Phase Diagram of near Equiatomic Zr-Pd Alloy
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
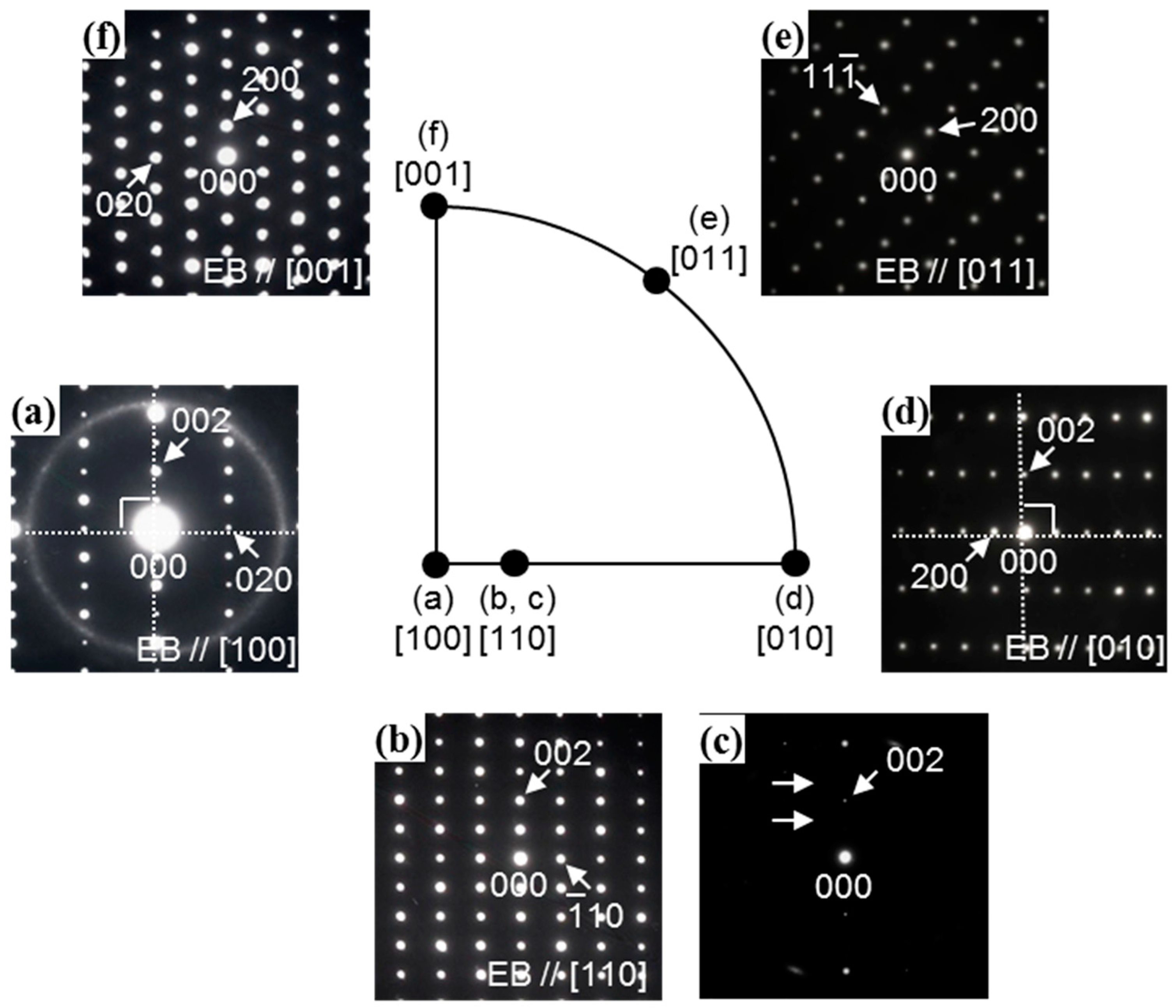
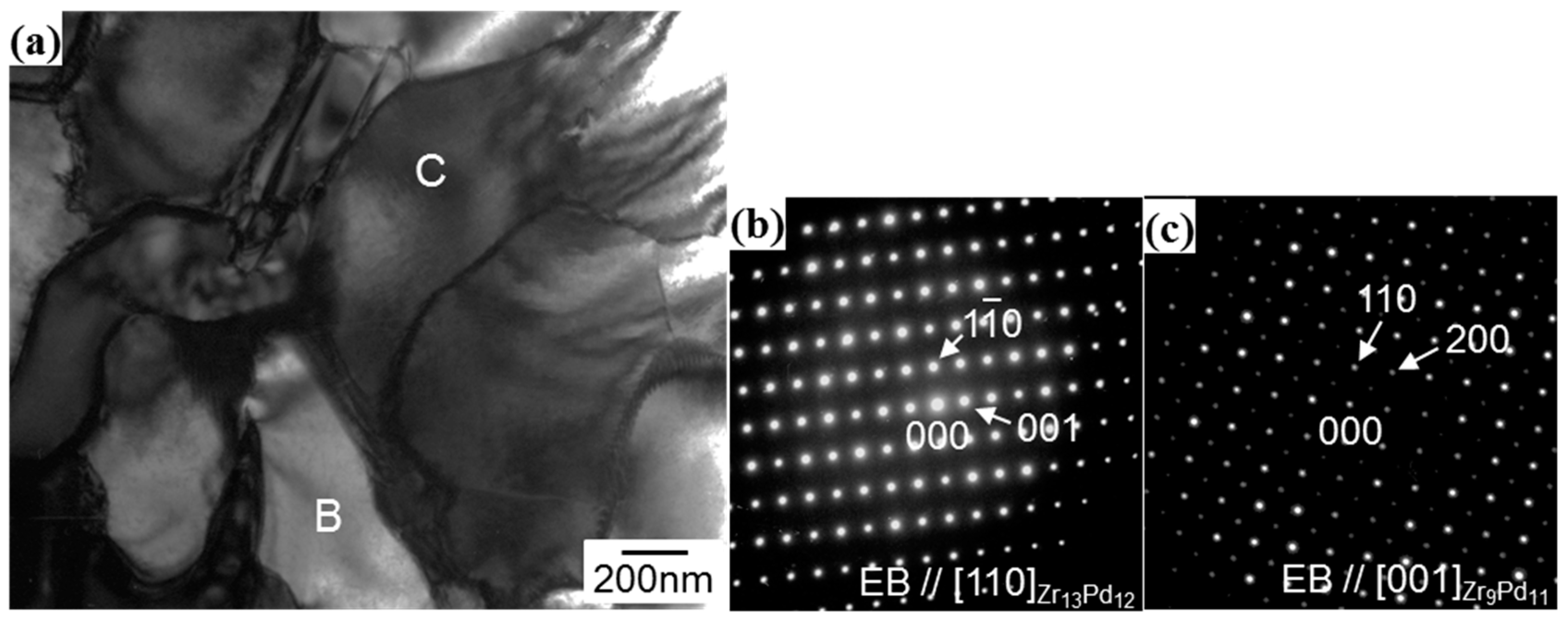
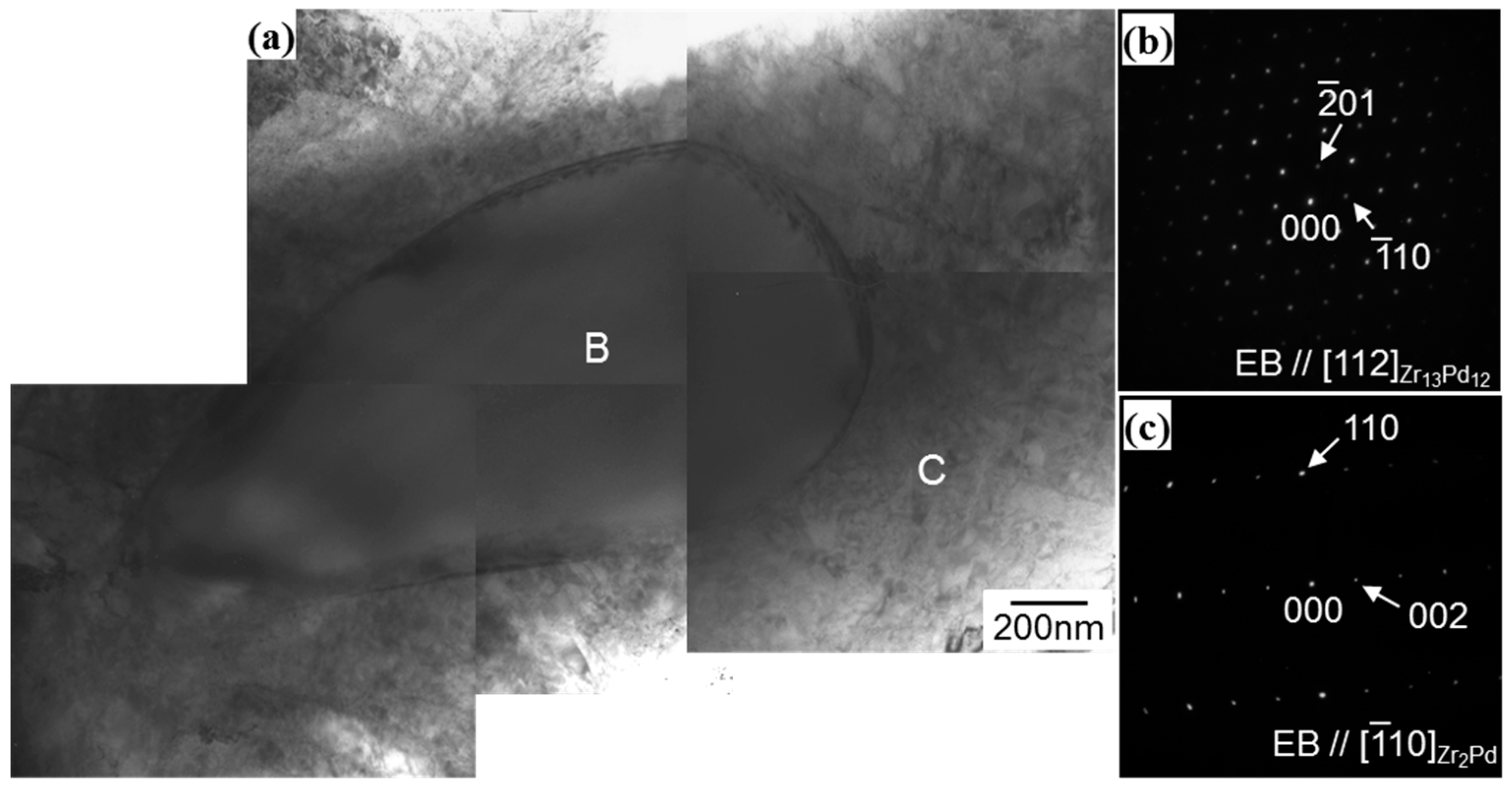
3.1. Microstructural Analysis of Zr13Pd12 Compound by the Electron Diffraction Experiments
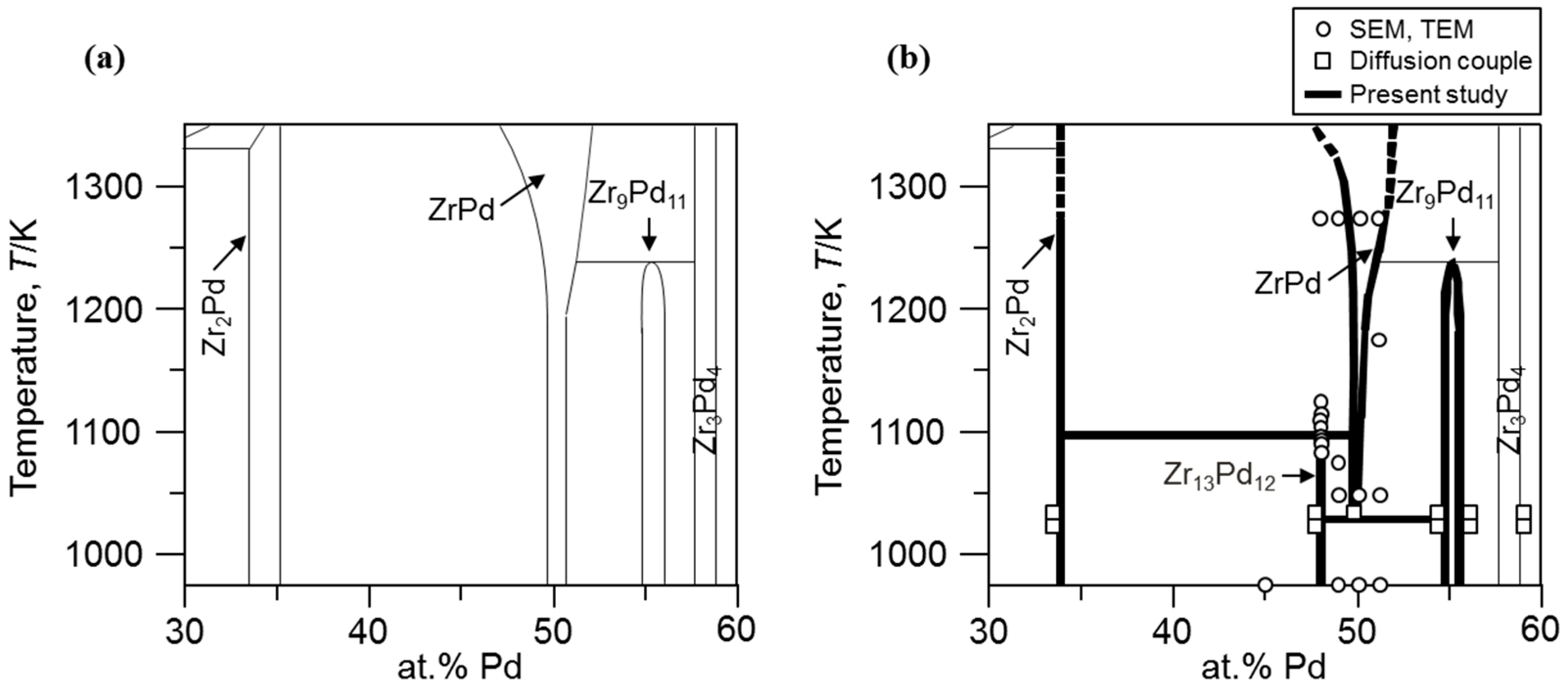
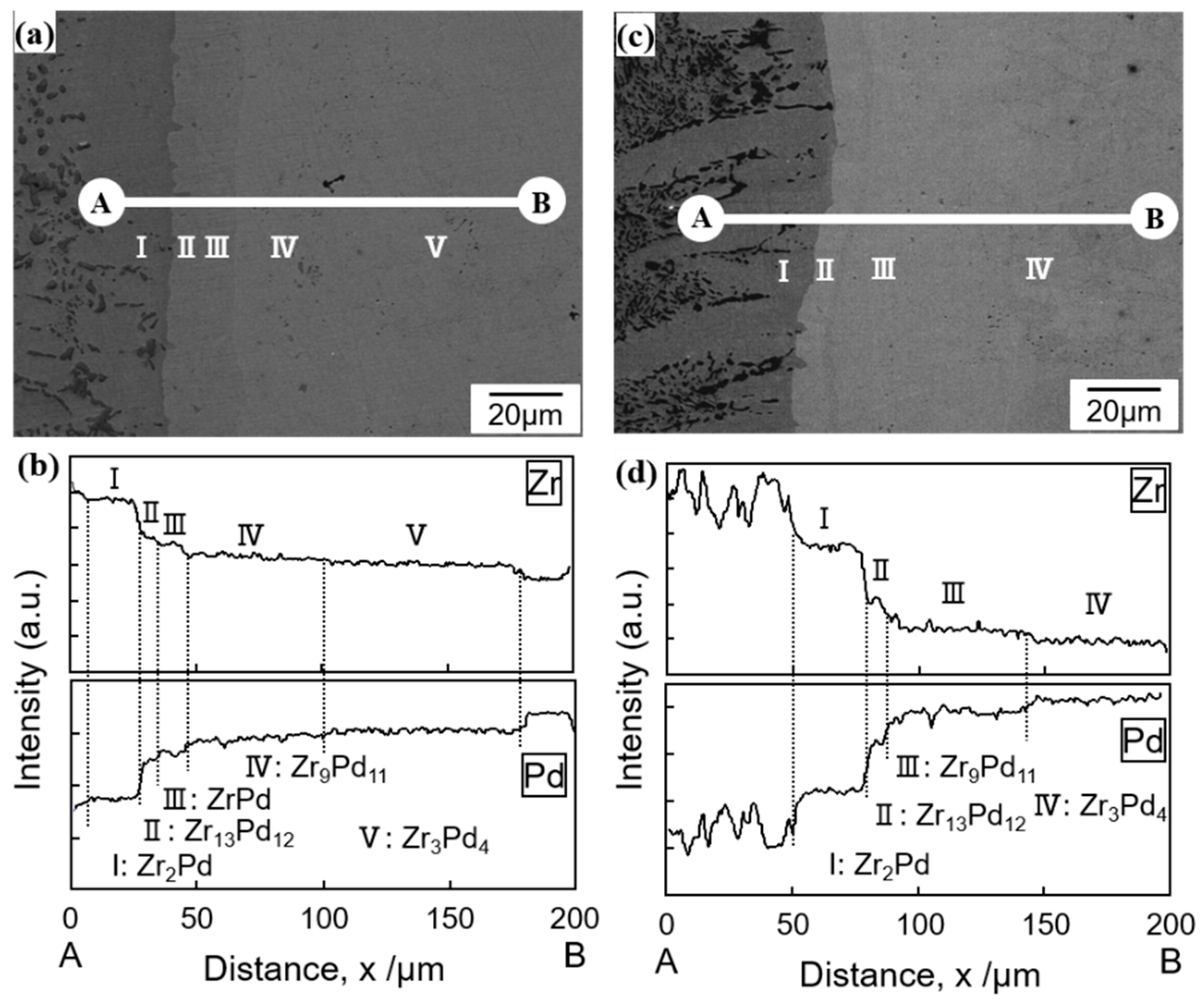
3.2. Determination of Eutectoid and Peritectoid Temperatures
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ma, J.; Karaman, I.; Noebe, R.D. High temperature shape memory alloys. Int. Mater. Rev. 2010, 55, 257–315. [Google Scholar] [CrossRef]
- Humbeeck, J.V. Shape memory alloys with high transformation temperatures. Mater. Res. Bull. 2012, 47, 2966–2968. [Google Scholar] [CrossRef]
- Jani, J.M.; Leary, M.; Subic, A.; Gibson, M.A. A review of shape memory alloy research, applications and opportunities. Mater. Des. 2014, 56, 1078–1113. [Google Scholar] [CrossRef]
- Bendersky, L.A.; Stalick, J.K.; Portier, R.; Waterstrat, R.M. Crystallogrphic structures and phase transformations in ZrPd. J. Alloys Compd. 1996, 236, 19–25. [Google Scholar] [CrossRef]
- Waterstrat, R.M.; Shapiro, A.; Jeremie, A. The palladium-zirconium phase diagram. J. Alloys Compd. 1999, 290, 63–70. [Google Scholar] [CrossRef]
- Cuiping, G.; Zhenmin, D.; Changrong, L. Thermodynamic modeling of the Pd-Zr system. Calphad 2006, 30, 482–488. [Google Scholar]
- Nishiura, T.; Yamamuro, T.; Hashimoto, D.; Morizono, Y.; Nishida, M. Martensitic transformation and phase equilibrium in near equiatomic Zr-Pd alloys. Mater. Sci. Eng. A 2006, 438–440, 852–856. [Google Scholar] [CrossRef]
- Maeland, A.J.; Lukacevic, E.; Rush, J.J.; Santoro, A. Neutron powder diffraction and inelastic scattering study of the structures of Zr2Pd, Zr2PdD1.70 and Zr2PdD1.96. J. Less Common Met. 1987, 129, 77–91. [Google Scholar] [CrossRef]
- Matsuki, H.; Ibuka, H.; Saka, H. TEM observation of interfaces in a solder joint in a semiconductor device. Sci. Technol. Adv. Mater. 2002, 3, 261–270. [Google Scholar] [CrossRef]






© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuda, M.; Nishiura, T.; Yamamuro, T.; Nishida, M. Phase Diagram of near Equiatomic Zr-Pd Alloy. Metals 2018, 8, 366. https://doi.org/10.3390/met8050366
Matsuda M, Nishiura T, Yamamuro T, Nishida M. Phase Diagram of near Equiatomic Zr-Pd Alloy. Metals. 2018; 8(5):366. https://doi.org/10.3390/met8050366
Chicago/Turabian StyleMatsuda, Mitsuhiro, Tomohiro Nishiura, Takateru Yamamuro, and Minoru Nishida. 2018. "Phase Diagram of near Equiatomic Zr-Pd Alloy" Metals 8, no. 5: 366. https://doi.org/10.3390/met8050366
APA StyleMatsuda, M., Nishiura, T., Yamamuro, T., & Nishida, M. (2018). Phase Diagram of near Equiatomic Zr-Pd Alloy. Metals, 8(5), 366. https://doi.org/10.3390/met8050366



