Effect of Heat Treatment on the Corrosion Resistance of AlFeCoNiMo0.2 High-Entropy Alloy in NaCl and H2SO4 Solutions
Abstract
1. Introduction
2. Materials and Methods
3. Experiments and Results
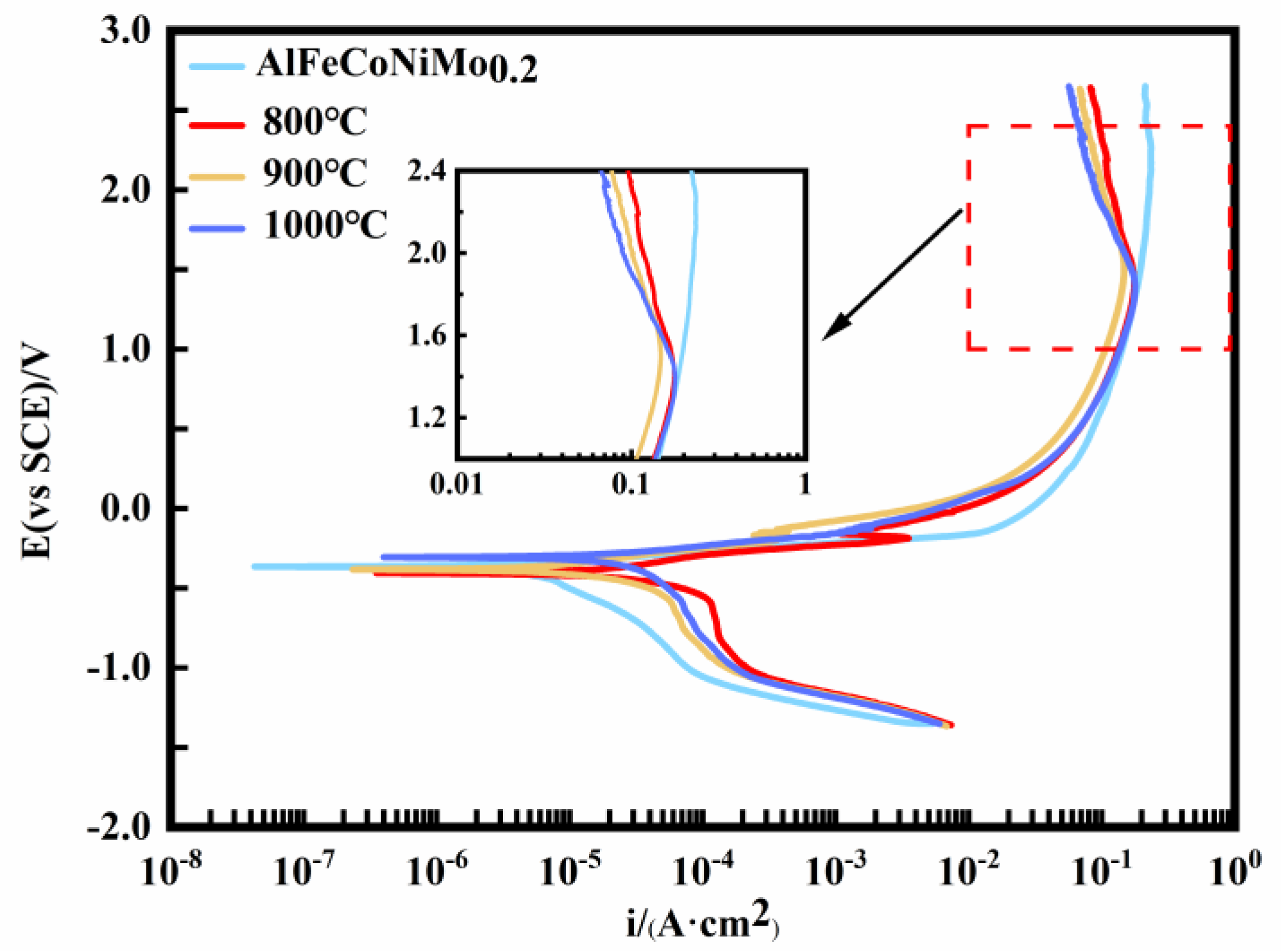
3.1. AlFeCoNiMo0.2 High-Entropy Alloy Corrosion Morphology and Electrochemical Behavior in 3.5 wt% NaCl Solution
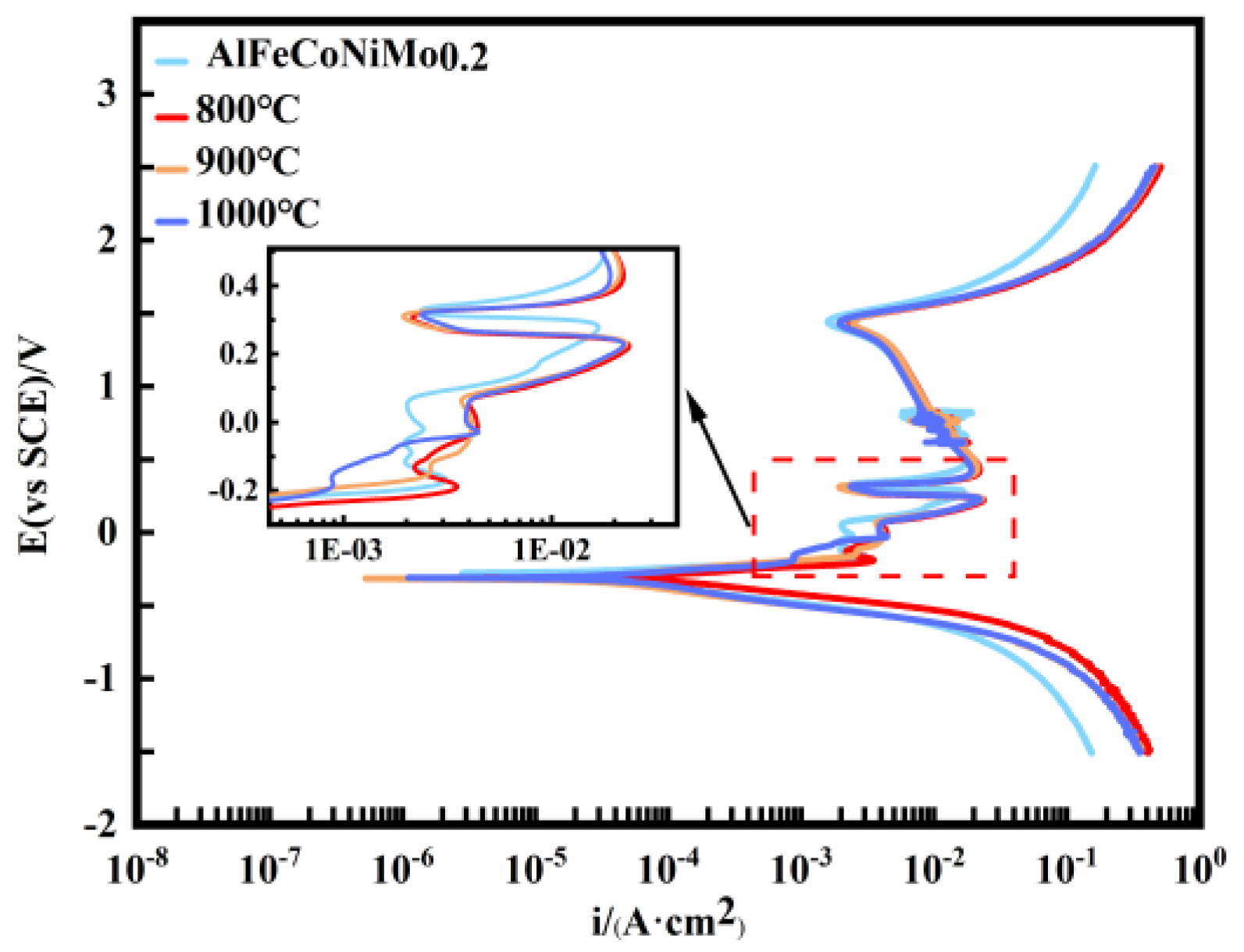
3.2. AlFeCoNiMo0.2 High-Entropy Alloy Corrosion Morphology and Electrochemical Behavior in 0.5 M H2SO4 Solution
4. Analysis and Discussion
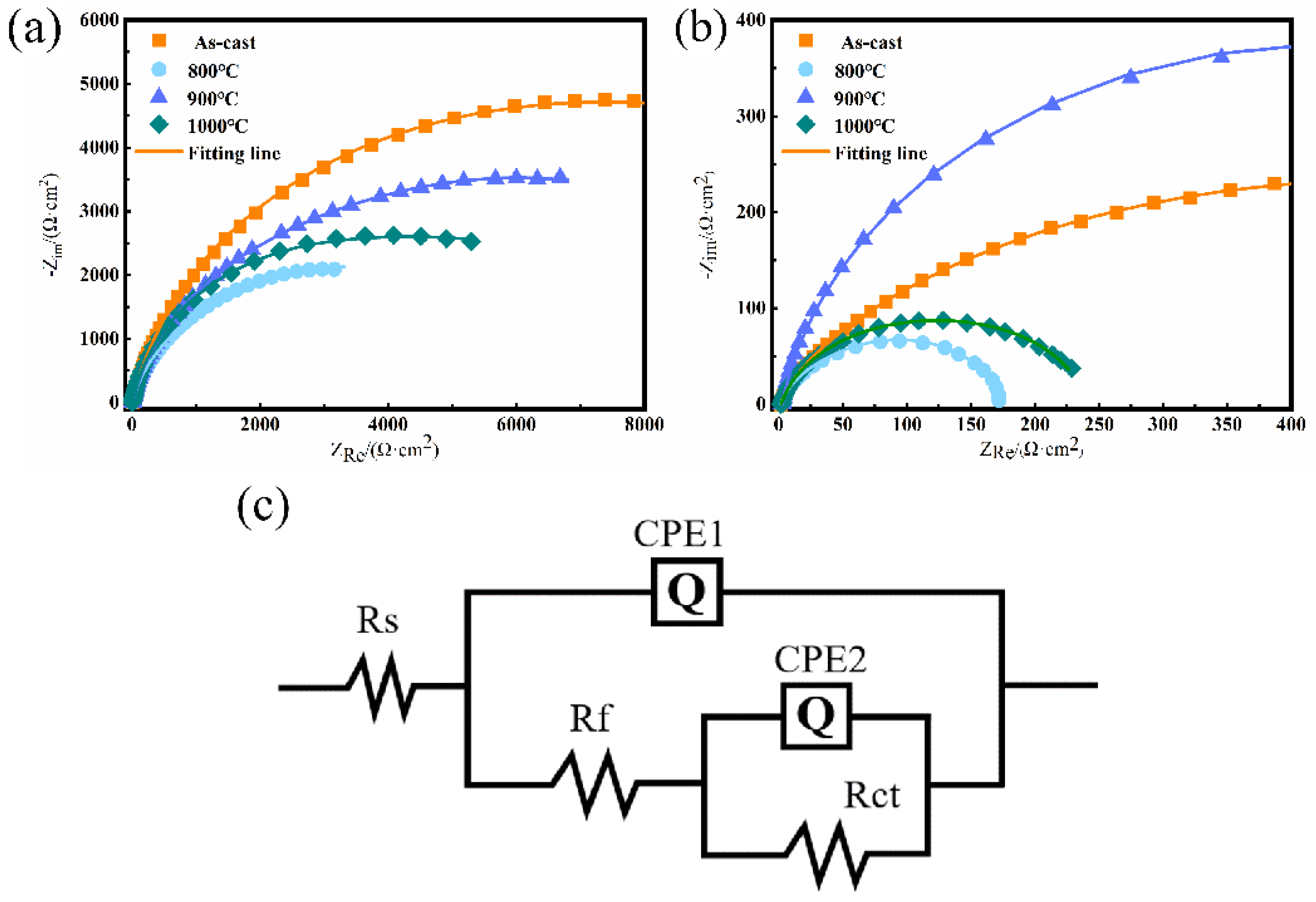
4.1. Electrochemical Impedance Spectroscopy (EIS) Analysis
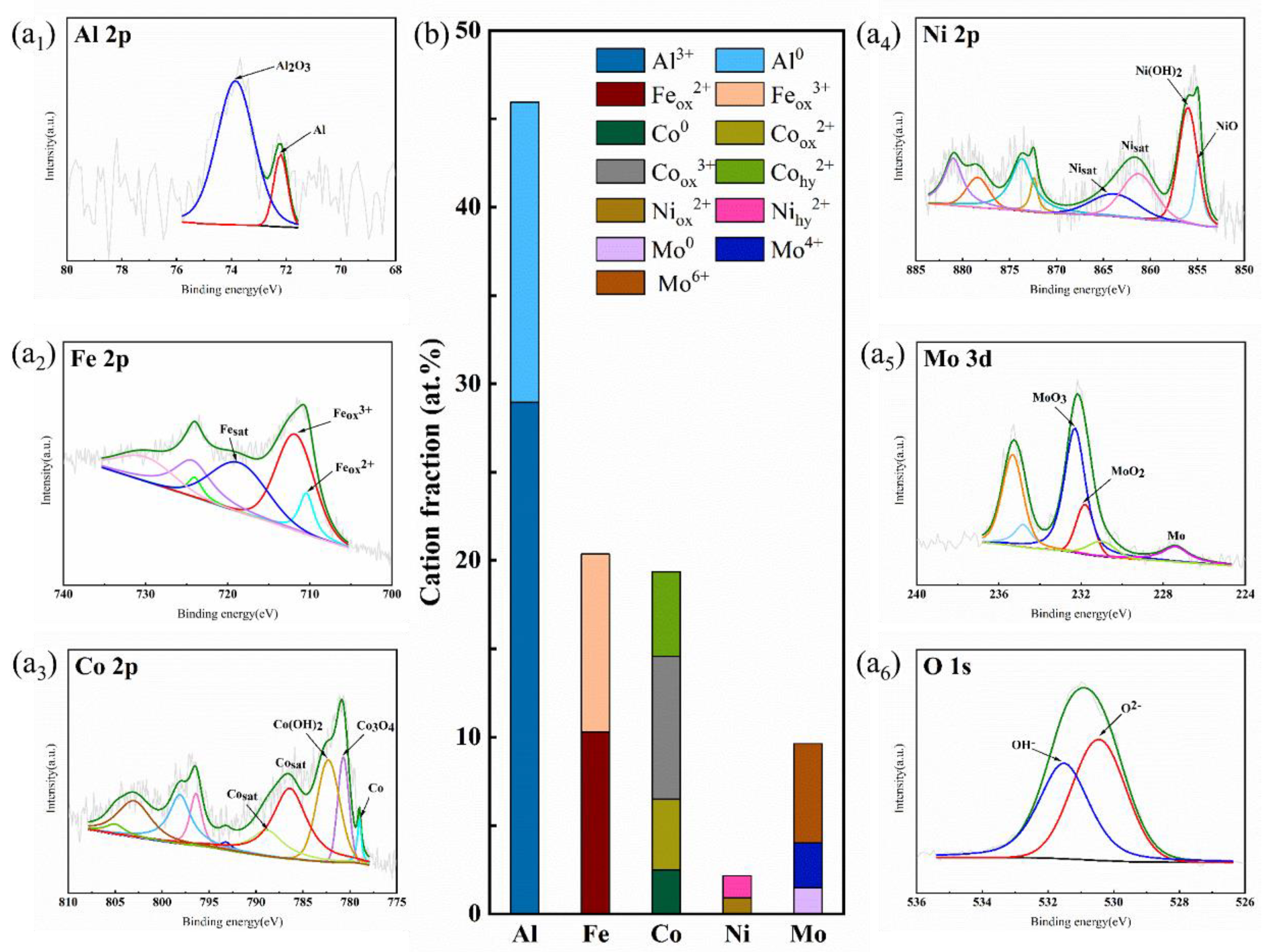
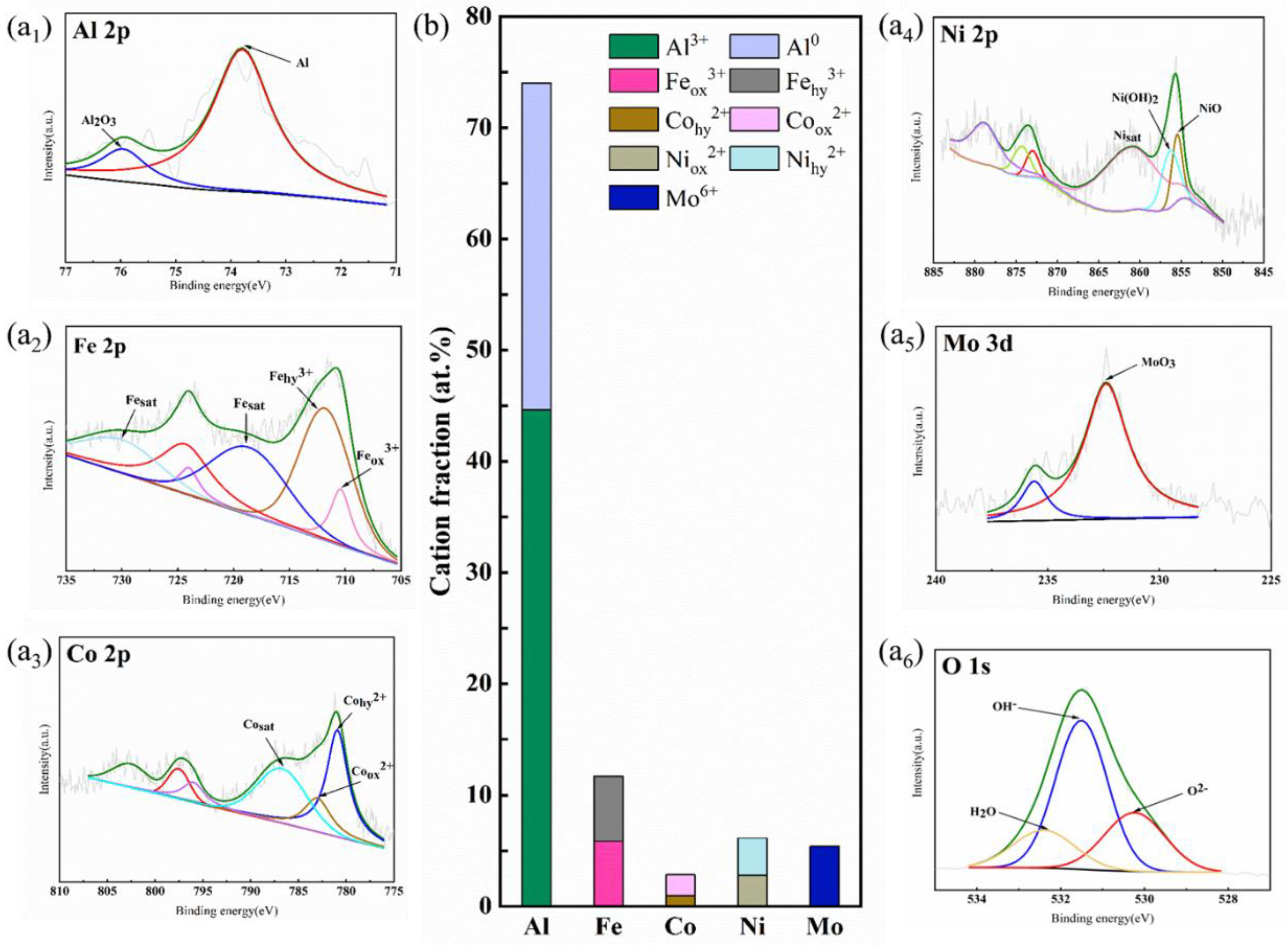
4.2. XPS Analysis of Alloy Passive Films
5. Conclusions
- In the Cl− environment, the AlFeCoNiMo0.2 high-entropy alloy has a much lower corrosion current density in the as-cast state and significantly higher corrosion resistance than the annealed alloy. The reason for this is mainly due to the severe segregation of elements Al and Mo in the grain and grain boundaries after annealing, which enhances the occurrence of galvanic corrosion and leads to pitting corrosion in the grain;
- In the SO42− environment, the 900 °C annealed state AlFeCoNiMo0.2 high-entropy alloy has better corrosion resistance than other alloys. The electrochemical test results show that the alloys all undergo secondary passivation, producing two successive product films to protect the metal substrate. The corrosion morphology shows that the priority corrosion areas are mainly concentrated in the grain boundaries and nearby dendritic areas, mainly due to the enrichment of Mo elements at the grain boundaries of the alloy after annealing. However, Mo reduces the corrosion resistance of the alloy in sulphuric acid solution;
- XPS results of cast AlFeCoNiMo0.2 after constant potential polarization in 3.5 wt% NaCl solution show that Al, Fe, Co, and Mo are mainly present on the alloy surface as oxides and Ni as oxides and hydroxides. The component Al in the alloy is an active metal and will form complexes with halogen atoms. The Cl− ions in the solution will destroy the dense film structure of Al2O3, making Al3+ more soluble in solution and causing corrosive dissolution of the passive film, resulting in no significant passivation of AlFeCoNiMo0.2 in the Cl− solution;
- XPS results of cast AlFeCoNiMo0.2 after constant potential polarization in 0.5 mol/L H2SO4 solution show that Al and Mo are mainly present on the alloy surface as oxides; Fe, Co, and Ni are present on the alloy surface as oxides and hydroxides; and the passive film is an oxide film with a high mixture of multi-element hydroxides and oxides. The Mo element in the passive film inhibits the activation of Fe to dissolve into iron oxide and generate the protective component MoO3 in the passive film, which inhibits the dissolution of the alloy and can improve the stability of the passive film.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yeh, J.W.; Chen, S.K.; Lin, S.J.; Gan, J.Y.; Chin, T.S.; Shun, T.T.; Tsau, C.H.; Chang, S.Y. Nanostructured high-entropy alloys with multiple principal elements: Novel alloy design concepts and outcomes. Adv. Eng. Mater. 2004, 6, 299–303. [Google Scholar] [CrossRef]
- Cantor, B.; Chang, I.T.H.; Knight, P.; Vincent, A.J.B. Microstructural development in equiatomic multicomponent alloys. Mat. Sci. Eng. A 2004, 375, 213–218. [Google Scholar] [CrossRef]
- Tsai, M.H.; Yeh, J.W. High-entropy alloys: A critical review. Mater. Res. Lett. 2014, 2, 107–123. [Google Scholar] [CrossRef]
- Ye, Y.F.; Wang, Q.; Lu, J.; Liu, C.T.; Yang, Y. High-entropy alloy: Challenges and prospects. Mater. Today 2016, 19, 349–362. [Google Scholar] [CrossRef]
- Xiao, Y.; Zou, Y.; Ma, H.; Sologubenko, A.S.; Maeder, X.; Spolenak, R.; Wheeler, J.M. Nanostructured NbMoTaW high entropy alloy thin films: High strength and enhanced fracture toughness. Scr. Mater. 2019, 168, 51–55. [Google Scholar] [CrossRef]
- Li, D.; Zhang, Y. The ultrahigh charpy impact toughness of forged AlxCoCrFeNi high entropy alloys at room and cryogenic temperatures. Intermetallics 2016, 70, 24–28. [Google Scholar] [CrossRef]
- Ma, S.G. Creep resistance and strain-rate sensitivity of a CoCrFeNiAl0.3 high-entropy alloy by nanoindentation. Mater. Res. Express 2019, 6, 126508. [Google Scholar] [CrossRef]
- Cao, T.; Chang, J.; Zhao, J.; Cheng, C.; Wang, R.; Wang, H. The influence of Al elements on the structure and the creep behavior of AlxCoCrFeNi high entropy alloys. Mater. Lett. 2016, 164, 344–347. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Yeh, J.W.; Chen, S.K.; Shun, T.T. Wear resistance and high-temperature compression strength of FCC CuCoNiCrAl0.5Fe alloy with boron addition. Metall. Mater. Trans. A 2004, 35, 1465–1469. [Google Scholar] [CrossRef]
- Nene, S.S.; Frank, M.; Liu, K.; Sinha, S.; Mishra, R.S.; McWilliams, B.A.; Cho, K.C. Corrosion-resistant high entropy alloy with high strength and ductility. Scr. Mater. 2019, 166, 168–172. [Google Scholar] [CrossRef]
- Chou, Y.L.; Yeh, J.W.; Shih, H.C. The effect of molybdenum on the corrosion behaviour of the high-entropy alloys Co1.5CrFeNi1.5Ti0.5Mox in aqueous environments. Corros. Sci. 2010, 52, 2571–2581. [Google Scholar] [CrossRef]
- Chou, H.P.; Chang, Y.S.; Chen, S.K.; Yeh, J.W. Microstructure, thermophysical and electrical properties in AlxCoCrFeNi (0 ≤ x ≤ 2) high-entropy alloys. Mat. Sci. Eng. B 2009, 163, 184–189. [Google Scholar] [CrossRef]
- Wang, W.R.; Wang, W.L.; Wang, S.C.; Tsai, Y.C.; Lai, C.H.; Yeh, J.W. Effects of Al addition on the microstructure and mechanical property of AlxCoCrFeNi high-entropy alloys. Intermetallics 2012, 26, 44–51. [Google Scholar] [CrossRef]
- Kao, Y.F.; Chen, S.K.; Chen, T.J.; Chu, P.C.; Yeh, J.W.; Lin, S.J. Electrical, magnetic, and hall properties of AlxCoCrFeNi high-entropy alloys. J. Alloys Compd. 2011, 509, 1607–1614. [Google Scholar] [CrossRef]
- Yang, H.O.; Shang, X.L.; Wang, L.L.; Wang, Z.J.; Wang, J.C.; Lin, X. Effect of constituent elements on the corrosion resistance of single-phase CoCrFeNi high-entropy alloys in NaCl solution. Acta Metall. Sin. 2018, 54, 905–910. [Google Scholar]
- Niu, Z.Z.; Wang, Y.Z.; Geng, C.; Xu, J.; Wang, Y. Microstructural evolution, mechanical and corrosion behaviors of as-annealed CoCrFeNiMox (x = 0, 0.2, 0.5, 0.8, 1) high entropy alloys. J. Alloys Compd. 2020, 820, 153273. [Google Scholar] [CrossRef]
- Wang, W.; Wang, J.; Sun, Z.H.; Li, J.T.; Li, L.F.; Song, X.; Wen, X.D.; Xie, L.; Yang, X. Effect of Mo and aging temperature on corrosion behavior of (CoCrFeNi)100−xMox high-entropy alloys. J. Alloys Compd. 2020, 812, 152139. [Google Scholar] [CrossRef]
- Lin, C.M.; Tsai, H.L.; Bor, H.Y. Effect of aging treatment on microstructure and properties of high-entropy Cu0.5CoCrFeNi alloy. Intermetallics 2010, 18, 1244–1250. [Google Scholar] [CrossRef]
- Wen, X.; Jin, G.; Pang, X.J.; Cai, Z.B.; Zhang, Z.H.; Cui, X.F.; Wang, H.D.; Xu, B.S. Effect of heat treatment on microstructure and corrosion resistance of NiCrCoTiV high-entropy alloy prepared by vacuum hot-pressing sintering. Mater. Rep. B 2017, 31, 79–83. [Google Scholar]
- Zhang, X.; Guo, J.; Zhang, X.; Song, Y.; Li, Z.; Xing, X.; Kong, D. Influence of remelting and annealing treatment on corrosion resistance of AlFeNiCoCuCr high entropy alloy in 3.5wt% NaCl solution. J. Alloys Compd. 2018, 775, 565–570. [Google Scholar] [CrossRef]
- Fernandes, D.M.; Montes, R.H.O.; Almeida, E.S.; Nascimento, A.N.; Oliveira, P.V.; Richter, E.M.; Munoz, R.A.A. Storage stability and corrosive character of stabilised biodiesel exposed to carbon and galvanised steels. Fuel 2013, 107, 609–614. [Google Scholar] [CrossRef]
- Sugimoto, K.; Sawada, Y. The role of molybdenum additions to austenitic stainless steels in the inhibition of pitting in acid chloride solutions. Corros. Sci. 1997, 17, 425–445. [Google Scholar] [CrossRef]
- Lu, P.; Saal, J.E.; Olson, G.B.; Li, T.S.; Sahu, S.; Swanso, O.J.; Frankel, G.S.; Gerard, A.; Scully, J.R. Computational design and initial corrosion assessment of a series of non-equimolar high entropy alloys. Scr. Mater. 2019, 172, 12–16. [Google Scholar] [CrossRef]
- Ming, J.; Wu, M.; Shi, J.J. Passive film modification by concrete carbonation: Re-visiting a corrosion-resistant steel with Cr and Mo. Cement. Concr. Comp. 2021, 123, 104178. [Google Scholar] [CrossRef]
- Olsson, C.O.A.; Landolt, D. Passive films on stainless steels—Chemistry, structure and growth. Electrochim. Acta 2003, 48, 1093–1104. [Google Scholar] [CrossRef]
- Jiang, R.J.; Wang, Y.W.; Wen, X.; Chen, C.F.; Zhao, J.M. Effect of time on the characteristics of passive film formed on stainless steel. Appl. Surf. Sci. 2017, 412, 214–222. [Google Scholar] [CrossRef]
- Abdallah, M.; Salem, M.M.; Zaafarany, I.A. Corrosion performance of stainless steel and nickel alloys in aqueous sodium hydroxide as revealed from cyclic voltammetry and potentiodynamic anodic polarization. Orient. J. Chem. 2017, 33, 2875–2883. [Google Scholar] [CrossRef]
- Pourbaix, M. Atlas of electrochemical equilibria in aqueous solutions. Electroanal. Chem. 1967, 13, 471. [Google Scholar]
- Aliyu, A.; Srivastava, C. Corrosion behavior and protective film constitution of AlNiCoFeCu and AlCrNiCoFeCu high entropy alloy coatings. Surf. Interfaces 2021, 27, 101481. [Google Scholar] [CrossRef]
- Nascimento, C.B.; Donatus, U.; Ríos, C.T.; Antunes, R.A. Passive film composition and stability of CoCrFeNi and CoCrFeNiAl high entropy alloys in chloride solution. Mater. Chem. Phys. 2021, 267, 124582. [Google Scholar] [CrossRef]
- Luo, H.; Zou, S.W.; Chen, Y.H.; Li, Z.M.; Du, C.W.; Li, X.G. Influence of carbon on the corrosion behaviour of interstitial equiatomic CoCrFeMnNi high-entropy alloys in a chlorinated concrete solution. Corros. Sci. 2020, 163, 108287. [Google Scholar] [CrossRef]
- Okamoto, G. Passive film of 18-8 stainless steel structure and its function. Corros. Sci. 1973, 13, 471–489. [Google Scholar] [CrossRef]
- Dai, C.D.; Zhao, T.L.; Du, C.W.; Liu, Z.Y.; Zhang, D.W. Effect of molybdenum content on the microstructure and corrosion behavior of FeCoCrNiMox high-entropy alloys. J. Mater. Sci. Technol. 2020, 46, 64–73. [Google Scholar] [CrossRef]
- Falkenberg, F.; Raja, V.S.; Ahlberg, E. An alternative explanation for the apparent passivation of molybdenum in 1 mol/L hydrochloric acid. J. Electrochem. Soc. 2001, 148, B132. [Google Scholar] [CrossRef]
- Hashimoto, K.; Asami, K.; Kawashima, A.; Habazaki, H.; Akiyama, E. The role of corrosion-resistant alloying elements in passivity. Corros. Sci. 2007, 49, 42–52. [Google Scholar] [CrossRef]




| Al | Fe | Co | Ni | Mo |
|---|---|---|---|---|
| 12.28 | 25.40 | 26.86 | 26.721 | 8.78 |
| Material | Temperature (°C) | Ecorr (mV) | Icorr (μA·cm−2) |
|---|---|---|---|
| AlFeCoNiMo0.2 | as-cast | −337.83 (±4.88) | 3.90 (±0.31) |
| 800 °C | −347.87 (±4.21) | 31.07 (±0.48) | |
| 900 °C | −325.91 (±4.24) | 13.92 (±0.44) | |
| 1000 °C | −310.15 (±3.92) | 22.48 (±0.39) |
| Material | Temperature (°C) | Ecorr (mV) | Icorr (μA·cm−2) |
|---|---|---|---|
| AlFeCoNiMo0.2 | as-cast | −275.33 (±3.81) | 20.58 (±0.81) |
| 800 °C | −304.64 (±4.02) | 51.73 (±1.01) | |
| 900 °C | −305.08 (±3.96) | 17.04 (±0.76) | |
| 1000 °C | −293.88 (±4.18) | 27.23 (±0.98) |
| Condition | Solution | Rs (ohm) | Rf (ohm) | Rct (ohm) | CPE1 | CPE2 | ||
|---|---|---|---|---|---|---|---|---|
| Y1 (F) | n1 | Y2 (F) | n2 | |||||
| As-cast | 3.5 wt% NaCl | 8.098 | 9656 | 5448 | 1.039 × 10−4 | 0.8583 | 5.469 × 10−4 | 0.7459 |
| 800-1 h | 7.751 | 1944 | 5762 | 7.516 × 10−5 | 0.8778 | 1.558 × 10−4 | 0.4889 | |
| 900-1 h | 10.05 | 5630 | 6143 | 8.886 × 10−5 | 0.8614 | 2.941 × 10−4 | 0.6862 | |
| 1000-1 h | 8.041 | 5438 | 3527 | 8.641 × 10−5 | 0.8769 | 6.602 × 10−4 | 0.7862 | |
| Condition | Solution | Rs (ohm) | Rf (ohm) | Rct (ohm) | CPE1 | CPE2 | ||
|---|---|---|---|---|---|---|---|---|
| Y1 (F) | n1 | Y2 (F) | n2 | |||||
| As-cast | 0.5 mol/L H2SO4 | 1.841 | 353.7 | 1063 | 1.051 × 10−4 | 0.9341 | 8.139 × 10−4 | 0.8713 |
| 800-1 h | 1.857 | 117 | 55.63 | 3.804 × 10−4 | 0.8675 | 6.648 × 10−4 | 0.9155 | |
| 900-1 h | 2.705 | 866.1 | 22.85 | 2.62 × 10−4 | 0.9074 | 2.807 × 10−2 | 0.9787 | |
| 1000-1 h | 3.193 | 208.6 | 31.18 | 2.938 × 10−4 | 0.8456 | 2.538 × 10−4 | 0.9586 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, Y.; Zhou, G.; Han, J.; Li, J.; Zhang, H.; Zhang, S.; Lin, L.; Chen, L.; Cao, X. Effect of Heat Treatment on the Corrosion Resistance of AlFeCoNiMo0.2 High-Entropy Alloy in NaCl and H2SO4 Solutions. Metals 2023, 13, 849. https://doi.org/10.3390/met13050849
Peng Y, Zhou G, Han J, Li J, Zhang H, Zhang S, Lin L, Chen L, Cao X. Effect of Heat Treatment on the Corrosion Resistance of AlFeCoNiMo0.2 High-Entropy Alloy in NaCl and H2SO4 Solutions. Metals. 2023; 13(5):849. https://doi.org/10.3390/met13050849
Chicago/Turabian StylePeng, Yuhan, Ge Zhou, Jinke Han, Jianlin Li, Haoyu Zhang, Siqian Zhang, Li Lin, Lijia Chen, and Xue Cao. 2023. "Effect of Heat Treatment on the Corrosion Resistance of AlFeCoNiMo0.2 High-Entropy Alloy in NaCl and H2SO4 Solutions" Metals 13, no. 5: 849. https://doi.org/10.3390/met13050849
APA StylePeng, Y., Zhou, G., Han, J., Li, J., Zhang, H., Zhang, S., Lin, L., Chen, L., & Cao, X. (2023). Effect of Heat Treatment on the Corrosion Resistance of AlFeCoNiMo0.2 High-Entropy Alloy in NaCl and H2SO4 Solutions. Metals, 13(5), 849. https://doi.org/10.3390/met13050849








