Dynamic Strain Aging Behaviour in AISI 316L Austenitic Stainless Steel under As-Received and As-Welded Conditions
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
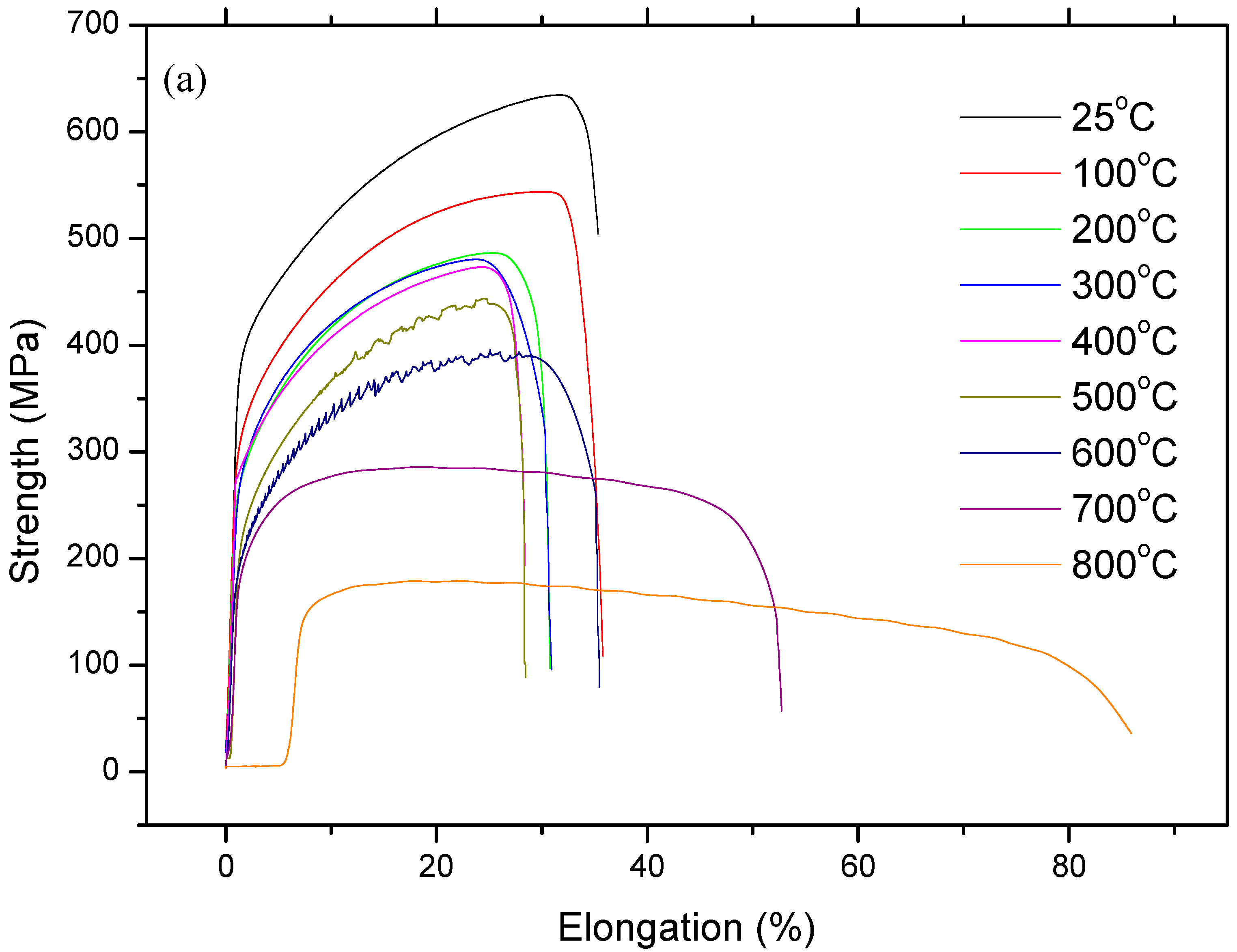
- DSA occurs in 316L austenitic stainless steel under as-received and as-welded conditions which revealed small variation in YS and UTS values up to 600 °C corresponding to the DSA region.
- As-welded samples are more susceptible to DSA compared to the as-received samples, due to the partial dissolution of carbides in the HAZ next to the fusion line during welding.
- As-received samples of 316L austenitic stainless steel achieved higher work hardening index n values than as-welded samples because of good formability. However, the as-received samples reached a larger reduction in work hardening index (5%) while as-welded samples showed an increase in work hardening index (23%) at the peak ageing temperature of 500 °C, compared to those in the room temperature testing conditions.
- 316L austenitic stainless steel has high susceptibility to deformation induced martensite after testing at 25 °C. The results obtained from present study indicated that the martensite occurred at the shear bands.
- Fracture surface analysis indicated that as-received and as-welded samples showed ductile fracture mode after testing at 25 °C, 200 °C and 800 °C. However, as-received and as-welded samples revealed mixed type fracture of cleavage facets and dimples at 400 °C and 600 °C. The reduction in area is also decreased at the same temperatures of 400 °C or 600 °C, which suggests that fracture occurs more easily under DSA conditions.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mcguire, M. Stainless Steel for Design Engineering, 1st ed.; ASM International: Geauga County, OH, USA, 2008; Chapter 6; pp. 69–78. ISBN 978-0-87170-717-8. [Google Scholar]
- Lippold, J.C.; Kotecki, D.J. Welding Metallurgy and Weldability of Stainless Steels, 1st ed.; John Wiley & Sons Inc.: Hoboken, NJ, USA, 2005; pp. 20–44. ISBN 978-0-471-47379-4. [Google Scholar]
- Ehrnsten, U.; Toivonen, A.; Ivanchenko, M.; Nevdacha, V.; Yagozinskyy, Y.; Haenninen, H. Dynamic strain ageing of deformed nitrogen-alloyed AISI 316 stainless steels. In Proceedings of the EUROCORR 2004—European Corrosion Conference: Long Term Prediction and Modeling of Corrosion, Paris, France, 12–16 September 2004. [Google Scholar]
- Wang, D.Q.; Zhu, M.L.; Xuan, F.Z. Correlation of local strain with microstructures around fusion zone of a CrNi-Mo-V steel welded joint. Mater. Sci. Eng. A 2017, 685, 205–212. [Google Scholar] [CrossRef]
- Neuenschwander, M.; Knobloch, M.; Fontana, M. Elevated temperature mechanical properties of solid section structural steel. Constr. Build. Mater. 2017, 149, 186–201. [Google Scholar] [CrossRef]
- Ivanchenko, M. Dynamic Strain Ageing of Austenitic Stainless Steels and Ni-based Alloys. Ph.D. Thesis, Aalto University, Espoo, Finland, 19 November 2010. [Google Scholar]
- Gopinath, K.; Gogia, A.K.; Kamat, S.V.; Ramamurty, U. Dynamic strain ageing in Ni-base superalloy 720Li. Acta Mater. 2009, 57, 1243–1253. [Google Scholar] [CrossRef]
- Yu, D.; Yu, W.; Chen, G.; Jin, F.; Chen, X. Role of dynamic strain aging in the tensile property, cyclic deformation and fatigue behavior of Z2CND18.12N stainless steel between 293 K and 723 K. Mater. Sci. Eng. A 2012, 558, 730–736. [Google Scholar] [CrossRef]
- Dieter, G.E. Mechanical Metallurgy, 3rd ed.; McGraw-Hill: Edmonton, AB, Canada, 2013; pp. 100–130. ISBN 10: 1-25-906479-4. [Google Scholar]
- Ananthakrishna, G. Current theoretical approaches to collective behavior of dislocations. Phys. Rep. 2007, 440, 113–259. [Google Scholar] [CrossRef]
- Vargas-Arista, B.; Angeles-Chavez, C.; Albiter, A.; Hallen, J.M. Metallurgical investigation of the aging process on tensile fracture welded joints in pipeline steel. Mater. Charact. 2009, 60, 1561–1568. [Google Scholar] [CrossRef]
- Hutchinson, B.; Komenda, J.; Rohrer, G.S.; Beladi, H. Heat affected zone microstructures and their influence on toughness in two microalloyed HSLA steels. Acta Mater. 2015, 97, 380–391. [Google Scholar] [CrossRef]
- Buono, V.T.L.; Gonzales, B.M.; Andrade, M.S. Strain aging of AISI 430 ferritic stainless steel. Scr. Mater. 1997, 38, 185–190. [Google Scholar] [CrossRef]
- Hong, S.G.; Lee, S.B. The tensile and low-cycle fatigue behavior of cold worked 316L stainless steel: influence of dynamic strain aging. Int. J. Fatigue 2004, 26, 899–910. [Google Scholar] [CrossRef]
- Gündüz, S. Dynamic strain aging effects in niobium microalloyed steel. Ironmak. Steelmak. 2002, 29, 341–346. [Google Scholar] [CrossRef]
- Huang, Z.; Wagner, D.; Bathias, C. Some metallurgical aspects of Dynamic Strain Aging effect on the Low Cycle Fatigue behavior of C-Mn steels. Int. J. Fatigue 2015, 80, 113–120. [Google Scholar] [CrossRef]
- Kou, S. Welding Metallurgy, 2nd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003; pp. 143–147. ISBN 0-471-43491-4. [Google Scholar]
- Kožuh, S.; Gojić, M.; Kosec, L. Mechanical properties and microstructure of austenitic stainless steel after welding and post-weld heat treatment. Kovove Mater. 2007, 47, 253–262. [Google Scholar]
- Kožuh, S.; Gojić, M.; Kosec, L. The effect of annealing on properties of AISI 316L base and weld metals. Mater. Geoenviron. 2007, 54, 331–344. [Google Scholar]
- Lee, H.J.; Lee, H.W. Effect of Cr content on microstructure and mechanical properties of low carbon steel welds. Int. J. Electrochem. Sci. 2015, 10, 8028–8040. [Google Scholar]
- Odnobokova, M.; Kipelova, A.; Belyakov, A.; Kaibyshev, R. Microstructure evolution in a 316L stainless steel subjected to multidirectional forging and unidirectional bar rolling. IOP Conf. Ser. Mater. Sci. Eng. 2014, 63, 012060. [Google Scholar] [CrossRef]
- Solomon, N.; Solomon, I. Deformation induced martensite in AISI 316 stainless steel. Rev. Metal. 2010, 46, 121–128. [Google Scholar] [CrossRef]
- Song, R.B.; Xiang, J.Y.; Hou, D.P. Characteristics of mechanical properties an microstructure for 316L austenitic stainless steel. J. Iron Steel Res. Int. 2011, 18, 53–59. [Google Scholar] [CrossRef]
- Karcı, F.; Kaçar, R.; Gündüz, S. The effect of process parameter on the properties of spot welded cold deformed AISI304 grade austenitic stainless steel. J. Mater. Proc. Technol. 2009, 209, 4011–4019. [Google Scholar] [CrossRef]
- Callister, W.D.; Rethwisch, D.G. Materials Science and Engineering, 8th ed.; John Wiley and Sons: New York, NY, USA, 2011; pp. 237–250. ISBN 13: 978-0-471-73696-7. [Google Scholar]












| Materials | C | Si | Mn | Ni | Cr | Mo | N | S | P |
|---|---|---|---|---|---|---|---|---|---|
| 316L | 0.024 | 0.38 | 1.30 | 10.10 | 16.57 | 2.03 | 0.041 | 0.004 | 0.029 |
| Welding Wire | 0.03 | 0.85 | 1.70 | 12.5 | 18.5 | 2.75 | - | 0.003 | 0.0025 |
| Current (A) | Voltage (V) | Welding Speed (cm/min) | Gas Flow Rate (lt/min) | Heat Input (kj/mm) |
|---|---|---|---|---|
| 240 | 23 | 35 | 14 | 0.95 |
| Test Temperatures (°C) | Yield Strength (0.2%, MPa) | Tensile Strength (MPa) | Elongation (%) |
|---|---|---|---|
| 25 | 388 ± 8 | 650 ± 13 | 66 ± 2.3 |
| 100 | 265 ± 5 | 539 ± 11 | 45 ± 1.9 |
| 200 | 216 ± 4 | 483 ± 10 | 38 ± 1.8 |
| 300 | 206 ± 4 | 470 ± 9 | 36 ± 1.7 |
| 400 | 202 ± 4 | 473 ± 9 | 32 ± 1.6 |
| 500 | 180 ± 3 | 437 ± 9 | 34 ± 1.7 |
| 600 | 160 ± 3 | 401 ± 8 | 43 ± 1.9 |
| 700 | 142 ± 3 | 282 ± 6 | 70 ± 2,4 |
| 800 | 125 ± 2 | 178 ± 4 | 112 ± 3.2 |
| Test Temperatures (°C) | Yield Strength (0.2%, MPa) | Tensile Strength (MPa) | Elongation (%) |
|---|---|---|---|
| 25 | 368 ± 8 | 635 ± 13 | 34 ± 1.7 |
| 100 | 306 ± 6 | 544 ± 11 | 36 ± 1.7 |
| 200 | 254 ± 5 | 487 ± 10 | 30 ± 1.6 |
| 300 | 269 ± 5 | 481 ± 10 | 31 ± 1.6 |
| 400 | 283 ± 6 | 473 ± 10 | 28 ± 1.5 |
| 500 | 260 ± 5 | 444 ± 9 | 28 ± 1.5 |
| 600 | 231 ± 5 | 409 ± 8 | 27 ± 1.5 |
| 700 | 181 ± 5 | 286 ± 6 | 52 ± 2.1 |
| 800 | 138 ± 4 | 179 ± 4 | 80 ± 2.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muhamed, G.A.; Gündüz, S.; Erden, M.A.; Taştemur, D. Dynamic Strain Aging Behaviour in AISI 316L Austenitic Stainless Steel under As-Received and As-Welded Conditions. Metals 2017, 7, 362. https://doi.org/10.3390/met7090362
Muhamed GA, Gündüz S, Erden MA, Taştemur D. Dynamic Strain Aging Behaviour in AISI 316L Austenitic Stainless Steel under As-Received and As-Welded Conditions. Metals. 2017; 7(9):362. https://doi.org/10.3390/met7090362
Chicago/Turabian StyleMuhamed, Guma Alnaji, Süleyman Gündüz, Mehmet Akif Erden, and Demet Taştemur. 2017. "Dynamic Strain Aging Behaviour in AISI 316L Austenitic Stainless Steel under As-Received and As-Welded Conditions" Metals 7, no. 9: 362. https://doi.org/10.3390/met7090362
APA StyleMuhamed, G. A., Gündüz, S., Erden, M. A., & Taştemur, D. (2017). Dynamic Strain Aging Behaviour in AISI 316L Austenitic Stainless Steel under As-Received and As-Welded Conditions. Metals, 7(9), 362. https://doi.org/10.3390/met7090362




