1. Introduction
Zirconium is an important alloying addition for aluminum alloys, mainly because of the thermal stability Al
3Zr phase in the Al matrix [
1]. Al
3Zr precipitates are coarsening resistant up to temperatures greater than 673 K (400 °C) [
1], which is ~200 K higher than commonly used precipitation strengthening additions in commercial alloys (Cu, Si, Mg, etc.). The low solubility of Zr in Al limits the effectiveness of Al
3Zr precipitates as a primary strengthening method [
1,
2], but this does not stop Zr from being widely researched and put to use in several key applications. The desirable thermal stability of Al
3Zr has led to Zr being added in hyperperitectic concentrations to numerous Al alloys to pin grain boundaries for use in superplastic deformation [
1,
2] and to refine grains in cold worked alloys [
3,
4]. It has been demonstrated as a complementary addition to the diluted Al-Sc alloy system for coherent nanoscale precipitation strengthening [
5,
6,
7], and it is used in higher concentrations to form bulk metallic glass [
8,
9,
10].
Throughout history, the majority of Al-Zr research has focused on alloys with Zr concentrations higher than the maximum solubility limit of Zr in the Al matrix phase, with the intent of forming large primary precipitates at the grain boundaries. The notable exception to this is the research focused on Al-Sc-Zr alloys, which relies on the absence of primary precipitates to achieve the maximum potential precipitate strengthening for Al3(Sc, Zr) precipitates. This work focuses on the interaction between Al-Cu and small Al-Zr precipitates, and thus does not require concentrations high enough to form primary Al3Zr precipitates.
The primary goal of these experiments was to assess whether diluted Zr additions to Al-Cu alloys could increase the thermal stability of Al-Cu precipitation strengthening. If the refinement of microstructures associated with nanoscale Al3Zr precipitates in Al-Cu alloys was shown to be partly because of a decrease in the coarsening kinetics of Al-Cu precipitates, it could be possible to increase the strength of more thermally stable Al alloys. To examine this effect, two sets of experiments were run, each comparing Al-Cu and Al-Cu-Zr alloys: (1) an isothermal heat treatment at 473 K (200 °C), quenching and taking hardness measurements periodically throughout the hold; and (2) a multi-step heat treatment that increased in temperature from 443 K (170 °C) to 643 K (370 °C) with five hour holds at each temperature, quenching and taking hardness measurements between each step.
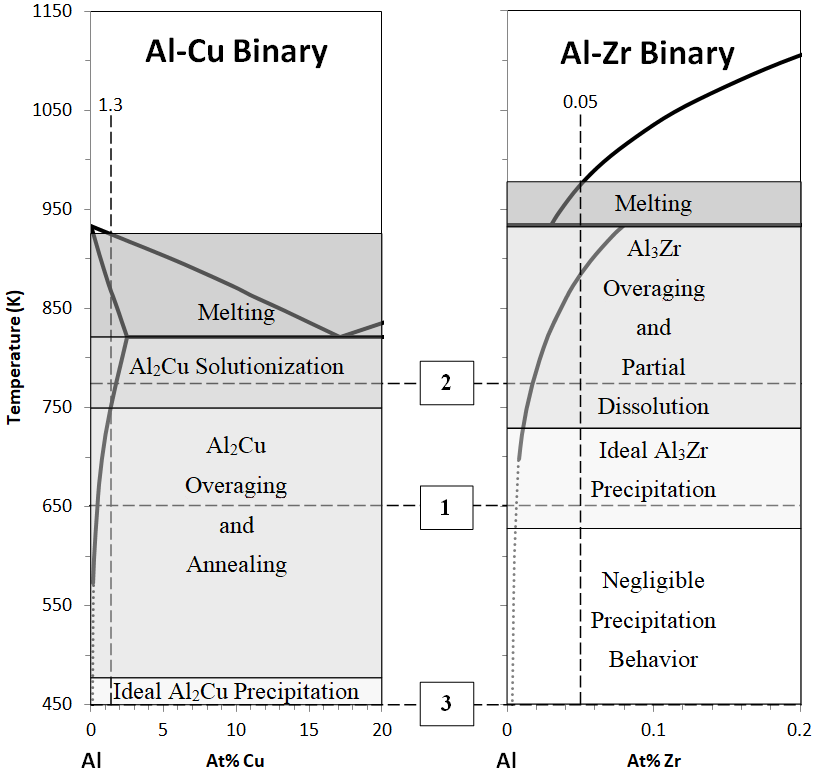
As the goal of these experiments was to observe the effect of Al
3Zr nanoprecipitates on Al-Cu precipitation, two different concentrations of Zr were observed: 0.05 at % Zr (primary precipitate free) and 0.2 at % (primary precipitate forming). It was necessary to nucleate and grow the desired nanoprecipitates with an initial heat treatment procedure before holding at 443 K (170 °C). Relevant phase diagrams were consulted to determine appropriate heat treatments for these alloys, with the end goal of having the majority of Zr solute atoms precipitated out of solution into coherent Al
3Zr nanoprecipitates, while retaining the Cu solute atoms in solution in the matrix. The concentration levels of Zr and Cu in these alloys are low enough that the respective binary phase diagrams can approximate the trends expected in the ternary alloy. These phase diagrams (
Figure 1), along with the listed points that immediately follow this paragraph, describe the heat treatment steps that were decided on and the reasoning behind their selection.
The first required heat treatment step was designed to nucleate Al
3Zr nanoprecipitates. The literature review indicates that precipitation strengthening behavior occurs in Al-Zr alloys at and above ~648 K (375 °C) [
5,
6]. The ideal temperature for this step was taken to be at the low end of the range, 648 K (375 °C), to reduce the final size of the Al
3Zr precipitates. This goal was made more challenging by the requirements of the second step.
As the temperature of the first step was sufficiently high enough to fully precipitate and anneal Al-Cu precipitates, the second required step was designed to re-solutionize the Cu solute atoms, such that preferred microstructural arrangements and strengthening could be achieved at a later step. To reduce overaging of the Al3Zr precipitates, it was desired to keep this step as low in temperature as possible while still achieving solutionization of Cu. With this in mind, 773 K (500 °C) was chosen.
Once the initial heat treatment steps were completed and the microstructure was created, the aging and overaging steps were performed, as discussed in
Section 2. Aging steps were selected to be below 473 K (200 °C), closely approximating the artificial aging treatments of binary Al-Cu alloys.
Looking at the phase diagrams in
Figure 1 [
11,
12], several other notable conclusions about the proposed processing can be drawn. For one, the high liquidus temperature range of the Al-Zr phase diagram makes it necessary to reach much higher temperatures with the melting of Zr bearing alloys in order to fully dissolve Al
3Zr precipitates into the liquid. This effect is even more pronounced when adding Zr from master alloys with higher comparative Zr contents, as the large Zr precipitates sink and can create regions of the melt with disproportionately high liquidus temperatures.
Another notable conclusion is that it will be impossible to solutionize the Zr once these alloys are created. The relevant temperatures for solutionizing Zr are well above those necessary to begin melting in the Al-Cu system. Therefore, proper care must be taken with these alloys, as they are less forgiving of failed heat treatments than current precipitation strengthening alloys.
Finally, it can be noted that at the Al-Cu solutionizing temperature of 773 K (500 °C), Al
3Zr precipitates have been observed to partially dissolve into the solid solution because of an increased solubility of Zr in Al, indicated in the literature by a decrease in electrical conductivity after undergoing heat treatments at elevated temperatures [
5,
6]. This dissolution behavior is not ideal, but it also cannot be reasonably avoided. The Al-Cu solutionizing temperature can in fact be lowered to a temperature that would better preserve the Al
3Zr precipitates, but the only way to achieve this in this ternary system is to decrease the alloy Cu concentration, and in turn the potential for Al-Cu strengthening (see the solvus line for the Al-Cu binary in
Figure 1).
2. Materials and Methods
In preparation for casting the new alloys, master alloys were cut up using a band saw, cleaned thoroughly using an ultrasonic bath with acetone, and weighed out in the proper amounts to make ~600 g charges of the compositions listed in
Table 1. The resulting charges were then cast in a vacuum induction melter (VIM, constructed at Michigan Technological University, Houghton, MI, USA) into a bottom-fed permanent mold to create cylindrical bars approximately 19mm in diameter. After evacuation of the chamber, the charges were held in a liquid state at 1173 K (900 °C) for 10 min under a 99.999% purity argon atmosphere at ~684 torr. After solidification, samples with a thickness of ~1 cm were sectioned from the center of one cylindrical bar from each casting using an abrasive cutoff disc, and mounted in QuickSet epoxy. The mounted samples were then ground and polished to a final finishing step of 0.04 micron colloidal silica.
A hole was also drilled in each casting to create chips that could be dissolved for ICP-OES analysis. For each sample, approximately 0.4 g of these chips was dissolved in HCl and HNO3 using an Anton Paar microwave digestion setup, followed by dilution with water into a final solution volume of 100 mL. ICP-OES was performed with a Perkin Elmer Optima 7000DV to verify the Cu and Zr concentrations for each casting. Typical detection limits for ICP-OES on this machine, according to Perkin Elmer documentation, range between 0.15 and 4.5 μg/L for reported elements. Coincidentally, both Al and Cu have the same reported detection limit of 0.9 μg/L. The detection limit of Zr is not reported, thus it was estimated here to match the highest reported detection limit of 4.5 μg/L. As each sample consisted of 0.4 g of alloy dissolved and diluted into a 100 mL solution, these detection limits correspond to minimum detectable alloy concentrations of 2.25 × 10−4 wt % for Al and Cu and 1.125 × 10−3 wt % for Zr, which are well below the alloy concentrations used in this study.
Vickers hardness testing was performed for each sample using a LECO MHT Series 200 hardness tester (LECO Corporation, St. Joseph, MI, USA). Using the automated XY stage, hardness indents were created with 10 g of force and a 15 s hold time in a large grid across the sample. For the isothermal aging study (
Table 2), indents were performed in a 2 × 5 grid for a total of 10 points, and for the stepped aging study (
Table 2), indents were performed in a 3 × 10 grid for a total of 30 points. The hardness data reported in
Section 3 represent the average value of one grid of hardness indents (10 or 30 points, depending on the study), with error bars indicating the 95% confidence interval within the hardness grid.
After evaluating these indents, the epoxy was broken off of the sample in preparation for heat treatment using a bench-mounted vice, taking care not to deform the relatively soft aluminum sample. Heat treatment was performed on the samples in a Thermolyne box furnace with a Furnatrol 53,600 Controller, according to the schedules depicted in
Table 2. Between each heat treatment step, the samples were quenched in water as they were removed, and epoxy was then re-applied to the samples in such a way that minimal polishing was required to achieve a freshly polished surface. Another round of 30 hardness tests were performed, offset from the previous indent grids to avoid unwanted interactions, and then the sample was again broken out of the epoxy and placed back in the furnace at the next temperature. This process (mount
→ hardness test
→ unmount
→ heat treat
→ repeat) was repeated until all heat treatment steps were complete.
After completion of the stepped aging heat treatment described in
Table 2, transmission electron microscope (TEM) samples were created from each of the specimens. TEM sample preparation was performed as follows: 0.5 mm thick slices were cut from the sample using a Japax Lux3 Wire EDM (Japax, Japan). Each slice was then polished on both sides to a mirror surface, and 3 mm diameter discs were punched out using a specialized hole punch. Using a Gatan Disc Grinder system, these discs were polished further on each side to yield a final thickness of approximately 50 microns, and a final surface polished with 0.04 micron colloidal silica. These discs were then perforated with a Metalthin Twin Jet Electropolishing System (South Bay Technology Inc., San Clemente, CA, USA), using a solution of 29% reagent grade nitric acid and 71% reagent grade methanol. The solution was kept below 243 K (−30 °C) with a Multicool recirculating methanol refrigeration unit. The relevant operating settings for the Metalthin instrument were as follows: 20–30 volts, jet speed of 4, sensitivity of 7–8.
Successfully prepared TEM specimens were analyzed on a JEOL JEM-2010 high resolution transmission electron microscope (JEOL, Tokyo, Japan). All images were captured with a Gatan Orius SC200 high-speed digital camera, and chemical analysis of nanoscale features was performed using an Oxford energy dispersive spectrometer (EDS). TEM analysis of these specimens yielded images of hundreds of precipitates for each heat treated specimen. The length and width of all resolvable precipitates were measured manually using the freely available image processing software ImageJ.
3. Results and Discussion
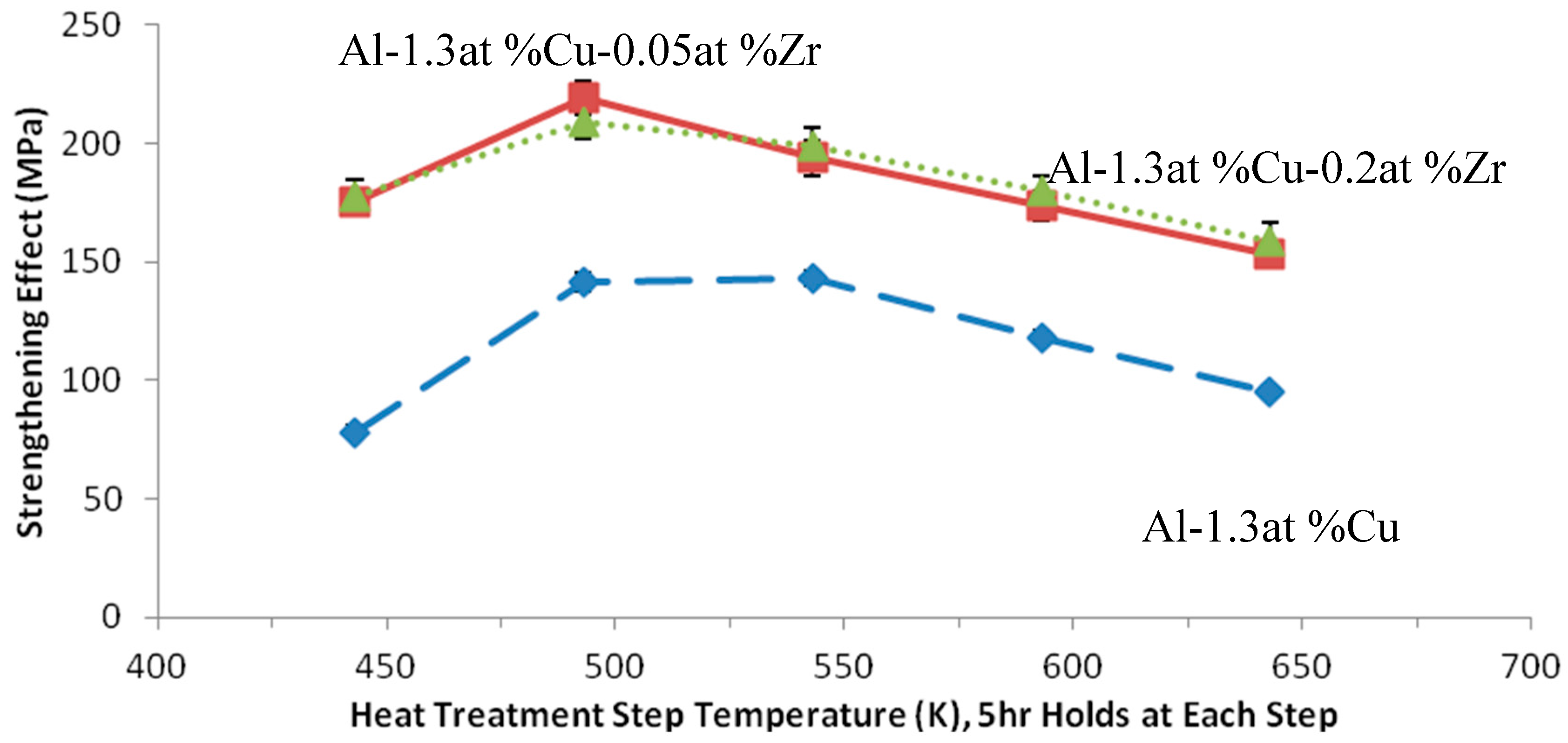
Figure 2 and
Figure 3 describe the strengthening behavior observed for the isothermal and stepped aging experiments, respectively. From these results, it could immediately be seen that all Zr-bearing Al-Cu alloys invariably maintained a higher strength than the binary Al-Cu alloys, by what appears to be a constant strengthening increment for each set of experimental parameters. This strengthening increment appears to be thermally stable at elevated temperatures that would normally coarsen and overage Al-Cu (primarily Al
2Cu) precipitates. This information taken by itself was promising, especially considering that the alloy containing only 0.05 at % Zr was shown to have a similar strengthening effect as the alloy containing 0.2 at % Zr, implying that aged, nanoscale Al
3Zr precipitates are equally as effective as the larger primary precipitates. However, the results did not indicate any noticeable increase in thermal stability of the total precipitation strengthening that would be expected to accompany any improvement in coarsening resistance of the Al-Cu precipitate phases.
Without consulting TEM images of the sample microstructure, the false assumption could be reached that the Zr additions do not significantly affect the Al-Cu precipitation behavior, and that the observed thermally stable strengthening increment is entirely because of strengthening contributions of Al
3Zr nanoprecipitates. However, TEM images of the samples taken after completion of stepped aging (
Table 2) negate this possibility. In these images, Al-Cu precipitates found in the Al-1.3 at % Cu sample have measured lengths ~3× longer than the precipitates found in both of the Al-1.3 at % Cu-(0.05, 0.2) at % Zr samples, suggesting that the addition of Zr does significantly affect the precipitation behavior of Al-Cu precipitates. Representative images are shown in
Figure 4.
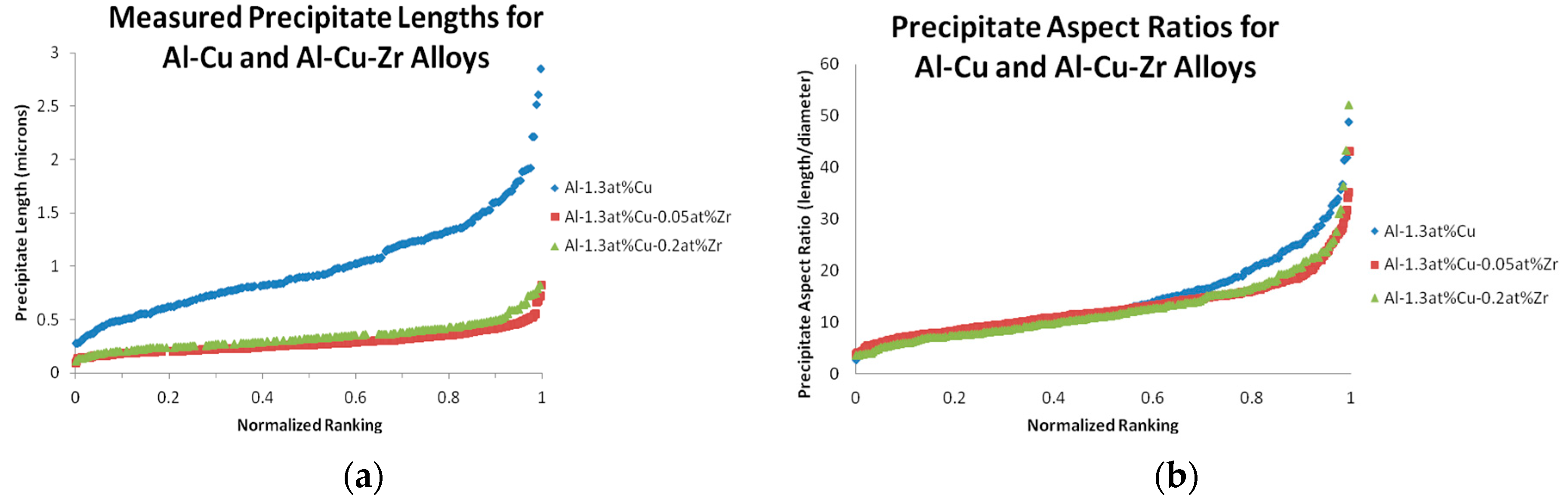
A comparison of observed precipitate lengths and aspect ratios between the three alloys that underwent the stepped aging heat treatment (
Table 2) can be seen in
Figure 5. This comparison indicates that the Al-Cu precipitate aspect ratio distributions are consistent between Al-Cu and Al-Cu-Zr alloys. It also shows that the average Al-Cu precipitate length, while similar in both observed Al-Cu-Zr alloys, is noticeably larger in the binary Al-Cu alloy.
Orowan strengthening contributions of the oblong θ’ precipitates can be estimated using Equation (1) [
13], where
M = 3.06 is the Taylor factor [
14],
G = 25.4 GPa is the shear modulus of the matrix [
15],
b = 0.286 nm is the Burgers vector magnitude of the matrix [
15], and
= 0.572 nm is the inner dislocation cut-off radius [
16]. Image analysis of the TEM images yields average precipitate plate diameter (
) and an estimate of the thickness (
), although the actual thickness can be assumed to be slightly lower than this estimate because of unknown disorientation of the TEM images. The volume fraction (
) can be assumed to be at equilibrium and estimated from the solvus composition for a given alloy.
Using this method, Orowan strengthening contributions of the θ’ precipitates were estimated to be 23.9 and 63.9 MPa for Al-1.3 at % Cu and Al-1.3 at % Cu-0.05 at % Zr alloys, respectively, after the final stepped heat treatment step of 643 K (370 °C). The difference between these two estimates is ~40 MPa, which accounts for a significant amount of the observed difference between these alloys of ~58 MPa.
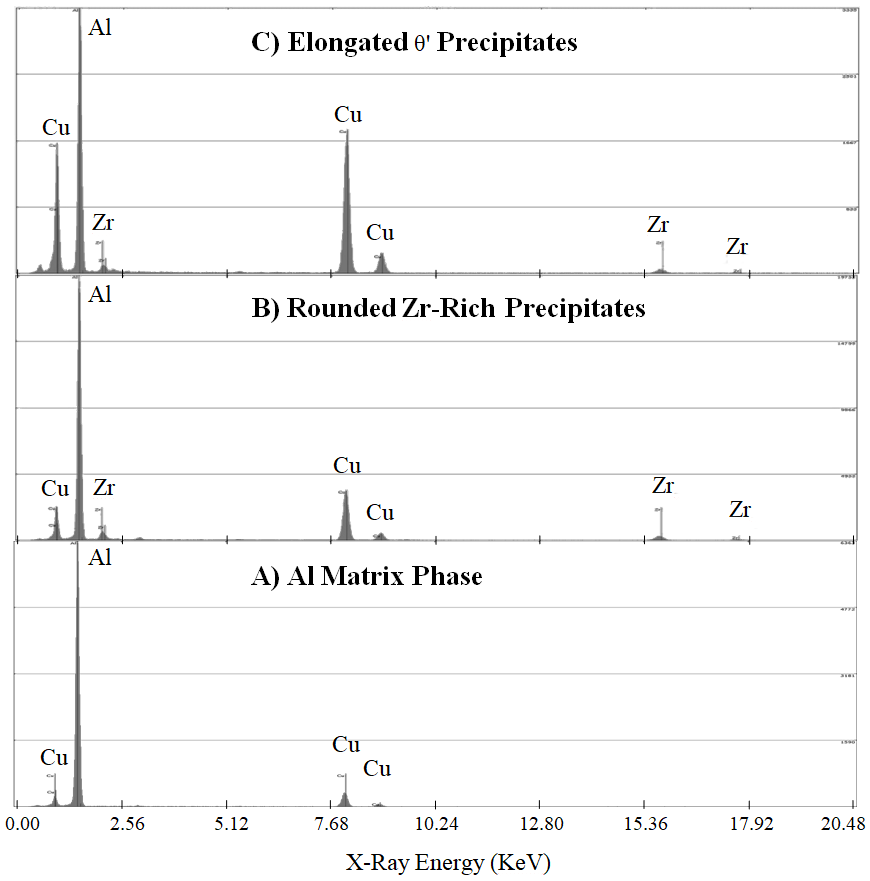
Upon closer inspection of the TEM samples for the Al-Cu-Zr alloys, a fine dispersion of rounded Zr-rich nanoprecipitates becomes apparent among the larger θ’ precipitates, as seen in
Figure 6. When analyzed with EDS, these rounded precipitates contain not only Al and Zr, as would be expected with simple Al
3Zr precipitate phase, but significant quantities of Cu as well, when compared with the matrix (
Figure 7).
The formation of these Zr-rich precipitates could only have occurred during the initial heat treatment step, which was intended to solutionize Cu and simultaneously form the Al
3Zr phase. It is unclear whether this phenomenon is because of an attraction of Cu atoms to the stable Al
3Zr precipitates at these higher temperatures, or to the formation of a stable, ternary Al-Cu-Zr phase. While many Al-Cu-Zr intermetallic phases are known to exist, only the
phase (Al-15.7 at % Cu-17.1 at % Zr) is predicted to potentially be stable with the given alloy composition at 773 K (500 °C) [
8,
17].
One consequence of forming Al-Cu-Zr precipitates, as opposed to the simple Al3Zr phase, is that a small amount of Cu is depleted from the matrix and made unavailable for θ’ precipitates. However, assuming all Zr matrix content is used to form precipitates, the maximum loss in matrix Cu content would still only be ~0.046 at % Cu for the Al-1.3 at % Cu-0.05 at % Zr alloy and ~0.072 at % Cu for the Al-1.3 at % Cu-0.2 at % Zr alloy (assuming a Zr solubility limit of 0.078 at % Zr). This is expected to result in a decrease in the final θ’ precipitate phase fraction of up to ~5% when compared with the Al-1.3 at % Cu binary alloy.
It is also interesting to note that the θ’ and Zr-rich precipitates intercept each other in several instances within
Figure 6. These instances are representative of all such behavior observed in other Al-Cu-Zr TEM images, in that the elongated θ’ precipitates tend to terminate in or on a Zr-rich precipitate when they intercept. This behavior may be indicative of the nucleation of θ’ precipitates on the previously formed Zr-rich precipitates [
17]. Nucleation on Al
3Zr precipitates could account for the Cu-enriched nature of the Zr-rich phase. This nucleation phenomenon contributes to an increased number density of θ’ precipitates, which in turn results in lower average dimensions of θ’ precipitates in the Zr-bearing alloys when compared with the binary alloys.
Comparison of TEM EDS scans for the three phases indicated in
Figure 6 can be found in
Figure 7. The difference in Cu:Zr intensity ratios between the θ’ and
precipitates in
Figure 7 clearly indicates that the two precipitate phases are of different compositional makeup. It should also be noted that the Zr content in the scan for the θ’ precipitate is not universal; some of the θ’ precipitates observed showed no noticeable Zr content, while others still showed Zr quantities between the matrix and Al
3Zr concentrations. This discrepancy could potentially be due to inaccuracies in measuring low concentration elements in relatively thin precipitates, or to the unknowing contamination of signal from small, nearby Zr-rich precipitates.