Determination of Final Ferrite Grain Size During Multiple-Stage Controlled Cooling of Low-Carbon, Low-Alloy Steels
Abstract
1. Introduction
2. Materials and Methodology
2.1. Materials
2.2. Methodology
- Continuous Cooling (CC): Samples were cooled from 950 °C to room temperature at rates ranging from 0.12 °C/s to 50 °C/s.
- Isothermal Hold Transformation (IH): Samples were rapidly cooled at a rate of 100 °C/s from 950 °C to various temperatures (termed transition points (TP)) between 600 °C and 700 °C, then held isothermally until transformation was complete.
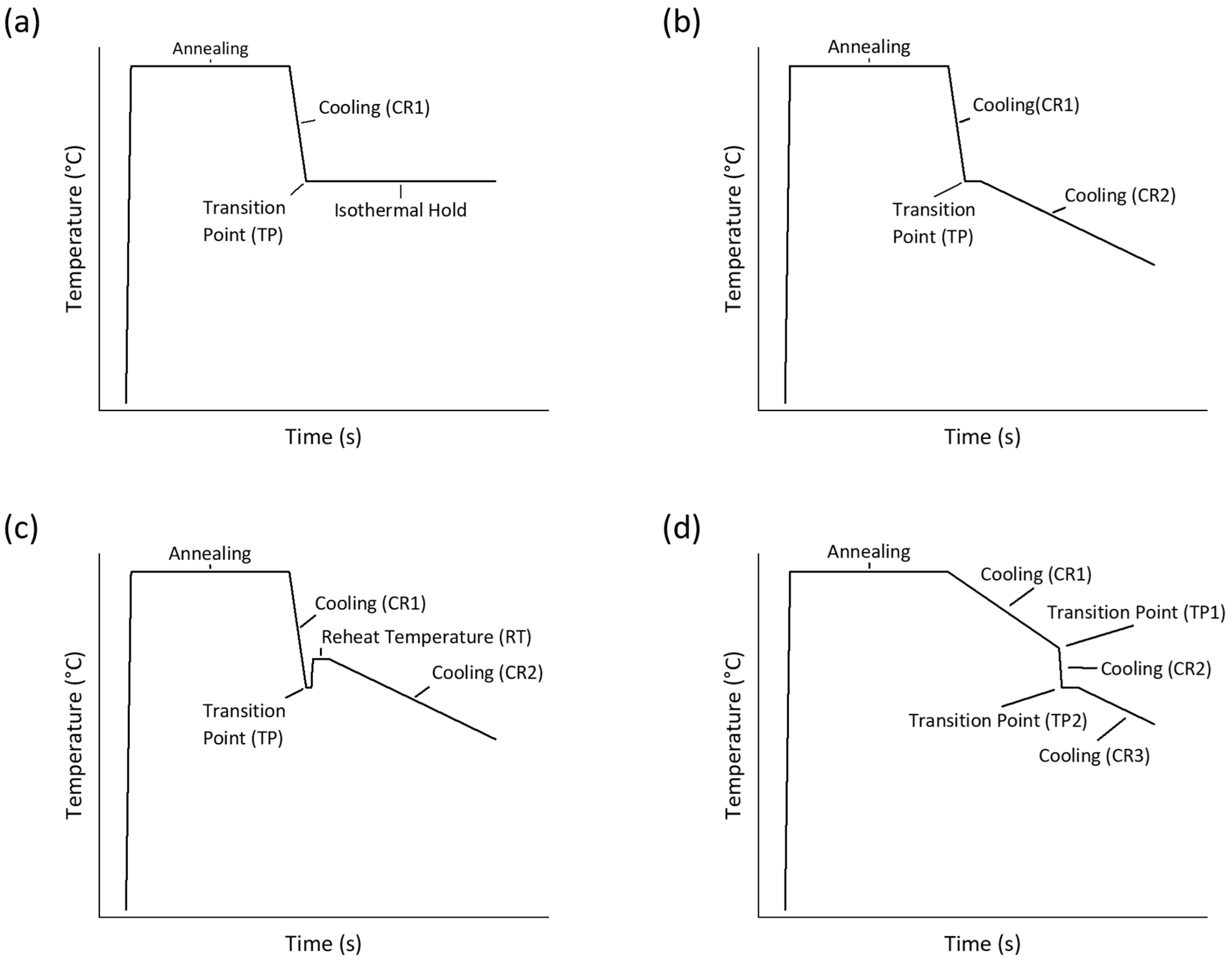
- Cooling and Isothermal Hold Transformation (CI): Samples were cooled from 950 °C at rates ranging from 1 °C/s to 50 °C/s to TP temperatures above the bainite formation temperature. Once this temperature was reached, the samples were held isothermally until transformation was completed. Shown in Figure 1a.
- Fast-Slow (FS): Samples were cooled from 950 °C at rates ranging from 1 °C/s to 20 °C/s to TP temperatures above the bainite formation temperature. Afterward, the cooling rate was reduced to 0.5 °C/s until room temperature was reached. Shown in Figure 1b.
- ‘Bounce Back’ (BB): Samples were cooled from 950 °C at rates ranging from 5 °C/s to 15 °C/s to TP temperatures above the bainite formation temperature. The samples were then reheated by 10 °C to 40 °C at a heating rate of 10 °C/s and slowly cooled at 0.5 °C/s to room temperature. Shown in Figure 1c.
- Slow-Fast (SF): Samples were cooled from 950 °C at rates of 0.12 °C/s and 1 °C/s to TP1 temperatures above the bainite formation temperature. Afterwards, the cooling rate was increased to 20 °C/s until 650 °C had been reached. Once at 650 °C, the cooling rate was decreased to 0.5 °C/s until full ferrite–pearlite formation had been achieved. Shown in Figure 1d.
3. Results and Discussion
3.1. Austenization Behavior
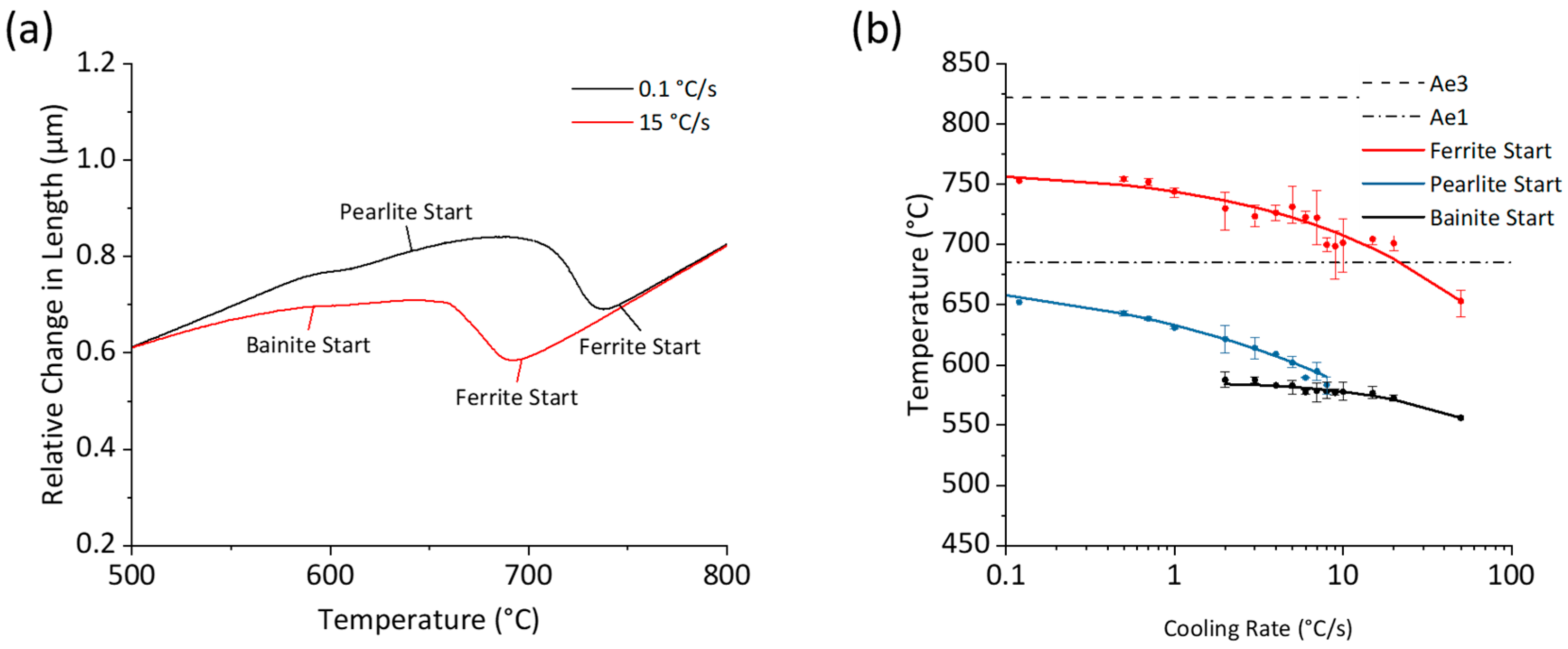
3.2. Continuous Cooling (CC) Transformation Behavior
3.3. Isothermal (IH) Transformation Behavior
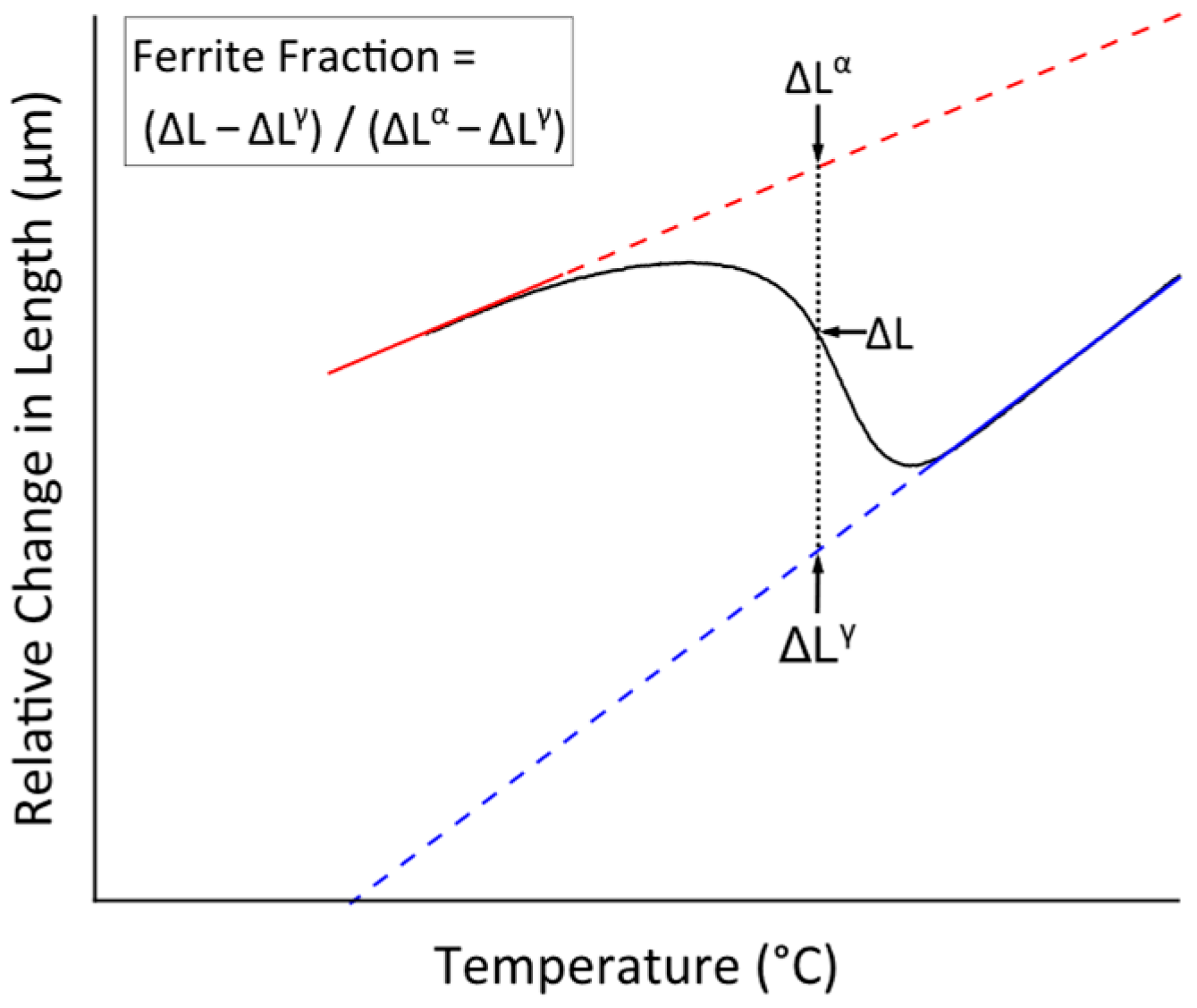
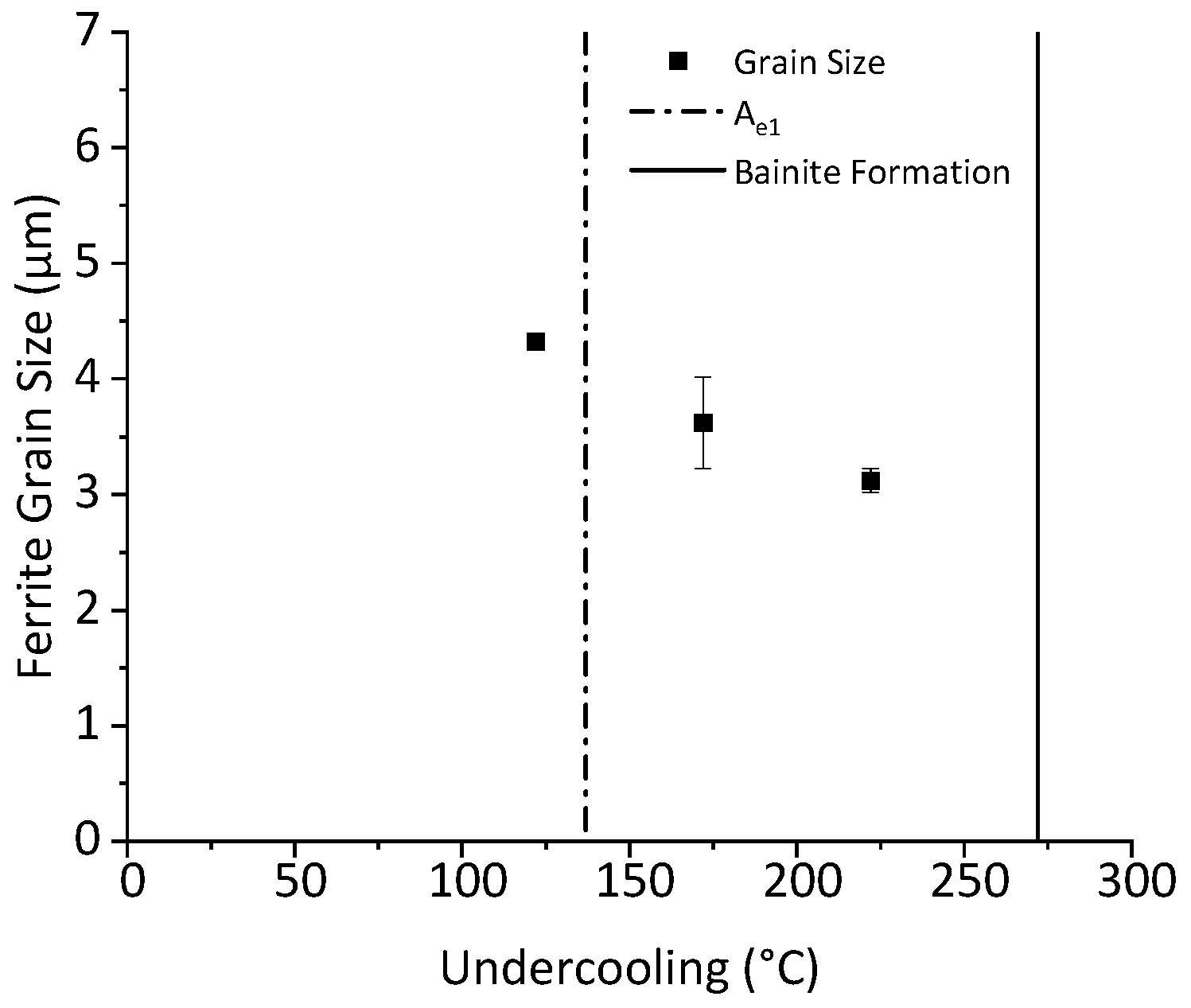
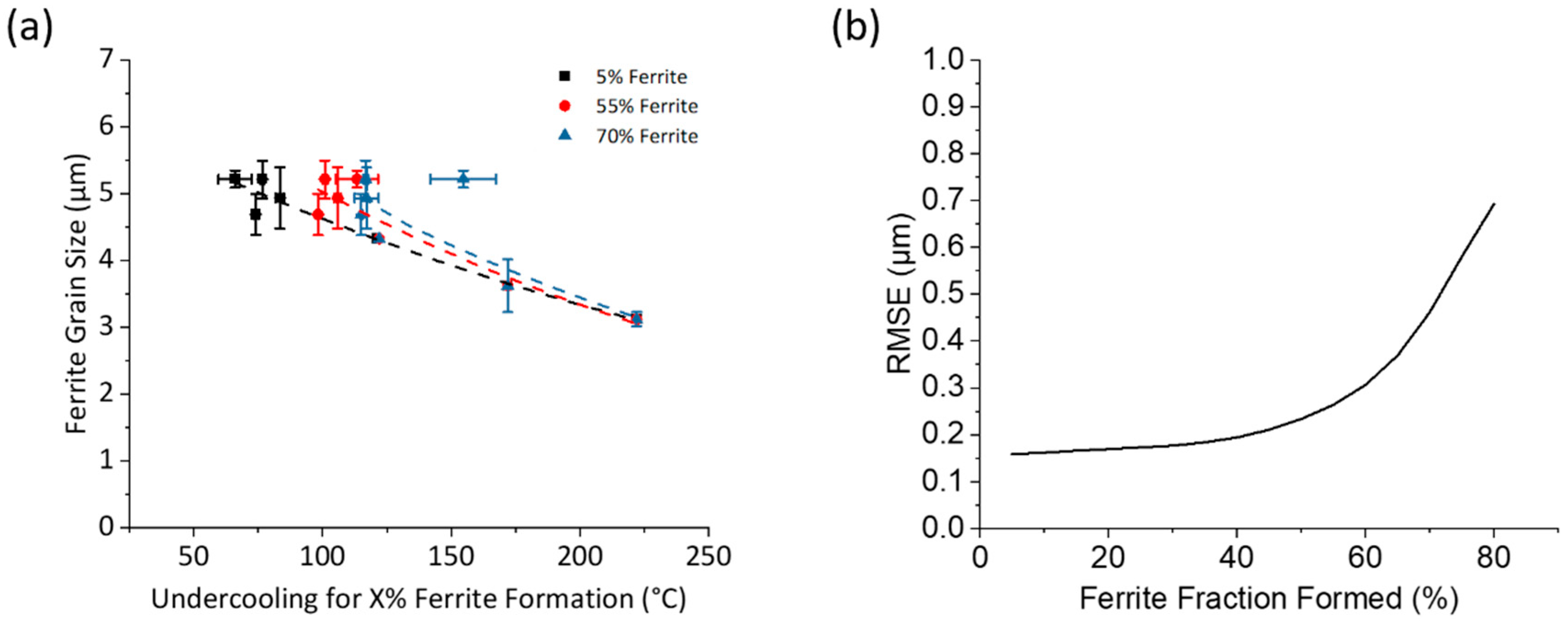
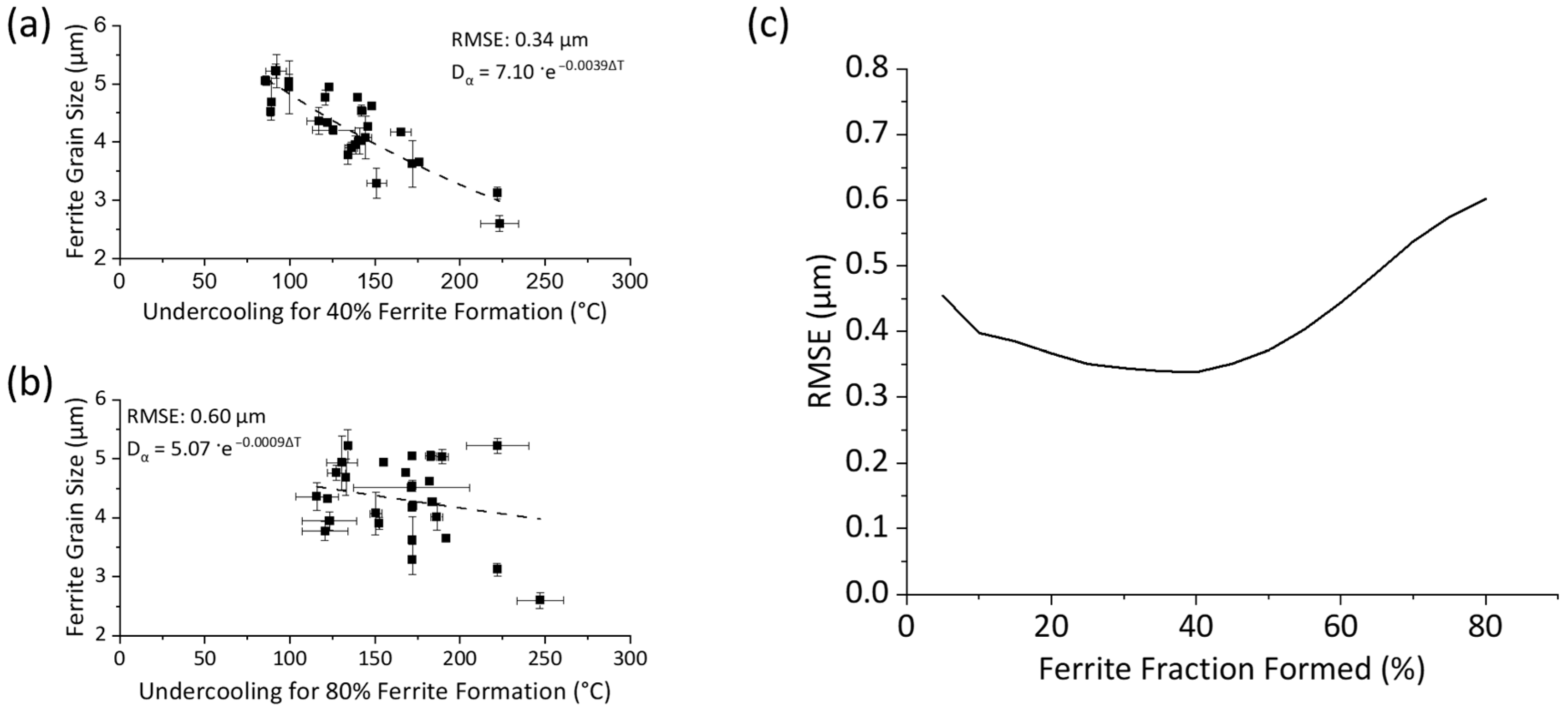
3.4. Determination of Critical Ferrite Percentage
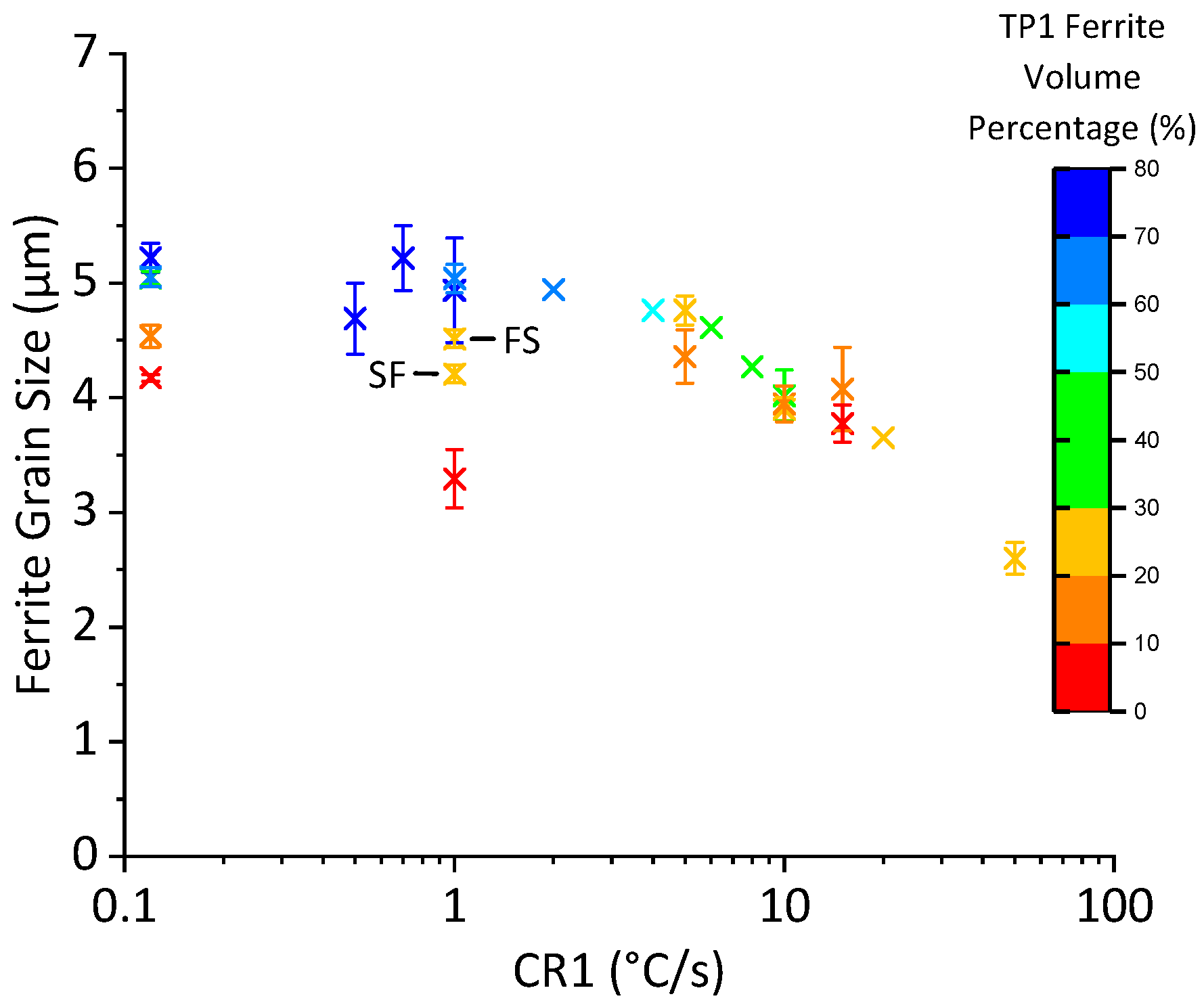
3.5. Complex Cooling Routines
4. Conclusions
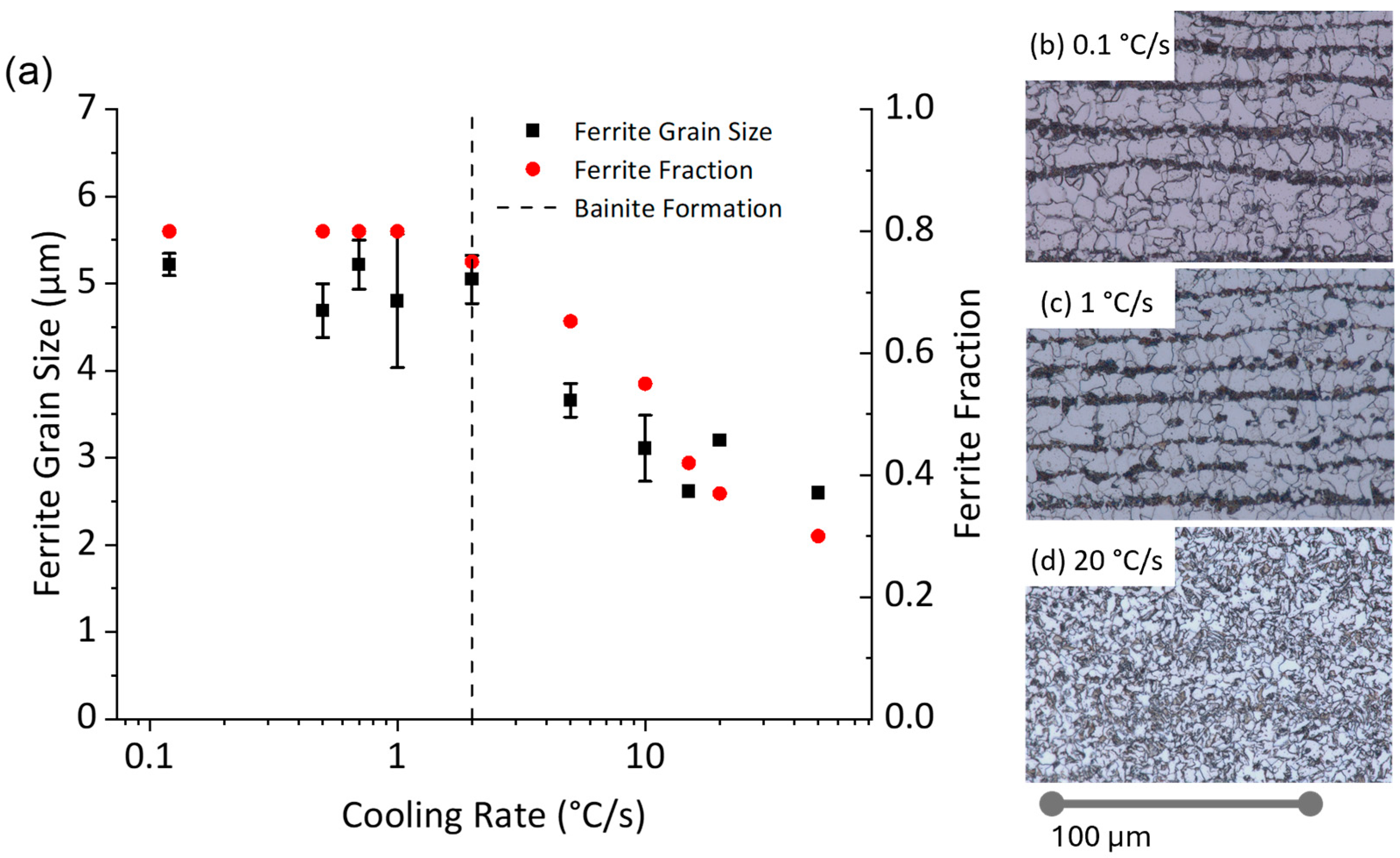
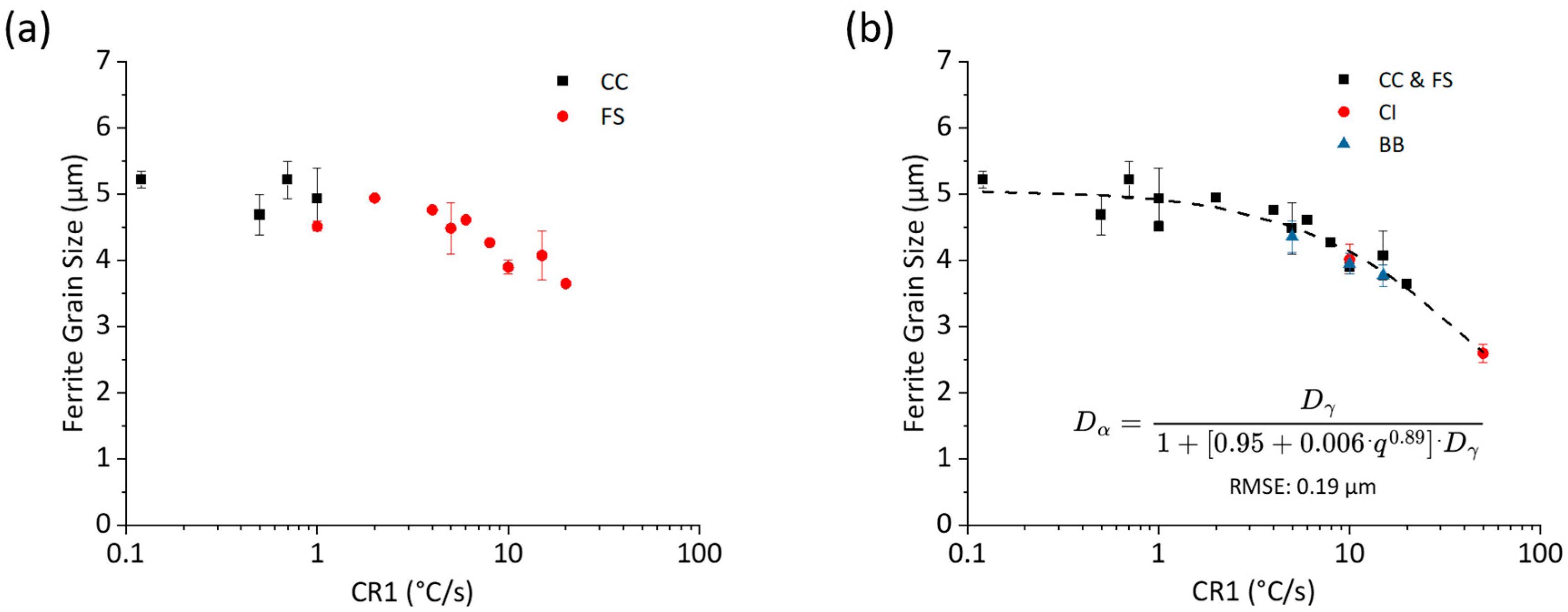
- Impact of Cooling Rate on Continuous Cooling: During continuous cooling, ferrite–pearlite microstructures were achieved only at cooling rates below 2 °C/s. Ferrite grain size was refined from 5.2 μm to 4.8 µm with a cooling rate increase from 0.12 °C/s to 1 °C/s.
- Influence of Isothermal Transformation Temperature: During isothermal transformations, ferrite grain sizes were refined from 4.3 µm to 3.0 µm with a decrease in transformation temperature from 700 °C to 600 °C.
- Early Transformation Determines Final Grain Size: During controlled cooling, the final ferrite grain size was primarily determined by the thermal profile during the first 40% of ferrite formation. The initial stage of transformation was critical for determining grain size, the thermal profile after 40% ferrite formation had a negligible effect on grain size. Therefore, to maximize refinement by stimulating nucleation and to reduce initial ferrite growth, transformation should be initiated at the lowest possible temperature. Utilizing this finding, grain refinement from 4.8 μm to 2.6 μm can be achieved by increasing the initial cooling rate from 1 °C/s to 50 °C/s with 40% ferrite undercoolings of 100 °C and 223 °C, respectively.
- Negligible Effect of Subsequent Cooling: The thermal profile after initial cooling was found to not have a noticeable effect on the final ferrite grain size, provided 40% ferrite was formed during the initial cooling stage. This held true as long as the sample was not reheated above Ae1 or cooled to permit bainite formation.
- Mechanism of Grain Size Determination: In ferrite–pearlite S-grade steels, ferrite grain size is thought to be determined during the early stages of transformation by the saturation or consumption of nucleation sites (i.e., austenite grain boundaries and triple points). Once these sites are saturated, the remaining transformation is primarily governed by growth and impingement.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World-Steel-Association. World Steel in Figures 2024; Worldsteel.org; World-Steel-Association: Brussels, Belgium, 2024. [Google Scholar]
- BSI. Hot Finished Structural Hollow Sections of Non-Alloy and Fine Grain Steels; BSI: Boon, Germany, 2006. [Google Scholar]
- Tovee, J.P. Microstructural Influence on the Effects of Forward and Reverse Mechanical Deformation in Hsla X65 And X80 Linepipe Steels. In Department of Metallurgy and Materials; University of Birmingham: Birmingham, UK, 2014. [Google Scholar]
- Wang, Y.D.; Tang, Z.-H.; Xiao, S.-F.; Siyasiya, C.W. Effects of final rolling temperature and coiling temperature on precipitates and microstructure of high-strength low-alloy pipeline steel. J. Iron Steel Res. Int. 2022, 29, 1236–1244. [Google Scholar] [CrossRef]
- Kaijalainen, A.; Mourujärvi, J.; Tulonen, J.; Steen, P.; Kömi, J.I. Improvement on Impact Toughness of Cold Formed S420 Steel by Direct Quenching; Trans Tech Publications Ltd.: Bäch, Switzerland, 2021; Volume 1016, pp. 648–653. [Google Scholar]
- Tiwari, S.; Ahn, H.; Reddy, M.H.; Park, N.; Reddy, N.G.S. Mechanical Property Prediction of Industrial Low-Carbon Hot-Rolled Steels Using Artificial Neural Networks. Materials 2025, 18, 2966. [Google Scholar] [CrossRef]
- Chen, D.; Zhang, R.; Li, Y.; Li, Z.; Yuan, G. Online cooling system and improved similar self-adaptive strategy for hot-rolled seamless steel tube. ISIJ Int. 2021, 61, 2135–2142. [Google Scholar] [CrossRef]
- Abathun, M.Z.; Han, J.; Yu, W. Effects of manufacturing methods and production routes on residual stresses of rectangular and square hollow steel sections: A review. Arch. Civ. Mech. Eng. 2021, 21, 100. [Google Scholar] [CrossRef]
- Siddique, M. A Knowledge-Based System for Process Planning in a Seamless Steel Tube Plant. In Mechanical and Production Engineering; Aston University: Birmingham, UK, 1990. [Google Scholar]
- Manohar, P.A.; Chandra, T. Continuous cooling transformation behaviour of high strength microalloyed steels for linepipe applications. ISIJ Int. 1998, 38, 766–774. [Google Scholar] [CrossRef]
- Amirjani, N.; Ketabchi, M.; Eskandari, M.; Hizombor, M. Effect of cooling rate and finish rolling temperature on structure and strength of API 5LX70 linepipe steel plate. J. Mater. Eng. Perform. 2020, 29, 4275–4285. [Google Scholar] [CrossRef]
- Militzer, M.; Pandi, R.; Hawbolt, E.B. Ferrite nucleation and growth during continuous cooling. Metall. Mater. Trans. A 1996, 27, 1547–1556. [Google Scholar] [CrossRef]
- Tamura, I. Some Fundamental Steps in Thermomechanical Processing of Steels. Trans. Iron Steel Inst. Jpn. 1987, 27, 763–779. [Google Scholar] [CrossRef]
- Umemoto, M.; Hai Guo, Z.; Tamura, I. Effect of cooling rate on grain size of ferrite in a carbon steel. Mater. Sci. Technol. 1987, 3, 249–255. [Google Scholar] [CrossRef]
- Choquet, P.; Fabreque, P.; Giusti, J.; Chamont, B.; Pezant, J.N.; Blanchet, F. Mathematical Modelling of Hot Rolling of Steel; Yue, S., Ed.; Publisher: Hamilton, ON, Canada, 1990; pp. 34–43. [Google Scholar]
- Abe, T.; Honda, T.; Ishizaki, S.; Wada, H.; Shikanai, N.; Okita, T. Mathematical Modelling of Hot Rolling of Steel; Yue, S., Ed.; Publisher: Hamilton, ON, Canada, 1990; pp. 66–75. [Google Scholar]
- Militzer, M.; Giumelli, A.; Hawbolt, E.B.; Meadowcroft, T.R. Austenite and ferrite grain size evolution in plain carbon steel. In Proceedings of the Mechanical Working and Steel Processing Conference, Baltimore, MD, USA, 16–19 October 1994; American Iron and Steel Instituate: Pittsburgh, PA, USA, 1995. [Google Scholar]
- Siwecki, T. Modelling of microstructure evolution during recrystallization controlled rolling. ISIJ Int. 1992, 32, 368–376. [Google Scholar] [CrossRef]
- Suehiro, M.; Sato, K.; Tsukano, Y.; Yada, H.; Senuma, T.; Matsumura, Y. Computer Modeling of Microstructural Change and Strength of Low Carbon Steel in Hot Strip Rolling. Trans. Iron Steel Inst. Jpn. 1987, 27, 439–445. [Google Scholar] [CrossRef]
- Liu, D.; Fazeli, F.; Militzer, M. Modeling of Microstructure Evolution during Hot Strip Rolling of Dual Phase Steels. ISIJ Int. 2007, 47, 1789–1798. [Google Scholar] [CrossRef]
- Militzer, M.; Hawbolt, E.B.; Meadowcroft, T.R. Microstructural model for hot strip rolling of high-strength low-alloy steels. Metall. Mater. Trans. A 2000, 31, 1247–1259. [Google Scholar] [CrossRef]
- Sokolov, D.F.; Vasil’ev, A.A.; Kolbasnikov, N.G.; Sokolov, S.F. Predicting the Ferrite Grain Size in Low Alloy Steels. STEEL Transl. 2012, 42, 462–466. [Google Scholar] [CrossRef]
- Duan, J.; Farrugia, D.; Slater, C.; Li, Z.; Davis, C. Microstructure development during multi-pass deformation in a low carbon steel with a leaner composition, finer grain size, and higher strength. J. Mater. Res. Technol. 2025, 36, 10373–10382. [Google Scholar] [CrossRef]
- Chae, J.-Y.; Qin, R. Topology of the deformation of a non-uniform grain structure. ISIJ Int. 2009, 49, 115–118. [Google Scholar] [CrossRef]
- Priestner, R.; Hodgson, P.D. Ferrite grain coarsening during transformation of thermomechanically processed C–Mn–Nb austenite. Mater. Sci. Technol. 1992, 8, 849–854. [Google Scholar] [CrossRef]
- Dong, Z.; Chen, D.; Long, M.; Li, W.; Chen, H. Computation of Phase Fractions in Austenite Transformation with the Dilation Curve for Various Cooling Regimens in Continuous Casting. Metall. Mater. Trans. B 2016, 47, 1553–1564. [Google Scholar] [CrossRef]
- Suh, D.W.; Oh, C.S.; Han, H.N.; Kim, S.J. Dilatometric analysis of austenite decomposition considering the effect of non-isotropic volume change. Acta Mater. 2007, 55, 2659–2669. [Google Scholar] [CrossRef]
- Sun, L.-y.; Liu, X.; Xu, X.; Lei, S.-W.; Li, H.-G.; Zhai, Q.-J. Review on niobium application in microalloyed steel. J. Iron Steel Res. Int. 2022, 29, 1513–1525. [Google Scholar] [CrossRef]
- Zhang, L.C.; Wen, X.L.; Liu, Y.Z. Effect of Precipitates on austenite grain growth behavior in a low-carbon Nb-V microalloyed steel. In Materials Science Forum; Trans Tech Publication: Pfaffikon, Switzerland, 2017. [Google Scholar]
- Martín, S.D.; Cock, D.T.; García-Junceda, A.; Caballero, F.G.; Capdevila, C.; De Andrés, C.G. Effect of heating rate on reaustenitisation of low carbon niobium microalloyed steel. Mater. Sci. Technol. 2008, 24, 266–272. [Google Scholar] [CrossRef]
- Slater, C.; Bandi, B.; Dastur, P.; Davis, C. Segregation neutralised steels: Microstructural banding elimination from dual-phase steel through alloy design accounting for inherent segregation. Metall. Mater. Trans. A 2022, 53, 2286–2299. [Google Scholar] [CrossRef]
- Khulka, K.; Aleksandrov, S. Promising tube steels for gas pipelines. Metallurgist 2006, 50, 137–143. [Google Scholar] [CrossRef]
- Matrosov, Y.I.; Tskitishvili, E.O.; Popov, E.S.; Konovalov, G.N.; Kholodnyi, A.A. Accelerated cooling after controlled rolling during heavy plate pipe steel manufacture in 3600 mill at the azovstal metallurgical combine. Metallurgist 2014, 57, 837–844. [Google Scholar] [CrossRef]
- Matrosov, M.Y.; Efron, L.I.; Il, V.I.; Severinets, I.Y.; Lipunov, Y.I. Use of accelerated cooling to improve the mechanical and processing properties of rolled plates used to make large-diameter gas-line pipe. Metallurgist 2005, 49, 220–229. [Google Scholar] [CrossRef]
- Matrosov, Y.; Kichkina, A.A.; Efimov, A.A.; Efron, L.I.; Bagmet, O.A. Simulating structure-forming processes in tube steels during controlled rolling with accelerated cooling. Metallurgist 2007, 51, 367–376. [Google Scholar] [CrossRef]




| C | Si | Mn | Nb |
|---|---|---|---|
| 0.15 | 0.19 | 1.40 | 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dixon, N.; Slater, C.; Du, J.; Davis, C. Determination of Final Ferrite Grain Size During Multiple-Stage Controlled Cooling of Low-Carbon, Low-Alloy Steels. Metals 2025, 15, 956. https://doi.org/10.3390/met15090956
Dixon N, Slater C, Du J, Davis C. Determination of Final Ferrite Grain Size During Multiple-Stage Controlled Cooling of Low-Carbon, Low-Alloy Steels. Metals. 2025; 15(9):956. https://doi.org/10.3390/met15090956
Chicago/Turabian StyleDixon, Nathan, Carl Slater, Jinlong Du, and Claire Davis. 2025. "Determination of Final Ferrite Grain Size During Multiple-Stage Controlled Cooling of Low-Carbon, Low-Alloy Steels" Metals 15, no. 9: 956. https://doi.org/10.3390/met15090956
APA StyleDixon, N., Slater, C., Du, J., & Davis, C. (2025). Determination of Final Ferrite Grain Size During Multiple-Stage Controlled Cooling of Low-Carbon, Low-Alloy Steels. Metals, 15(9), 956. https://doi.org/10.3390/met15090956








