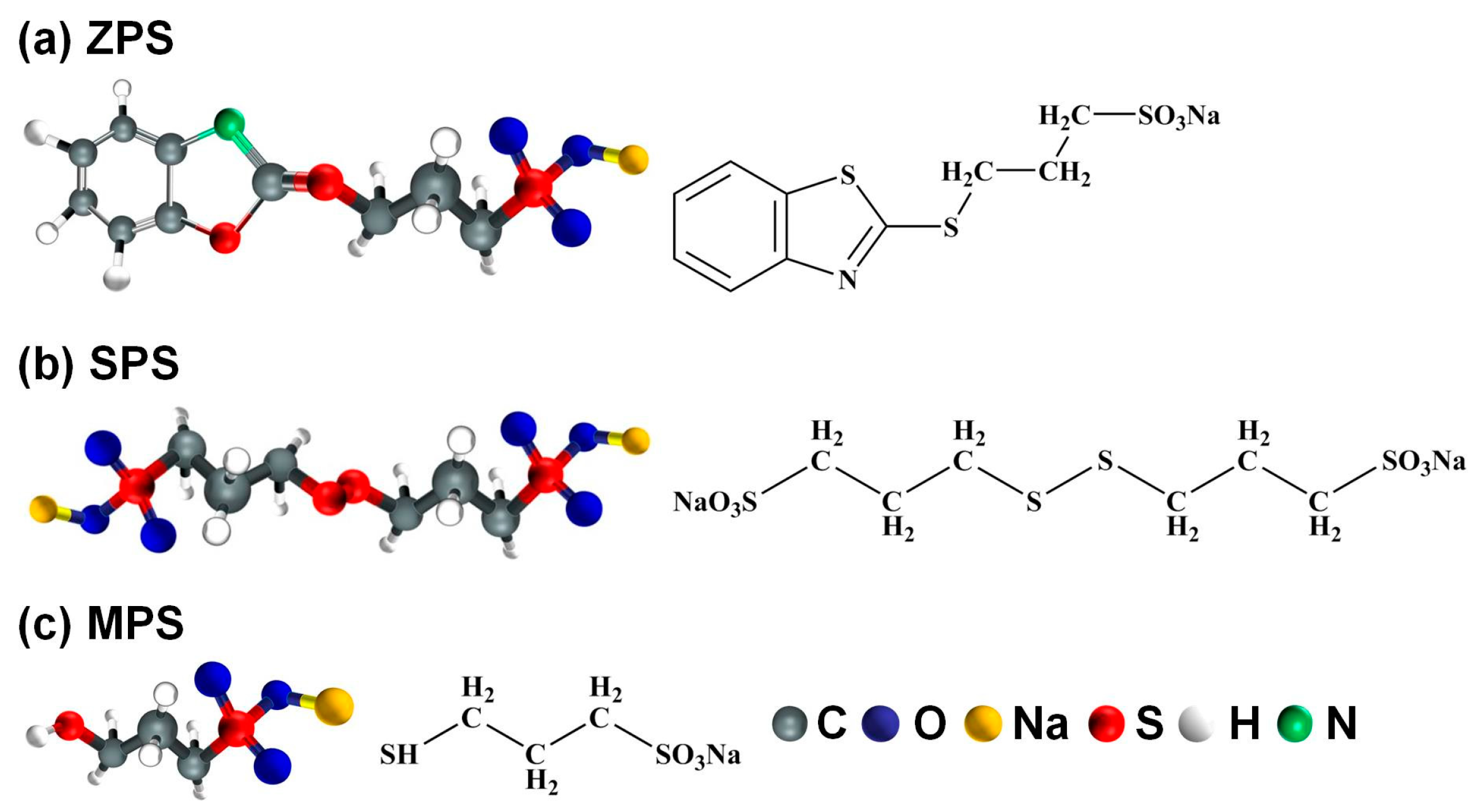
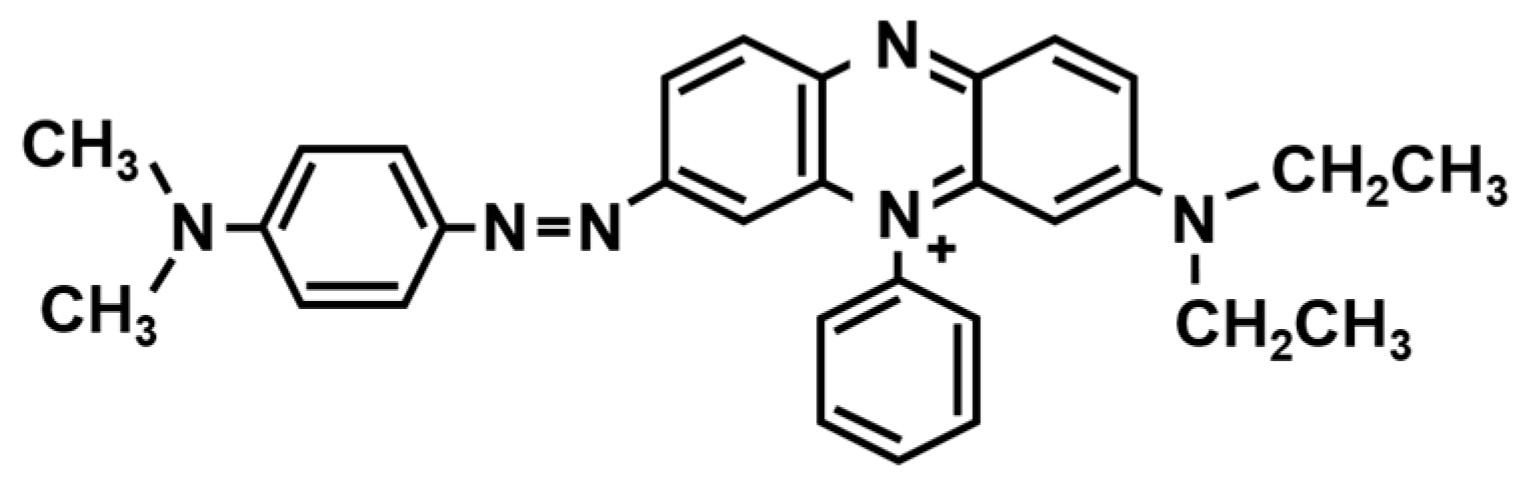
2.1.1. Suppressor
2.1.2. Accelerator
2.1.3. Leveler
2.2. Impurity Residues in Electroplated Cu Layer
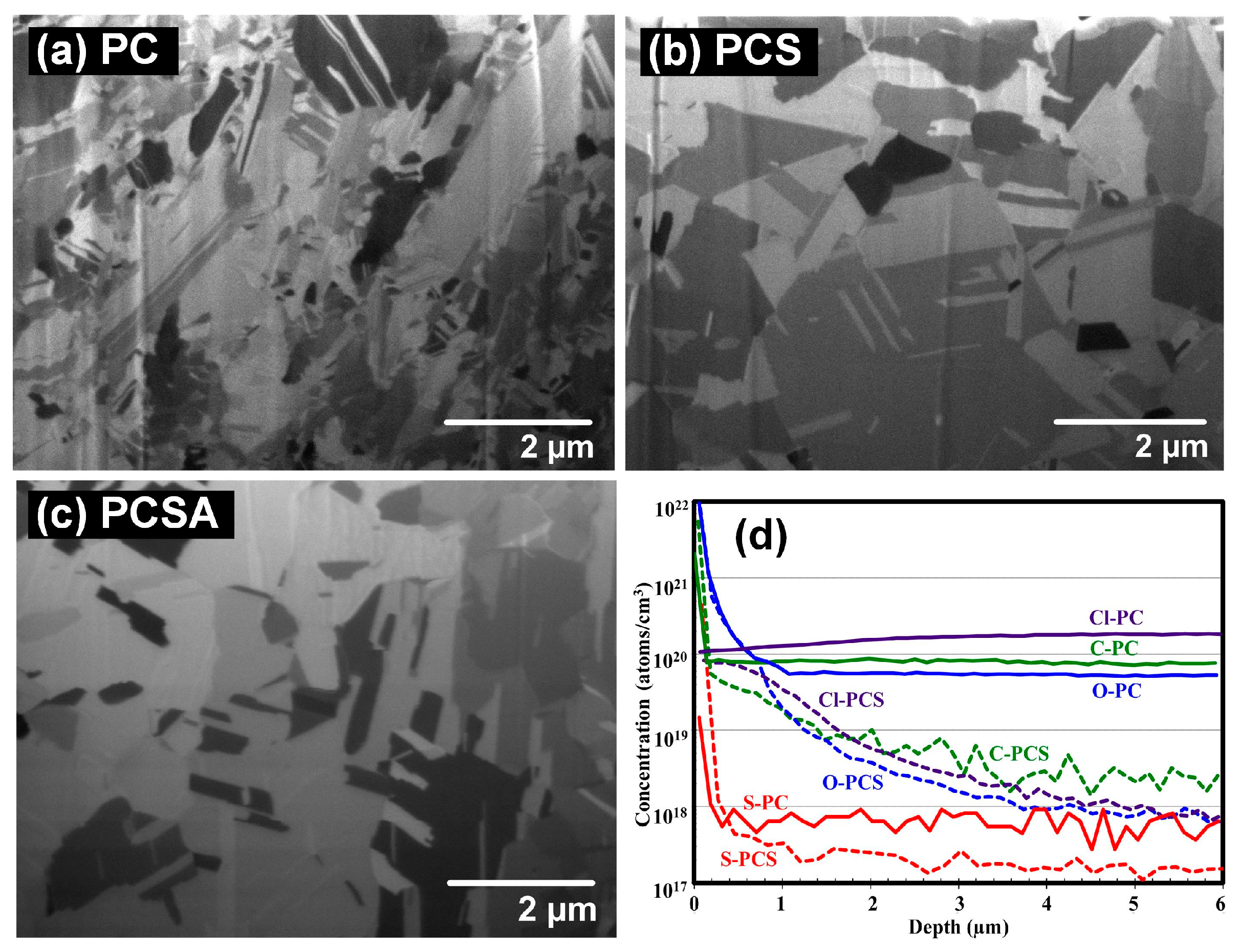
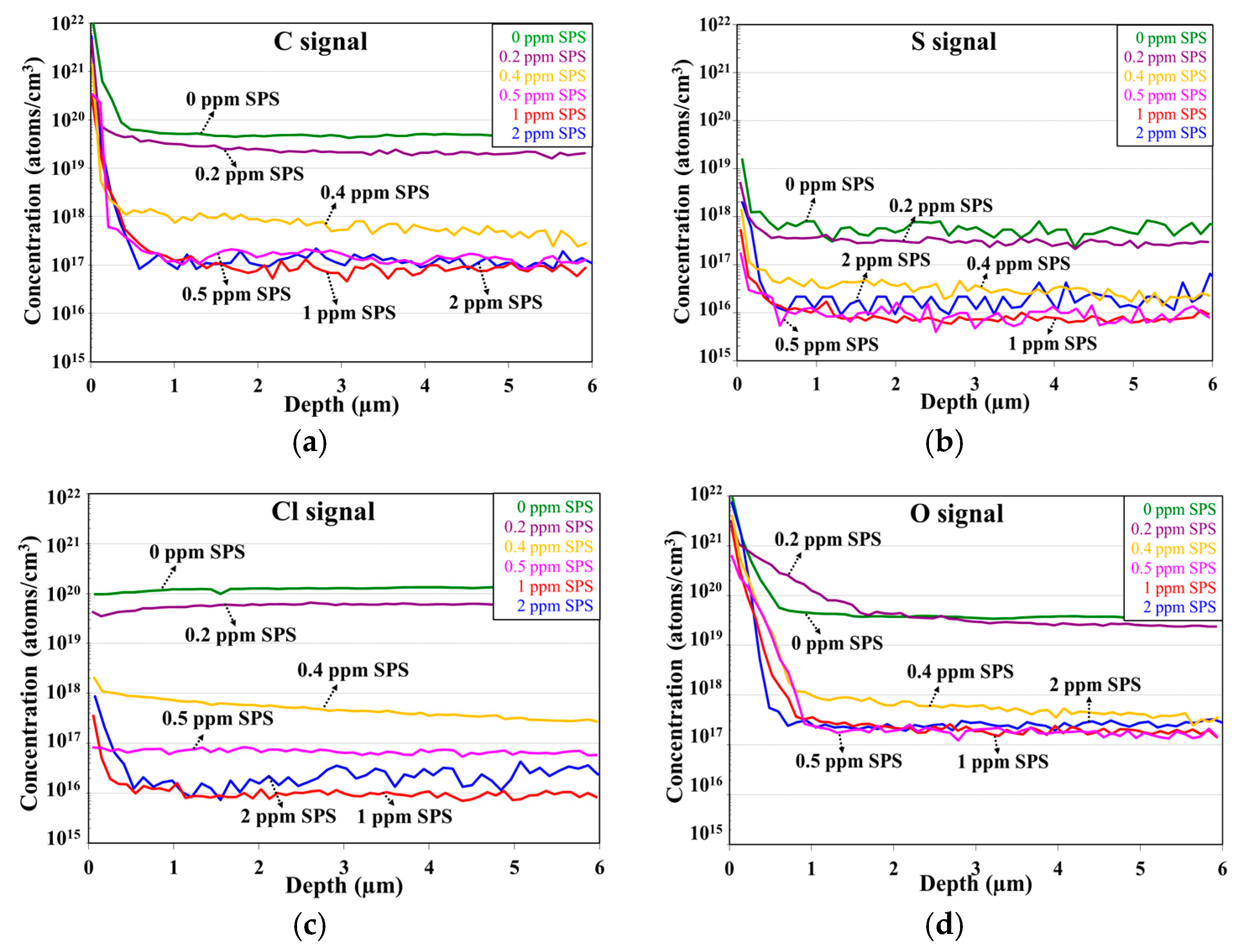
2.2.1. Effects of Plating Parameter and Additive Formula on Impurity Incorporation
2.2.2. Effects of Impurity Residues on Electroplated Cu Layer
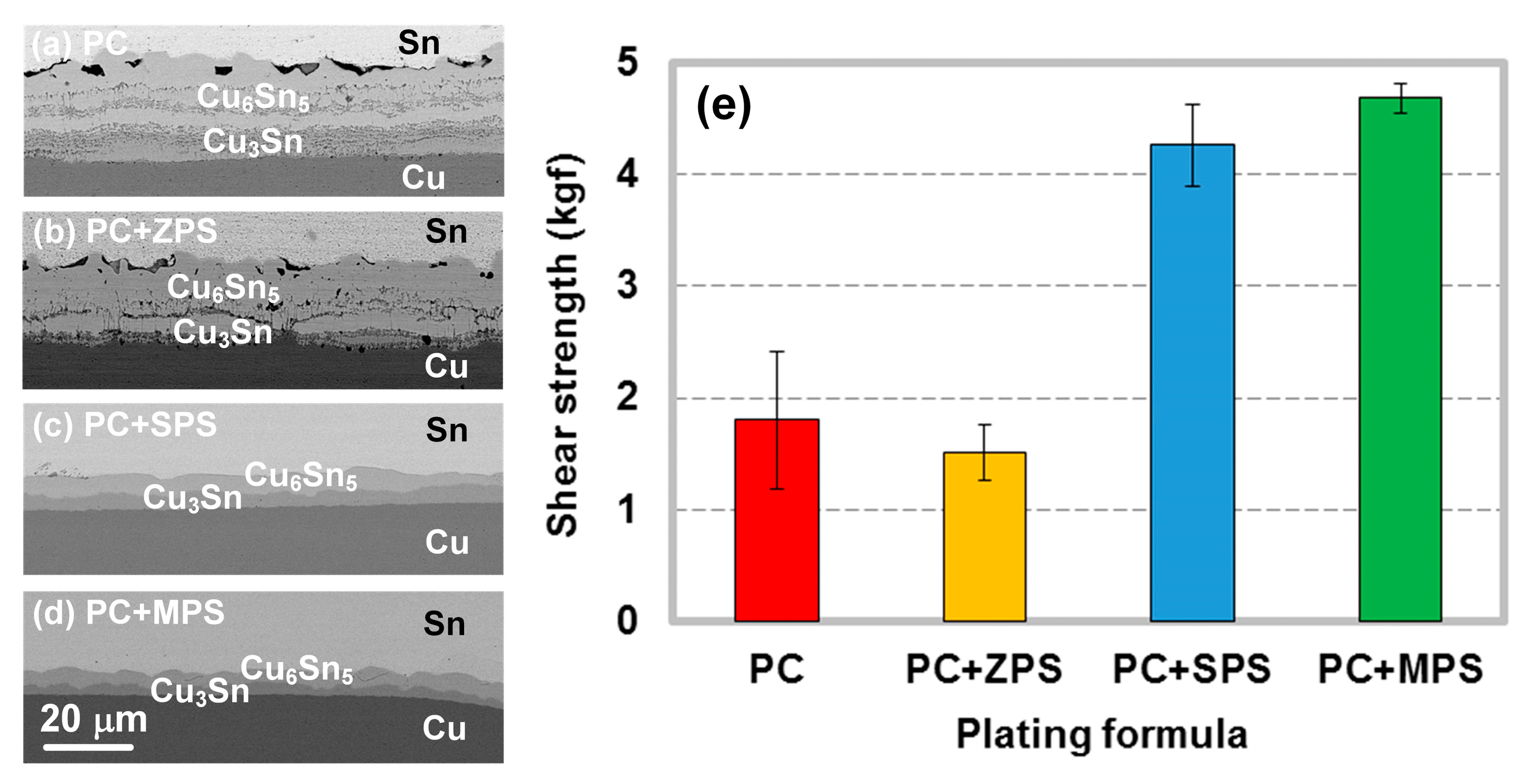
3. Effects of Impurity on Interfacial Reactions of Electroplated-Cu Solder Joints
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andricacos, P.C.; Uzoh, C.; Dukovic, J.O.; Horkans, J.; Deligianni, H. Damascene copper electroplating for chip interconnections. IBM J. Res. Dev. 1998, 42, 567–574. [Google Scholar] [CrossRef]
- Van Olmen, J.; Huyghebaert, C.; Coenen, J.; Van Aelst, J.; Sleeckx, E.; Van Ammel, A.; Armini, S.; Katti, G.; Vaes, J.; Dehaene, W. Integration challenges of copper through silicon via (TSV) metallization for 3D-stacked IC integration. Microelectron. Eng. 2011, 88, 745–748. [Google Scholar] [CrossRef]
- Hofmann, L.; Ecke, R.; Schulz, S.E.; Gessner, T. Investigations regarding through silicon via filling for 3D integration by periodic pulse reverse plating with and without additives. Microelectron. Eng. 2011, 88, 705–708. [Google Scholar] [CrossRef]
- Moffat, T.; Josell, D. Extreme bottom-up superfilling of through-silicon-vias by damascene processing: Suppressor disruption, positive feedback and turing patterns. J. Electrochem. Soc. 2012, 159, D208–D216. [Google Scholar] [CrossRef]
- Sun, J.-J.; Kondo, K.; Okamura, T.; Oh, S.; Tomisaka, M.; Yonemura, H.; Hoshino, M.; Takahashi, K. High-aspect-ratio copper via filling used for three-dimensional chip stacking. J. Electrochem. Soc. 2003, 150, G355–G358. [Google Scholar] [CrossRef]
- Dow, W.-P.; Yen, M.-Y.; Lin, W.-B.; Ho, S.-W. Influence of molecular weight of polyethylene glycol on microvia filling by copper electroplating. J. Electrochem. Soc. 2005, 152, C769–C775. [Google Scholar] [CrossRef]
- Feng, Z.V.; Li, X.; Gewirth, A.A. Inhibition due to the interaction of polyethylene glycol, chloride, and copper in plating baths: A surface-enhanced raman study. J. Phys. Chem. B 2003, 107, 9415–9423. [Google Scholar] [CrossRef]
- Kelly, J.J.; West, A.C. Copper deposition in the presence of polyethylene glycol I. Quartz crystal microbalance study. J. Electrochem. Soc. 1998, 145, 3472–3476. [Google Scholar] [CrossRef]
- Hayase, M.; Taketani, M.; Aizawa, K.; Hatsuzawa, T.; Hayabusa, K. Copper bottom-up deposition by breakdown of PEG-Cl inhibition. Electrochem. Solid State Lett. 2002, 5, C98–C101. [Google Scholar] [CrossRef]
- Beica, R.; Sharbono, C.; Ritzdorf, T. Through silicon via copper electrodeposition for 3D integration. In Proceedings of the 58th Electronic Components and Technology Conference, ECTC 2008, Lake Buena Vista, FL, USA, 27–30 May 2008; IEEE: Piscataway, NJ, USA, 2008; pp. 577–583. [Google Scholar]
- Chiu, Y.-D.; Dow, W.-P. Accelerator screening by cyclic voltammetry for microvia filling by copper electroplating. J. Electrochem. Soc. 2013, 160, D3021–D3027. [Google Scholar] [CrossRef]
- Tan, M.; Harb, J.N. Additive behavior during copper electrodeposition in solutions containing Cl−, PEG, and SPS. J. Electrochem. Soc. 2003, 150, C420–C425. [Google Scholar] [CrossRef]
- Jin, Y.; Sui, Y.; Wen, L.; Ye, F.; Sun, M.; Wang, Q. Competitive adsorption of PEG and SPS on copper surface in acidic electrolyte containing Cl−. J. Electrochem. Soc. 2013, 160, D20–D27. [Google Scholar] [CrossRef]
- Farndon, E.E.; Campbell, S.A.; Walsh, F.C. Effect of thiourea, benzotriazole and 4,5-dithiaoctane-1,8-disulphonic acid on the kinetics of copper deposition from dilute acid sulfate solutions. J. Appl. Electrochem. 1995, 25, 574–583. [Google Scholar] [CrossRef]
- Kelly, J.J.; West, A.C. Copper deposition in the presence of polyethylene glycol II. Electrochemical impedance spectroscopy. J. Electrochem. Soc. 1998, 145, 3477–3481. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, J.; Yin, L.; Kondos, P.; Parks, C.; Borgesen, P.; Henderson, D.; Cotts, E.; Dimitrov, N. Influence of plating parameters and solution chemistry on the voiding propensity at electroplated copper-solder interface. J. Appl. Electrochem. 2008, 38, 1695–1705. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, L.; Bliznakov, S.; Kondos, P.; Borgesen, P.; Henderson, D.W.; Parks, C.; Wang, J.; Cotts, E.J.; Dimitrov, N. Improving copper electrodeposition in the microelectronics industry. IEEE Trans. Compon. Packag. Technol. 2010, 33, 127–137. [Google Scholar] [CrossRef]
- Yu, T.-Y.; Lee, H.; Hsu, H.-L.; Dow, W.-P.; Cheng, H.-K.; Liu, K.-C.; Chen, C.-M. Effects of Cu electroplating formulas on the interfacial microstructures of Sn/Cu joints. J. Electrochem. Soc. 2016, 163, D734–D741. [Google Scholar] [CrossRef]
- Stangl, M.; Acker, J.; Oswald, S.; Uhlemann, M.; Gemming, T.; Baunack, S.; Wetzig, K. Incorporation of sulfur, chlorine, and carbon into electroplated Cu thin films. Microelectron. Eng. 2007, 84, 54–59. [Google Scholar] [CrossRef]
- Reed-Hill, R.E.; Abbaschian, R. Physical Metallurgy Principles, 3rd ed.; PWS Publishing Company: Boston, MA, USA, 1994; pp. 262–263. ISBN 0-534-92173-6. [Google Scholar]
- Chen, C.-C.; Hsieh, C.-H.; Lee, Y.-W.; Yang, C.-H.; Ho, C.-E. Formation mechanism of pinholes in electroplated Cu films and its mitigation. Thin Solid Films 2015, 596, 209–215. [Google Scholar] [CrossRef]
- Cheng, H.-K.; Huang, C.-W.; Lee, H.; Wang, Y.-L.; Liu, T.-F.; Chen, C.-M. Interfacial reactions between Cu and SnAgCu solder doped with minor Ni. J. Alloys Compd. 2015, 622, 529–534. [Google Scholar] [CrossRef]
- Lin, C.-P.; Chen, C.-M.; Yen, Y.-W. Enhanced growth of the Cu6Sn5 phase in the Sn/Ag/Cu and Sn/Cu multilayers subjected to applied strain. J. Alloys Compd. 2014, 591, 297–303. [Google Scholar] [CrossRef]
- Zeng, K.; Stierman, R.; Chiu, T.-C.; Edwards, D.; Ano, K.; Tu, K. Kirkendall void formation in eutectic SnPb solder joints on bare Cu and its effect on joint reliability. J. Appl. Phys. 2005, 97, 024508. [Google Scholar] [CrossRef]
- Wang, Y.-W.; Lin, Y.; Kao, C.R. Kirkendall voids formation in the reaction between Ni-doped snag lead-free solders and different Cu substrates. Microelectron. Reliab. 2009, 49, 248–252. [Google Scholar] [CrossRef]
- Ho, C.E.; Kuo, T.T.; Wang, C.C.; Wu, W.H. Inhibiting the growth of Cu3Sn and kirkendall voids in the Cu/Sn-Ag-Cu system by minor Pd alloying. Electron. Mater. Lett. 2012, 8, 495–501. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, Y.; Tu, C.; Kao, C. Effects of minor Fe, Co, and Ni additions on the reaction between SnAgCu solder and Cu. J. Alloys Compd. 2009, 478, 121–127. [Google Scholar] [CrossRef]
- Yin, L.; Borgesen, P. On the root cause of kirkendall voiding in Cu3Sn. J. Mater. Res. 2011, 26, 455–466. [Google Scholar] [CrossRef]
- Yu, J.; Kim, J. Effects of residual S on kirkendall void formation at Cu/Sn–3.5Ag solder joints. Acta Mater. 2008, 56, 5514–5523. [Google Scholar] [CrossRef]
- Wu, J.-Y.; Lee, H.; Wu, C.-H.; Lin, C.-F.; Dow, W.-P.; Chen, C.-M. Effects of electroplating additives on the interfacial reactions between Sn and Cu electroplated layers. J. Electrochem. Soc. 2014, 161, D522–D527. [Google Scholar] [CrossRef]
- Lee, H.; Yu, T.-Y.; Cheng, H.-K.; Liu, K.-C.; Chan, P.-F.; Dow, W.-P.; Chen, C.-M. Impurity incorporation in the Cu electrodeposit and its effects on the microstructural evolution of the Sn/Cu solder joints. J. Electrochem. Soc. 2017, 164, D457–D462. [Google Scholar] [CrossRef]
- Ross, G.; Vuorinen, V.; Paulasto-Kröckel, M. Void formation and its impact on CuSn intermetallic compound formation. J. Alloys Compd. 2016, 677, 127–138. [Google Scholar] [CrossRef]
- Wafula, F.; Liu, Y.; Yin, L.; Bliznakov, S.; Borgesen, P.; Cotts, E.; Dimitrov, N. Impact of key deposition parameters on the voiding sporadically occurring in solder joints with electroplated copper. J. Electrochem. Soc. 2010, 157, D111–D118. [Google Scholar] [CrossRef]
- Wafula, F.; Liu, Y.; Yin, L.; Borgesen, P.; Cotts, E.; Dimitrov, N. Effect of the deposition parameters on the voiding propensity of solder joints with Cu electroplated in a hull cell. J. Appl. Electrochem. 2011, 41, 469–480. [Google Scholar] [CrossRef]
- Wafula, F.; Yin, L.; Borgesen, P.; Andala, D.; Dimitrov, N. Influence of poly(ethylene glycol) degradation on voiding sporadically occurring in solder joints with electroplated Cu. J. Electron. Mater. 2012, 41, 1898–1906. [Google Scholar] [CrossRef]
- Cheng, H.K.; Lin, Y.J.; Chen, C.M.; Liu, K.C.; Wang, Y.L.; Liu, T.F. Microstructural evolution of Cu/solder/Cu pillar-type structures with different diffusion barriers. Metall. Mater. Trans. A 2016, 47, 3971–3980. [Google Scholar] [CrossRef]
- Jones, T.D.A.; Bernassau, A.; Flynn, D.; Price, D.; Beadel, M.; Desmulliez, M.P.Y. Copper electroplating of PCB interconnects using megasonic acoustic streaming. Ultrason. Sonochem. 2018, 42, 434–444. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, M. Copper Electrodepositon. In Copper Electrodeposition for Nanofabrication of Electronics Devices, Nanostructure Science and Technology; Kondo, K., Akolkar, R.N., Barkey, D.P., Yokoi, M., Eds.; Springer: New York, NY, USA, 2014; pp. 3–25. [Google Scholar]
- Dow, W.-P.; Huang, H.-S.; Yen, M.-Y.; Huang, H.-C. Influence of convection-dependent adsorption of additives on microvia filling by copper electroplating. J. Electrochem. Soc. 2005, 152, C425–C434. [Google Scholar] [CrossRef]
- Kondo, K.; Matsumoto, T.; Watanabe, K. Role of additives for copper damascene electrodeposition experimental study on inhibition and acceleration effects. J. Electrochem. Soc. 2004, 151, C250–C255. [Google Scholar] [CrossRef]
- Reid, J. Copper electrodeposition: Principles and recent progress. Jpn. J. Appl. Phys. 2001, 40, 2650–2657. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.-B. Effect of Cl− on the adsorption-desorption behavior of PEG. J. Electrochem. Soc. 2008, 155, D263–D269. [Google Scholar] [CrossRef]
- Moffat, T.P.; Wheeler, D.; Josell, D. Electrodeposition of copper in the SPS-PEG-Cl additive system I. Kinetic measurements: Influence of SPS. J. Electrochem. Soc. 2004, 151, C262–C271. [Google Scholar] [CrossRef]
- Gallaway, J.W.; West, A.C. PEG, PPG, and their triblock copolymers as suppressors in copper electroplating. J. Electrochem. Soc. 2008, 155, D632–D639. [Google Scholar] [CrossRef]
- Choe, S.; Kim, M.J.; Kim, H.C.; Cho, S.K.; Ahn, S.H.; Kim, S.-K.; Kim, J.J. Degradation of bis (3-sulfopropyl) disulfide and its influence on copper electrodeposition for feature filling. J. Electrochem. Soc. 2013, 160, D3179–D3185. [Google Scholar] [CrossRef]
- Chen, T.-C.; Tsai, Y.-L.; Hsu, C.-F.; Dow, W.-P.; Hashimoto, Y. Effects of brighteners in a copper plating bath on throwing power and thermal reliability of plated through holes. Electrochim. Acta 2016, 212, 572–582. [Google Scholar] [CrossRef]
- Hau-Riege, S.P.; Thompson, C.V. In situ transmission electron microscope studies of the kinetics of abnormal grain growth in electroplated copper films. Appl. Phys. Lett. 2000, 76, 309–311. [Google Scholar] [CrossRef]
- Lagrange, S.; Brongersma, S.; Judelewicz, M.; Saerens, A.; Vervoort, I.; Richard, E.; Palmans, R.; Maex, K. Self-annealing characterization of electroplated copper films. Microelectron. Eng. 2000, 50, 449–457. [Google Scholar] [CrossRef]
- Ho, C.-E.; Chen, C.-C.; Lu, M.-K.; Lee, Y.-W.; Wu, Y.-S. In-situ study on the self-annealing behavior of electroplated Cu through the cantilever method, XRD, and EBSD. Surf. Coat. Technol. 2016, 303, 86–93. [Google Scholar] [CrossRef]
- Harper, J.; Cabral, C., Jr.; Andricacos, P.; Gignac, L.; Noyan, I.; Rodbell, K.; Hu, C. Mechanisms for microstructure evolution in electroplated copper thin films near room temperature. J. Appl. Phys. 1999, 86, 2516–2525. [Google Scholar] [CrossRef]
- Cheng, M.-Y.; Chen, K.-W.; Liu, T.-F.; Wang, Y.-L.; Feng, H.-P. Effects of direct current and pulse-reverse copper plating waveforms on the incubation behavior of self-annealing. Thin Solid Films 2010, 518, 7468–7474. [Google Scholar] [CrossRef]
- Strehle, S.; Reiche, R.; Hoffmann, V.; Acker, J.; Gemming, T.; Wetzig, K. Sulfur incorporation in electroplated Cu (Ag) thin films. Microchim. Acta 2006, 156, 167–172. [Google Scholar] [CrossRef]
- Huang, Q.; Avekians, A.; Ahmed, S.; Parks, C.; Baker-O’Neal, B.; Kitayaporn, S.; Sahin, A.; Sun, Y.; Cheng, T. Impurities in the electroplated sub-50 nm Cu lines: The effects of the plating additives. J. Electrochem. Soc. 2014, 161, D388–D394. [Google Scholar] [CrossRef]
- Kitayaporn, S.; Huang, Q.; Hopstaken, M.; Baker-O’Neal, B. Tin incorporated in copper films during damascene copper electrodeposition. J. Electrochem. Soc. 2015, 162, D74–D81. [Google Scholar] [CrossRef]
- Chen, C.-C.; Yang, C.-H.; Wu, Y.-S.; Ho, C.-E. Depth-dependent self-annealing behavior of electroplated Cu. Surf. Coat. Technol. 2017, 320, 489–496. [Google Scholar] [CrossRef]
- Lee, P.; Wu, Y.; Lin, P.; Chen, C.; Hsieh, W.; Ho, C. High-speed Cu electrodeposition and its solderability. Surf. Coat. Technol. 2017, 320, 559–567. [Google Scholar] [CrossRef]
- Kim, J.; Yu, J.; Kim, S. Effects of sulfide-forming element additions on the kirkendall void formation and drop impact reliability of Cu/Sn-3.5Ag solder joints. Acta Mater. 2009, 57, 5001–5012. [Google Scholar] [CrossRef]
- Kim, S.H.; Yu, J. Secondary IMC formation induced by Kirkendall voiding in Cu/Sn-3.5Ag solder joints. J. Mater. Res. 2010, 25, 1854–1858. [Google Scholar] [CrossRef]




| Baths | Primary Constituents (g/L) | Applications |
|---|---|---|
| Conventional bath | CuSO4·5H2O (180–250) H2SO4 (60–40) | Decorative plating |
| Copper refining | ||
| Electroforming, etc. | ||
| High-through bath | CuSO4·5H2O (60–100) | Printed wiring board plating |
| H2SO4 (250–200) | Through-hole plating | |
| Via filling bath | CuSO4·5H2O (150–230) | Build-up PWB plating |
| H2SO4 (15–50) | ||
| Nanoplating bath | CuSO4·5H2O (60–100) | LSI trench/via filling |
| H2SO4 (200–10) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Chen, C.-M. Impurity Effects in Electroplated-Copper Solder Joints. Metals 2018, 8, 388. https://doi.org/10.3390/met8060388
Lee H, Chen C-M. Impurity Effects in Electroplated-Copper Solder Joints. Metals. 2018; 8(6):388. https://doi.org/10.3390/met8060388
Chicago/Turabian StyleLee, Hsuan, and Chih-Ming Chen. 2018. "Impurity Effects in Electroplated-Copper Solder Joints" Metals 8, no. 6: 388. https://doi.org/10.3390/met8060388
APA StyleLee, H., & Chen, C.-M. (2018). Impurity Effects in Electroplated-Copper Solder Joints. Metals, 8(6), 388. https://doi.org/10.3390/met8060388






