Fatigue Crack Growth Behavior of Austempered AISI 4140 Steel with Dissolved Hydrogen
Abstract
:1. Introduction
2. Experimental Procedure
2.1. Material
2.2. Heat Treatment and Tensile Testing
2.3. Pre-Cracking of CT Specimens
2.4. External Hydrogen Charging
2.5. Hydrogen Concentration Analysis
2.6. Fatigue Testing
2.7. Fracture Toughness Test
3. Results and Discussion
3.1. Microstructure
3.2. Mechanical Properties
3.3. Hydrogen Concentration
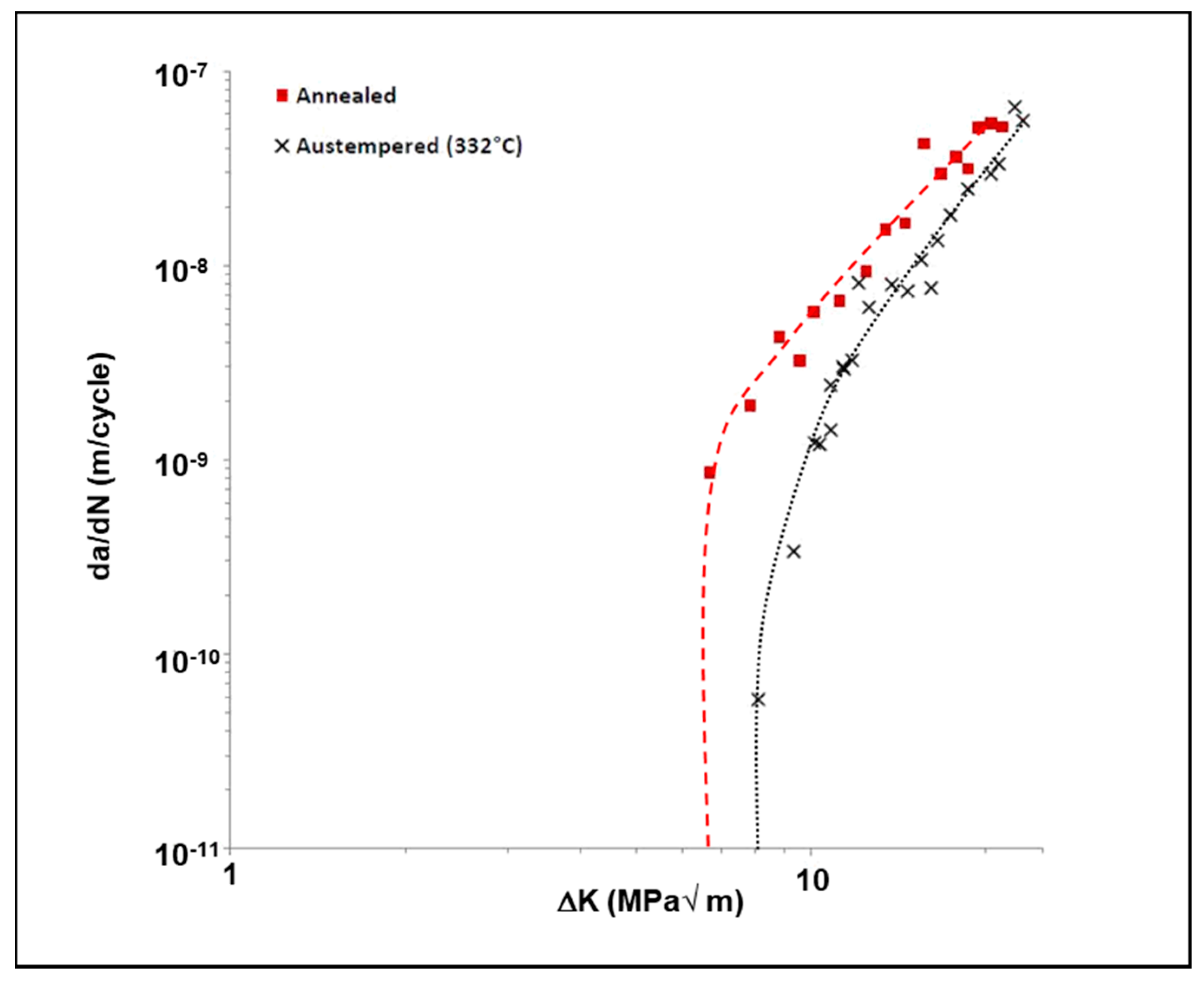
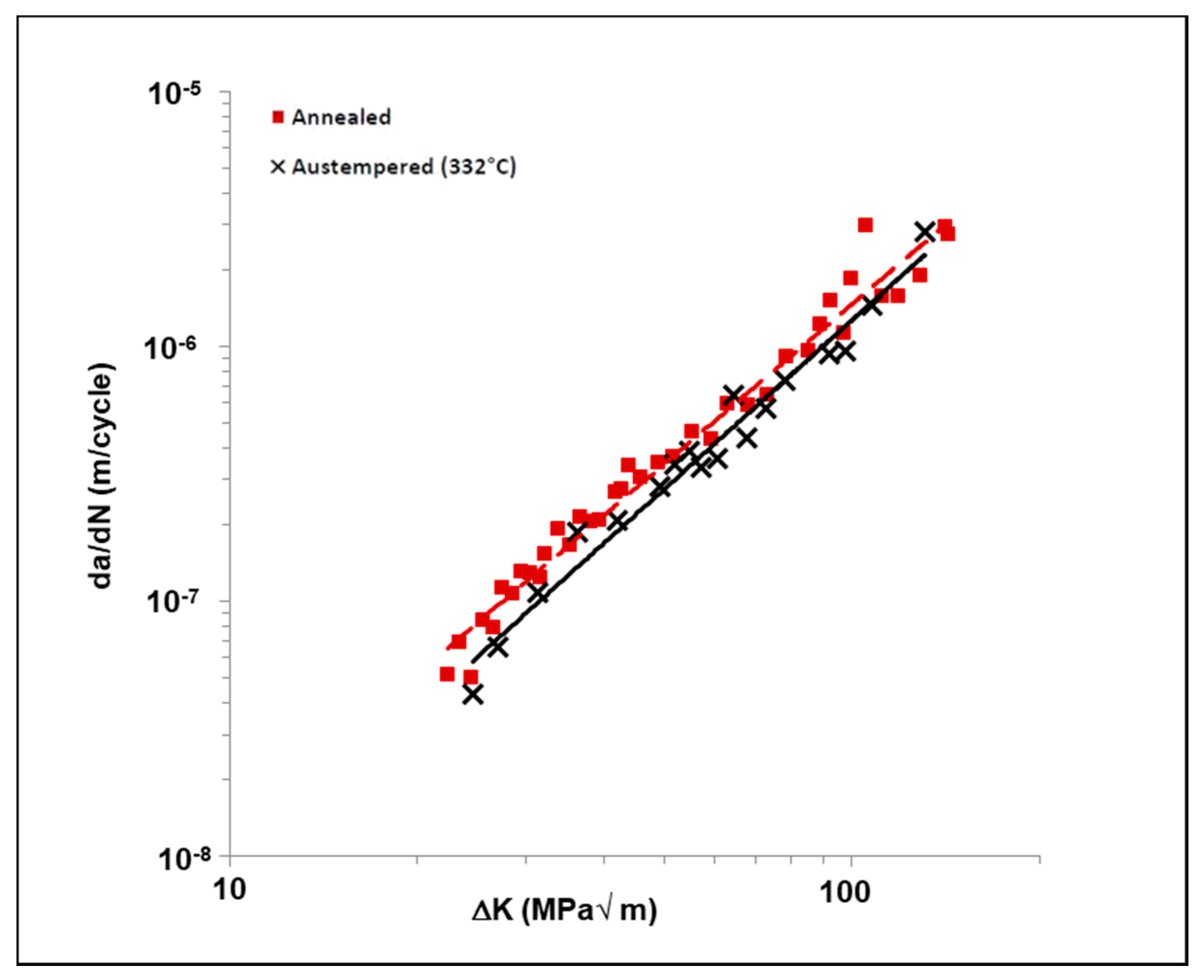
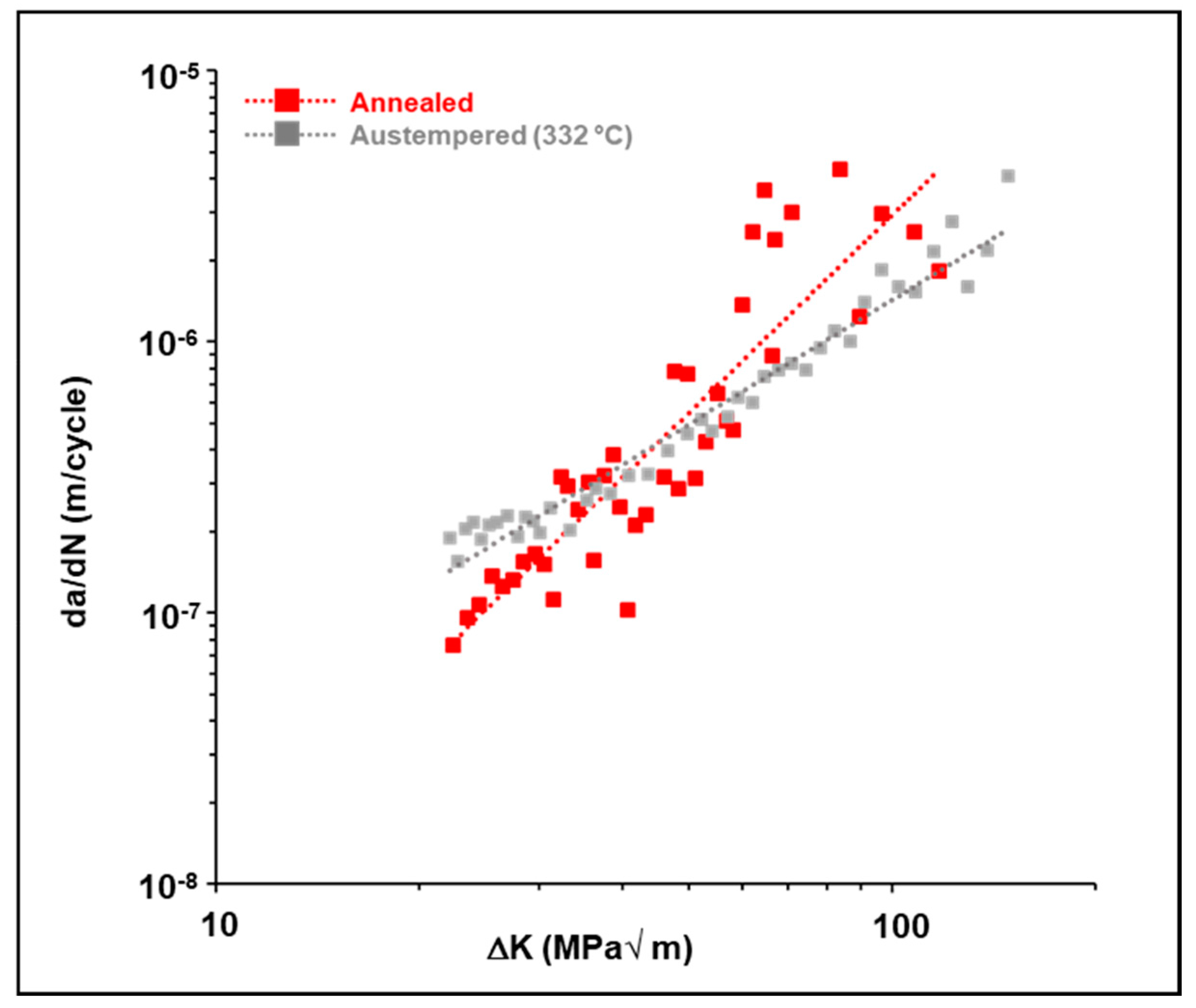
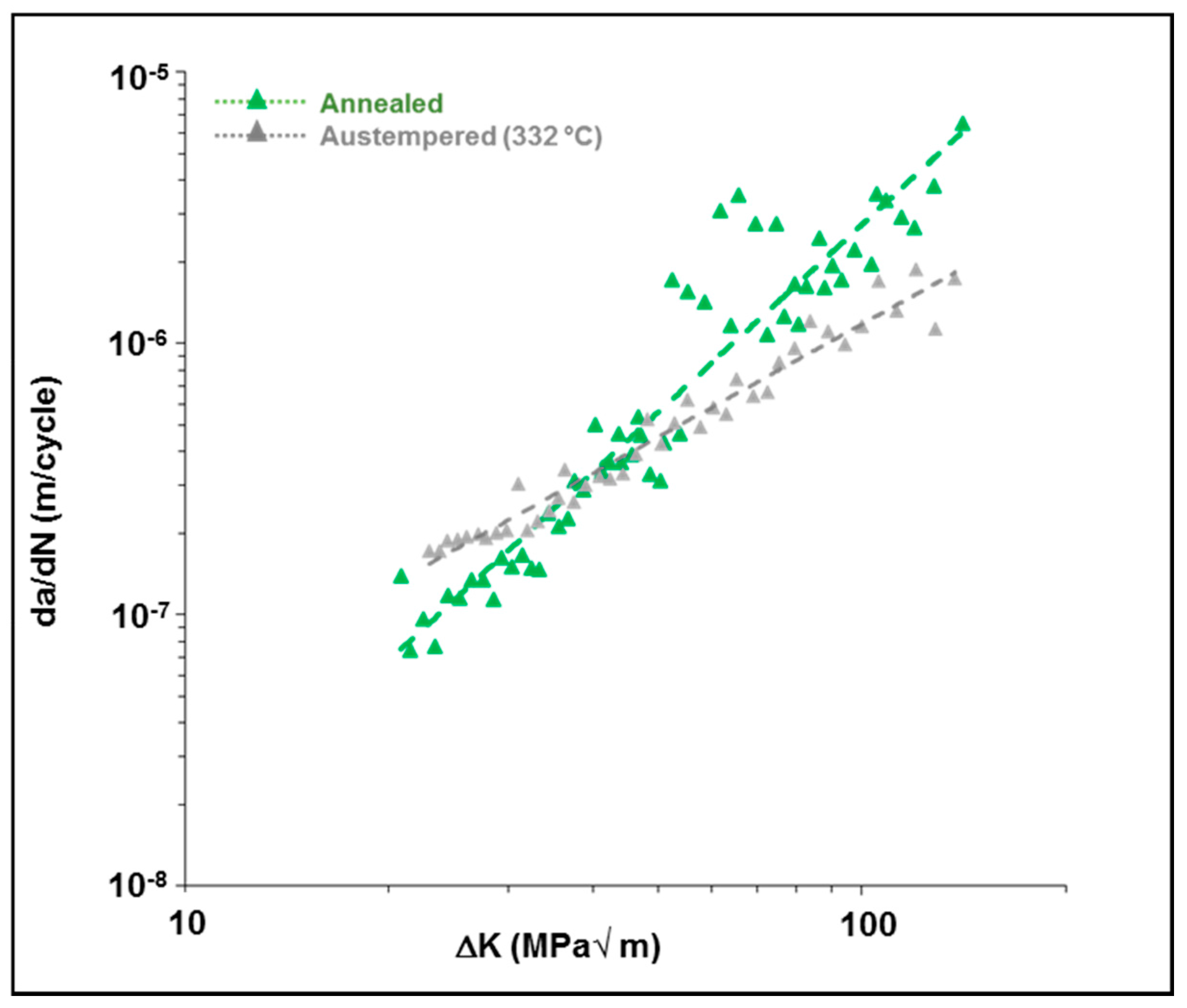
3.4. Influence of Austempering on the Crack Growth Behavior of Uncharged 4140 Steel
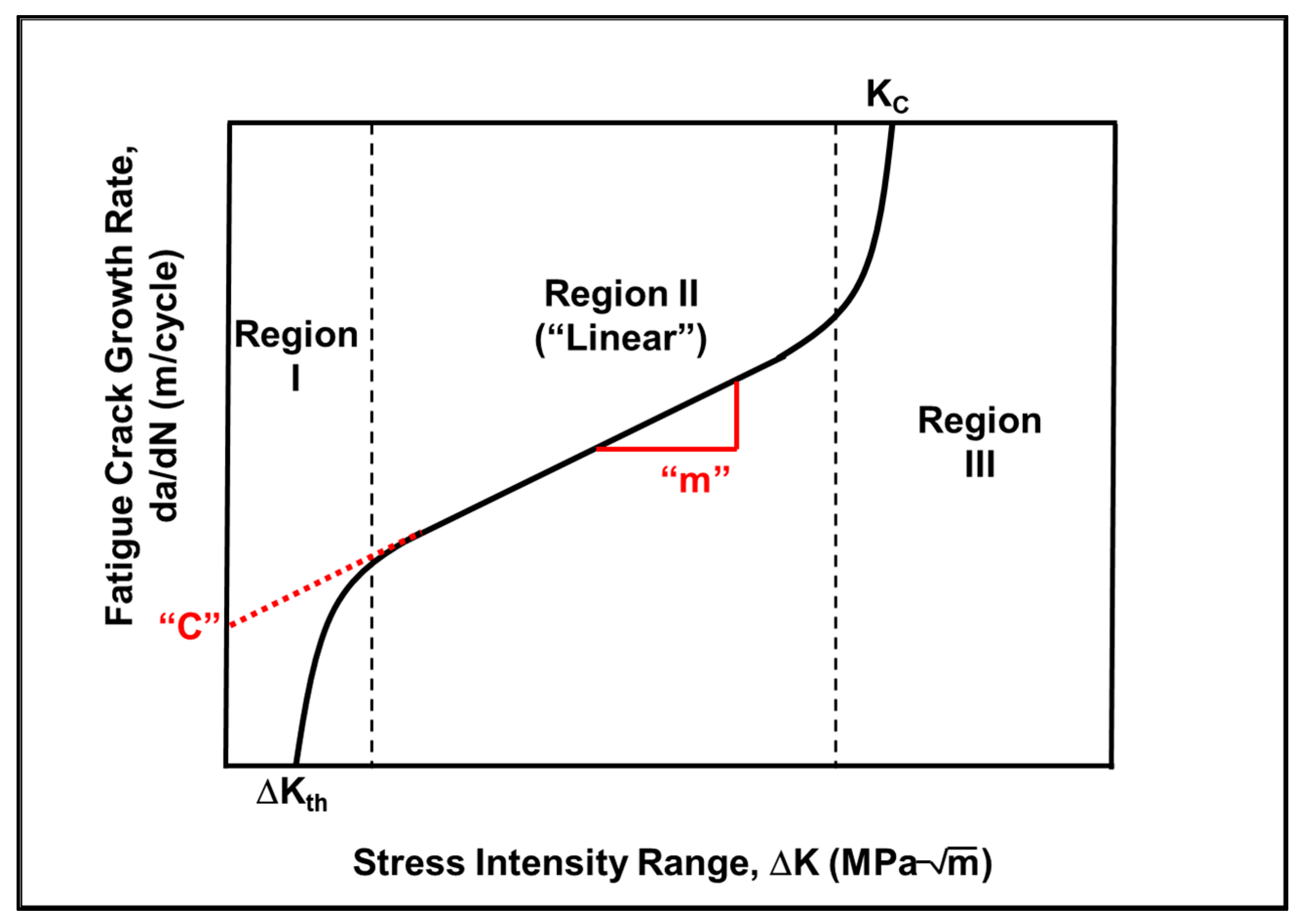
- First, the austempering process has increased the fracture toughness of the material due to the presence of a microstructure containing a large amount of lower bainite. The lower bainitic microstructure increases the fracture toughness of the material [3,4,5]. The higher fracture toughness is indicative of a greater crack growth resistance in this material. This, in turn, causes a lower fatigue crack growth rate and a higher fatigue threshold in the material.
- Secondly, the austempered samples had a very fine-scale microstructure consisting of lower bainite with a limited amount of tempered martensite. A lower bainitic structure has a much finer grain size than upper bainite or pearlite. This creates additional resistance to crack growth since the crack tip encounters large number of fine scale grain boundaries. This, in turn, reduces the crack propagation rate because the crack grows along a longer, more torturous path.
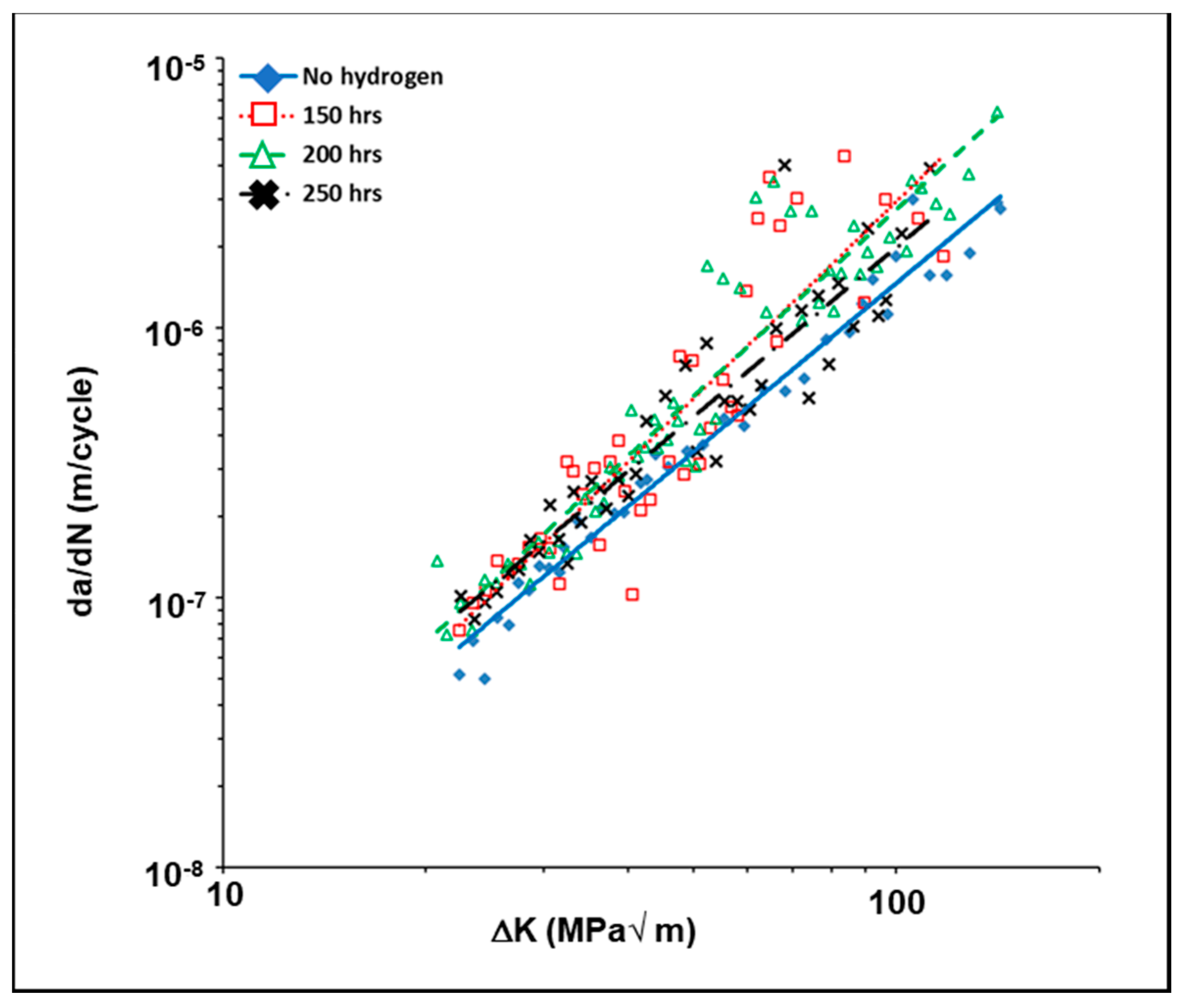
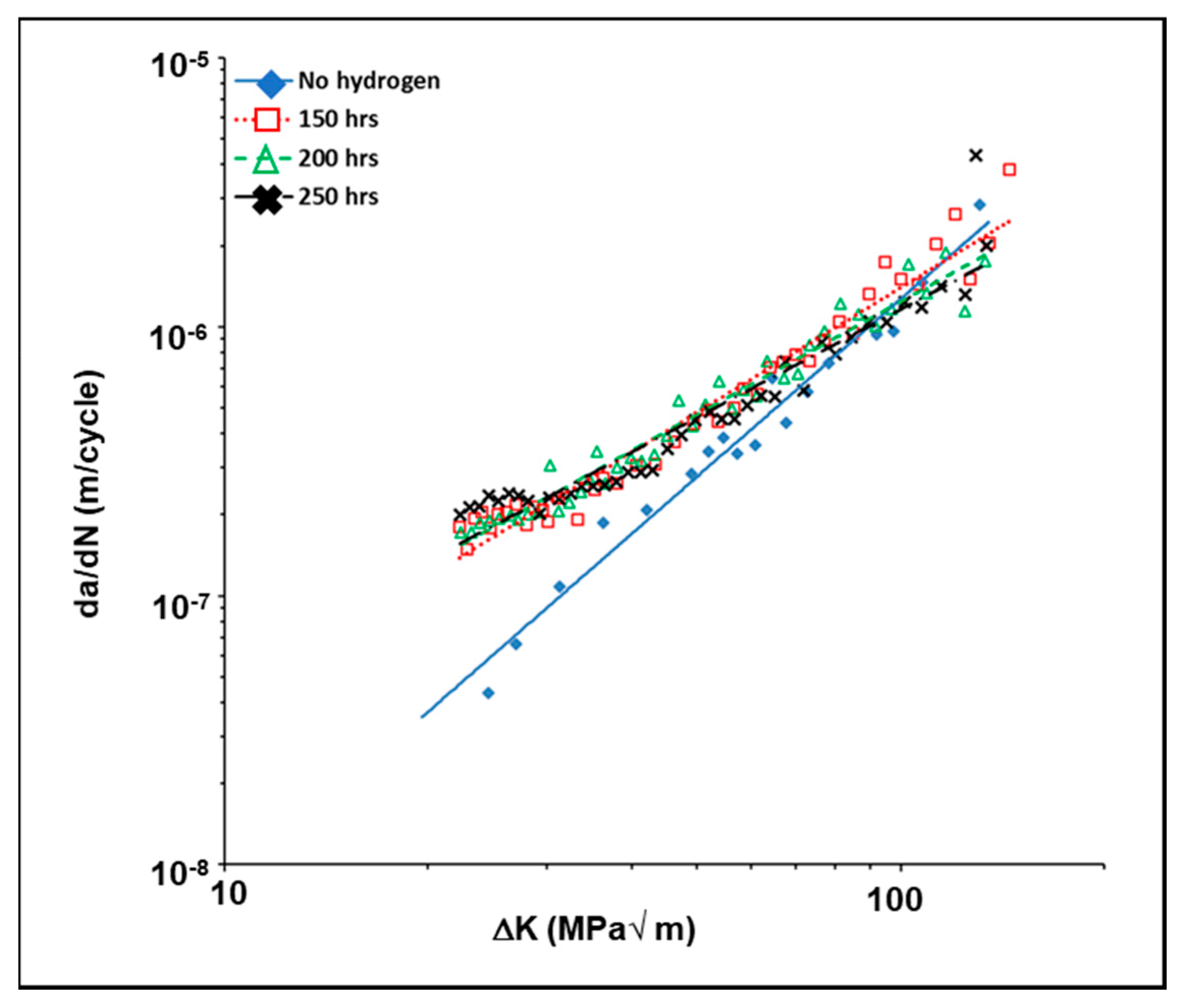
3.5. Influence of Hydrogen on the Fatigue Crack Gowth Behavior of 4140 Steel
3.6. Fractography
4. Conclusions and Future Work
- Austempering in the lower bainitic temperature range has significantly increased the mechanical properties and the fracture toughness of AISI 4140 steel as compared to the as-received (annealed) condition.
- In the absence of any charged hydrogen, the austempered samples had a much lower average crack growth rate and higher fatigue threshold than the as-received (annealed) samples.
- The presence of dissolved hydrogen increased the average crack growth rate in the austempered as well as in the as-received (annealed) samples.
- There is a transition stress intensity factor value of approximately 40–50 MPa√m; below this value, the presence of dissolved hydrogen causes the crack growth rate to be higher in the austempered samples when compared to annealed samples.
- In presence of dissolved hydrogen, above the transition stress intensity factor value, the crack growth rate was increasingly greater in the annealed specimens as compared to the austempered specimens.
- When compared to the as-received (annealed) condition, austempering of 4140 steel appears to provide a processing route by which the strength, hardness, and fracture toughness of the material can be increased with little or no degradation in the ductility and fatigue crack growth behavior.
Author Contributions
Conflicts of Interest
References
- Reed-Hill, R.E.; Abbaschian, R. Principles of Physical Metallurgy; PWS–Kent Publishing Company: Boston, MA, USA, 2010. [Google Scholar]
- Mangoonan, P.L. The Principle of Material Selection in Engineering Design; Prentice Hall: Salle River, NJ, USA, 1999. [Google Scholar]
- Putatunda, S.K. Fracture Toughness of a high carbon high silicon steel. Mater. Sci. Eng. A 2001, 297, 31–43. [Google Scholar] [CrossRef]
- Putatunda, S.K.; Martis, C.; Boileau, J. Influence of austempering temperature on the mechanical properties of low carbon low alloy steel. Mater. Sci. Eng. A 2011, 528, 5053–5059. [Google Scholar] [CrossRef]
- Putatunda, S.K.; Singar, A.V.; Tackett, R.; Lawes, G. Development of a high strength high toughness ausferritic steel. Mater. Sci. Eng. A 2009, 513–514, 329–339. [Google Scholar] [CrossRef]
- ASTM E-399. Standard Test Method for determination of Plain Strain Fracture Toughness of Metallic Materials. In Annual Book of ASTM Standards; ASTM: Philadelphia, PA, USA, 2016; pp. 589–611. [Google Scholar]
- Paris, P.C.; Erdogan, F. A critical analysis of crack propagation laws. J. Basic Eng. 1963, 85, 528–534. [Google Scholar] [CrossRef]
- Cwiek, J. Hydrogen degradation of high-strength steels. J. Achiev. Mater. Manuf. Eng. 2009, 37, 193–212. [Google Scholar]
- Lukacs, J. Fatigue crack propagation limit curves for different metallic and non-metallic materials. Mater. Sci. Forum 2003, 31, 414–415. [Google Scholar] [CrossRef]
- Philips, T.V.; McCaffrey, T.J. Ultra High-Strength Steels. In Metals Handbook, 10th ed.; ASM: Geauga County, OH, USA, 1990; Volume 1, pp. 430–448. [Google Scholar]
- Banerjee, K.; Chatterjee, U.K. Effect of microstructure on hydrogen embrittlement of weld-simulated HSLA-80 and HSLA-100 steels. Metall. Mater. Trans. A 2003, 34, 1297–1309. [Google Scholar] [CrossRef]
- Jha, A.K.; Narayanan, P.R.; Sreekumar, K.; Mittal, M.C.; Ninan, K.N. Hydrogen embrittlement of 3.5Ni-1.5Cr-0.5Mo steel fastener. Eng. Fail. Anal. 2008, 15, 431–439. [Google Scholar] [CrossRef]
- Lee, H.; Uhlig, H. Corrosion fatigue of type 4140 high strength steel. Metall. Mater. Trans. B 1972, 3, 2949–2957. [Google Scholar] [CrossRef]
- Murakami, Y.; Matusuoka, S. Effect of Hydrogen on fatigue crack growth of metals. Eng. Fract. Mech. 2010, 77, 1926–1940. [Google Scholar] [CrossRef]
- Chuang, J.H.; Tsay, L.W.; Chen, C. Crack growth behaviour of heat-treated 4140 steel in air and gaseous hydrogen. Int. J. Fatigue 1998, 20, 531–536. [Google Scholar] [CrossRef]
- Vergani, L.; Columbo, C.; Gobbi, G.; Bolzoni, F.M.; Fumagalli, G. Hydrogen effect on fatigue behavior of a quenched and tempered steel. Procedia Eng. 2014, 74, 468–471. [Google Scholar] [CrossRef]
- Rao, P.P.; Putatunda, S.K. Comparative study of fracture toughness of austempered ductile irons with upper and lower ausferrite microstructures. Mater. Sci. Technol. 1998, 14, 1257–1265. [Google Scholar] [CrossRef]
- Ramasagara Nagarajan, V.; Putatunda, S.K. Influence of dissolved hydrogen on the fatigue crack growth behavior of a low alloy (AISI 4140) steel. Int. J. Fatigue Special Issue Fatigue Damage Struct. Mater. IX 2014, 62, 236–248. [Google Scholar]
- ASTM E-8. Standard Test Method for Tensile Testing of Materials. In Annual Book of ASTM Standards; ASTM: Philadelphia, PA, USA, 2016. [Google Scholar]
- ASTM E-647. Standard Test Method for Fatigue Crack Growth Behavior of Materials. In Annual Book of ASTM Standards; ASTM: Philadelphia, PA, USA, 2016. [Google Scholar]
- Karim, R.A. Cathodic hydrogen charging of high strength low alloy 4140 steel. Mater. Perform. 2006, 45, 58–63. [Google Scholar]
- Hertzberg, R.W. Deformation of Fracture Mechanics of Engineering Materials, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 1989. [Google Scholar]
- Parvathavarthini, N.; Saroja, S.; Dayal, R.K.; Khatak, H.S. Studies on hydrogen permeability of 2.25% Cr-1% Mo ferritic steel: Correlation with microstructure. J. Nucl. Mater. 2001, 288, 187–196. [Google Scholar]
- Darken, L.S.; Smith, R.P. Behaviour of hydrogen in steel during and after immersion in acid. Corrosion 1949, 5, 1–16. [Google Scholar] [CrossRef]
- Dong, C.F.; Li, X.G.; Liu, Z.Y.; Zhang, Y.R. Hydrogen—Induced cracking and healing behavior of X70 steel. J. Alloys Compd. 2009, 484, 966–972. [Google Scholar] [CrossRef]
- Kim, C.D.; Longinow, A.W. Techniques for investigating hydrogen—Induced cracking of steels with high yield strength. Corrosion 1968, 24, 313–318. [Google Scholar] [CrossRef]
- Rice, J.R. Fatigue Crack Propagation; ASTM STP 415; ASTM: Philadelphia, PA, USA, 1967; p. 247. [Google Scholar]
- Nykyforchyn, H. Effect of hydrogen on the kinetics and mechanism of fatigue crack growth in structural steels. Mater. Sci. 1997, 33, 504–515. [Google Scholar] [CrossRef]
- Tsay, L.W.; Liu, C.C.; Shieh, Y.H. Fatigue crack propagation in 2.25Cr-1.0Mo steel weldments in air and hydrogen. Mater. Sci. Eng. A 2001, 299, 16–26. [Google Scholar] [CrossRef]








| Element | Composition (wt %) |
|---|---|
| C | 0.40 |
| Cr | 1.12 |
| Mo | 0.18 |
| Mn | 0.93 |
| Si | 0.33 |
| S | 0.031 |
| P | 0.025 |
| Cu | 0.02 |
| Ni | 0.17 |
| Fe | Balance |
| Set | Material | Externally Charged with Hydrogen? |
|---|---|---|
| “A” | Annealed | No |
| “B” | Annealed | Yes |
| “C” | Austempered (332 °C/1 h) | No |
| “D” | Austempered (332 °C/1 h) | Yes |
| Material Condition | Yield Strength (MPa) | Ultimate Tensile Strength (MPa) | % Elongation | Hardness (HRc) | Fracture Toughness (MPa√m) |
|---|---|---|---|---|---|
| Annealed | 757 ± 20 | 1031 ± 5 | 5.6 | 28 ± 1 | 65 ± 2 |
| Austempered (332 °C/1 h) | 1481 ± 12 | 1646 ± 9 | 5.5 | 45 ± 1 | 72 ± 6 |
| Material Condition | Hydrogen Charging Time (Hours) | Measured Hydrogen Content (ppm) |
|---|---|---|
| Annealed | 150 | 0.7 |
| 200 | 1.5 | |
| 250 | 2.9 | |
| Austempered (332 °C/1 h) | 150 | 1.6 |
| 200 | 3.6 | |
| 250 | 8.1 |
| Material Condition | ∆Kth (MPa√m) |
|---|---|
| Annealed | 6.68 |
| Austempered (332 °C/1 h) | 8.12 |
| Material Condition | Paris Law Constant | |
|---|---|---|
| C | m | |
| Annealed | 1 × 10−10 | 2.08 |
| Austempered (332 °C/1 h) | 5 × 10−11 | 2.14 |
| Material Condition | Hydrogen Charging Time (Hours) | Hydrogen Concentration (ppm) | ∆Kth (MPa√m) |
|---|---|---|---|
| Annealed | 0 | --- | 6.68 |
| 150 | 0.7 | 5.58 | |
| 200 | 1.5 | 5.49 | |
| 250 | 2.9 | 7.85 | |
| Austempered (332 °C/1 h) | 0 | --- | 8.12 |
| 150 | 1.6 | 6.59 | |
| 200 | 3.6 | 6.17 | |
| 250 | 8.1 | 6.01 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagarajan, V.R.; Putatunda, S.K.; Boileau, J. Fatigue Crack Growth Behavior of Austempered AISI 4140 Steel with Dissolved Hydrogen. Metals 2017, 7, 466. https://doi.org/10.3390/met7110466
Nagarajan VR, Putatunda SK, Boileau J. Fatigue Crack Growth Behavior of Austempered AISI 4140 Steel with Dissolved Hydrogen. Metals. 2017; 7(11):466. https://doi.org/10.3390/met7110466
Chicago/Turabian StyleNagarajan, Varun Ramasagara, Susil K. Putatunda, and James Boileau. 2017. "Fatigue Crack Growth Behavior of Austempered AISI 4140 Steel with Dissolved Hydrogen" Metals 7, no. 11: 466. https://doi.org/10.3390/met7110466
APA StyleNagarajan, V. R., Putatunda, S. K., & Boileau, J. (2017). Fatigue Crack Growth Behavior of Austempered AISI 4140 Steel with Dissolved Hydrogen. Metals, 7(11), 466. https://doi.org/10.3390/met7110466




