3.2. Effect of Strain on Microcracks
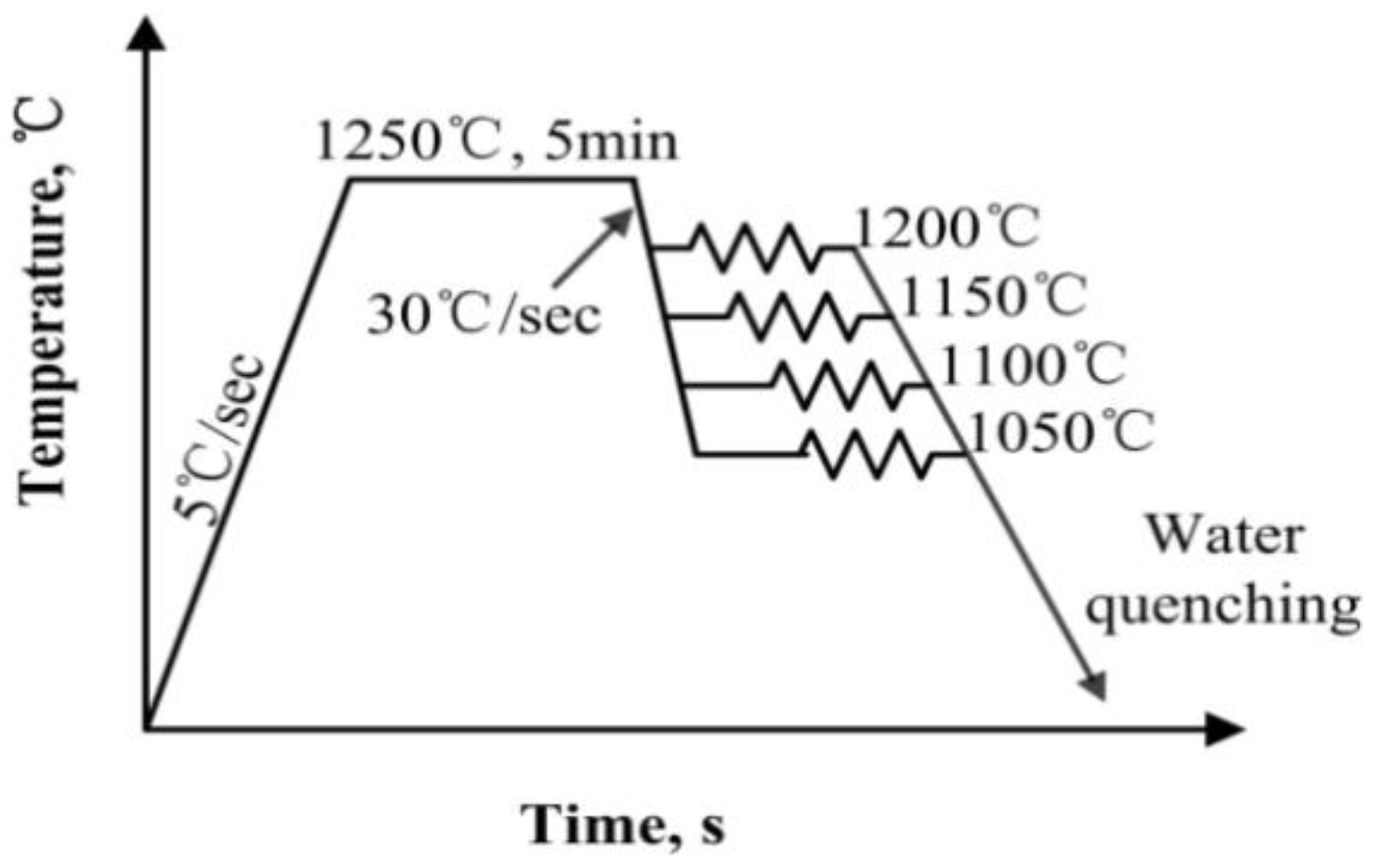
Based on the schematic diagrams shown in
Figure 1, the hot compression tests were carried out to simulate the hot rolling process.
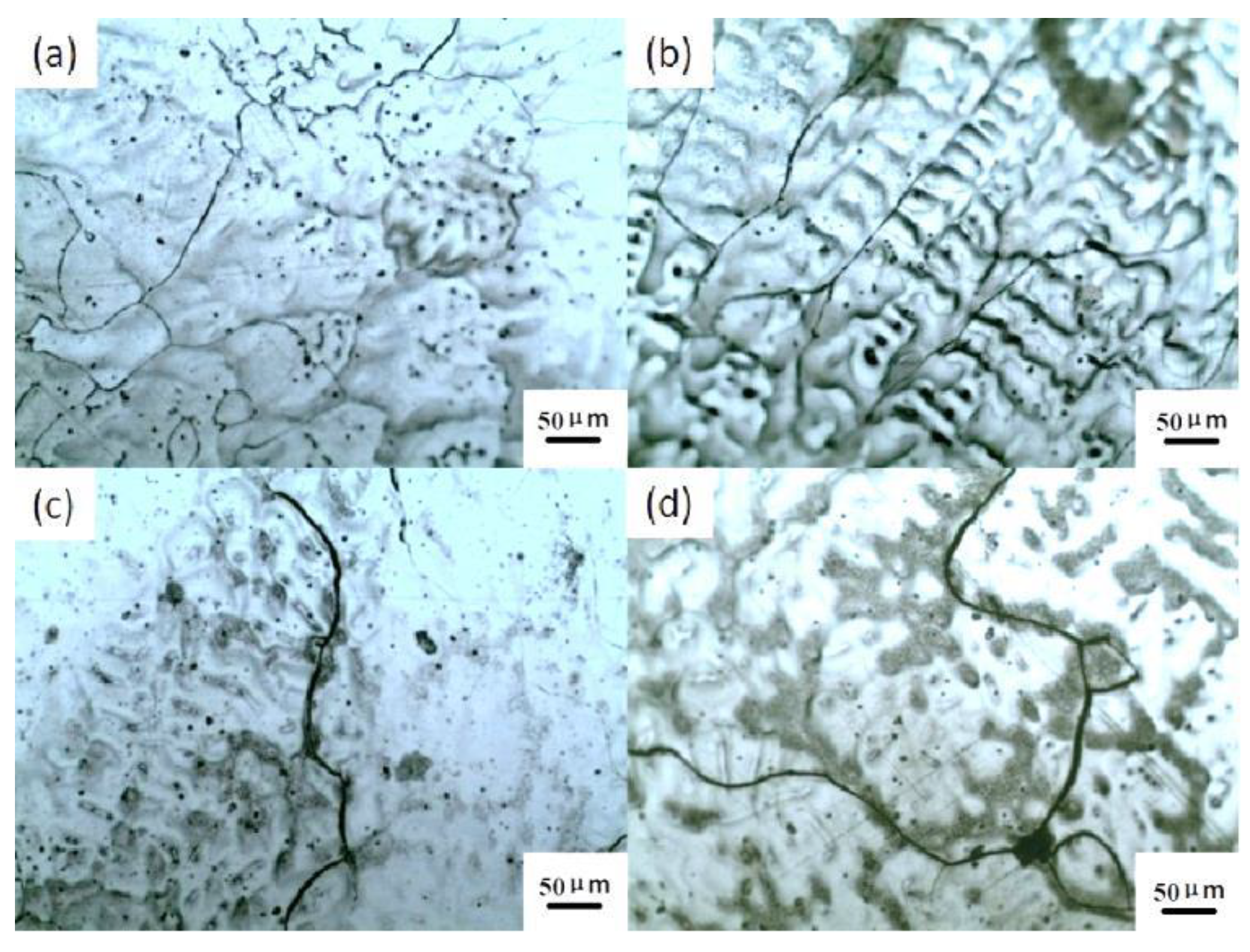
Figure 4 shows the development of microcracks at 1150 °C during the hot deformation process. It can be seen that no microcracks form in the samples compressed at the strain of 0.2, but the discontinuous segregation of precipitates appears, as shown in
Figure 4a. When the strain reaches 0.5, apparent microcracks nucleate, coalesce and propagate, as shown in
Figure 4b, and eventually evolve into microcracks.
To further study the formation mechanism of microcracks, field emission scanning electron microscopy (FESEM) with energy dispersive spectroscopy (EDS) was used to elucidate the origins of microcrack formation.
Figure 5a shows a SEM micrograph of the precipitates for the Cu-bearing austenite stainless steel after the hot compression test with the strain of 0.2 at 1050 °C; it can be seen that no cracking occurs.
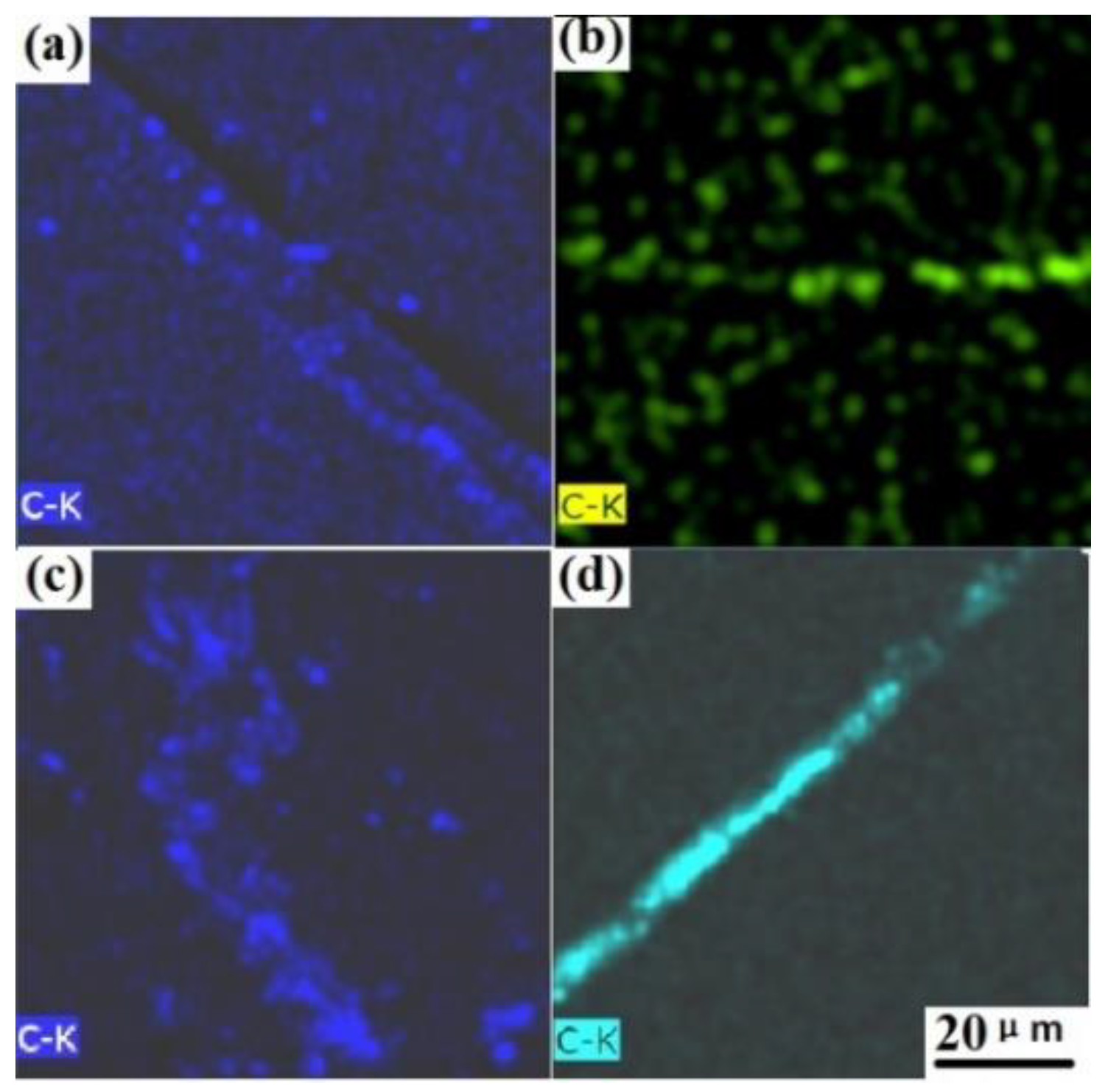
Figure 5b shows the EDS-X-ray maps corresponding to
Figure 5a. Through the examination of the EDS-X-ray maps of C, S and Si shown in
Figure 5f,g,h, respectively, these precipitates are identified as carbide and oxide inclusions, and no apparent Cr-rich and Cu-rich zone is observed(
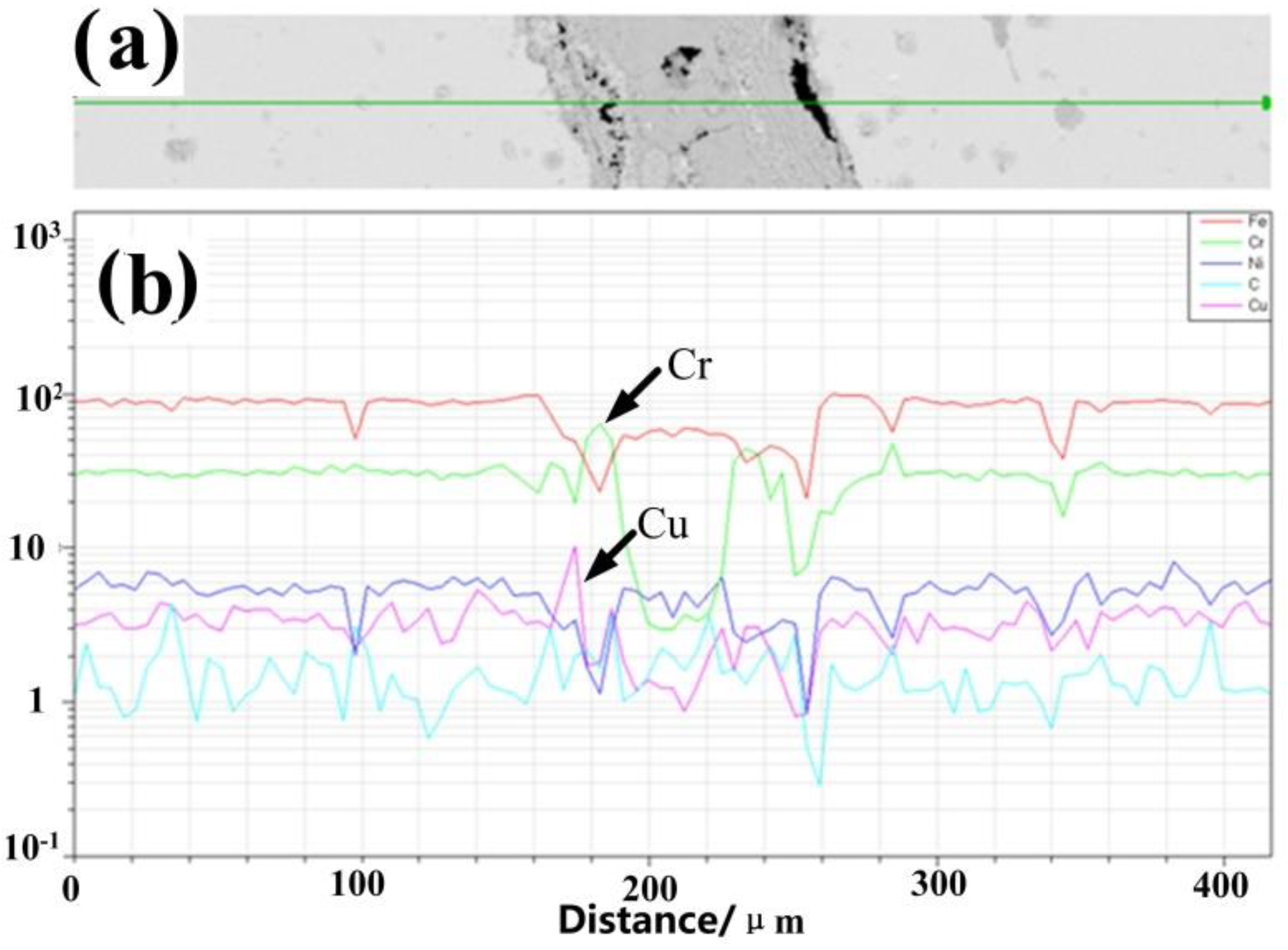
Figure 5d,e). To further illustrate the distribution of Cu in deformed materials, a SEM-EDS elemental line-scan map is obtained in the process of hot deformation at the strain of 0.2, with the results shown in
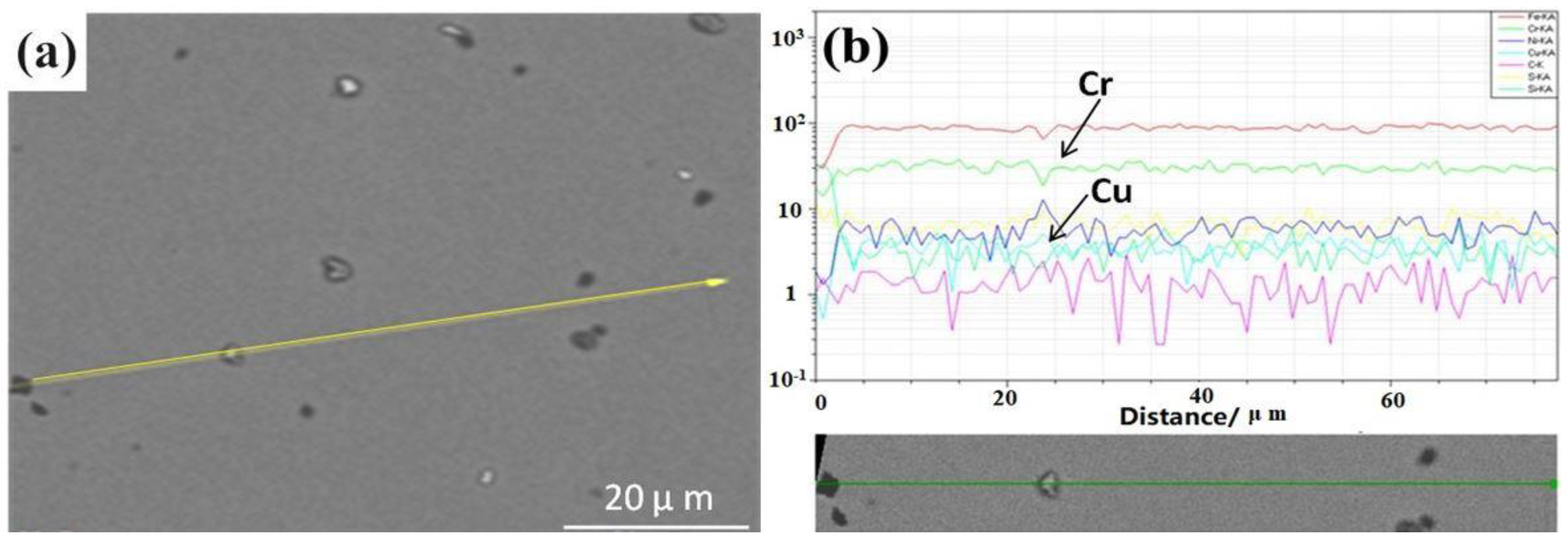
Figure 6a. It can be seen from the maps of
Figure 6b that the peak and trough values of Cu show little difference with the change in the distance. The values of vibration are almost the same and are approximately in the range of 1–2 μm, which accounts for the homogeneous Cu distribution along the line and the possibility of the dispersed distribution of the particles.
From the standpoint of fracture mechanics, the formation of microcracks is closely related to the second-phase particles and precipitates contained in steel. The second-phase inclusions contained in the material induce a cavity that nucleates, grows, and polymerizes until it is connected to the macroscopic cracks, and this process is considered to be the root cause of the crack propagation and the fracture of the material. With the strain reaching 0.5, the nucleation and propagation of microcracks arises.
Figure 7a shows the area of cavity formation and aggregation, in which many small cavities in the left and a cavity after aggregation are marked, and a SEM-EDS elemental line-scan map is carried out along a line. It can be seen from
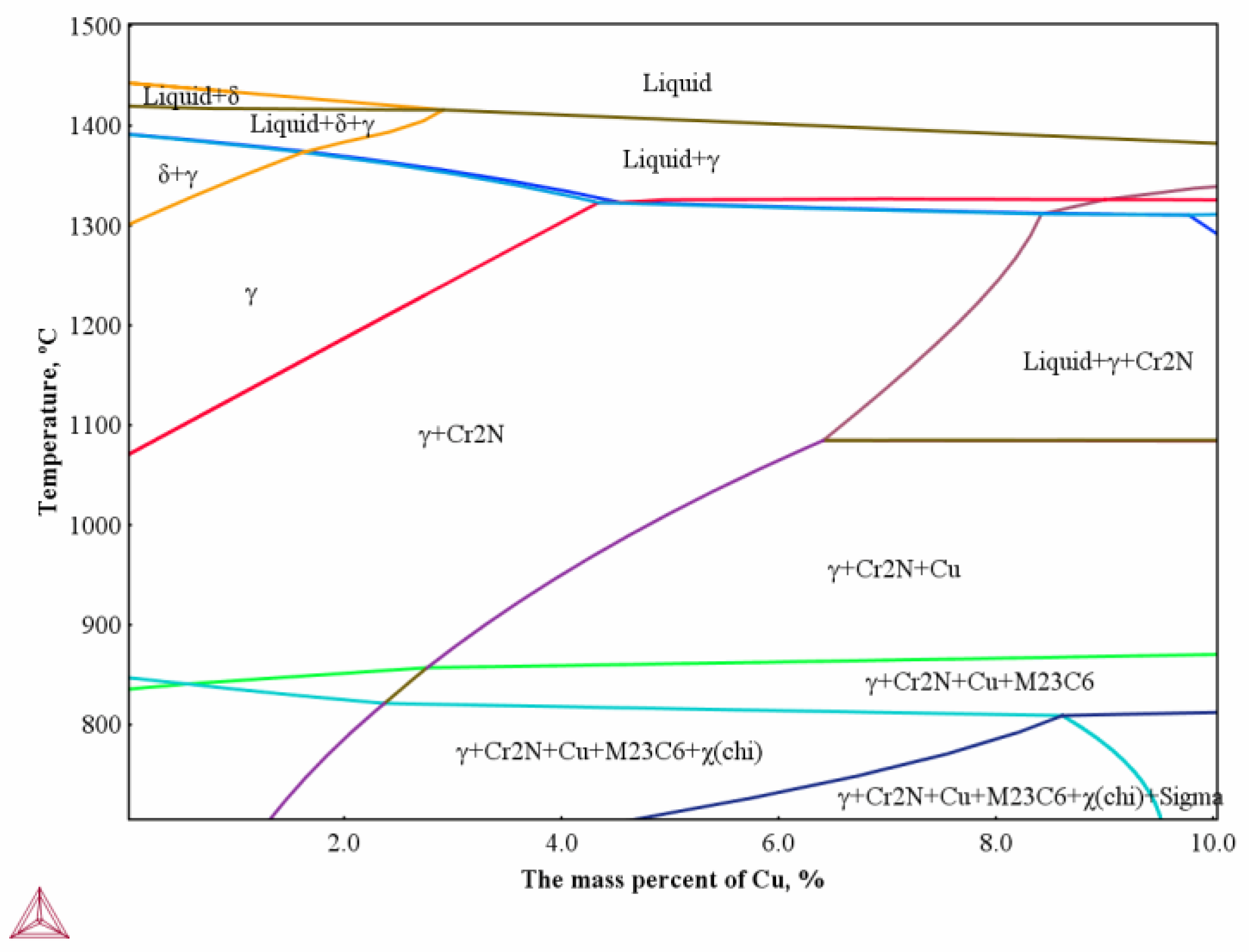
Figure 7b that the Cr-rich areas appear around cavities and the distribution of Cu along the line is inhomogeneous. The peak and trough values of Cu show large differences, and a chromium-depleted zone appears in the middle. It can be seen from the above phenomena that the rich-Cu phase increases with the strain increasing, and the chromium-rich zone is generated due to the Cr(C, N) precipitates in the grain boundary.
The dislocations and the twin crystals of the samples deformed at 1050 °C are shown in
Figure 8. Various dislocation configurations, including free dislocations, dislocation walls and twins, were observed in deformed grains, further confirming that the dislocations also play an important role in crack formation. However, as shown in
Figure 8a, for the sample deformed with the strain of 0.2 at 1050 °C, the dislocation walls are observed obviously, and there are fewer free dislocations. While deformed at the strain of 0.5, it is difficult for the dislocations to move, and stress concentration becomes severe. The twins are observed as a complement to slip at the stress concentration zone (
Figure 8b).
Yeli and Auger [
15] have proposed that Cr
2N precipitates out in advance around the grain boundary, offering the inhomogeneous nucleation location for Cu. The Cu-rich precipitates increase in the deformation process, and some Cu nucleates around Cr
2N distributed in the grain boundary, leading to the segregation of Cu and weakening of the grain boundary. As shown in
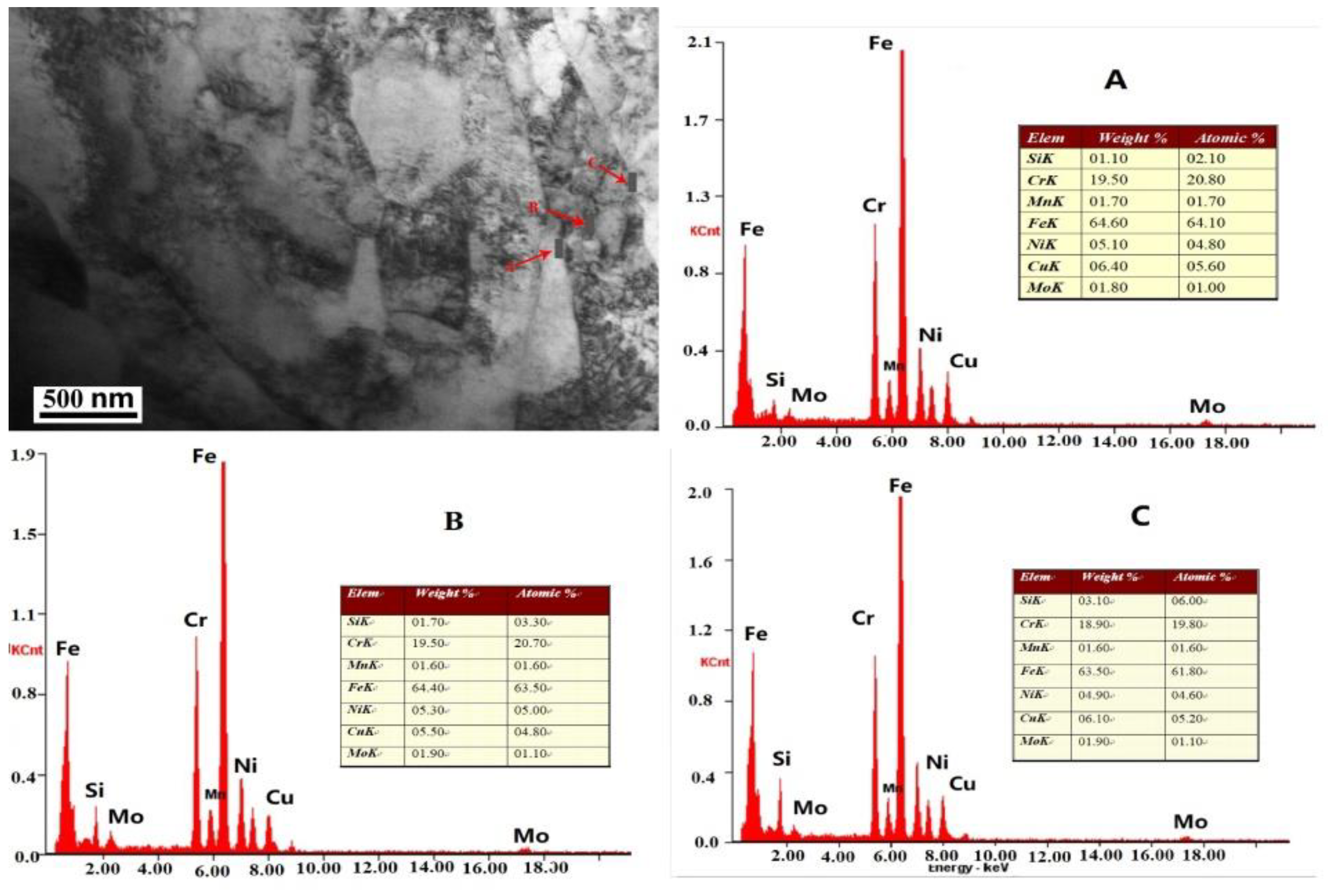
Figure 9, regions A and C are the grain boundaries, and B is the region inside the grain. The copper content is 6.4 wt.% in region A, 5.5 wt.% in region B and 6.1 wt.% in region C, from which it can be seen that the Cu content in the grain boundary is higher than that of Cu inside the grain. On the other hand, it is also revealed that the intercrystalline crack formation is related to intercrystalline Cr
2N precipitation [
16]. When hot deformation is conducted, dislocations move along the slip plane. Nevertheless, intercrystalline Cr
2N hinders the dislocation movement, leading to dislocation pile-up and stress concentration. Therefore, microcracks arise during hot compression due to the synergistic effect of intergranular Cu-rich and Cr
2N precipitation. According to literature 17, Nitrogen is detected exclusively in the form of CrN
+ by APT, the interconnected structure displayed is possibly due to the migration of N to dislocations and matrix defects, which explains the results of the experiment.
3.3. Effect of Deformation Temperature on Microcracks
To further study the origin of crack formation, hot compression tests were carried out for the Cu-bearing austenite stainless steel at the strain of 0.5 and temperature range of 1050–1200 °C.
The samples were cut along the direction perpendicular to the axial direction and polished. It can be seen from
Figure 10 that a large number of cracks in the interior of the sample occur at the deformation temperature of 1100 °C, while less quantity is observed at other temperatures.
From the microscopic point of view, the formation of cracks is strongly related to the precipitation of the second phase. However, conditions of undeformed austenite are close to those of homogeneous nucleation. The driving force of precipitation is only the supersaturated energy of micro-alloying elements in austenite, and carbon nitrides can be hardly obtained in actual production. Nevertheless, the strain accelerates the precipitation process of strain-induced precipitation. After the hot deformation at the different strains was investigated, it was necessary to investigate the element distribution of the samples under different deformation temperatures.
Figure 11 shows the SEM images of the microcrack zones and the cavity zones obtained by hot deformation at the different temperatures. In addition, the elements are analyzed by EDS-X-ray maps. The result in
Figure 12,
Figure 13 and
Figure 14 represent the distributions of Cu, Cr, and C, respectively, in the zone marked in
Figure 11. It can be seen that Cu-rich, Cr-rich and carbide phases appear in the microcrack and cavity areas and increase with the increase in the temperature of the hot compression process.
EBSD analysis of the test specimens with the strain of 0.5 at 1100 °C (
Figure 15a) shows that the compressed sample has a plastic deformation zone on the cut surface. Micropores can easily form, expand, and link with each other in this region, leading to crack propagation.
Figure 15b shows that a very small amount of ferrite forms, it is possible to be δ-ferrite formed during the hot rolling process, which easily distributes along the grain boundary in the plastic deformation zone being prone to crack propagation along the ferrite.
Figure 15c shows coincidence site lattice (CSL) distribution, it means that the low-angle Σ3 grain boundary accounts for more than 66%, the grain boundary energy is low, and it is easy to form cracks in the compression process.
The actual cracked sample rolling at 1100 °C was taken, and TEM analysis was performed. Block precipitates along the grain boundary in the
Figure 16a were a composite of carbide, and the combined transmission analysis (selected area electron diffraction, SAED) showed mainly M
23C
6 carbide in the
Figure 16b.
The elements mainly precipitate in the form of the Cu-rich phase and the Cr(C, N) phase, which weaken the grain boundary so that the cavities form around the second phase. Meanwhile, the inclusions hinder dislocation movement, and the dislocation pile-up is generated, which leads to the concentration of stress. The stress concentration results in cavity aggregation and growth, and eventually the microcracks form. The microcrack extension under large strain leads to the fracture of materials. However, it can be seen from
Figure 10 that the most cracks occur at 1100 °C, and the least number of cracks is observed at higher temperature and at lower temperature. That is mainly because an increased temperature accelerates the velocity of the dislocations and reduces the stress concentration phenomenon caused by the dislocation pile-up. Furthermore, the second phase seldom precipitates at a lower temperature during the process of deformation, and few cavities are formed. When the hot compression temperature is higher, the speed of the dislocations increases, and the stress concentration phenomenon due to dislocation pile-up is weakened. Briefly, the macroscopic cracks decrease at lower and higher temperatures in the hot deformation process.
To further study the relationship of second phase precipitation and the microcrack formation, the external stress
required in the process of cavity formation around the second-phase particles is introduced, and its expression is obtained as [
17]:
where
q is the average stress concentration factor around the second phase,
E is the elastic modulus of the matrix, γ
S is the specific surface energy of the matrix, σ
s is the yield strength of the matrix, and α is the proportionality coefficient and its value in the steel is 0.5.
V is the volume of the second-phase particles, and Δ
V is the volume of the plastic deformation zone of the matrix around the second-phase particles. Equation (1) shows that the required stress is inversely proportional to the precipitate size. The stress that cavity formation has is unrelated to the shape of the second phase, but rather, mainly depends on size. Cavities are more likely to form around the second phase with large dimensions. The Cu-rich precipitates, carbides and nitrides increase with the increase in strain, and the external stress σ required in the process of forming cavities decreases, making it easy to form cavities. Furthermore, the deformation temperature and strain rate have a significant influence. A relatively low temperature can give rise to a rapid increase in the yield strength σ
s due to the slow movement of dislocations under the low temperature, when the strain rate reaches a constant value. According to Equation (1), the external stress σ increases.
According to the energy consumption analysis method of the crack propagation process, the fracture toughness of materials
GIC is obtained [
17]:
where γ
S is the specific surface energy of the matrix, and γ
p is the consumed plastic energy during the formation of the microcracks per unit area. Because of the poor toughness for most second phases compared to the matrix, it is impossible for plastic deformation to occur, which restricts the size of the plastic zone near the crack tip; therefore, γ
p decreases, and crack propagation is promoted through the formation, growth and coalescence of cavities. Hence, the increase in the second phase and inclusion volume fraction is detrimental to the fracture toughness of materials. In addition, the size, shape and distribution of the precipitates have a remarkable influence on the fracture toughness. Furthermore, the existence of segregation element weakening of the grain boundary significantly reduces the strength of the interface and the binding force of both sides of the crystal interface but has no obvious influence on the yield strength of materials. Fracture plastic energy γ
p of the micro-zone adjacent to the interface is so low that it is easy for microcracks to form and extend along the grain boundary, thus significantly reducing the tensile strength. In this experiment, it is clearly observed that with the strain of 0.5 at each temperature, microcracks exist along the grains and extend through the grain boundary, destroying the microstructure, resulting in the material failure. The reason is that most intergranular fractures are brittle fractures, and the toughness is low, which is due to the element segregation, the second phase and stress concentration interaction with weakening the grain boundary.