High Corrosion Resistance of Ti3C2Tx/Al6061 Composites Achieved via Equal Channel Angular Pressing
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
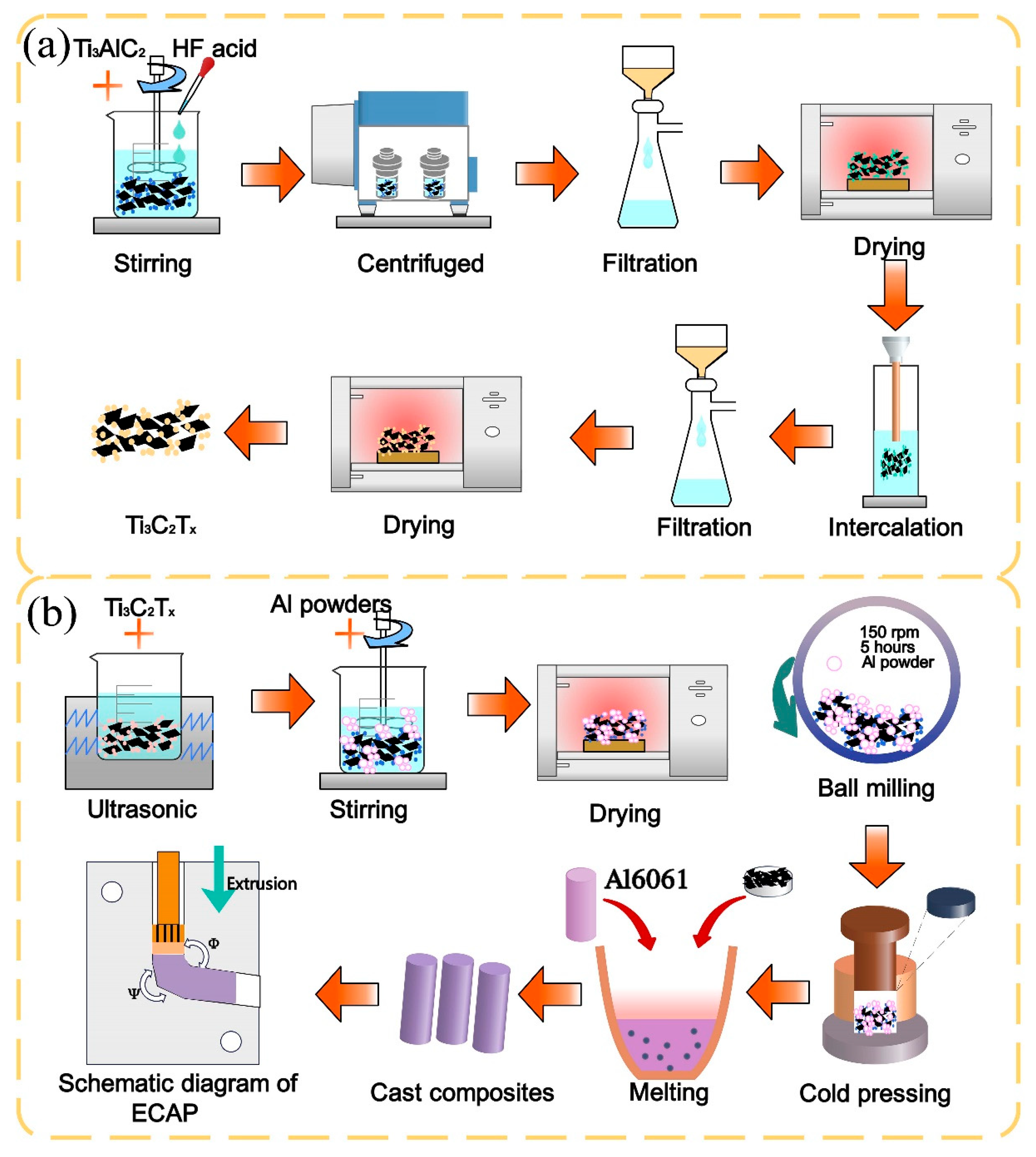
2.2. Preparation of Ti3C2Tx
2.3. Preparation of Ti3C2Tx/Al6061 Composites
2.4. Electrochemical Measurements and Immersion Corrosion Tests
2.5. Density, Porosity, and Hardness Tests
2.6. Microstructural Characterization
3. Results and Discussion
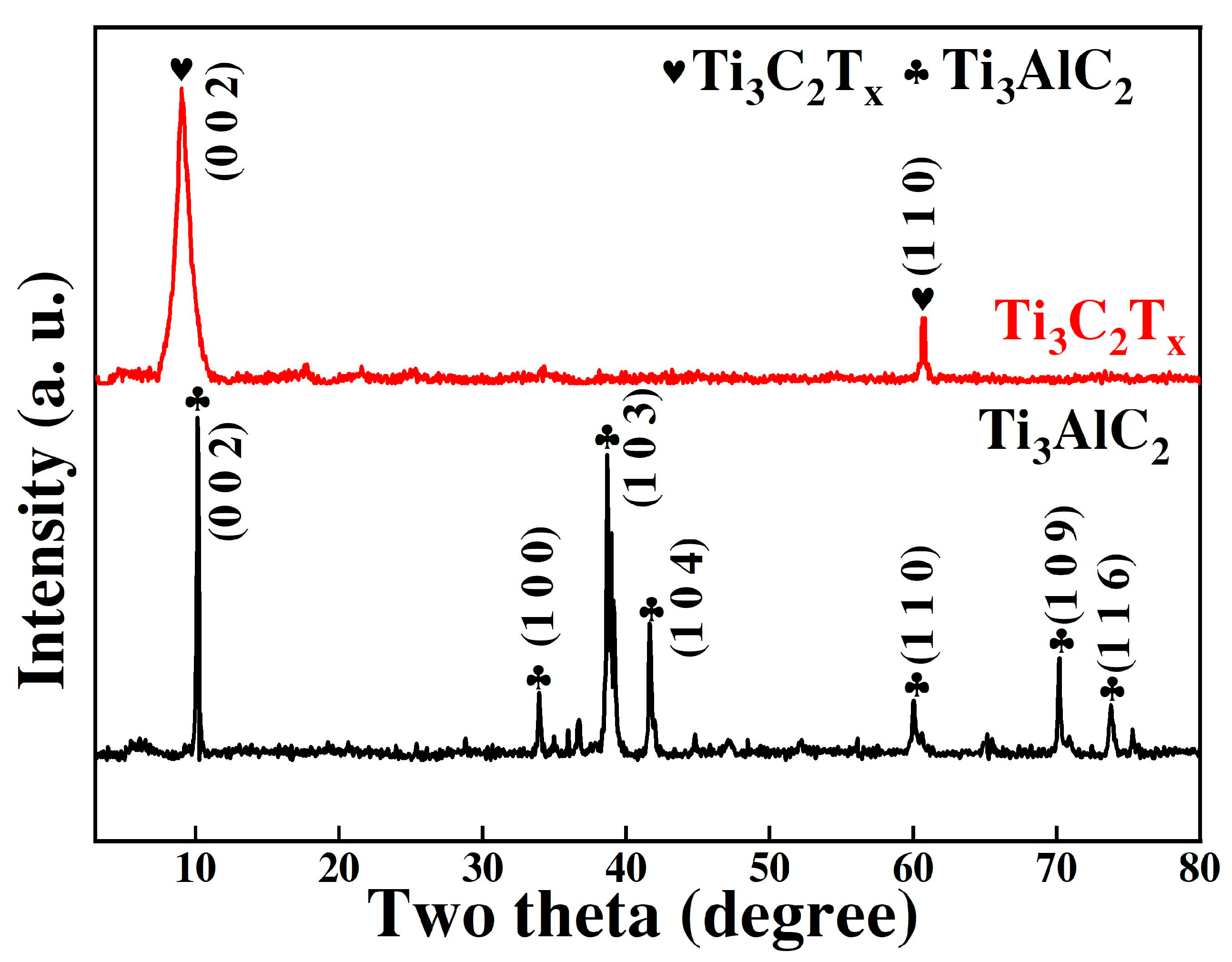
3.1. Microstructure and Physical Phase Analysis of Ti3C2Tx
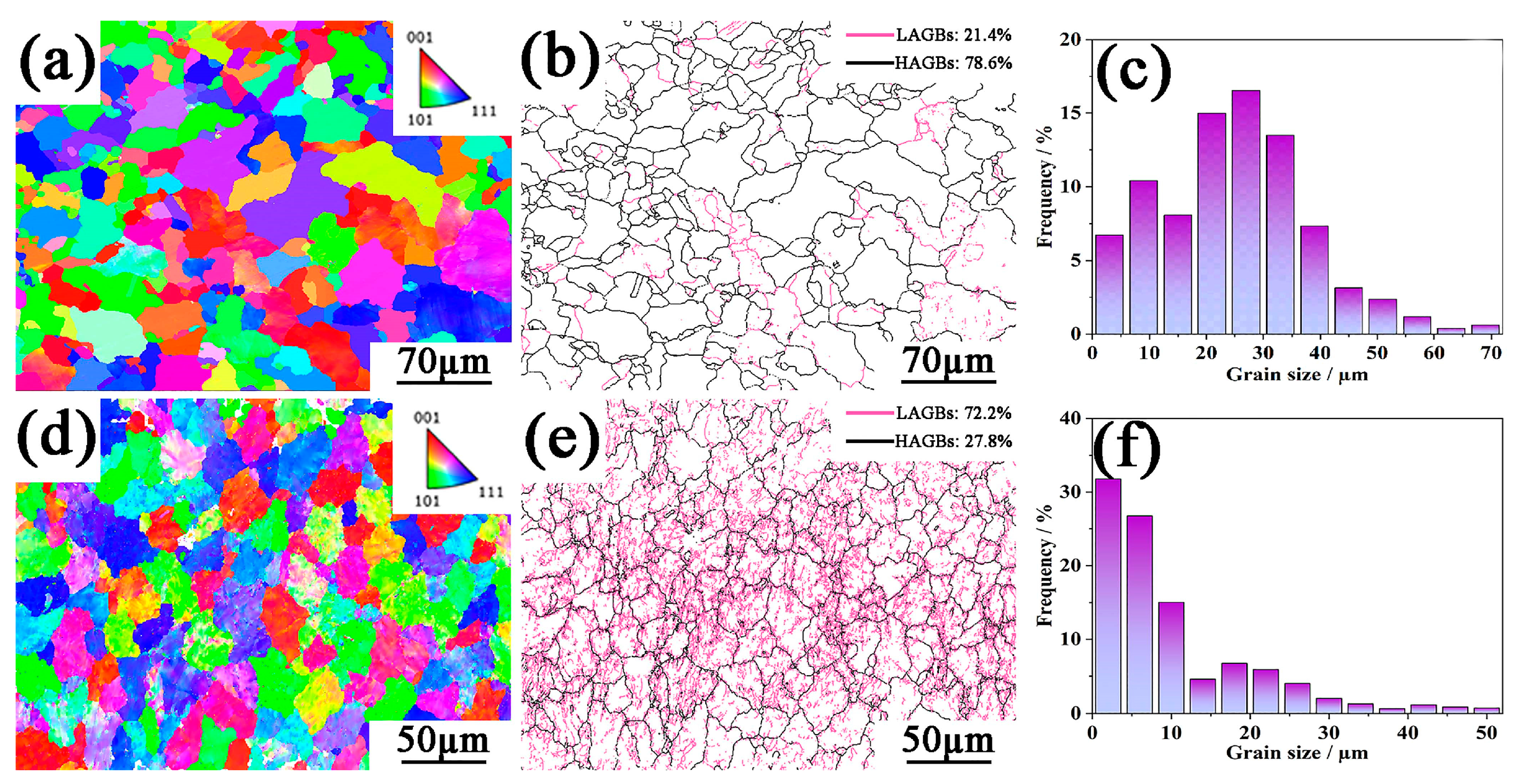
3.2. Microstructural Analysis of Ti3C2Tx/Al6061 Composites
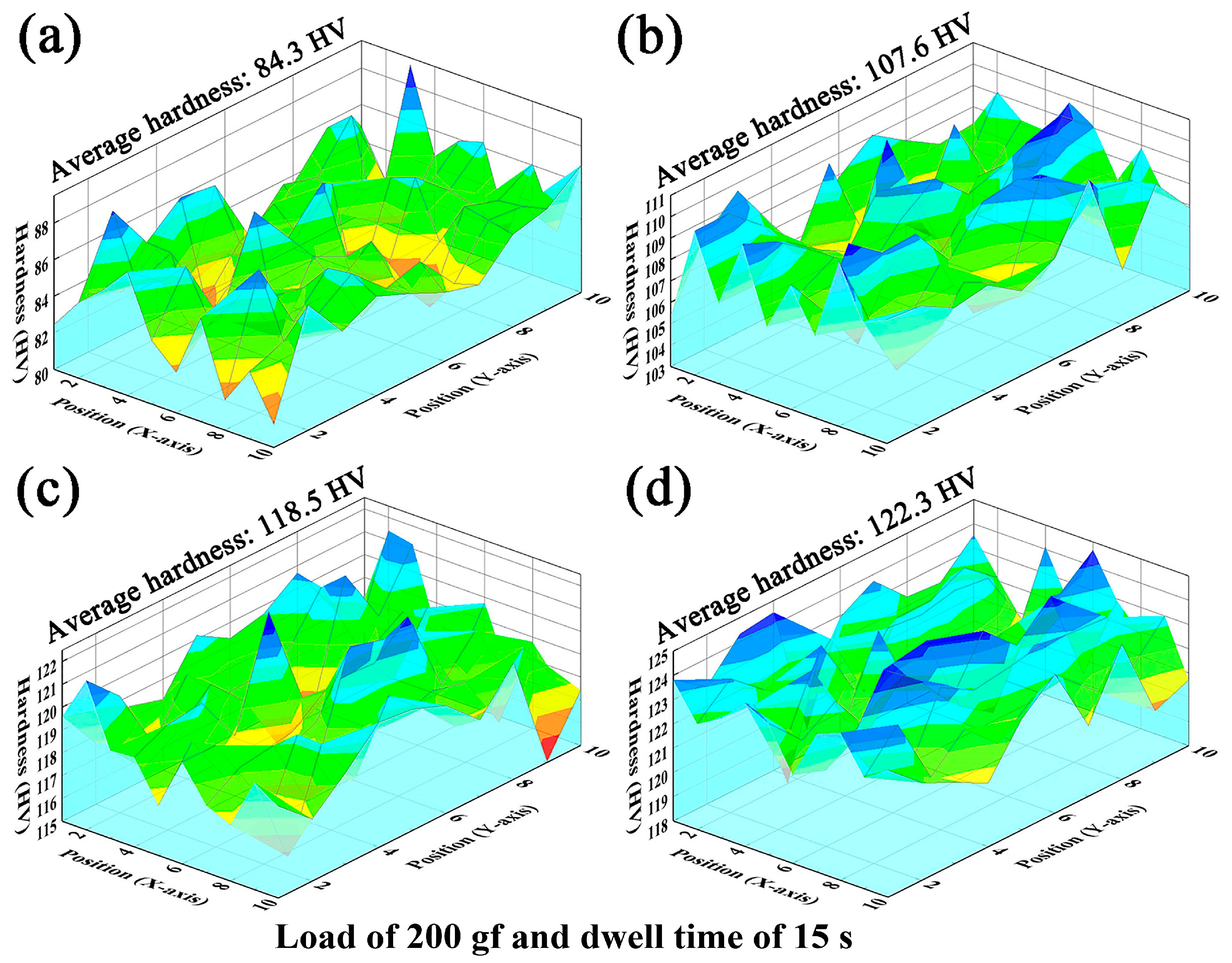
3.3. Densification and Hardness Analysis
3.4. Surface Morphology Analysis After Corrosion
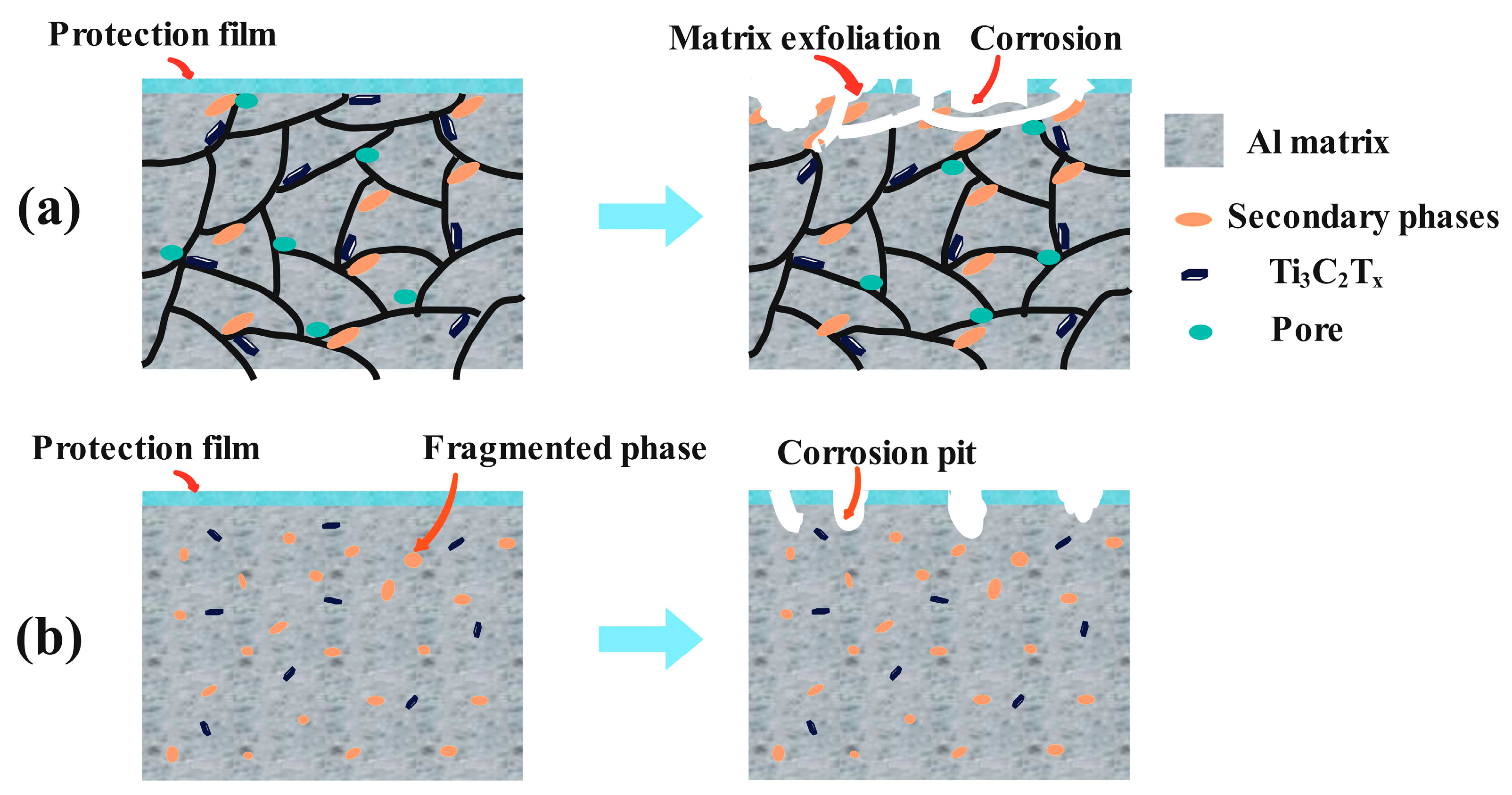
3.5. Corrosion Mechanism
4. Conclusions
- (1)
- Following ECAP treatment, SPD induces fragmentation and redistribution of secondary/reinforcing phases. Increasing ECAP passes enhances grain refinement and orientation texture, with EBSD confirming an average grain size of 8.7 µm after three passes.
- (2)
- ECAP significantly improves hardness: microhardness increases to 107.6 HV (one-pass), 118.5 HV (two-pass), and 122.3 HV (three-pass)—representing 40.6% and 45.1% increases versus as-cast for two-pass and three-pass, respectively. Density increases while porosity decreases (0.72% after three passes).
- (3)
- ECAP processing reduces corrosion susceptibility by (i) diminishing Icorr and achieving a 3.4-fold Rct increase through grain refinement and homogeneous phase dispersion, which suppress electrochemical heterogeneity and micro-galvanic coupling; (ii) reducing corrosion groove depth from 37.58 µm (as-cast) to 16.73 µm (three-pass) by mitigating interfacial strain mismatch; and (iii) impeding electrolyte infiltration via porosity reduction.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ning, W.; Zhang, W.; Zhao, Y. Phase-field simulation for evolution of iron-rich phase during solidification of Al–Si–Fe alloys. J. Mater. Res. Technol. 2024, 29, 5495–5506. [Google Scholar] [CrossRef]
- Xi, H.L.; Xiao, W.L.; Li, H.; Fu, Y.; Yi, G.; Qie, J.; Ma, X.; Ma, C. Effects of submicron-sized TiC particles on the microstructure modification and mechanical properties of Al-Si-Mg alloy. J. Alloys Compd. 2023, 968, 171963. [Google Scholar] [CrossRef]
- Chen, J.C.; Huang, Z.W.; Hu, Q.; Yan, H. Enhanced interfacial bonding and mechanical properties of CuO@GO/2024Al composites. Mater. Sci. Eng. A 2025, 923, 147732. [Google Scholar] [CrossRef]
- Xiang, K.; Qin, L.; Zhao, Y.; Huang, S.; Du, W.; Boller, E.; Rack, A.; Li, M.; Mi, J. Operando study of the dynamic evolution of multiple Fe-rich intermetallics of an Al recycled alloy in solidification by synchrotron X-ray and machine learning. Acta Mater. 2024, 279, 120267. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, P.; He, S.; Xu, Q.; Dou, Z.; Wang, H. Effect of beryllium content and heat treatment on microstructure and yield strength in Be/6061Al composites. J. Alloys Compd. 2018, 743, 746–755. [Google Scholar] [CrossRef]
- Liu, Z.; Luo, Z.; Zhang, X.; Feng, Y.; Wang, M.; Yang, J. Study on microstructure and mechanical properties of (TiC+B4C)/6061Al composites prepared by vacuum hot-press sintering method. J. Manuf. Process. 2024, 131, 670–681. [Google Scholar] [CrossRef]
- Wan, H.; Shuai, L.; Ling, L.; Hu, Z.; Yan, H. Mechanical Properties and Corrosion Resistance of La2O3/A356 Composites Fabricated by Ultrasonic-Assisted Casting. Metals 2025, 15, 184. [Google Scholar] [CrossRef]
- Zhou, W.; Xue, F.; Li, M. Corrosion Behavior of Al-Mg Alloys with Different Alloying Element Contents in 3.5% NaCl Solution. Metals 2025, 15, 327. [Google Scholar] [CrossRef]
- Fang, N.; Wang, H.; Dong, D.; Wei, Z.; Zhu, L.; Zhu, D.; Wang, Z. Unraveling the pressure-induced microstructural origin of enhanced corrosion resistance in a high-strength Al-Si-Cu-Mg alloy. Corros. Sci. 2025, 252, 112972. [Google Scholar] [CrossRef]
- Zhang, J.; Hu, J.; Lu, Y.; Gu, J.; Yang, X.; Zhao, Y.; Qi, L.; Wei, W. The enhancement of corrosion resistance for Al-Cu alloy reinforced by ZrB2 particles with different scales. J. Alloys Compd. 2025, 1026, 180357. [Google Scholar] [CrossRef]
- Lv, Y.; Meng, X.; Dong, Z.; Ma, Y.; Zhang, X. Localized corrosion evolution associated with intermetallic particles in Al-Cu-Li alloys. Mater. Charact. 2023, 204, 113144. [Google Scholar] [CrossRef]
- Lokhande, P.; Rednam, U.; Khasim, S.; Hamdalla, T.; Vedpathak, A.; Kumar, D.; Singh, K. Etching duration as a key parameter for tailoring Ti3C2Tx MXene electrochemical properties. J. Phys. Chem. Solids 2025, 207, 112902. [Google Scholar] [CrossRef]
- Guo, W.; Wang, B.; Shu, Q. An Efficient and Stable MXene-Immobilized, Cobalt-Based Catalyst for Hydrogen Evolution Reaction. Metals 2024, 14, 922. [Google Scholar] [CrossRef]
- Cao, S.; Shen, B.; Tong, T.; Fu, J.; Yu, J. 2D/2D heterojunction of ultrathin MXene/Bi2WO6 nanosheets for improved photocatalytic CO2 reduction. Adv. Funct. Mater. 2018, 28, 1800136. [Google Scholar] [CrossRef]
- Eid, K.; Lu, Q.; Abdel-Azeim, S.; Soliman, A.; Abdullah, A.M.; Abdelgwad, A.M.; Forbes, R.P.; Ozoemena, K.I.; Varma, R.S.; Shibl, M.F. Highly exfoliated Ti3C2Tx MXene nanosheets atomically doped with Cu for efficient electrochemical CO2 reduction: An experimental and theoretical study. J. Mater. Chem. A 2022, 10, 1965–1975. [Google Scholar] [CrossRef]
- Gong, B.; Ma, X.; Wang, T.; Hou, J.; Ji, S.; Shen, X.; Min, Y.; Xu, Q.; Cao, H. Enhanced corrosion resistance of MXene/Zn-Al LDH coating on Al alloy under dry/humid and temperature alternating environments. Corros. Sci. 2025, 255, 113088. [Google Scholar] [CrossRef]
- Yan, H.; Cai, M.; Li, W.; Fan, X.; Zhu, M. Amino-functionalized Ti3C2Tx with anti-corrosive/wear function for waterborne epoxy coating. J. Mater. Sci. Technol. 2020, 54, 144–159. [Google Scholar] [CrossRef]
- Gazizov, M.R.; Mironov, S.Y.; Holmestad, R.; Gazizova, M.Y.; Kaibyshev, R.O. Effect of ECAP and aging on microstructure of an Al-Cu-Mg-Si alloy. Mater. Charact. 2024, 218, 114500. [Google Scholar] [CrossRef]
- Jia, H.; Bjørge, R.; Cao, L.; Song, H.; Marthinsen, K.; Li, Y. Quantifying the grain boundary segregation strengthening induced by post-ECAP aging in an Al-5Cu alloy. Acta Mater. 2018, 155, 199–213. [Google Scholar] [CrossRef]
- Sabirov, I.; Murashkin, M.Y.; Valiev, R.Z. Nanostructured aluminium alloys produced by severe plastic deformation: New horizons in development. Mater. Sci. Eng. A 2013, 560, 1–24. [Google Scholar] [CrossRef]
- Chen, Y.L.; Liu, Y.; Zhang, J.; Liu, M.; Li, H.; Ding, L.; Jia, Z.H.; Liu, X. Deformation-mediated cyclic evolution of precipitates in Al-Mg-Si-Cu alloy by multi-pass ECAP and thermal treatments. J. Mater. Sci. Technol. 2025, 213, 42–54. [Google Scholar] [CrossRef]
- Li, G.S.; Pan, X.Y.; Jiang, J.; Li, J.H.; Xie, L.L.; Liu, H.T.; Zhang, M.Y. Achieving ultra-fine grains and high corrosion resistance of Al–Zn–Mg–Cu alloy by ECAP and post cold rolling. J. Mater. Res. Technol. 2023, 26, 7354–7368. [Google Scholar] [CrossRef]
- Elhefnawey, M.; Shuai, G.L.; Li, Z.; Zhang, D.T.; Tawfik, M.M.; Li, L. On achieving ultra-high strength and improved wear resistance in Al–Zn–Mg alloy via ECAP. Tribol. Int. 2021, 163, 107188. [Google Scholar] [CrossRef]
- Ciemiorek, M.; Chromiński, W.; Jasiński, C.; Lewandowska, M. Microstructural changes and formability of Al–Mg ultrafine-grained aluminum plates processed by multi-turn ECAP and upsetting. Mater. Sci. Eng. A 2022, 831, 142202. [Google Scholar] [CrossRef]
- Li, J.; He, T.; Du, X.Y.; Vereschaka, A.; Zhang, J.J. Regulating hardness homogeneity and corrosion resistance of Al-Zn-Mg-Cu alloy via ECAP combined with inter-pass aging. Mater. Charact. 2024, 218, 114489. [Google Scholar] [CrossRef]
- Downes, M.; Shuck, C.E.; McBride, B.; Busa, J.; Gogotsi, Y. Comprehensive synthesis of Ti3C2Tx from MAX phase to MXene. Nat. Protoc. 2024, 19, 1807–1834. [Google Scholar] [CrossRef]
- Scepanovic, J.; Asanovic, V.; Herenda, S.; Vuksanovic, D.; Radonjic, D.; Korac, F. Microstructural characteristics, mechanical properties, fracture analysis and corrosion behavior of hypereutectic Al-13.5Si alloy. Int. J. Met. 2019, 13, 700–704. [Google Scholar] [CrossRef]
- Xu, R.; Tan, Z.Q.; Xiong, D.; Fan, G.; Guo, Q.; Zhang, J.; Su, Y.S.; Li, Z.; Zhang, D. Balanced strength and ductility in CNT/Al composites achieved by flake powder metallurgy via shift-speed ball milling. Compos. Part A Appl. Sci. Manuf. 2017, 96, 57–66. [Google Scholar] [CrossRef]
- Chegini, M.; Shaeri, M.H.; Taghiabadi, R.; Chegini, S.; Djavanroodi, F. The correlation of microstructure and mechanical properties of in-situ Al-Mg2Si cast composite processed by equal channel angular pressing. Materials 2019, 12, 1553. [Google Scholar] [CrossRef]
- Gao, H.M.; Yao, A.; Shi, Y.H.; Noor, N.; Zeb, A.; Li, M.L.; Li, H.L. Preparation and properties of hierarchical Al–Mg layered double hydroxides as UV resistant hydrotalcite. Mater. Chem. Phys. 2020, 256, 123630. [Google Scholar] [CrossRef]
- Ibrahim, M.; Sadawy, M.M. Influence of ECAP as grain refinement technique on microstructure evolution, mechanical properties and corrosion behavior of pure aluminum. Trans. Nonferrous Met. Soc. China 2015, 25, 3865–3876. [Google Scholar] [CrossRef]
- Rogachev, S.O.; Naumova, E.A.; Inozemtseva, O.V.; Andreev, V.A.; Karelin, R.D.; Komarov, V.S.; Tabachkova, N.Y.; Khatkevich, V.M.; Bondareva, S.A. Effect of number of ECAP passes on structure and mechanical properties of Al–Ca–Mn–Fe alloy. Mater. Today Commun. 2024, 38, 107762. [Google Scholar] [CrossRef]
- Zha, M.; Li, Y.; Mathiesen, R.; BJØRGE, R.; Roven, H. Microstructure, hardness evolution and thermal stability of binary Al-7Mg alloy processed by ECAP with intermediate annealing. Trans. Nonferrous Met. Soc. China 2014, 24, 2301–2306. [Google Scholar] [CrossRef]
- Sekhar, A.; Mandal, A.; Das, D. Mechanical properties and corrosion behavior of artificially aged Al-Mg-Si alloy. J. Mater. Res. Technol. 2020, 9, 1005–1024. [Google Scholar] [CrossRef]
- Xing, X.; Sui, Y.; Xu, X.; Zhou, D.; Li, Q.; Xu, Y.; Tang, E.; Liu, S. Preparation of Ti3C2Tx MXene based composite corrosion inhibitor and anticorrosive properties of enhanced coatings. Diam. Relat. Mater. 2023, 140, 110546. [Google Scholar] [CrossRef]
- Zhu, H.; Huang, Z.; Jin, G.; Gao, M. Effect of temperature on galvanic corrosion of Al 6061-SS 304 in nitric acid. Energy Rep. 2022, 8, 112–123. [Google Scholar] [CrossRef]






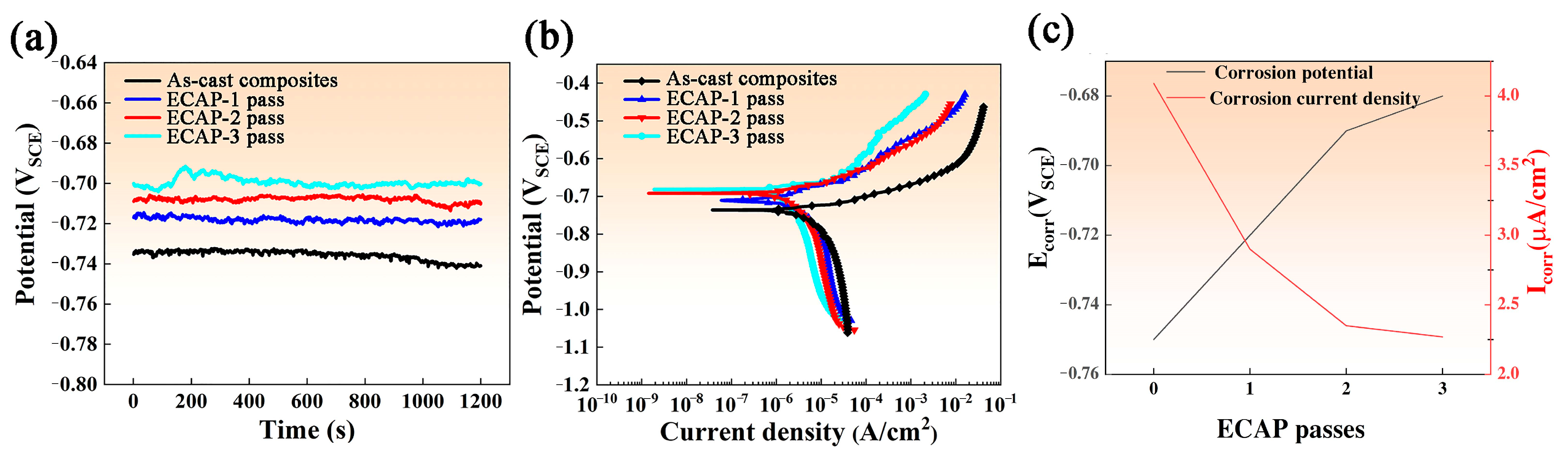

| Specimen | Rs (Ω·cm2) | Rct (Ω·cm2) | CPE-T (F·cm−2) | n |
|---|---|---|---|---|
| As-cast composites | 26.07 | 7274 | 1.75 × 10−5 | 0.87 |
| ECAP 1-pass | 18.84 | 14,253 | 4.91 × 10−6 | 0.83 |
| ECAP 2-pass | 22.23 | 20,199 | 8.24 × 10−6 | 0.86 |
| ECAP 3-pass | 21.86 | 24,584 | 8.85 × 10−6 | 0.85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.; Hu, W.; Hu, Q.; Liu, Z.; Yan, H. High Corrosion Resistance of Ti3C2Tx/Al6061 Composites Achieved via Equal Channel Angular Pressing. Metals 2025, 15, 954. https://doi.org/10.3390/met15090954
Chen J, Hu W, Hu Q, Liu Z, Yan H. High Corrosion Resistance of Ti3C2Tx/Al6061 Composites Achieved via Equal Channel Angular Pressing. Metals. 2025; 15(9):954. https://doi.org/10.3390/met15090954
Chicago/Turabian StyleChen, Jianchao, Wenjie Hu, Qihong Hu, Zhibin Liu, and Hong Yan. 2025. "High Corrosion Resistance of Ti3C2Tx/Al6061 Composites Achieved via Equal Channel Angular Pressing" Metals 15, no. 9: 954. https://doi.org/10.3390/met15090954
APA StyleChen, J., Hu, W., Hu, Q., Liu, Z., & Yan, H. (2025). High Corrosion Resistance of Ti3C2Tx/Al6061 Composites Achieved via Equal Channel Angular Pressing. Metals, 15(9), 954. https://doi.org/10.3390/met15090954






