Review of the Soft Sparking Issues in Plasma Electrolytic Oxidation
Abstract
:1. Introduction
2. Observations and Factors in Soft Sparking
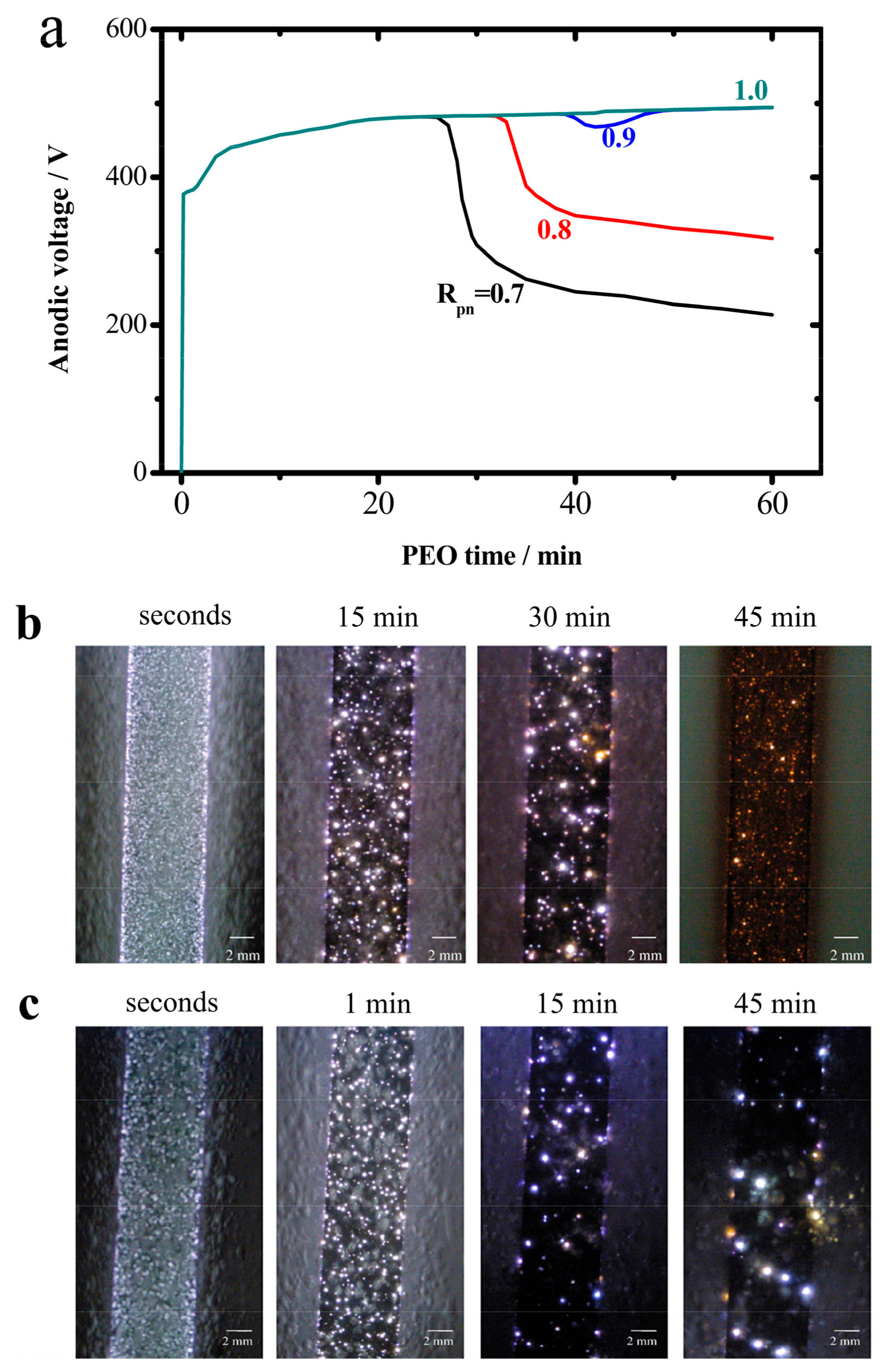
2.1. Transition Phenomena
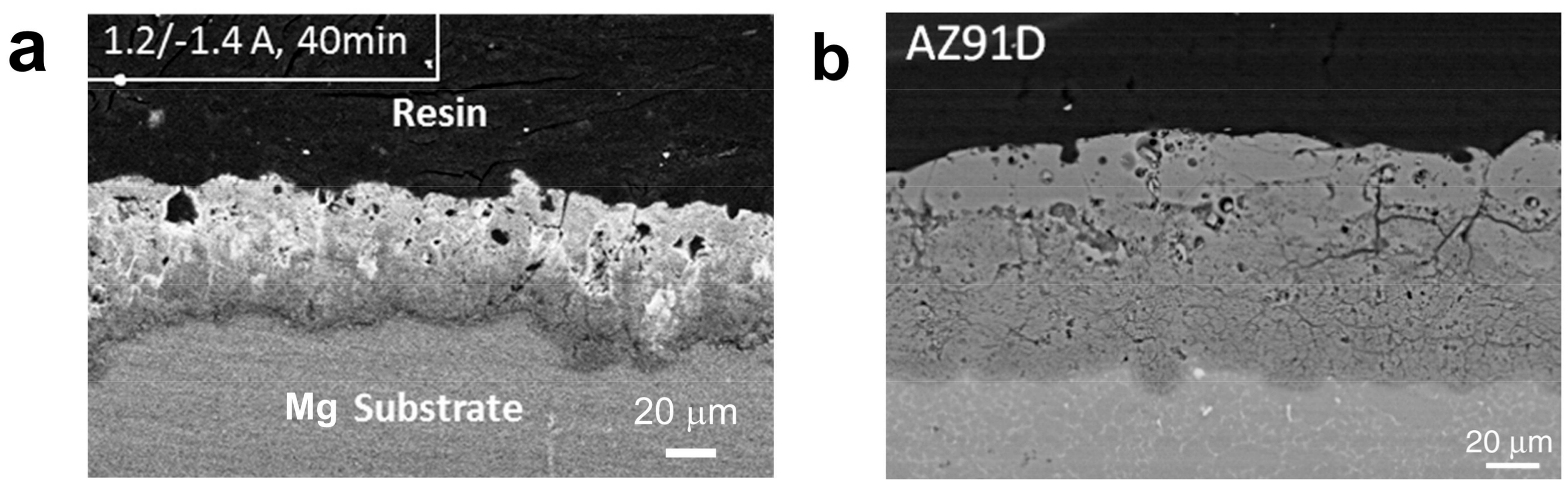
2.2. Microstructure Features
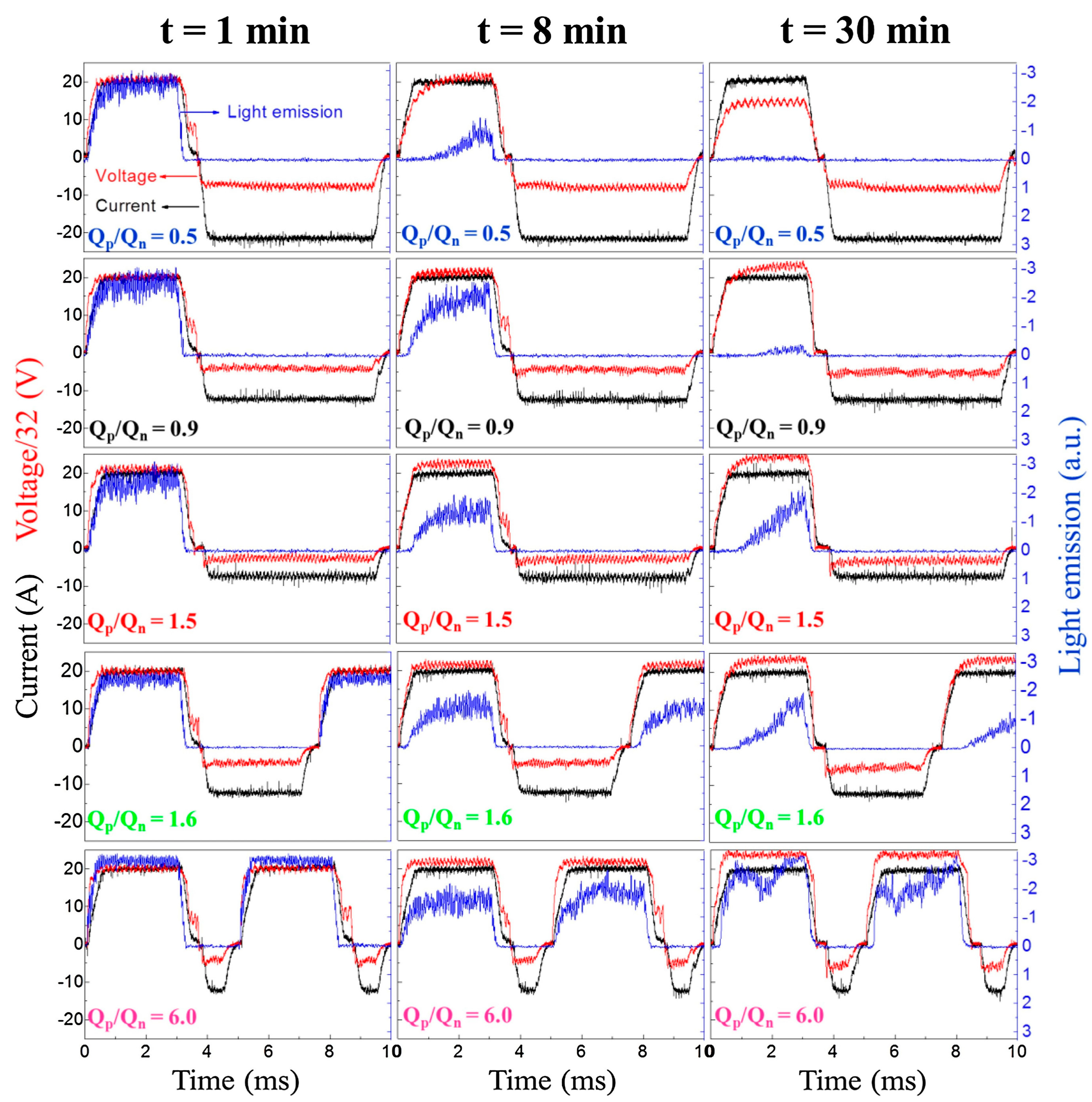
2.3. Plasma Softening
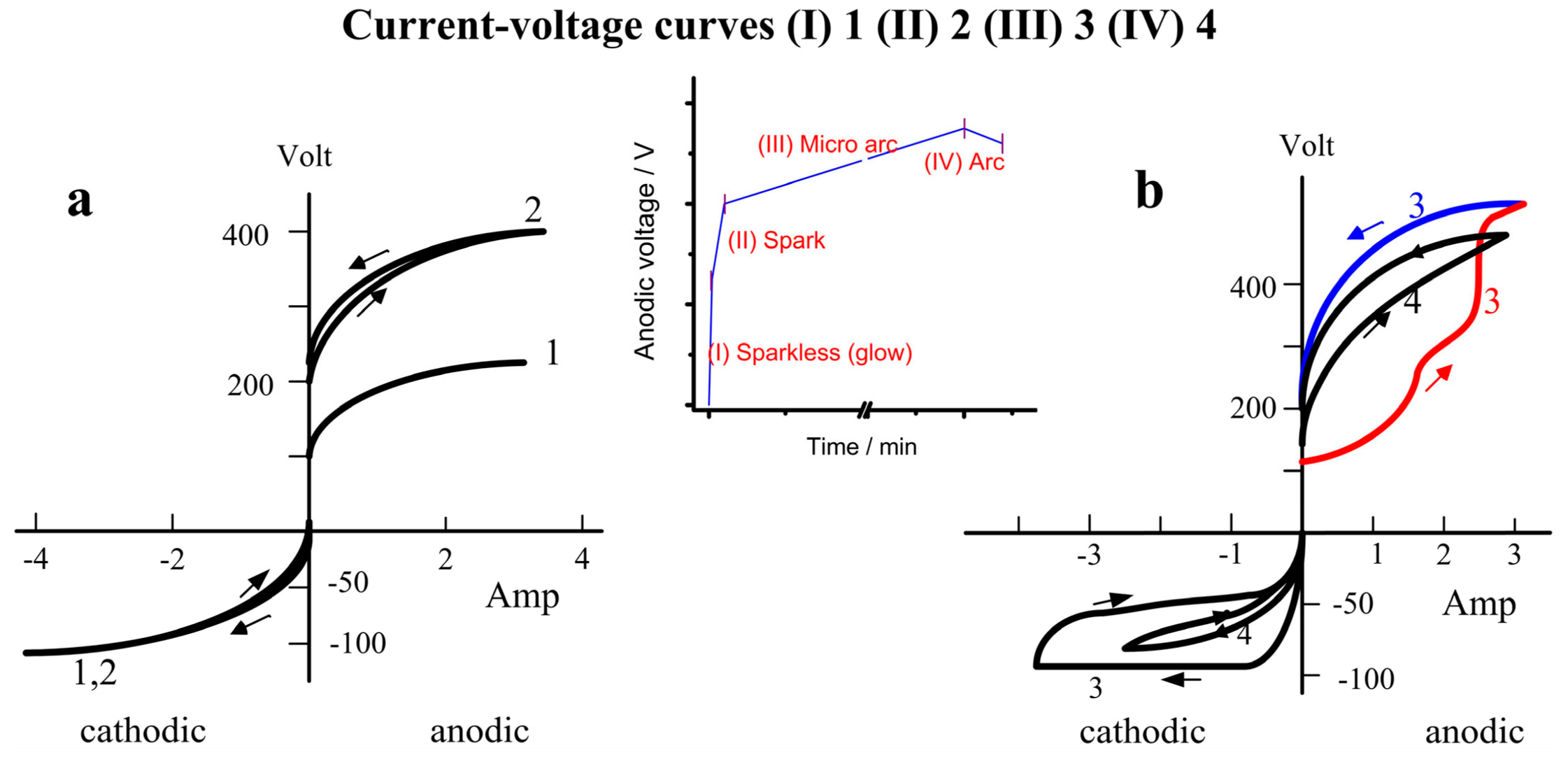
2.4. The Cathodic Current Sets the Tone
- (1)
- Sparkless (glow) stage, CVC-1. The sample glows in this stage, up to the spark voltage ~255 V; its oxide growth is similar to that of anodizing. No hysteresis is observed in CVC. The oxide thin film could be incomplete, or, even if complete, have a large number of defects. This period is very brief.
- (2)
- Spark stage, CVC-2. In this stage, small sparks emerge, migrate, and distribute on the metal surface homogeneously. The anodic voltage rises to ~400 V. The voltage difference between the positive and the negative polarization can be attributed to the Schottky barrier and the gas envelope of the spark. A small hysteresis exists in both anodic and cathodic CVCs.
- (3)
- Micro-arc stage, CVC-3. The sparks increase in size and intensity, and migrate to the metal surface. The anodic voltage rises slowly to ~550–570 V. Hysteresis in the current–voltage plot is most significant during the micro-arc stage. The micro-arc stage could last 45–60 min. If thickness is not the primary goal (<70–80 µm), the operation could end in this stage.
- (4)
- Arc stage, CVC-4. The arc size further increases. A portion of the coating is destroyed. The anodic voltage could drop a little, down to ~500–520 V. A coating of good quality ought to avoid entering the arc stage since those strong electric discharges could damage the oxide coating. Such damage is difficult to repair.
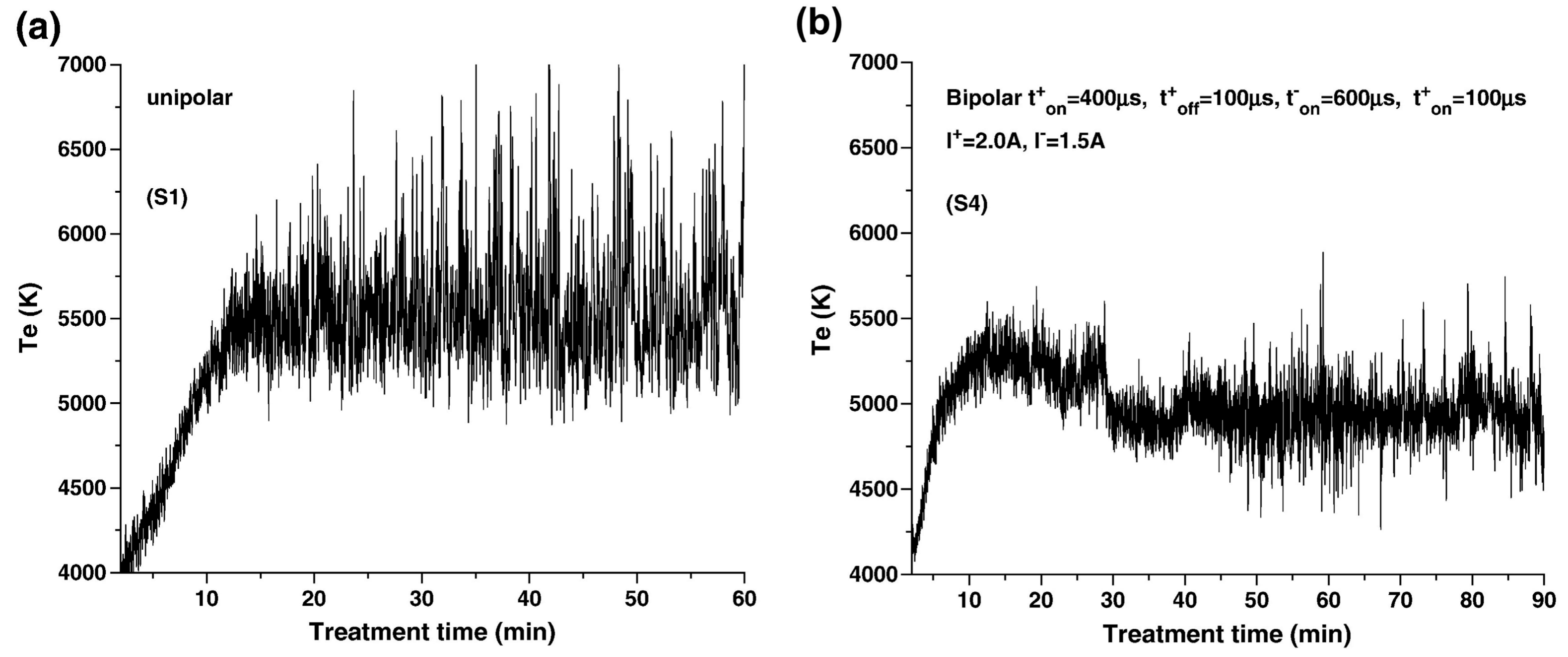
2.5. Plasma State in Electron Temperature
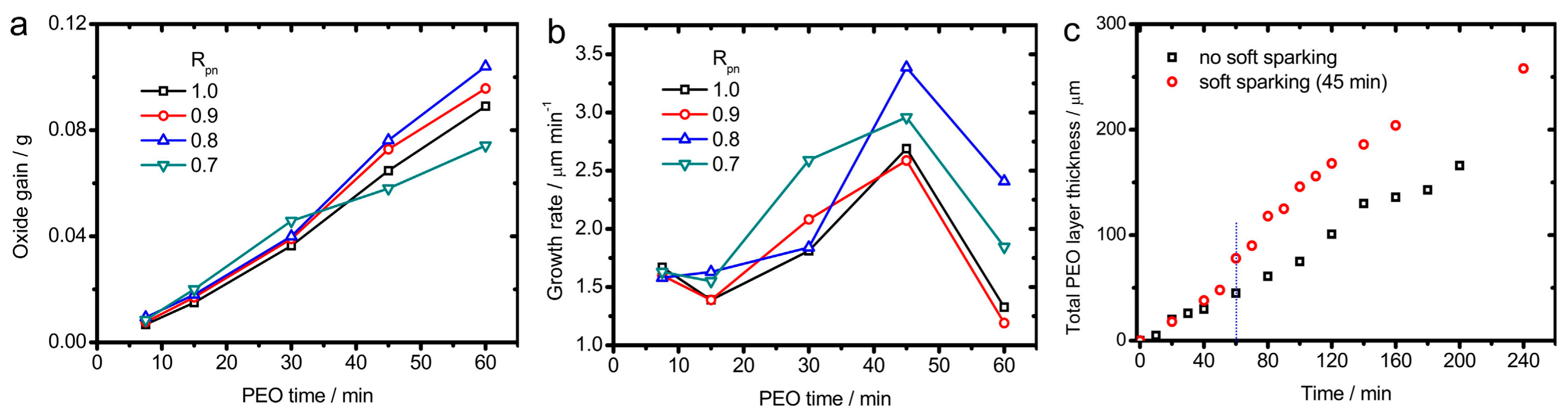
2.6. Amassed Oxide and Energy Consumption
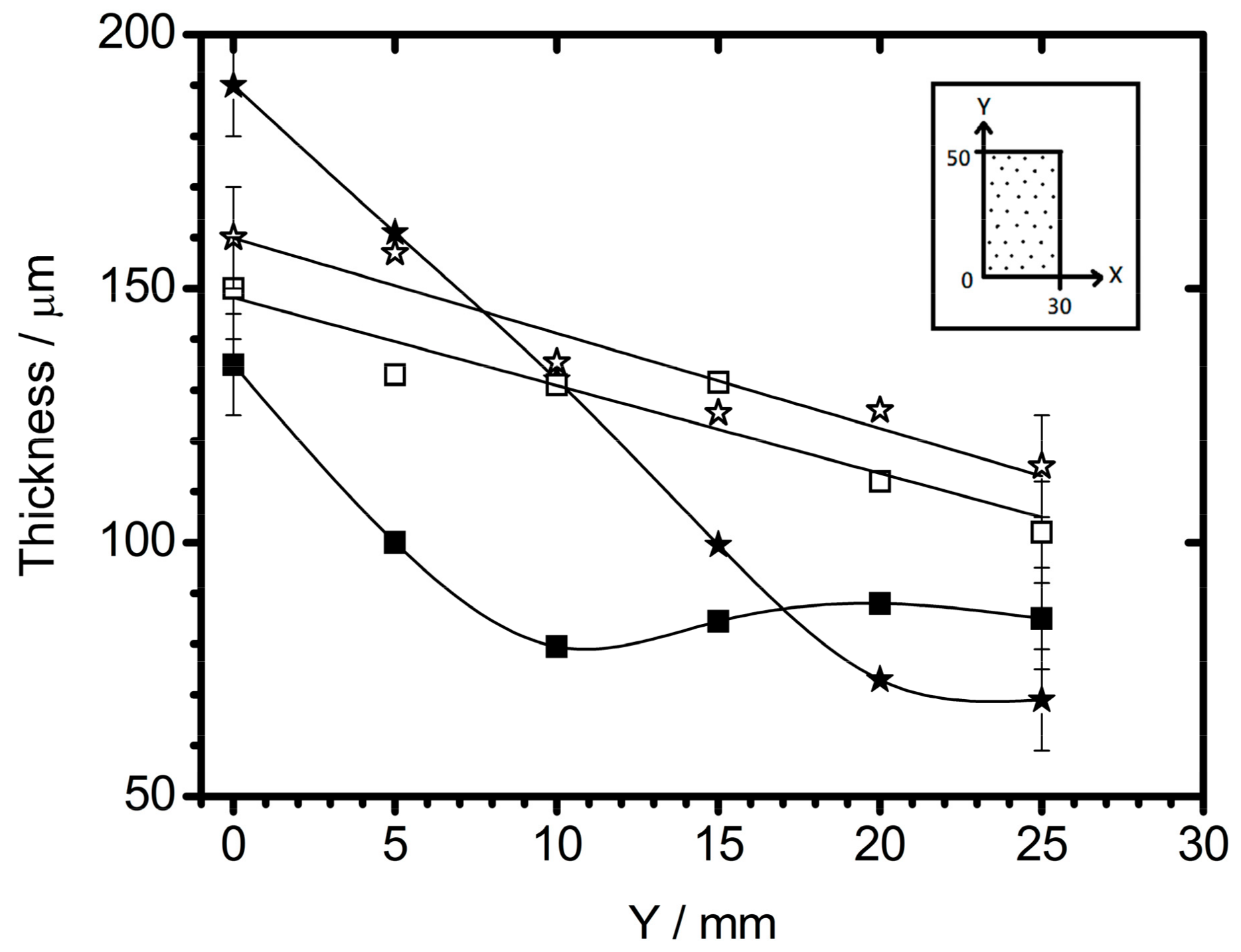
2.7. Coating Growth and Uniformity
3. Mg- and Ti-Based Alloys
3.1. Soft Sparking on Mg-Based Alloys
3.2. Soft Sparking on Ti-Based Alloys
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lohrengel, M.M. Thin anodic oxide layers on aluminum and other valve metals. Mater. Sci. Eng. 1993, R11, 243–294. [Google Scholar] [CrossRef]
- Dyer, C.K. Electrolytic rectification and cathodic charge reversibility of some valve metals. Electrocompon. Sci. Technol. 1974, 1, 121–127. [Google Scholar] [CrossRef]
- Middelhoek, A. The mechanism of electrolytic rectification. J. Electrochem. Soc. 1964, 111, 379–380. [Google Scholar] [CrossRef]
- Blawert, C.; Dietzel, W.; Ghali, E.; Song, G. Anodizing treatments for magnesium alloys and their effect on corrosion resistance in various environments. Adv. Eng. Mater. 2006, 8, 511–533. [Google Scholar] [CrossRef]
- Diggle, J.W.; Downie, T.C.; Goulding, C.W. Anodic oxide films on aluminum. Chem. Rev. 1969, 69, 365–405. [Google Scholar] [CrossRef]
- Paez, M.A.; Bustos, O.; Thompson, G.E.; Skeldon, P.; Shimizu, K.; Wood, G.C. Porous anodic film formation on an Al-3.5 wt % Cu alloy. J. Electrochem. Soc. 2000, 147, 1015–1020. [Google Scholar] [CrossRef]
- Dickey, J.R.; Davidson, J.L.; Tzeng, Y. Improved dielectric properties for anodic aluminum oxide films by soft/hard two-step electrolytic anodization. J. Electrochem. Soc. 1989, 136, 1772–1777. [Google Scholar] [CrossRef]
- Sunderland, R.J. Analysis of chromate conversion coatings on aluminum. Jpn. J. Appl. Phys. 1974, 13, 347–350. [Google Scholar] [CrossRef]
- Waldrop, J.R.; Kendig, M.W. Nucleation of chromate conversion coating on aluminum 2024-T3 investigated by atomic force microscopy. J. Electrochem. Soc. 1998, 145, L11–L13. [Google Scholar] [CrossRef]
- Xia, L.; McCreery, R.L. Chemistry of a chromate conversion coating on aluminum alloy AA 2024-T3 probed by vibrational spectroscopy. J. Electrochem. Soc. 1998, 145, 3083–3089. [Google Scholar] [CrossRef]
- Hagans, P.L.; Haas, C.M. Chromate conversion coatings. In Surface Engineering ASM Handbook; Cotell, C.M., Sprague, J.A., Schmidt, F.A., Jr., Eds.; ASM International: Cleveland, OH, USA, 1994; Volume 5, pp. 405–411. ISBN 978-0-87170-384-2. [Google Scholar]
- ECHA Annex XVII to REACH—Conditions of Restriction; Entry 47–Chromium VI Compounds. Available online: https://echa.europa.eu/documents/10162/1f775bd4-b1b0-4847-937f-d6a37e2c0c98 (accessed on 24 November 2015).
- Matykina, E.; Arrabal, R.; Skeldon, P.; Thompson, G.E. Optimization of the plasma electrolytic oxidation process efficiency on aluminum. Surf. Interface Anal. 2010, 42, 221–226. [Google Scholar] [CrossRef]
- Zhang, X.M.; Tian, X.B.; Yang, S.Q.; Gong, C.Z.; Fu, R.K.Y.; Chu, P.K. Low energy-consumption plasma electrolytic oxidation based on grid cathode. Rev. Sci. Instrum. 2010, 81, 103504. [Google Scholar] [CrossRef] [PubMed]
- Snizhko, L.O.; Yerokhin, A.L.; Pilkington, A.; Gurevina, N.L.; Misnyankin, D.O.; Leyland, A.; Matthews, A. Anodic process in plasma electrolytic oxidation of aluminum in alkaline solutions. Electrochim. Acta 2004, 49, 2085–2095. [Google Scholar] [CrossRef]
- Troughton, S.C.; Nomine, A.; Nomine, A.V.; Henrion, G.; Clyne, T.W. Synchronised electrical monitoring and high speed video of bubble growth associated with individual discharges during plasma electrolytic oxidation. Appl. Surf. Sci. 2015, 359, 405–411. [Google Scholar] [CrossRef]
- Curran, J.A.; Clyne, T.W. Porosity in plasma electrolytic oxidation coatings. Acta Mater. 2006, 54, 1985–1993. [Google Scholar] [CrossRef]
- Dunleavy, C.S.; Golosnoy, I.O.; Curran, J.A.; Clyne, T.W. Characterization of discharge events during plasma electrolytic oxidation. Surf. Coat. Technol. 2009, 203, 3410–3419. [Google Scholar] [CrossRef]
- Dunleavy, C.S.; Curran, J.A.; Clyne, T.W. Self-similar scaling of discharge events through PEO coatings on aluminum. Surf. Coat. Technol. 2011, 206, 1051–1061. [Google Scholar] [CrossRef]
- Mia, T.; Jiang, B.; Liu, Z.; Fan, L. Plasma formation mechanism of microarc oxidation. Electrochim. Acta 2014, 123, 369–377. [Google Scholar] [CrossRef]
- Nomine, A.; Troughton, S.C.; Nomine, A.V.; Henrion, G.; Clyne, T.W. High speed video evidence for localized discharge cascades during plasma electrolytic oxidation. Surf. Coat. Technol. 2015, 269, 125–130. [Google Scholar] [CrossRef]
- Troughton, S.C.; Nomine, A.; Dean, J.; Clyne, T.W. Effect of individual discharge cascades on the microstructure of plasma electrolytic oxidation. Appl. Surf. Sci. 2016, 389, 260–269. [Google Scholar] [CrossRef]
- Dean, J.; Gu, T.; Clyne, T.W. Evaluation of residual stress levels in plasma electrolytic oxidation coatings using a curvature method. Surf. Coat. Technol. 2015, 269, 47–53. [Google Scholar] [CrossRef]
- Khan, R.H.U.; Yerokhin, A.; Matthews, A. Structural characteristics and residual stresses in oxide films produced on Ti by pulsed unipolar plasma electrolytic oxidation. Philos. Mag. 2008, 88, 795–807. [Google Scholar] [CrossRef]
- Gordienko, P.S.; Korkosh, S.V.; Kharchenko, U.V.; Panin, E.S.; Dostovalov, V.A.; Bulanova, S.B. Micro-arc oxidation of metals and alloys in electrolytes with energy control within the breakdown zones. Pac. Sci. Rev. 2008, 10, 307–312. [Google Scholar]
- Jiang, B.L.; Wang, Y.M. Plasma electrolytic oxidation treatment of aluminum and titanium alloys. In Surface Engineering of Light Alloys; Dong, H., Ed.; Woodhead Publishing Limited, CRC Press: New York, NY, USA, 2010; Chapter 5; pp. 110–153. ISBN 978-1-84569-537-8. [Google Scholar]
- Hussein, R.O.; Northwood, D.O. Production of anti-corrosion coatings on light alloys (Al, Mg, Ti) by plasma electrolytic oxidation (PEO). In Developments in Corrosion Protection; Aliofkhazraei, M., Ed.; InTech: London, UK, 2014; Chapter 11; pp. 201–239. ISBN 978-953-51-1223-5. [Google Scholar]
- Malyshev, V.N. Modification of friction knots work surfaces on the basis of micro arc oxidation method. Int. J. Sci. Res. Sci. Eng. Technol. 2016, 2, 464–480. [Google Scholar]
- Narayanan, T.S.N.S.; Park, I.S.; Lee, M.H. Strategies to improve the corrosion resistance of micro arc oxidation coated magnesium alloys for degradable implants: Prospects and challenges. Prog. Mater. Sci. 2014, 60, 1–71. [Google Scholar] [CrossRef]
- Lu, X.; Mohedano, M.; Blawert, C.; Matykina, E.; Arrabal, R.; Kainer, K.U.; Zheludkevich, M.L. Plasma electrolytic oxidation coatings with particle additions—A review. Surf. Coat. Technol. 2016, 307, 1165–1182. [Google Scholar] [CrossRef]
- Parfenov, E.V.; Yerokhin, A.; Nevyantseva, R.R.; Gorbatkov, M.V.; Liang, C.-J.; Matthews, A. Towards smart electrolytic plasma technologies. Surf. Coat. Technol. 2015, 269, 2–22. [Google Scholar] [CrossRef]
- Gupta, P.; Tenhundfeld, G.; Daigle, E.O.; Ryabkov, D. Electrolytic plasma technology: Science and engineering—An overview. Surf. Coat. Technol. 2007, 201, 8746–8760. [Google Scholar] [CrossRef]
- Yerokhin, A.L.; Nie, X.; Leyland, A.; Matthews, A.; Dowey, S.J. Plasma electrolysis for surface engineering. Surf. Coat. Technol. 1999, 122, 73–93. [Google Scholar] [CrossRef]
- Schultze, J.W.; Lohrengel, M.M. Stability, reactivity and breakdown of passive films. Problems of recent and future research. Electrochim. Acta 2000, 45, 2499–2513. [Google Scholar] [CrossRef]
- Jaspard-Mecuson, F.; Czerwiec, T.; Henrion, G.; Belmonte, T.; Dujardin, L.; Viola, A.; Beauvir, J. Tailored aluminum oxide layers by bipolar current adjustment in the plasma electrolytic oxidation process. Surf. Coat. Technol. 2007, 201, 8677–8682. [Google Scholar] [CrossRef]
- Martin, J.; Melhem, A.; Shchedrina, I.; Duchanoy, T.; Nominé, A.; Henrion, G.; Czerwiec, T.; Belmonte, T. Effects of electrical parameters on plasma electrolytic oxidation of aluminum. Surf. Coat. Technol. 2013, 221, 70–76. [Google Scholar] [CrossRef]
- Xin, S.; Song, L.; Zhao, R.; Hu, X. Influence of cathodic current on composition, structure and properties of Al2O3 coatings on aluminum alloy prepared by micro-arc oxidation process. Thin Solid Films 2006, 515, 326–332. [Google Scholar] [CrossRef]
- Sah, S.P.; Tsuji, E.; Aoki, Y.; Habazaki, H. Cathodic pulse breakdown of anodic films on aluminum in alkaline silicate electrolyte. Corros. Sci. 2012, 55, 90–96. [Google Scholar] [CrossRef]
- Aliasghari, S.; Skeldon, P.; Thompson, G.E. Plasma electrolytic oxidation of titanium in a phosphate/silicate electrolyte and tribological performance of the coatings. Appl. Surf. Sci. 2014, 316, 463–476. [Google Scholar] [CrossRef]
- Gebarowski, W.; Pietrzyk, S. Influence of the cathodic pulse on the formation and morphology of oxide coatings on aluminum produced by plasma electrolytic oxidation. Arch. Metall. Mater. 2013, 58, 241–245. [Google Scholar] [CrossRef]
- Gebarowski, W.; Pietrzyk, S. Growth characteristics of the oxide layer on aluminum in the process of plasma electrolytic oxidation. Arch. Metall. Mater. 2014, 59, 407–411. [Google Scholar] [CrossRef]
- Samir, H.A.; Qian, H.C. Effects of cathodic component of current on porosity and hardness characteristics of micro plasma oxidation coatings on aluminum alloy. Trans. Nonferrous Met. Soc. China 2005, 15, 113–118. [Google Scholar]
- Xue, W.; Deng, Z.; Chen, R.; Zhang, T. Growth regularity of ceramic coatings formed by micro arc oxidation on Al-Cu-Mg alloy. Thin Solid Films 2000, 372, 114–117. [Google Scholar] [CrossRef]
- Xue, W.; Deng, Z.; Lai, Y.; Chen, R. Analysis of phase distribution for ceramic coatings formed by microarc oxidation on aluminum alloy. J. Am. Ceram. Soc. 1998, 81, 1365–1368. [Google Scholar] [CrossRef]
- Malyshev, V.N.; Markov, G.A.; Fedorov, V.A.; Petrosyants, A.A.; Terleeva, G.P. Features of the structure and properties of coatings applied by the method of micro arc oxidation. Chem. Pet. Eng. 1984, 20, 41–43. [Google Scholar] [CrossRef]
- Timoshenko, A.V.; Magurova, Y.V. Application of oxide coatings to metals in electrolytic solutions by microplasma methods. Rev. Metal. (Madrid) 2000, 36, 323–330. [Google Scholar] [CrossRef]
- Rakoch, A.G.; Bardin, I.V.; Kovalev, V.L.; Avanesyan, T.G. Microarc oxidation of light constructional alloys: Part 1. Main notions on the microarc oxidation of light constructional alloys. Russ. J. Nonferrous Metal. 2013, 54, 341–344. [Google Scholar] [CrossRef]
- Rakoch, A.G.; Bardin, I.V.; Kovalev, V.L.; Avanesyan, T.G.; Seferyan, A.G. Microarc oxidation of light constructional alloys: Part 2. Influence of the current waveform on the growth kinetics of microarc coatings on the surface of light construction alloys in alkali (pH ≤ 12.5) electrolytes. Russ. J. Nonferrous Metal. 2013, 54, 345–348. [Google Scholar] [CrossRef]
- Duan, H.; Li, Y.; Xie, Y.; Chen, S. Transient voltage-current characteristics: New insights into PEO process of aluminum alloy. Int. J. Electrochem. Sci. 2012, 7, 7619–7630. [Google Scholar]
- Martin, J.; Nomine, A.; Brochard, F.; Briancon, J.-L.; Noel, C.; Belmonte, T.; Czerwiec, T.; Henrion, G. Delays in micro-discharges appearance during PEO of Al: Evidence of a mechanism of charge accumulation at the oxide-electrolyte interface. Appl. Surf. Sci. 2017, 410, 29–41. [Google Scholar] [CrossRef]
- Rogov, A.B.; Shayapov, V.R. The role of cathodic current in PEO of aluminum: Influence of cationic electrolyte composition on the transient current-voltage curves and the discharges optical emission spectra. Appl. Surf. Sci. 2017, 394, 323–332. [Google Scholar] [CrossRef]
- Rogov, A.B.; Yerokhin, A.; Matthews, A. The role of cathodic current in PEO of aluminum: Phenomenological concepts of the soft sparking mode. Langmuir 2017, 33, 11059–11069. [Google Scholar] [CrossRef] [PubMed]
- Jovovic, J.; Stojadinovic, S.; Sisovic, N.M.; Konjevic, N. Spectroscopic study of plasma during electrolytic oxidation of magnesium and aluminum alloys. J. Quant. Spectrosc. Radiat. Trans. 2012, 113, 1928–1937. [Google Scholar] [CrossRef]
- Mecuson, F.; Czerwiec, T.; Belmonte, T.; Dujardin, L.; Viola, A.; Henrion, G. Diagnostics of an electrolytic micro arc process for aluminum alloy oxidation. Surf. Coat. Technol. 2005, 200, 804–808. [Google Scholar] [CrossRef]
- Rogov, A.B.; Shayapov, V.R. Correlations between the optical emission spectra and microstructure of microplasma coatings on aluminum 2024 alloy. Appl. Surf. Sci. 2012, 258, 4871–4876. [Google Scholar] [CrossRef]
- Sarvan, M.; Radic-Peric, J.; Kasalica, B.; Belca, I.; Stojadinovic, S.; Peric, M. Investigation of long-duration plasma electrolytic oxidation of aluminum by means of optical spectroscopy. Surf. Coat. Technol. 2014, 254, 270–276. [Google Scholar] [CrossRef]
- Hussein, R.O.; Nie, X.; Northwood, D.O.; Yerokhin, A.L.; Matthews, A. Spectroscopic study of electrolytic plasma and discharge behavior during the PEO process. J. Phys. D Appl. Phys. 2010, 43, 105203. [Google Scholar] [CrossRef]
- Liu, R.; Wu, J.; Xue, W.; Qu, Y.; Yang, C.; Wang, B.; Wu, X. Discharge behaviors during plasma electrolytic oxidation on aluminum alloy. Mater. Chem. Phys. 2014, 148, 284–292. [Google Scholar] [CrossRef]
- Yang, X.; Chen, L.; Qu, Y.; Liu, R.; Wei, K.; Xue, W. Optical emission spectroscopy of plasma electrolytic oxidation process on 7075 aluminum alloy. Surf. Coat. Technol. 2017, 324, 18–25. [Google Scholar] [CrossRef]
- Hussein, R.O.; Nie, X.; Northwood, D.O. Influence of process parameters on electrolytic plasma discharging behavior aluminum oxide coating microstructure. Surf. Coat. Technol. 2010, 205, 1659–1667. [Google Scholar] [CrossRef]
- Gao, Y.; Yerokhin, A.; Parfenov, E.; Matthews, A. Application of voltage pulse transient analysis during plasma electrolytic oxidation for assessment of characteristics and corrosion behavior of Ca- and P-containing coatings on magnesium. Electrochim. Acta 2014, 149, 218–230. [Google Scholar] [CrossRef]
- Fatkullin, A.R.; Parfenov, E.V.; Yerokhin, A.; Lazarev, D.M.; Matthews, A. Effect of positive and negative pulse voltages on surface properties and equivalent circuit of the plasma electrolytic oxidation process. Surf. Coat. Technol. 2015, 284, 427–437. [Google Scholar] [CrossRef]
- Fatkullin, A.R.; Parfenov, E.V.; Yerokhin, A. Equivalent circuit modelling for pulsed bipolar plasma electrolytic oxidation process. Int. J. Inform. Electron. Eng. 2015, 5, 63–67. [Google Scholar] [CrossRef]
- Boinet, M.; Verdier, S.; Maximovitch, S.; Dalard, F. Plasma electrolytic oxidation of AM60 magnesium alloy: Monitoring by acoustic emission technique. Surf. Coat. Technol. 2005, 199, 141–149. [Google Scholar] [CrossRef]
- Caire, J.-P.; Dalard, F.; Minko, W.S.; Boinet, M. Modeling of the plasma electrolytic oxidizing of aluminum and magnesium alloys. ECS Trans. 2007, 2, 1–21. [Google Scholar] [CrossRef]
- Wang, L.; Chen, L.; Yan, Z.; Fu, W. Optical emission spectroscopy studies of discharge mechanism and plasma characteristics during plasma electrolytic oxidation of magnesium in different electrolytes. Surf. Coat. Technol. 2010, 205, 1651–1658. [Google Scholar] [CrossRef]
- Kasalica, B.; Radic-Peric, J.; Peric, M.; Petkovic-Benazzouz, M.; Belca, I.; Sarvan, M. The mechanism of evolution of microdischarges at the beginning of the PEO process on aluminum. Surf. Coat. Technol. 2016, 298, 24–32. [Google Scholar] [CrossRef]
- Yao, Z.; Xia, Q.; Wei, H.; Li, D.; Sun, Q.; Jiang, Z. Study on coating growth characteristics during the electrolytic oxidation of a magnesium-lithium alloy by optical emission spectroscopy analysis. RSC Adv. 2015, 5, 68806–68814. [Google Scholar] [CrossRef]
- Yao, Z.; Xia, Q.; Ju, P.; Wang, J.; Su, P.; Li, D.; Jiang, Z. Investigation of absorptance and emissivity of thermal control coatings on Mg-Li alloys and OES analysis during PEO process. Sci. Rep. 2016, 6, 29563. [Google Scholar] [CrossRef] [PubMed]
- Lamouri, S.; Hamidouche, M.; Bouaouadja, N.; Belhouchet, H.; Garnier, V.; Fantozzi, G.; Trelkat, J.F. Control of the γ-alumina to α-alumina phase transformation for an optimized alumina densification. Boletín De La Sociedad Española De Cerámica Y Vidrio 2017, 56, 47–54. [Google Scholar] [CrossRef]
- Yalamac, E.; Trapani, A.; Alkurt, S. Sintering and microstructural investigation of gamma-alpha alumina powders. Eng. Sci. Technol. 2014, 17, 2–7. [Google Scholar] [CrossRef]
- Boumaza, A.; Favaro, L.; Ledion, J.; Sattonnay, G.; Brubach, J.B.; Berthet, P.; Huntz, A.M.; Roy, P.; Tetot, R. Transition alumina phases induced by heat treatment of boehmite: An X-ray diffraction and infrared spectroscopy study. J. Solid State Chem. 2009, 192, 1171–1176. [Google Scholar] [CrossRef]
- Tsai, D.S.; Hsieh, C.C. Controlled gelation and sintering of monolithic gels prepared from γ-alumina fume powder. J. Am. Ceram. Soc. 1991, 74, 830–836. [Google Scholar] [CrossRef]
- Wang, J.H.; Du, M.H.; Han, F.Z.; Yang, J. Effects of the ratio of anodic and cathodic currents on the characteristics of micro-arc oxidation ceramic coatings on Al alloys. Appl. Surf. Sci. 2014, 292, 658–664. [Google Scholar] [CrossRef]
- Yerokhin, A.L.; Snizhko, L.O.; Gurevina, N.L.; Leyland, A.; Pilkington, A.; Matthews, A. Discharge characterization in plasma electrolytic oxidation of aluminum. J. Phys. D Appl. Phys. 2003, 36, 2110–2120. [Google Scholar] [CrossRef]
- Xia, L.; Han, J.; Domblesky, J.P.; Yang, Z.; Li, W. Study of scanning micro arc oxidation and coating development. J. Mater. Eng. Perform. 2017, 26, 5323–5332. [Google Scholar] [CrossRef]
- Xia, L.; Han, J.; Domblesky, J.P.; Yang, Z.; Li, W. Investigation of the scanning microarc oxidation. Adv. Mater. Sci. Eng. 2017, 2017, 2416821. [Google Scholar] [CrossRef]
- Matykina, E.; Arrabal, R.; Skeldon, P.; Thompson, G.E.; Belenguer, P. AC PEO of aluminium with porous alumina precursor films. Surf. Coat. Technol. 2010, 205, 1668–1678. [Google Scholar] [CrossRef]
- Matykina, E.; Arrabal, R.; Mohamed, A.; Skeldon, P.; Thompson, G.E. Plasma electrolytic oxidation of pre-anodized aluminum. Corros. Sci. 2009, 51, 2897–2905. [Google Scholar] [CrossRef]
- Matykina, E.; Arrabal, R.; Pardo, A.; Mohedano, M.; Mingo, B.; Rodriguez, I.; Gonzalez, J. Energy-efficient PEO process of aluminum alloys. Mater. Lett. 2014, 127, 13–16. [Google Scholar] [CrossRef]
- Matykina, E.; Arrabal, R.; Mohedano, M.; Mingo, B.; Gonzalez, J.; Pardo, A.; Merino, M.C. Recent advances in energy efficient PEO processing of aluminum alloys. Trans. Nonferrous Met. Soc. China 2017, 27, 1439–1454. [Google Scholar] [CrossRef]
- Mohedano, M.; Matykina, E.; Arrabal, R.; Mingo, B.; Zheludkevich, M.L. PEO of rheocast A356 Al alloy: Energy efficiency and corrosion properties. Surf. Interface Anal. 2016, 48, 953–959. [Google Scholar] [CrossRef]
- Melhem, A.; Henrion, G.; Czerwiec, T.; Briançon, J.L.; Duchanoy, T.; Brochard, F.; Belmonte, T. Changes induced by process parameters in oxide layers grown by the PEO process on Al alloys. Surf. Coat. Technol. 2011, 205, S133–S136. [Google Scholar] [CrossRef]
- Darband, G.B.; Aliofkhazraei, M.; Hamghalam, P.; Valizade, N. Plasma electrolytic oxidation of magnesium and its alloys: Mechanism, properties and applications. J. Magnes. Alloys 2017, 5, 74–132. [Google Scholar] [CrossRef]
- Tjiang, F.; Ye, L.W.; Huang, Y.J.; Chou, C.C.; Tsai, D.S. Effect of processing parameters on soft regime behavior of plasma electrolytic oxidation of magnesium. Ceram. Int. 2017, 43, S567–S572. [Google Scholar] [CrossRef]
- Arrabal, R.; Matykina, E.; Hashimoto, T.; Skeldon, P.; Thompson, G.E. Characterization of AC PEO coatings on magnesium alloys. Surf. Coat. Technol. 2009, 203, 2207–2220. [Google Scholar] [CrossRef]
- Hussein, R.O.; Zhang, P.; Nie, X.; Xia, Y.; Northwood, D.O. The effect of current mode and discharge type on the corrosion resistance of plasma electrolytic oxidation coated magnesium alloy AJ62. Surf. Coat. Technol. 2011, 206, 1990–1997. [Google Scholar] [CrossRef]
- Hussein, R.O.; Nie, X.; Northwood, D.O. An investigation of ceramic coating growth mechanisms in plasma electrolytic oxidation processing. Electrochim. Acta 2013, 112, 111–119. [Google Scholar] [CrossRef]
- Hussein, R.O.; Northwood, D.O.; Nie, X. The influence of pulse timing and current mode on the microstructure and corrosion behavior of a plasma electrolytic oxidation (PEO) coated AM60B magnesium alloy. J. Alloys Compd. 2014, 541, 41–48. [Google Scholar] [CrossRef]
- Hussein, R.O.; Northwood, D.O.; Sue, J.F.; Nie, X. A study of the interactive effects of hybrid current modes on the tribological properties of a plasma electrolytic oxidation coated AM60B Mg-alloy. Surf. Coat. Technol. 2013, 215, 421–430. [Google Scholar] [CrossRef]
- Liang, J.; Guo, B.; Tian, J.; Liu, H.; Zhou, J.; Liu, W.; Xu, T. Effects of NaAlO2 on structure and corrosion resistance of microarc oxidation coatings formed on AM50B magnesium alloy in phosphate-KOH electrolyte. Surf. Coat. Technol. 2005, 199, 121–126. [Google Scholar] [CrossRef]
- Liang, J.; Srinivasan, P.B.; Blawert, C.; Dietzel, W. Influence of chlorine ion concentration on the electrochemical corrosion behavior of plasma electrolytic oxidation coated AM50 magnesium alloy. Electrochim. Acta 2010, 55, 6802–6811. [Google Scholar] [CrossRef]
- Srinivasan, P.B.; Liang, J.; Blawert, C.; Stormer, M.; Dietzel, W. Effect of current density on the microstructure and corrosion behavior of plasma electrolytic oxidation treated AM50 magnesium alloy. Appl. Surf. Sci. 2009, 255, 4212–4218. [Google Scholar] [CrossRef]
- Liang, J.; Srinivasan, P.B.; Blawert, C.; Stormer, M.; Dietzel, W. Electrochemical corrosion behavior of plasma electrolytic oxidation coatings on AM50 magnesium alloy formed in silicate and phosphate based electrolytes. Electrochim. Acta 2009, 54, 3842–3850. [Google Scholar] [CrossRef]
- Srinivasan, P.B.; Liang, J.; Balajeee, R.G.; Blawert, C.; Stormer, M.; Dietzel, W. Effect of pulse frequency on the microstructure, phase composition and corrosion performance of a phosphate- based plasma electrolytic oxidation coated AM50 magnesium alloy. Appl. Surf. Sci. 2010, 256, 3928–3935. [Google Scholar] [CrossRef]
- Gao, Y.; Yerokhin, A.; Matthews, A. Effect of current mode on PEO treatment of magnesium in Ca- and P-containing electrolyte and resulting coatings. Appl. Surf. Sci. 2014, 316, 558–567. [Google Scholar] [CrossRef]
- Pan, Y.K.; Chen, C.Z.; Wang, D.G.; Zhao, T.G. Improvement of corrosion and biological properties of micro arc oxidized coatings on Mg-Zn-Zr alloy by optimizing negative power density parameters. Colloids Surf. B Biointerfaces 2014, 113, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Krzakala, A.; Kazek-Kesik, A.; Simka, W. Application of plasma electrolytic oxidation to bioactive surface formation on titanium and its alloys. RSC Adv. 2013, 3, 19725–19743. [Google Scholar] [CrossRef]
- Nyan, M.; Tsutsumi, Y.; Oya, K.; Doi, H.; Nomura, N.; Kasugai, H.; Hanawa, T. Synthesis of novel oxide layers on titanium by combination of sputter deposition and micro-arc oxidation techniques. De. Mater. J. 2011, 30, 754–761. [Google Scholar] [CrossRef]
- Matykina, E.; Arrabal, R.; Mingo, B.; Mohedano, M.; Pardo, A.; Merino, M.C. In vitro corrosion performance of PEO coated Ti and Ti6Al4V used for dental and orthopedic implants. Surf. Coat. Technol. 2016, 307, 1255–1264. [Google Scholar] [CrossRef]
- Terleeva, O.P.; Slonova, A.I.; Mironov, I.V.; Rogov, A.B.; Sharkeev, Y.P. Microplasma synthesis of biocompatible coatings with additions of magnesium, silicon and silver on pure titanium from homogeneous electrolytes. Surf. Coat. Technol. 2016, 307, 1265–1273. [Google Scholar] [CrossRef]
- Rokosz, K.; Hryniewicz, T.; Gaiaschi, S.; Chapon, P.; Raaen, S.; Pietrzak, K.; Malorny, W. Characterization of calcium and phosphorus enriched porous coatings on CP titanium grade 2 fabricated by plasma electrolytic oxidation. Metals 2017, 7, 354. [Google Scholar] [CrossRef]
- Rokosz, K.; Hryniewicz, T.; Raaen, S. Development of plasma electrolytic oxidation for improved Ti6Al4V biomaterial surface properties. Int. J. Manuf. Technol. 2016, 85, 2425–2437. [Google Scholar] [CrossRef]
- Yeung, W.K.; Sukhorukova, I.V.; Shtansky, D.V.; Levashov, E.A.; Zhitnyak, I.Y.; Gloushankova, N.A.; Kiryukhantsev-Korneev, P.V.; Petrzhik, M.I.; Matthews, A.; Yerokhin, A. Characteristics and in vitro response of thin hydroxyapatite titania films produced by plasma electrolytic oxidation of Ti alloys in electrolytes with particle additions. RSC Adv. 2016, 6, 12688–12698. [Google Scholar] [CrossRef] [PubMed]
- Song, W.H.; Jun, Y.K.; Han, Y.; Hong, S.H. Biomimetic apatite coatings on micro-arc oxidized titania. Biomaterials 2004, 25, 3341–3349. [Google Scholar] [CrossRef] [PubMed]
- Kazek-Kesik, A.; Dercz, G.; Suchanek, K.; Kalemba-Rec, I.; Piotrowski, J.; Simka, W. Biofunctionalization Ti-13Nb-13Zr alloy surface by plasma electrolytic oxidation, Part I. Surf. Coat. Technol. 2015, 276, 59–69. [Google Scholar] [CrossRef]
- Kazek-Kesik, A.; Krok-Borkowicz, M.; Jakobik-Kolon, A.; Pamula, E.; Simka, W. Biofunctionalization Ti-13Nb-13Zr alloy surface by plasma electrolytic oxidation, Part II. Surf. Coat. Technol. 2015, 276, 23–30. [Google Scholar] [CrossRef]
- Yao, Z.; Jiang, Y.; Jia, F.; Jiang, Z.; Wang, F. Growth characteristics of plasma electrolytic oxidation ceramic coating on Ti-6Al-4V alloy. Appl. Surf. Sci. 2008, 254, 4084–4091. [Google Scholar] [CrossRef]
- Yao, Z.; Liu, Y.; Xu, Y.; Jiang, Z.; Wang, F. Effect of cathode pulse at high frequency on structure and composition of Al2TiO5 ceramic coatings on Ti alloy by plasma electrolytic oxidation. Mater. Chem. Phys. 2011, 126, 227–231. [Google Scholar] [CrossRef]
- Hussein, R.O.; Nie, X.; Northwood, D.O. A spectroscopic and microstructural study of oxide coatings produced on a Ti-6Al-4V alloy by plasma electrolytic oxidation. Mater. Chem. Phys. 2012, 134, 484–492. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, D.-S.; Chou, C.-C. Review of the Soft Sparking Issues in Plasma Electrolytic Oxidation. Metals 2018, 8, 105. https://doi.org/10.3390/met8020105
Tsai D-S, Chou C-C. Review of the Soft Sparking Issues in Plasma Electrolytic Oxidation. Metals. 2018; 8(2):105. https://doi.org/10.3390/met8020105
Chicago/Turabian StyleTsai, Dah-Shyang, and Chen-Chia Chou. 2018. "Review of the Soft Sparking Issues in Plasma Electrolytic Oxidation" Metals 8, no. 2: 105. https://doi.org/10.3390/met8020105
APA StyleTsai, D.-S., & Chou, C.-C. (2018). Review of the Soft Sparking Issues in Plasma Electrolytic Oxidation. Metals, 8(2), 105. https://doi.org/10.3390/met8020105





