Neutron Reflectometry for Studying Corrosion and Corrosion Inhibition
Abstract
1. Introduction
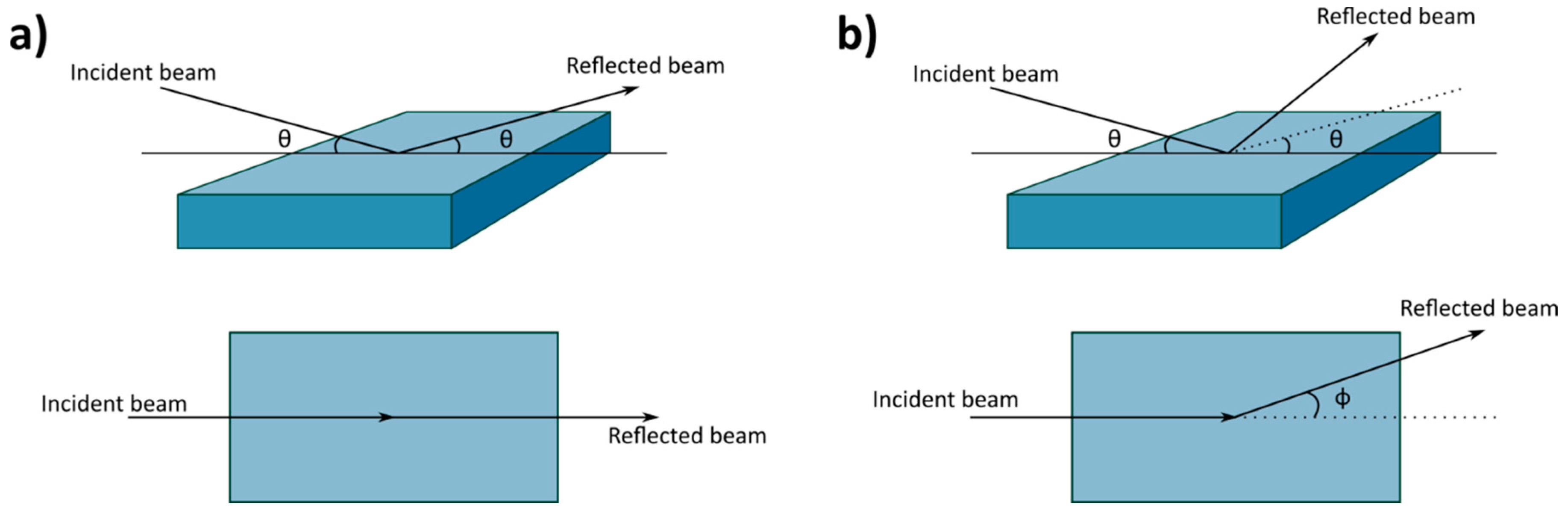
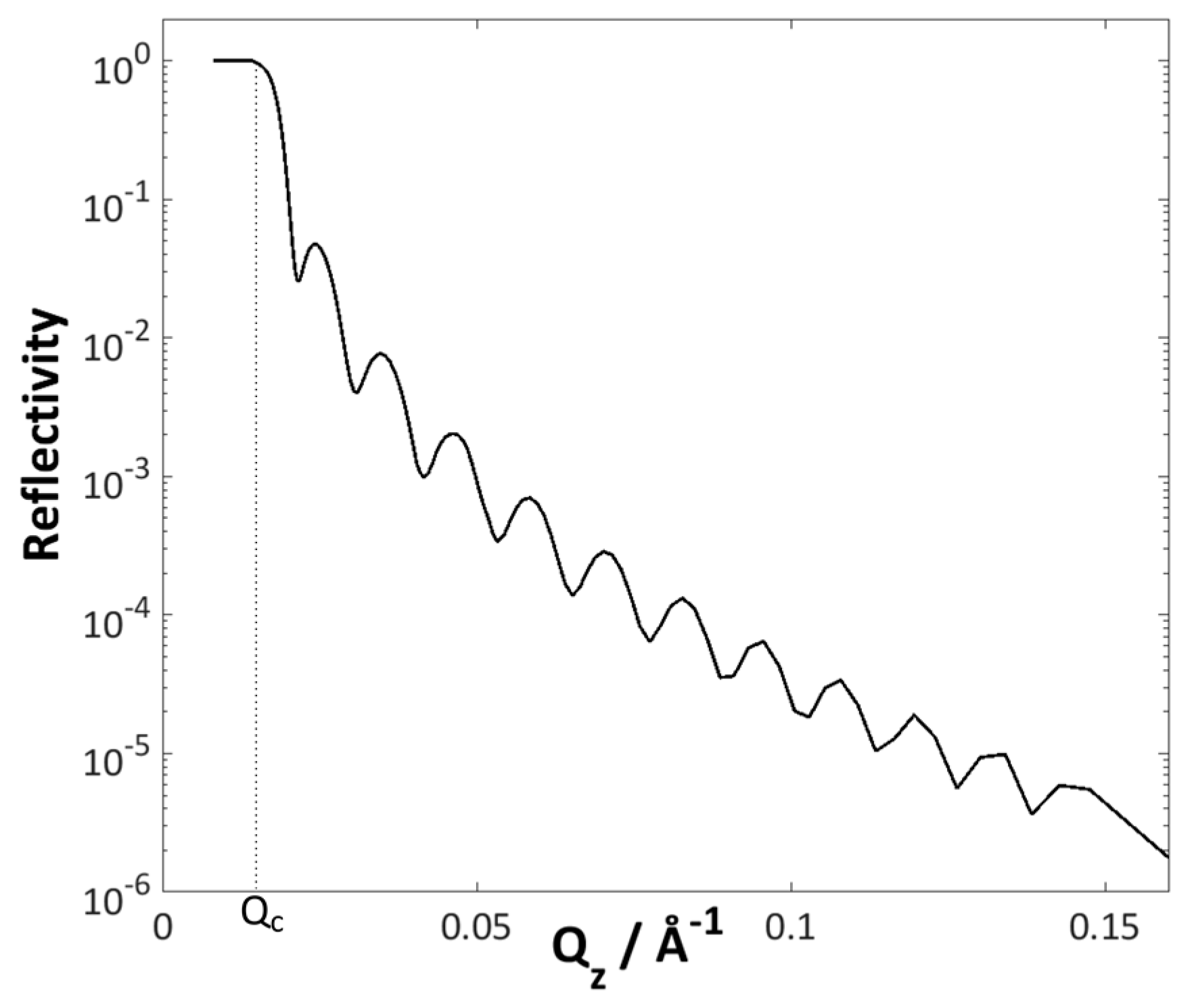
2. Basic Introduction to Neutron Reflectometry
- Q is varied at fixed incident λ and variable θ.
- Q is varied at fixed θ and variable λ.
3. SLD/Contrast Variation and Matching
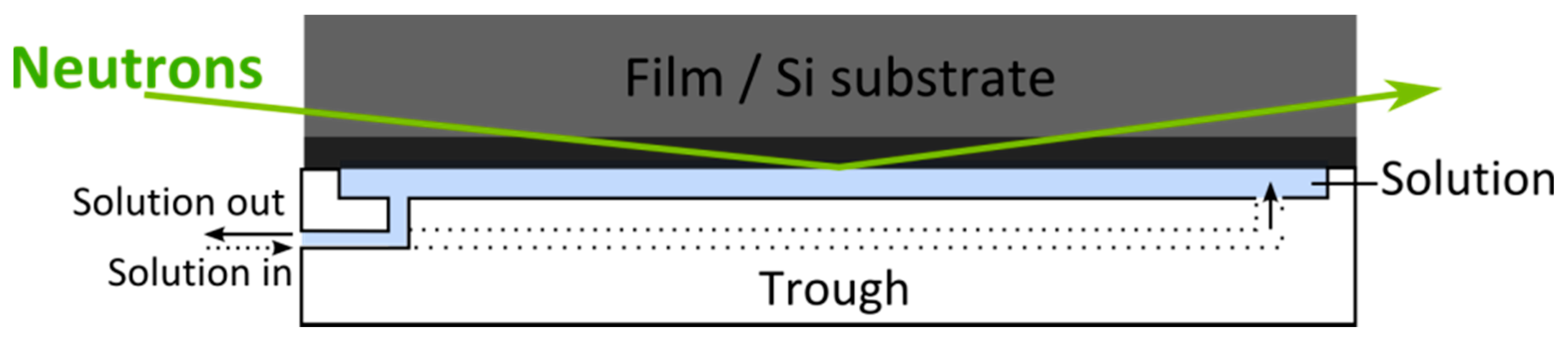
4. Experimental Setup
5. Substrates
6. Examples of Metal Corrosion Studies Using NR
6.1. Aluminium
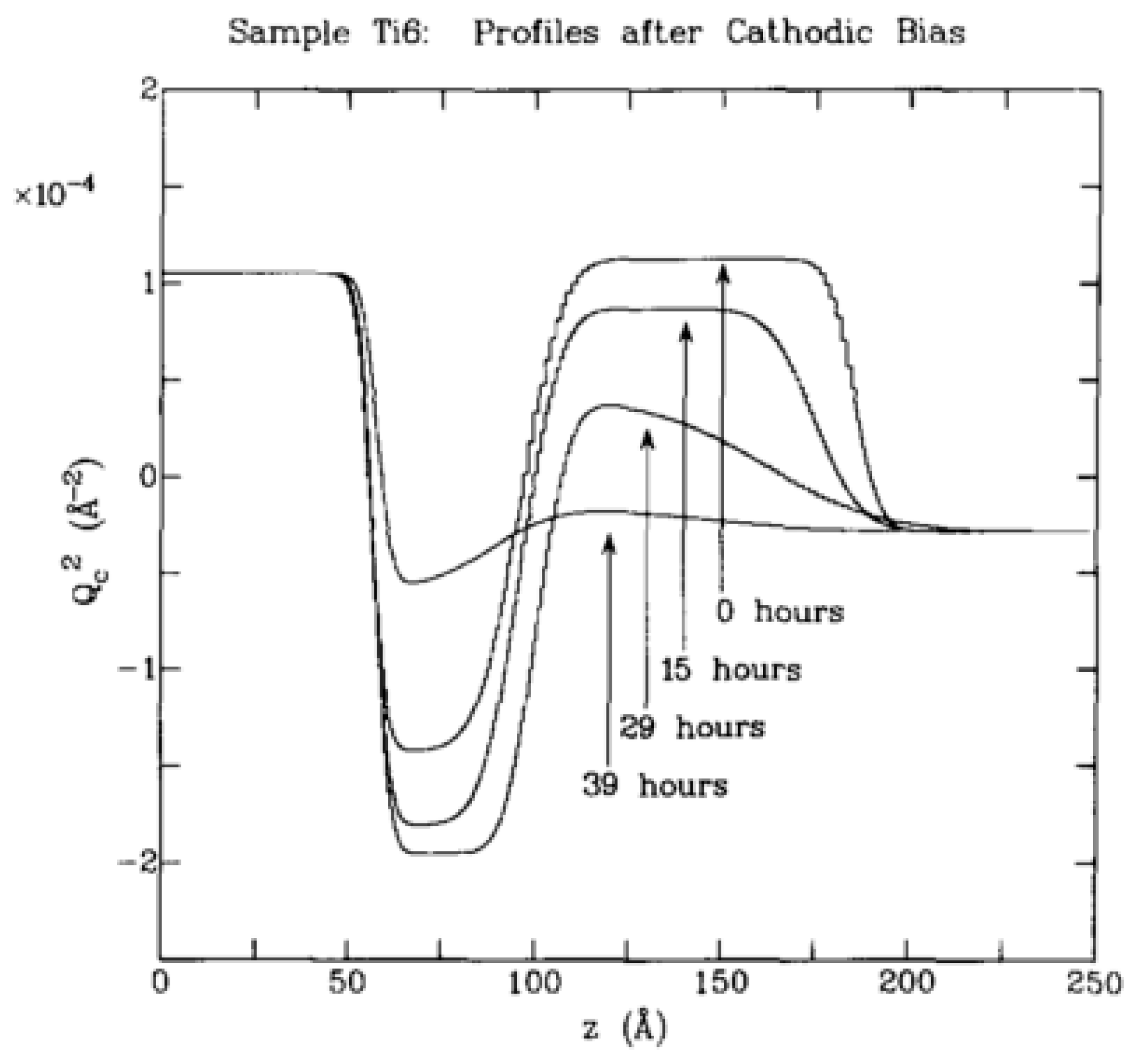
6.2. Titanium
6.3. Zirconium
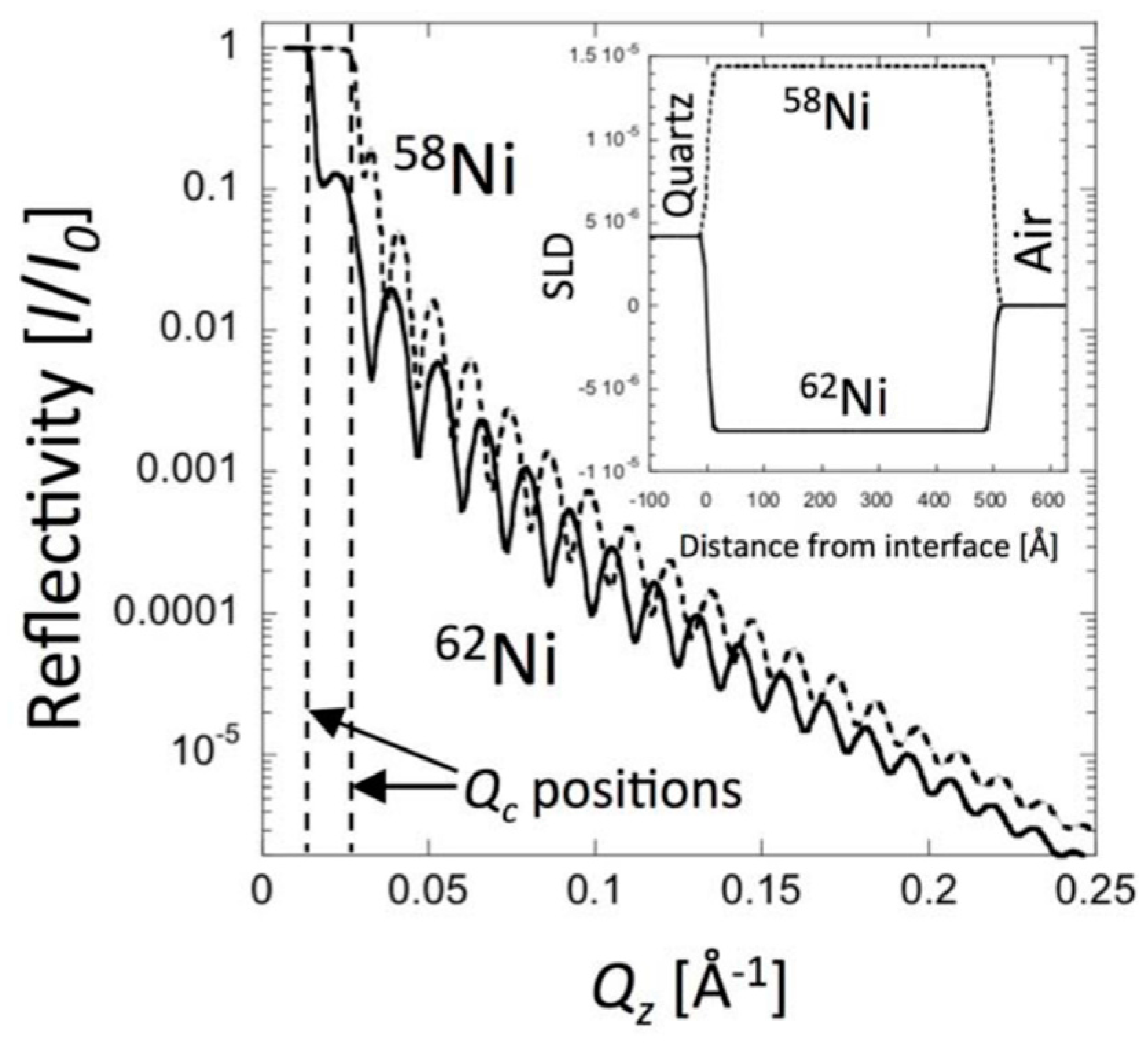
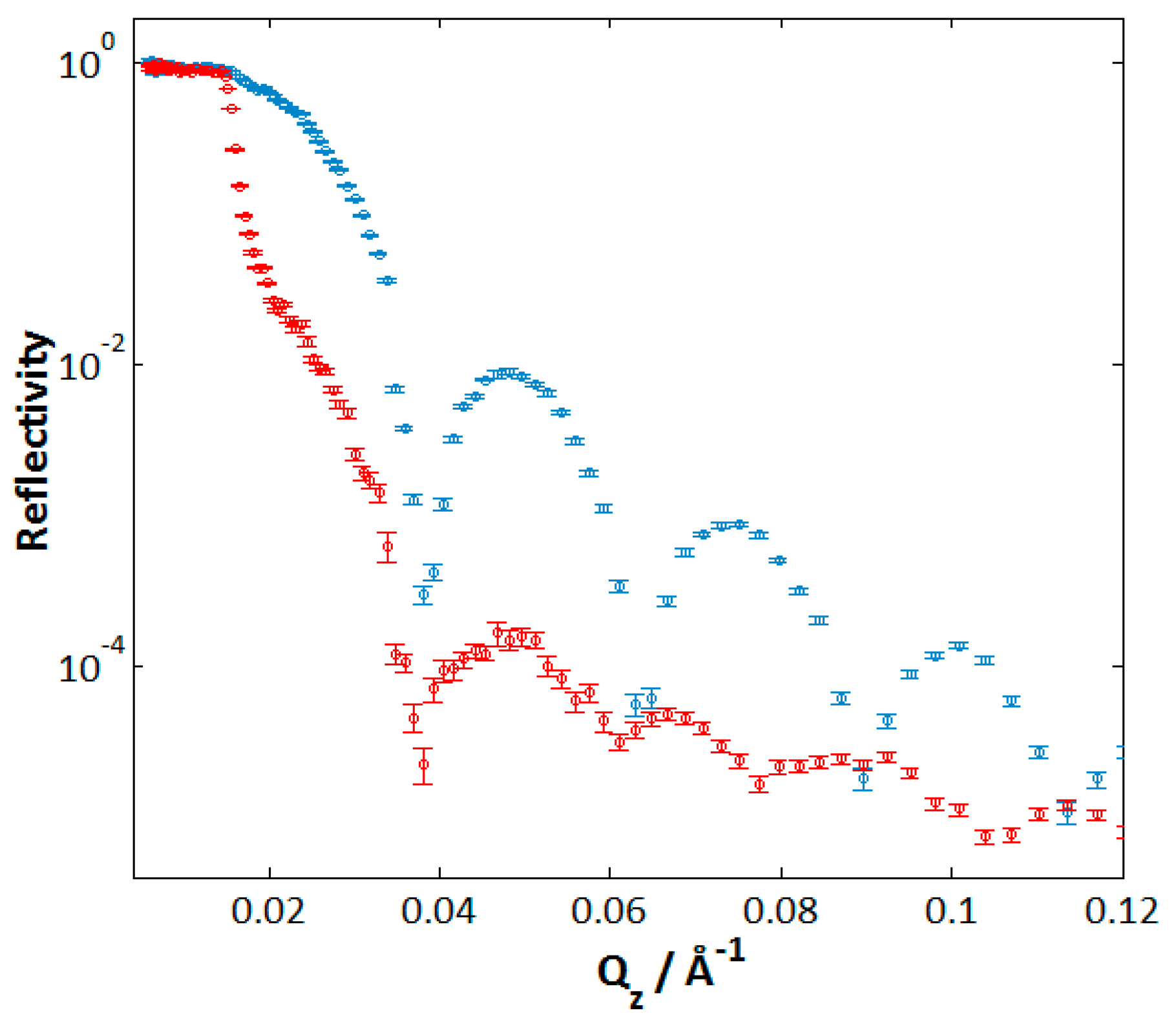
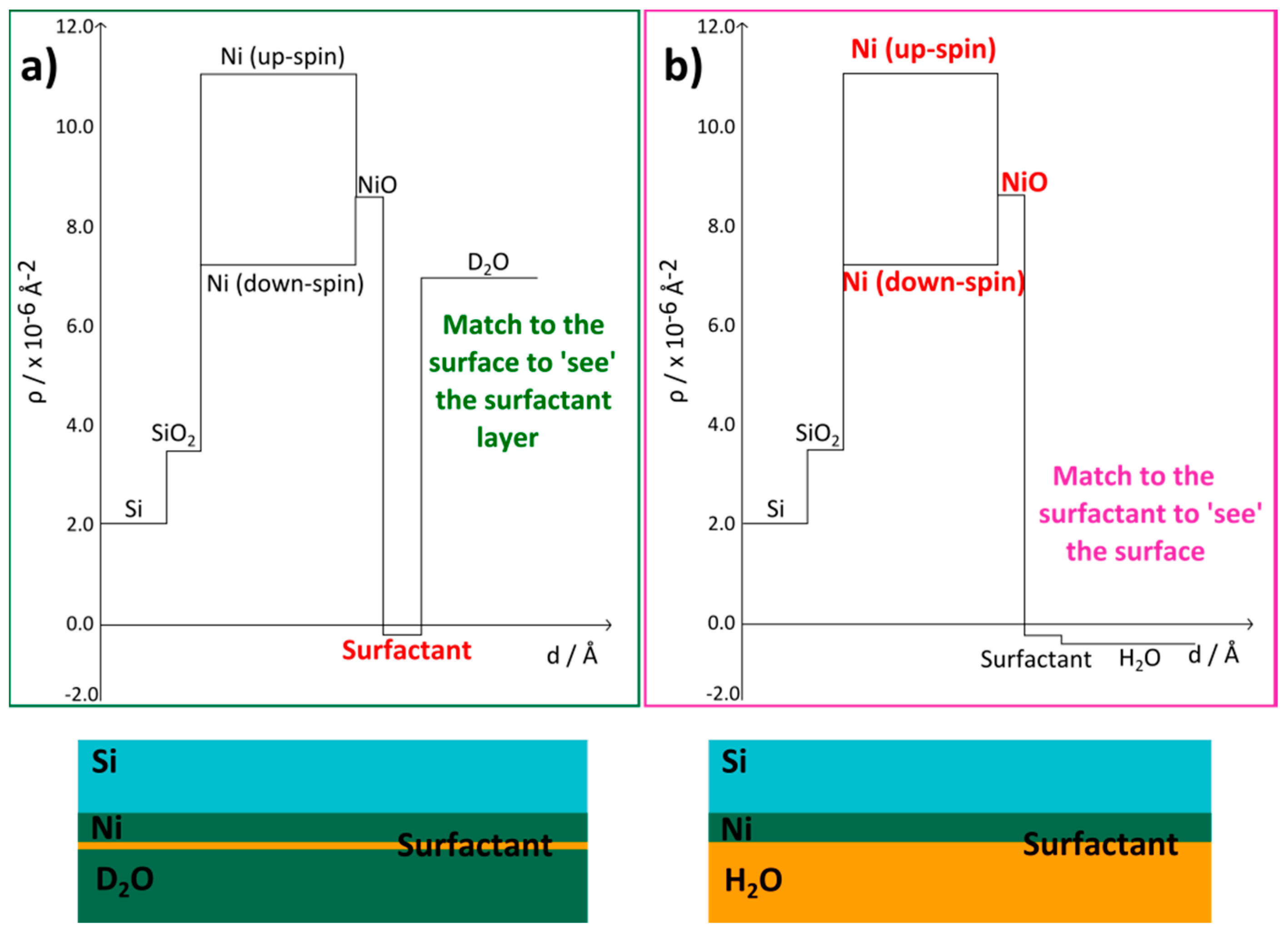
6.4. Nickel
6.5. Iron and Steel
6.6. Copper
6.7. Tantalum
7. Corrosion Related NR Work
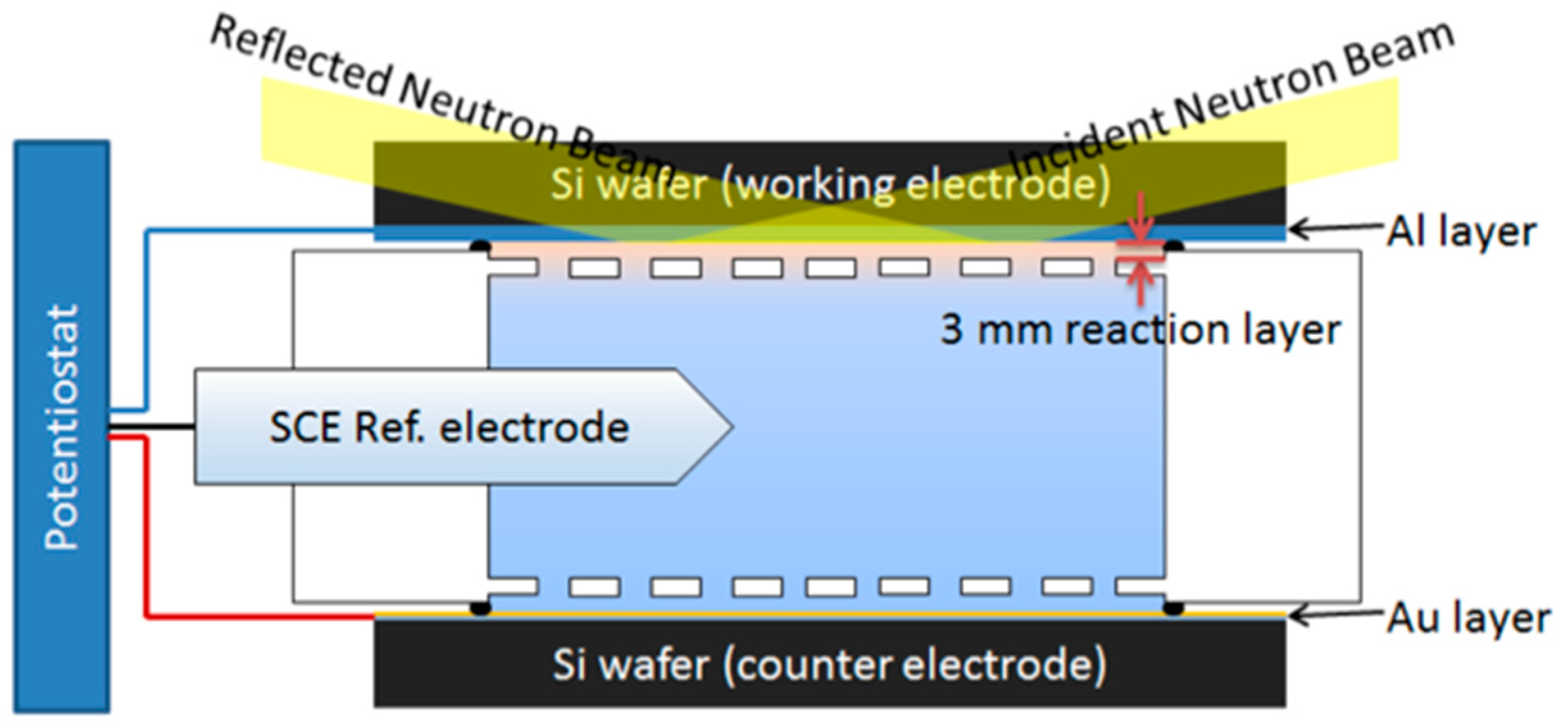
7.1. Electrochemical
7.2. Electroactive Polymer Films
7.3. Silane Barrier Films
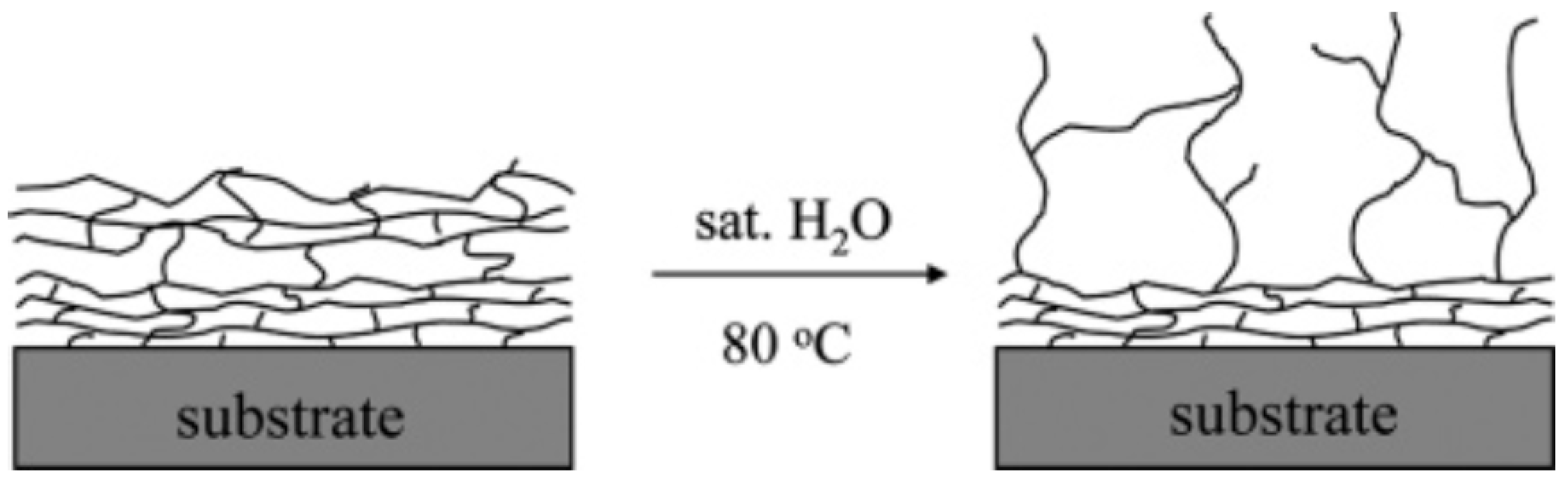
7.4. Other Corrosion-Inhibiting Coating Studies
7.5. Battery Material Studies
7.6. Ionic Liquids
8. Conclusions
Acknowledgments
Conflicts of Interest
References
- Noël, J.J. Oxide films and corrosion. In Neutron Reflectometry—A Probe for Materials Surfaces; International Atomic Energy Agency: Vienna, Austria, 2004; pp. 79–84. [Google Scholar]
- Mills, A.; Hazafy, D. UV-activated photocatalyst films and inks for cleaning tarnished metals. Chem. Commun. 2012, 48, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Scully, J.C. The Fundamentals of Corrosion, 3rd ed.; Pergamon Press: Oxford, UK, 1990. [Google Scholar]
- Schweitzer, P.A. Corrosion Engineering Handbook, 2nd ed.; Marcel Dekker, Inc.: New York, NY, USA, 1996. [Google Scholar]
- Dariva, C.G.; Galio, A.F. Corrosion inhibitors-principles, mechanisms and applications. In Developments in Corrosion Protection; Aliofkhazraei, M., Ed.; InTech: West Palm Beach, FL, USA, 2014. [Google Scholar]
- Anbarasi, C.M.; Rajendran, S.; Pandiarajan, M.; Krishnaveni, A. An encounter with corrosion inhibitors. Eur. Chem. Bull. 2013, 2, 197–207. [Google Scholar]
- Nii, K. On the dissolution behaviour of NiO. Corros. Sci. 1970, 10, 571–583. [Google Scholar]
- Khalifa, D.R.; Abdallah, S.M. Corrosion inhibition of some organic copmounds on low carbon steel in hydrochloric acid solution. Port. Electrochim. Acta 2011, 29, 47–56. [Google Scholar] [CrossRef]
- Kuznetsov, Y.I.; Mercer, A.D.; Thomas, J.G.N. Organic Inhibitors of Corrosion of Metals; Springer: New York, NY, USA, 1996. [Google Scholar]
- Welbourn, R.J.L.; Truscott, C.L.; Skoda, M.W.A.; Zarbakhsh, A.; Clarke, S.M. Corrosion and inhibition of copper in hydrocarbon solution on a molecular level investigated using neutron reflectometry and XPS. Corros. Sci. 2017, 115, 68–77. [Google Scholar] [CrossRef]
- Yilmazbayhan, A.; Motta, A.T.; Comstock, R.J.; Sabol, G.P.; Lai, B.; Cai, Z. Structure of zirconium alloy oxides formed in pure water studied with synchrotron radiation and optical microscopy: Relation to corrosion rate. J. Nucl. Mater. 2004, 324, 6–22. [Google Scholar] [CrossRef]
- Kelly, R.G.; Scully, J.R.; Shoesmith, D.W.; Buchheit, R.G. Electrochemical Techniques in Corrosion Science and Engineering; Marcel Dekker, Inc.: New York, NY, USA, 2003. [Google Scholar]
- Hayfield, P. Ellipsometry in corrosion technology. In Advances in Corrosion Science and Technology; Fontana, M.G., Staehle, R.W., Eds.; Springer: New York, NY, USA, 1972; pp. 43–113. [Google Scholar]
- Takahashi, Y.; Matsubara, E.; Suzuki, S.; Okamoto, Y.; Komatsu, T.; Konishi, H.; Mizuki, J.; Waseda, Y. In-situ X-ray diffraction of corrosion products formed on iron surfaces. Mater. Trans. 2005, 46, 637–642. [Google Scholar] [CrossRef]
- Lelièvre, G.; Fruchart, D.; Convert, P.; Lefèvre-Joud, F. Characterisation by neutron diffraction in high temperature pressurised water of the surface corrosion and hydrogen embrittlement of zircaloy-4. J. Alloys Compd. 2002, 347, 288–294. [Google Scholar] [CrossRef]
- Kobe, B.A.; Ramamurthy, S.; Biesinger, M.C.; McIntyre, N.S.; Brennenstühl, A.M. XPS imaging investigations of pitting corrosion mechanisms in Inconel 600. Surf. Interface Anal. 2005, 37, 478–494. [Google Scholar] [CrossRef]
- Miller, K.L. Neutron Reflectivity of Aqueous Mineral Amd Metal Oxide Interfaces. Ph.D. Thesis, University of Cambridge, Cambridge, UK, 2014. [Google Scholar]
- Sánchez, J.; Fullea, J.; Andrade, C.; Gaitero, J.J.; Porro, A. AFM study of the early corrosion of a high strength steel in a diluted sodium chloride solution. Corros. Sci. 2008, 50, 1820–1824. [Google Scholar] [CrossRef]
- Morsch, S.; Lyon, S.; Smith, S.D.; Gibbon, S.R. Mapping water uptake in an epoxy-phenolic coating. Prog. Org. Coat. 2015, 86, 173–180. [Google Scholar] [CrossRef]
- Speller, F.N.; Kendall, V.V. A new method of measuring corrosion in water. Ind. Eng. Chem. 1923, 15, 134–139. [Google Scholar] [CrossRef]
- Amin, M.A. Weight loss, polarisation, electrochemical impedance spectroscopy, SEM and EDX studies of the corrosion inhibition of copper in aerated NaCl solutions. J. Appl. Electrochem. 2006, 36, 215–226. [Google Scholar] [CrossRef]
- Lützenkirchen-Hecht, D.; Strehblow, H.-H. Synchtron methods for corrosion research. In Analytical Methods in Corrosion Science and Engineering; Marcus, P., Mansfeld, F.B., Eds.; CRC Press: New York, NY, USA, 2005; pp. 169–236. [Google Scholar]
- Chung, S.C.; Lin, A.S.; Chang, J.R.; Shih, H.C. EXAFS study of atmospheric corrosion products on zinc at the initial stage. Corros. Sci. 2000, 42, 1599–1610. [Google Scholar] [CrossRef]
- Lima, E.; Bosch, P.; Lara, V.; Villarreal, E.; Piña, C.; Torres, G.; Martín, S.; León, B. Metal corrosion in bones implanted with Zinalco—A SAXS and NMR study. J. Biomed. Mater. Res. B 2006, 76, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Song, C.H.; Choi, Y.; Shin, E.H.; Seong, B.S.; Han, Y.S. Analysis of organic-coated steels by using small-angle neutron scattering. Phys. Met. Metallogr. 2014, 115, 1333–1337. [Google Scholar] [CrossRef]
- Penfold, J. Principles of reflectometry with reactors and pulsed sources. In Neutron Reflectometry—A Probe for Materials Surfaces; International Atomic Energy Agency: Vienna, Austria, 2004; pp. 29–44. [Google Scholar]
- Penfold, J.; Thomas, R.K. The application of the specular reflection of neutrons to the study of surfaces and interfaces. J. Phys. Condens. Mater. 1990, 2, 1369–1412. [Google Scholar] [CrossRef]
- Lu, J.R.; Marrocco, A.; Su, T.J.; Thomas, R.K.; Penfold, J. Adsorption of dodecyl sulfate surfactants with monovalent metal counterions at the air-water interface studied by neutron reflection and surface tension. J. Colloid Interface Sci. 1993, 158, 303–316. [Google Scholar] [CrossRef]
- Li, Z.X.; Dong, C.C.; Thomas, R.K. Neutron reflectivity studies of the surface excess of gemini surfactants at the air-water interface. Langmuir 1999, 15, 4392–4396. [Google Scholar] [CrossRef]
- Eastoe, J.; Dalton, J.S. Dynamic surface tension and adsorption mechanisms of surfactants at the air-water interface. Adv. Colloid Interface. Sci. 2000, 85, 103–144. [Google Scholar] [CrossRef]
- Browning, K.L.; Griffin, L.R.; Gutfreund, P.; Barker, R.D.; Clifton, L.A.; Hughes, A.; Clarke, S.M. Specular neutron reflection at the mica/water interface-irreversible adsorption of a cationic dichain surfactant. J. Appl. Cryst. 2014, 47, 1638–1646. [Google Scholar] [CrossRef]
- Stocker, I.N.; Miller, K.L.; Lee, S.Y.; Welbourn, R.J.L.; Mannion, A.R.; Collins, I.R.; Webb, K.J.; Wildes, A.; Kinane, C.J.; Clarke, S.M. Neutron reflection at the calcite-water interface. Prog. Colloid Polym. Sci. 2012, 139, 91–99. [Google Scholar]
- Sferrazza, M.; Jones, R.A.L.; Penfold, J.; Bucknall, D.B.; Webster, J.R.P. Neutron reflectivity studies of the structure of polymer/polymer and polymer/substrate interfaces at the nanometer level. J. Mater. Chem. 2000, 10, 127–132. [Google Scholar] [CrossRef]
- Speranza, F.; Pilkington, G.A.; Dane, T.G.; Cresswell, P.T.; Li, P.; Jacobs, R.M.J.; Arnold, T.; Bouchenoire, L.; Thomas, R.K.; Briscoe, W.H. Quiescent bilayers at the mica-water interface. Soft Matter 2013, 9, 7028–7041. [Google Scholar] [CrossRef]
- Griffin, L.R.; Browning, K.L.; Truscott, C.L.; Clifton, L.A.; Clarke, S.M. Complete bilayer adsorption of C16TAB on the surface of mica using neutron reflection. J. Phys. Chem. B 2015, 119, 6457–6461. [Google Scholar] [CrossRef] [PubMed]
- Kreuzer, M.; Kaltofen, T.; Steitz, R.; Zehnder, B.H.; Dahint, R. Pressure cell for investigations of solid-liquid interfaces by neutron reflectivity. Rev. Sci. Instrum. 2011, 82, 023902. [Google Scholar] [CrossRef] [PubMed]
- Jeworrek, C.; Steitz, R.; Czeslik, C.; Winter, R. High pressure cell for neutron reflectivity measurements up to 2500 bar. Rev. Sci. Instrum. 2011, 82, 025106. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, J.R.; Rother, G.; Browning, J.F.; Ankner, J.F.; Banuelos, J.L.; Anovitz, L.M.; Wesolowski, D.J.; Cole, D.R. High-pressure cell for neutron reflectometry of supercritical and subcritical fluids at solid interfaces. Rev. Sci. Instrum. 2012, 83, 045108. [Google Scholar] [CrossRef] [PubMed]
- Welbourn, R.J.L. Adsorbed Layers under Challenging Conditions at the Solid-Liquid Interface. Ph.D. Thesis, University of Cambridge, Cambridge, UK, 2016. [Google Scholar]
- Baker, S.M.; Smith, G.; Pynn, R.; Butler, P.; Hayter, J.; Hamilton, W.; Magid, L. Shear cell for the study of liquid-solid interfaces by neutron scattering. Rev. Sci. Instrum. 1994, 65, 412–416. [Google Scholar] [CrossRef]
- Tun, Z.; Noël, J.J.; Shoesmith, D.W. Electrochemical modifications on the surface of a Ti film. Physica B 1998, 241–243, 1107–1109. [Google Scholar] [CrossRef]
- Burgess, I.; Zamlynny, V.; Szymanski, G.; Schwan, A.L.; Faragher, R.J.; Lipkowski, J.; Majewski, J.; Satija, S. Neutron reflectivity studies of field driven transformations in a monolayer of 4-pentadecyl pyridine at Au electrode surfaces. J. Electroanal. Chem. 2003, 550–551, 187–199. [Google Scholar] [CrossRef]
- Zamlynny, V.; Burgess, I.; Szymanski, G.; Lipkowski, J.; Majewski, J.; Smith, G.; Satija, S.; Ivkov, R. Electrochemical and neutron reflectivity studies of spontaneously formed amphiphilic surfactant bilayers at the gold-solution interface. Langmuir 2000, 16, 9861–9870. [Google Scholar] [CrossRef]
- Burgess, I.; Li, M.; Horswell, S.L.; Szymanski, G.; Lipkowski, J.; Majewski, J.; Satija, S. Electric field-driven transformations of a supported model biological membrane—An electrochemical and neutron reflectivity study. Biophys. J. 2004, 86, 1763–1776. [Google Scholar] [CrossRef]
- Richardson, R.M.; Swann, M.J.; Hillman, A.R.; Roser, S.J. In situ neutron reflectivity studies of electroactive films. Faraday Discuss. 1992, 94, 295–306. [Google Scholar] [CrossRef]
- Demkowicz, M.J.; Majewski, J.; Liss, K.-D. Probing interfaces in metals using neutron reflectometry. Metals 2016, 6, 20–37. [Google Scholar] [CrossRef]
- Felici, R.; Penfold, J.; Ward, R.C.; Williams, W.G. A polarised neutron reflectometer for studying surface magnetism. Appl. Phys. A 1988, 45, 169–174. [Google Scholar] [CrossRef]
- Jones, R.A.L.; Fletcher, P.; Thomas, R.K.; Roser, S.; Dickinson, E.; Bucknall, D.G.; Penfold, J.; Webster, J.R.P.; Zarbaksh, A.; Richardson, R.M.; et al. ICANS-SII and ESS-PM4; Bercher, R., Ed.; Paul Scherrer Institute: Villigen, Switzerland, 1995; p. 440. [Google Scholar]
- Webster, J.; Holt, S.; Dalgliesh, R. INTER: The chemical interfaces reflectometer on target station 2 at ISIS. Physica B 2006, 385–386, 1164–1166. [Google Scholar] [CrossRef]
- Charlton, T.R.; Coleman, R.L.S.; Dalgliesh, R.M.; Kinane, C.J.; Neylon, C.; Langridge, S.; Plomp, J.; Webb, N.G.J.; Webster, J.R.P. Advances in neutron reflectometry at ISIS. Neutron News 2011, 22, 15–18. [Google Scholar] [CrossRef]
- Cubitt, R.; Fragneto, G. D17: The new reflectometer at the ILL. Appl. Phys. A 2002, 74, S329–S331. [Google Scholar] [CrossRef]
- Campbell, R.A.; Wacklin, H.P.; Sutton, I.; Cubitt, R.; Fragneto, G. FIGARO: The new horizontal neutron reflectometer at the ILL. Eur. Phys. J. Phys. 2011, 126, 107–129. [Google Scholar] [CrossRef]
- Paul, A.; Krist, T.; Teichert, A.; Steitz, R. Specular and off-specular scattering with polarisation and polarisation analysis on reflectometer V6 at BER II, HZB. Physica B 2011, 406, 1598–1606. [Google Scholar] [CrossRef]
- Gupta, M.; Gutberlet, T.; Stahn, J.; Keller, P.; Clemens, D. AMOR—The time-of-flight neutron reflectometer at SINQ/PSI. Pramana-J. Phys. 2004, 63, 57–63. [Google Scholar] [CrossRef]
- Pan, G.; Yim, H.; Kent, M.S.; Majewski, J.; Schaefer, D.W. Neutron reflectivity investigation of bis-amino silane films. J. Adhes. Sci. Technol. 2003, 17, 2175–2189. [Google Scholar] [CrossRef]
- Griffin, L.R.; Browning, K.L.; Clarke, S.M.; Smith, A.M.; Perkin, S.; Skoda, M.W.A.; Norman, S.E. Direct measurements of ionic liquid layering at a single mica-liquid interface and in nano-films between two mica-liquid interfaces. Phys. Chem. Chem. Phys. 2017, 19, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Griffin, L.R.; Browning, K.L.; Lee, S.Y.; Skoda, M.W.A.; Rogers, S.E.; Clarke, S.M. Multilayering of calcium aerosol-OT at the mica/water interface studied with neutron reflection: Formation of a condensed lamellar phase at the CMC. Langmuir 2016, 32, 13054–13064. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A. Co-refinement of multiple-contrast neutron/X-ray reflectivity data using MOTOFIT. J. Appl. Cryst. 2006, 39, 273–276. [Google Scholar] [CrossRef]
- Björck, M.; Andersson, G. GenX: An extensible X-ray reflectivity refinement program utilising differential evolution. J. Appl. Cryst. 2007, 40, 1174–1178. [Google Scholar] [CrossRef]
- Majkrzak, C.F.; Berk, N.F. Exact determination of the phase in neutron reflectometry. Phys. Rev. B 1995, 52, 10827–10830. [Google Scholar] [CrossRef]
- Bowers, J.; Zarbakhsh, A.; Webster, J.R.P.; Hutchings, L.R.; Richards, R.W. Neutron reflectivity studies at liquid-liquid interfaces: Methodology and analysis. Langmuir 2001, 17, 140–145. [Google Scholar] [CrossRef]
- Zarbakhsh, A.; Querol, A.; Bowers, J.; Yaseen, M.; Lu, J.R.; Webster, J.R.P. Neutron reflection from the liquid-liquid interface: Adsorption of hexadecylphosphorylcholine to the hexadecane-aqueous solution interface. Langmuir 2005, 21, 11704–11709. [Google Scholar] [CrossRef] [PubMed]
- Wildes, A.R. Neutron Reflectometry—A Probe for Materials Surfaces; International Atomic Energy Agency: Vienna, Austria, 2004; pp. 145–160. [Google Scholar]
- Ankner, J.F.; Felcher, G.P. Polarised-neutron reflectometry. J. Magn. Magn. Mater. 1999, 200, 741–754. [Google Scholar] [CrossRef]
- Wood, M.H.; Welbourn, R.J.L.; Charlton, T.; Zarbakhsh, A.; Casford, M.T.; Clarke, S.M. Hexadecylamine adsorption at the iron oxide-oil interface. Langmuir 2013, 29, 13735–13742. [Google Scholar] [CrossRef] [PubMed]
- Rennie, A.R. Neutron reflection studies of solid/liquid interfaces. In Neutron Reflectometry—A Probe for Materials Surfaces; International Atomic Energy Agency: Vienna, Austria, 2004; pp. 85–94. [Google Scholar]
- Dong, X.; Argekar, S.; Wang, P.; Schaefer, D.W. In situ evolution of trivalent chromium process passive film on Al in a corrosive aqueous environment. Appl. Mater. Int. 2011, 3, 4206–4214. [Google Scholar] [CrossRef] [PubMed]
- Tun, Z.; Noël, J.J.; Shoesmith, D.W. Electrochemical modification of the passive oxide layer on a Ti film observed by in situ neutron reflectometry. J. Electrochem. Soc. 1999, 146, 988–994. [Google Scholar] [CrossRef]
- Hellsing, M.S.; Rennie, A.R.; Hughes, A.V. Effect of concentration and addition of ions on the adsorption of aerosol-OT to sapphire. Langmuir 2010, 26, 14567–14573. [Google Scholar] [CrossRef] [PubMed]
- Hellsing, M.S.; Rennie, A.R.; Hughes, A.V. Adsorption of aerosol-OT to sapphire: Lamellar structures studied with neutrons. Langmuir 2011, 27, 4669–4678. [Google Scholar] [CrossRef] [PubMed]
- Welbourn, R.J.L.; Lee, S.Y.; Gutfreund, P.; Hughes, A.; Zarbakhsh, A.; Clarke, S.M. Neutron reflection study of the adsorption of the phosphate surfactant NaDEHP onto alumina from water. Langmuir 2015, 31, 3377–3384. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Welbourn, R.; Clarke, S.M.; Skoda, M.W.A.; Clifton, L.; Zarbakhsh, A. Adsorption of sodium hexanoate on α-alumina. J. Colloid Int. Sci. 2013, 407, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Campana, M.; Teichert, A.; Clarke, S.; Steitz, R.; Webster, J.R.P.; Zarbakhsh, A. Surfactant adsorption at the metal-oil interface. Langmuir 2011, 27, 6085–6090. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.H.; Casford, M.T.; Steitz, R.; Zarbakhsh, A.; Welbourn, R.J.L.; Clarke, S.M. Comparative adsorption of saturated and unsaturated fatty acids at the iron oxide/oiil interface. Langmuir 2016, 32, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.H.; Welbourn, R.J.L.; Zarbakhsh, A.; Gutfreund, P.; Clarke, S.M. Polarised neutron reflectometry of nickel corrosion inhibitors. Langmuir 2015, 31, 7062–7072. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.H.; Browning, K.L.; Barker, R.D.; Clarke, S.M. Using neutron reflectometry to discern the structure of fibrinogen adsorption at the stainless steel/aqueous interface. J. Phys. Chem. B 2016, 120, 5405–5416. [Google Scholar] [CrossRef] [PubMed]
- Noël, J.J.; Shoesmith, D.W.; Tun, Z. Anodic oxide growth and hydrogen absorption on Zr in neutral aqueous solution: A comparison to Ti. J. Electrochem. Soc. 2008, 155, C444–C454. [Google Scholar] [CrossRef]
- Mitchell, W.J.; Burn, P.L.; Thomas, R.K.; Fragneto, G. Probing the polymer-electrode interface using neutron reflection. Appl. Phys. Lett. 2003, 82, 2724–2726. [Google Scholar] [CrossRef]
- Noël, J.J.; Jensen, H.L.; Tun, Z.; Shoesmith, D.W. Electrochemical modification of the passive oxide layer on Zr-2.5Nb observed by in situ neutron reflectometry. Electrochem. Solid State Lett. 2000, 3, 473–476. [Google Scholar] [CrossRef]
- Rieker, T.; Hubbard, P.; Majewski, J.; Smith, G.; Moody, N. Neutron reflectivity investigation of the effects of harsh environments on Ta2N thin films. Thin Solid Films 1999, 346, 116–119. [Google Scholar] [CrossRef]
- Kalisvaart, P.; Luber, E.; Fritzsche, H.; Mitlin, D. Effect of alloying magnesium with chromium and vanadium on hydrogenation kinetics studied with neutron reflectometry. Chem. Commun. 2011, 47, 4294–4296. [Google Scholar] [CrossRef] [PubMed]
- Watkins, E.B.; Majewski, J.; Baldwin, J.K.; Chen, Y.; Li, N.; Hoagland, R.G.; Yadav, S.K.; Liu, X.Y.; Beyerlein, I.J.; Mara, N.A. Neutron reflectometry investigations of interfacial structures of Ti/TiN layers deposited by magnetron sputtering. Thin Solid Films 2016, 616, 399–407. [Google Scholar] [CrossRef]
- Mayes, M.A.; Jagadamma, S.; Ambaye, H.; Petridis, L.; Lauter, V. Neutron reflectometry reveals the internal structure of organic compounds deposited on aluminium oxide. Geoderma 2013, 182–188. [Google Scholar] [CrossRef]
- Owejan, J.E.; Owejan, J.P.; DeCaluwe, S.C.; Dura, J.A. Solid electrolyte interphase in Li-ion batteries: Evolving structures measured in situ by neutron reflectometry. Chem. Mater. 2012, 24, 2133–2140. [Google Scholar] [CrossRef]
- Li, N.; Thomas, R.K.; Rennie, A.R. Adsorption of non-ionic surfactants to the sapphire/solution interface-effects of temperature and pH. J. Colloid Int. Sci. 2012, 369, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Li, N.N.; Thomas, R.K.; Rennie, A.R. Neutron reflectometry of anionic surfactants on sapphire: A strong maximum in the adsorption near the critical micelle concentration. J. Colloid Int. Sci. 2016, 471, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Junghans, A.; Chellappa, R.; Wang, P.; Majewski, J.; Luciano, G.; Marcelli, R.; Proietti, E. Neutron reflectometry studies of aluminium-saline water interface under hydrostatic pressure. Corros. Sci 2015, 90, 101–106. [Google Scholar] [CrossRef]
- Wang, P.; Dong, X.; Schaefer, D.W. Structure and water-barrier properties of vanadate-based corrosion inhibitor films. Corros. Sci. 2010, 52, 943–949. [Google Scholar] [CrossRef]
- Hu, N.; Dong, X.; He, X.; Browning, J.F.; Schaefer, D.W. Effect of sealing on the morphology of anodised aluminium oxide. Corros. Sci. 2015, 97, 17–24. [Google Scholar] [CrossRef]
- Armstrong, N.R.; Quinn, R.K. Auger and X-ray photoelectron spectroscopic and electrochemical characterisation of titanium thin film electrodes. Surf. Sci. 1977, 67, 451–468. [Google Scholar] [CrossRef]
- Wiesler, D.G.; Majkrzak, C.F. Neutron reflectometry studies of surface oxidation. Physica B 1994, 198, 181–186. [Google Scholar] [CrossRef]
- Wiesler, D.G.; Majkrzak, C.F. Growth and dissolution of protective oxide films on titanium: An in situ neutron reflectivity study. Manag. Res. Soc. Symp. Proc. 1995, 376, 247–257. [Google Scholar] [CrossRef]
- Noël, J.J.; Smith, J.M.; Vezvaie, M.; Tun, Z. In Situ Neutron Reflectometry Study of Hydrogen Absorption By Zirconium during Cathodic Polarization. In Proceedings of the 19th Pacific Basin Nuclear Conference, Vancouver, BC, Canada, 24–28 August 2014; p. 2821. [Google Scholar]
- Singh, S.; Basu, S.; Gupta, M.; Majkrzak, C.F.; Kienzle, P.A. Growth kinetics of intermetallic alloy phase at the interfaces of a Ni/Al multilayer using polarised neutron and X-ray reflectometry. Phys. Rev. B 2010, 81, 235413. [Google Scholar] [CrossRef]
- Singh, S.; Poswal, A.K.K.; Ghosh, S.K.K.; Basu, S. Scattering length density profile of Ni film under controlled corrosion: A study in neutron reflectometry. Pramana J. Phys. 2008, 71, 1097–1101. [Google Scholar] [CrossRef]
- Singh, S.; Basu, S. Microscopic characterisation of corrosion morphology: A study in specular and diffuse neutron reflectivity. Surf. Sci. 2006, 600, 493–496. [Google Scholar] [CrossRef]
- Singh, S.; Basu, S.; Poswal, A.K.K.; Tokas, R.B.B.; Ghosh, S.K.K. Electrochemically controlled pitting corrosion in Ni film: A study of AFM and neutron reflectometry. Corros. Sci. 2009, 51, 575–580. [Google Scholar] [CrossRef]
- Morales-Gil, P.; Walczak, M.S.; Cottis, R.A.; Romero, J.M.; Lindsay, R. Corrosion inhibitor binding in an acidic medium: Interaction of 2-mercaptobenizmidazole with carbon-steel in hydrochloric acid. Corros. Sci. 2014, 85, 109–114. [Google Scholar] [CrossRef]
- Molchan, I.S.; Thompson, G.E.; Lindsay, R.; Skeldon, P.; Likodimos, V.; Romanos, G.E.; Falaras, P.; Adamova, G.; Iliev, B.; Schubert, T.J.S. Corrosion behaviour of mild steel in 1-alkyl-3-methylimidazolium tricyanomethanide ionic liquids for CO2 capture applications. RSC Adv. 2014, 4, 5300–5311. [Google Scholar] [CrossRef]
- Saville, P.M.; Gonsalves, M.; Hillman, A.R.; Cubitt, R. Dynamic neutron reflectivity measurements during redox switching of nickel hydroxide films. J. Phys. Chem. B 1997, 101, 1–4. [Google Scholar] [CrossRef]
- Metelev, S.V.; Pleshanov, N.K.; Menelle, A.; Pusenkov, V.M.; Schebetov, A.F.; Soroko, Z.N.; Ul’yanov, V.A. The study of oxidation of thin metal films by neutron reflectometry. Physica B 2001, 297, 122–125. [Google Scholar] [CrossRef]
- Campana, M. Structural Studies of Surfactants at Interfaces. Ph.D. Thesis, University of London, London, UK, 2012. [Google Scholar]
- Wood, M.H. Adsorption at the Solid/Liquid Interface. Ph.D. Thesis, University of Cambridge, Cambridge, UK, 2015. [Google Scholar]
- Kruger, J.; Krebs, L.A.; Long, G.G.; Anker, J.F.; Majkrzak, C.F. Passivity of metals and alloys and its breakdown—New results from new non-electrochemical techniques. Mater. Sci. Forum 1995, 185–188, 367–376. [Google Scholar] [CrossRef]
- Feng, J. Characterization of Native Oxide and Passive Film on Iron and Iron-Chromium Alloy. Ph.D. Thesis, University of Cincinnati, Cincinnati, OH, USA, 2014. [Google Scholar]
- Wood, M.H.; Wood, T.J.; Poon, J.; Welbourn, R.J.L.; Madden, D.; Clarke, S.M. An X-ray and neutron reflectometry study of iron corrison in seawater at the angstrom level. 2017; in preparation. [Google Scholar]
- Jerliu, B.; Hüger, E.; Dörrer, L.; Seidlhofer, B.K.; Steitz, R.; Oberst, V.; Geckle, U.; Bruns, M.; Schmidt, H. Volume expansion during lithiation of amorphous silicon thin film electrodes studied by in-operando neutron reflectometry. J. Phys. Chem. C 2014, 118, 9395–9399. [Google Scholar] [CrossRef]
- Jerliu, B.; Dörrer, L.; Hüger, E.; Borchardt, G.; Steitz, R.; Geckle, U.; Oberst, V.; Bruns, M.; Schneider, O.; Schmidt, H. Neutron reflectometry studies of the lithiation of amorphous silicon electrodes in lithium-ion batteries. Phys. Chem. Chem. Phys. 2013, 15, 7777–7784. [Google Scholar] [CrossRef] [PubMed]
- Wagemaker, M.; Van de Krol, R.; Van Well, A.A. Nano-morphology of lithiated thin film TiO2 anatase probed with in situ neutron reflectometry. Physica B 2003, 336, 124–129. [Google Scholar] [CrossRef]
- Hirayama, M.; Yonemura, M.; Suzuki, K.; Torikai, N.; Smith, H.; Watkinsand, E.; Majewski, J.; Kanno, R. Surface characterisation of LiFePO4 epitaxial thin films by X-ray/neutron reflectometry. Electrochemistry 2010, 78, 413–415. [Google Scholar] [CrossRef]
- Browning, J.F.; Baggetto, L.; Jungjohann, K.L.; Wang, Y.; Tenhaeff, W.E.; Keum, J.K.; Wood, D.L.; Veith, G.M. In situ determination of the liquid/solid interface thickness and composition for the Li ion cathode LiMn1.5Ni0.5O4. Appl. Mater. Int. 2014, 6, 18569–18576. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.M.; Cubitt, R.; Dalgliesh, R.M.; Gadegaard, N.; Glidle, A.; Hillman, A.R.; Mortimer, R.J.; Ryder, K.S.; Smith, E.L. Dynamic in situ electrochemical neutron reflectivity measurements. J. Am. Chem. Soc. 2004, 126, 15362–15363. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.W.; Bailey, L.; Cubitt, R.; Gonsalves, M.; Glidle, A.; Hillman, A.R.; Vos, J.G.; Hogan, C.; Webster, J.R.P. A study of [Os(bipy)2(PVP)3.3(PS)6.7Cl]+ polymer film modified electrodes using neutron reflectivity. Phys. Chem. Chem. Phys. 1999, 1, 843–853. [Google Scholar] [CrossRef]
- Glidle, A.; Hillman, A.R.; Ryder, K.S.; Smith, E.L.; Cooper, J.; Gadegaard, N.; Webster, J.R.P.; Dalgliesh, R.; Cubitt, R. Use of neutron reflectivity to measure the dynamics of solvation and structural changes in polyvinylferrocene films during electrochemically controlled redox cycling. Langmuir 2009, 25, 4093–4103. [Google Scholar] [CrossRef] [PubMed]
- Lauw, Y.; Horne, M.D.; Rodopoulos, T.; Lockett, V.; Akgun, B.; Hamilton, W.A.; Nelson, A.R. Structure of [C4mpyr][NTf2] room-temperature ionic liquid at charged gold interfaces. Langmuir 2012, 28, 7374–7381. [Google Scholar] [CrossRef] [PubMed]
- Lauw, Y.; Rodopoulos, T.; Gross, M.; Nelson, A.; Gardner, R.; Horne, M.D. Electrochemical cell for neutron reflectometry studies of the structure of ionic liquids at electrified interface. Rev. Sci. Instrum. 2010, 81, 074101. [Google Scholar] [CrossRef] [PubMed]
- Itkis, D.M.; Velasco-Velez, J.J.; Knop-Gericke, A.; Vyalikh, A.; Avdeev, M.V.; Yashina, L.V. Probing operating electrochemical interfaces by photons and neutrons. ChemElectroChem 2015, 2, 1427–1445. [Google Scholar] [CrossRef]
- Wilson, R.W.; Cubitt, R.; Glidle, A.; Hillman, A.R.; Saville, P.M.; Vos, J.G. A neutron reflectivity study of [Os(bipy)2(PVP)10Cl]+ polymer film modified electrodes: Effect of pH and counterion. J. Electrochem. Soc. 1998, 145, 1454–1461. [Google Scholar] [CrossRef]
- Glidle, A.; Hadyoon, C.S.; Gadegaard, N.; Cooper, J.M.; Hillman, A.R.; Wilson, R.W.; Ryder, K.S.; Webster, J.R.P.; Cubitt, R. Evaluating the influence of deposition conditions on solvation of reactive conducting polymers with neutron reflectivity. J. Phys. Chem. B 2005, 109, 14335–14343. [Google Scholar] [CrossRef] [PubMed]
- Glidle, A.; Pearson, P.E.; Smith, E.L.; Cooper, J.M.; Cubitt, R.; Dalgliesh, R.M.; Hillman, A.R.; Ryder, K.S. Determining compositional profiles within conducting polymer films following reaction with vapour phase reagents. J. Phys. Chem. B 2007, 111, 4043–4053. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Watkins, E.; Ilavsky, J.; Metroke, T.L.; Wang, P.; Lee, B.; Schaefer, D.W. Water-barrier properties of mixed bis [trimethoxysilylpropyl] amine and vinyltriacetoxysilane films. J. Phys. Chem. B 2007, 111, 7041–7051. [Google Scholar] [CrossRef] [PubMed]
- Yim, H.; Kent, M.S.; Tallant, D.R.; Garcia, M.J.; Majewski, J. Hygrothermal Degradation of (3-glycidoxypropyl) trimethoxysilane films studied by neutron and X-ray reflectivity and attenuated total reflection infrared spectroscopy. Langmuir 2005, 21, 4382–4392. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Watkins, E.; Majewski, J.; Schaefer, D.W. Effect of thickness on the water-barrier properties of silane films. J. Phys. Chem. C 2007, 111, 15325–15330. [Google Scholar] [CrossRef]
- Payra, D.; Naito, M.; Fujii, Y.; Yamada, N.L.; Hiromoto, S.; Singh, A. Bioinspired adhesive polymer coatings for efficient and versatile corrosion resistance. RSC Adv. 2015, 5, 15977–159834. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, P.; Kohls, D.; Hamilton, W.A.; Schaefer, D.W. Water absorption and transport in bis-silane films. Phys. Chem. Chem. Phys. 2009, 11, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Schaefer, D.W. Salt exclusion in silane-laced epoxy coatings. Langmuir 2010, 26, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.; Schaefer, D.W. Morphology and water-barrier properties of silane films on aluminium and silicon. Thin Solid Films 2006, 503, 259–267. [Google Scholar] [CrossRef]
- Yim, H.; Kent, M.S.; Hall, J.S.; Benkoski, J.J.; Kramer, E.J. Probing the structure of organosilane films by solvent swelling and neutron and X-ray reflection. J. Phys. Chem. B 2002, 106, 2474–2481. [Google Scholar] [CrossRef]
- Lechenault, F.; Rountree, C.L.; Cousin, F.; Bouchaud, J.P.; Ponson, L.; Bouchaud, E. Damage of silicate glasses during stress corrosion. J. Phys. Conf. Ser. 2011, 319, 012005. [Google Scholar] [CrossRef]
- Barkhudarov, P.M.; Shah, P.B.; Watkins, E.B.; Doshi, D.A.; Brinker, C.J.; Majewski, J. Corrosion inhibition using superhydrophobic films. Corros. Sci. 2008, 50, 897–9902. [Google Scholar] [CrossRef]

| Atom | Scattering Length, bi/10−15 m | Atom | Scattering Length, bi/10−15 m |
|---|---|---|---|
| H | −3.74 | D | +6.67 |
| C | +6.65 | 6Li | +2.00 |
| O | +5.80 | 7Li | −2.22 |
| N | +9.36 | Al | +3.45 |
| Si | +4.15 | 58Ni | +14.40 |
| 54Fe | +4.20 | 60Ni | +2.80 |
| 56Fe | +9.94 | 61Ni | +7.60 |
| 57Fe | +2.30 | 62Ni | −8.70 |
| 58Fe | +15.70 | 64Ni | −0.37 |
| Species | SLD/10−6 Å−2 |
|---|---|
| H2O | −0.56 |
| D2O | +6.34 |
| H-Dodecane | −0.46 |
| Dodecane-d26 | +6.71 |
| Si | +2.07 |
| SiO2 | +3.63 |
| Fe (up-spin) | +12.99 |
| Fe (down-spin) | +3.05 |
| Fe (non-polarised) | +8.02 |
| Fe2O3 | +7.18 |
| Ni (up-spin) | +10.86 |
| Ni (down-spin) | +7.94 |
| Ni (non-polarised) | +9.41 |
| NiO | 8.66 |
| Ti | −1.91 |
| TiO2 | 2.63 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wood, M.H.; Clarke, S.M. Neutron Reflectometry for Studying Corrosion and Corrosion Inhibition. Metals 2017, 7, 304. https://doi.org/10.3390/met7080304
Wood MH, Clarke SM. Neutron Reflectometry for Studying Corrosion and Corrosion Inhibition. Metals. 2017; 7(8):304. https://doi.org/10.3390/met7080304
Chicago/Turabian StyleWood, Mary H., and Stuart M. Clarke. 2017. "Neutron Reflectometry for Studying Corrosion and Corrosion Inhibition" Metals 7, no. 8: 304. https://doi.org/10.3390/met7080304
APA StyleWood, M. H., & Clarke, S. M. (2017). Neutron Reflectometry for Studying Corrosion and Corrosion Inhibition. Metals, 7(8), 304. https://doi.org/10.3390/met7080304





