Hypereutectic Zn–Al Alloys: Microstructural Development under Unsteady-State Solidification Conditions, Eutectic Coupled Zone and Hardness
Abstract
:1. Introduction
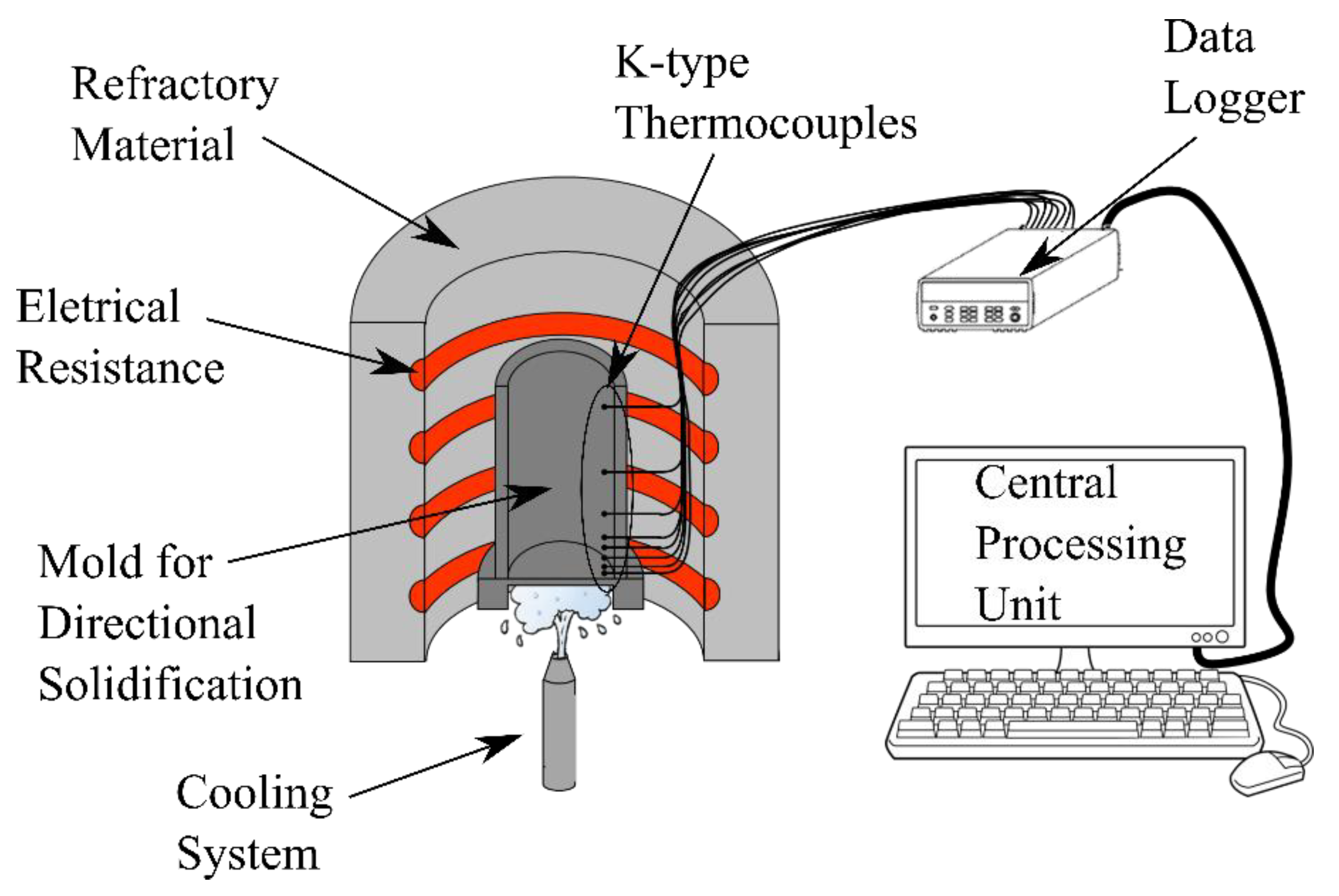
2. Materials and Methods
3. Results and Discussion
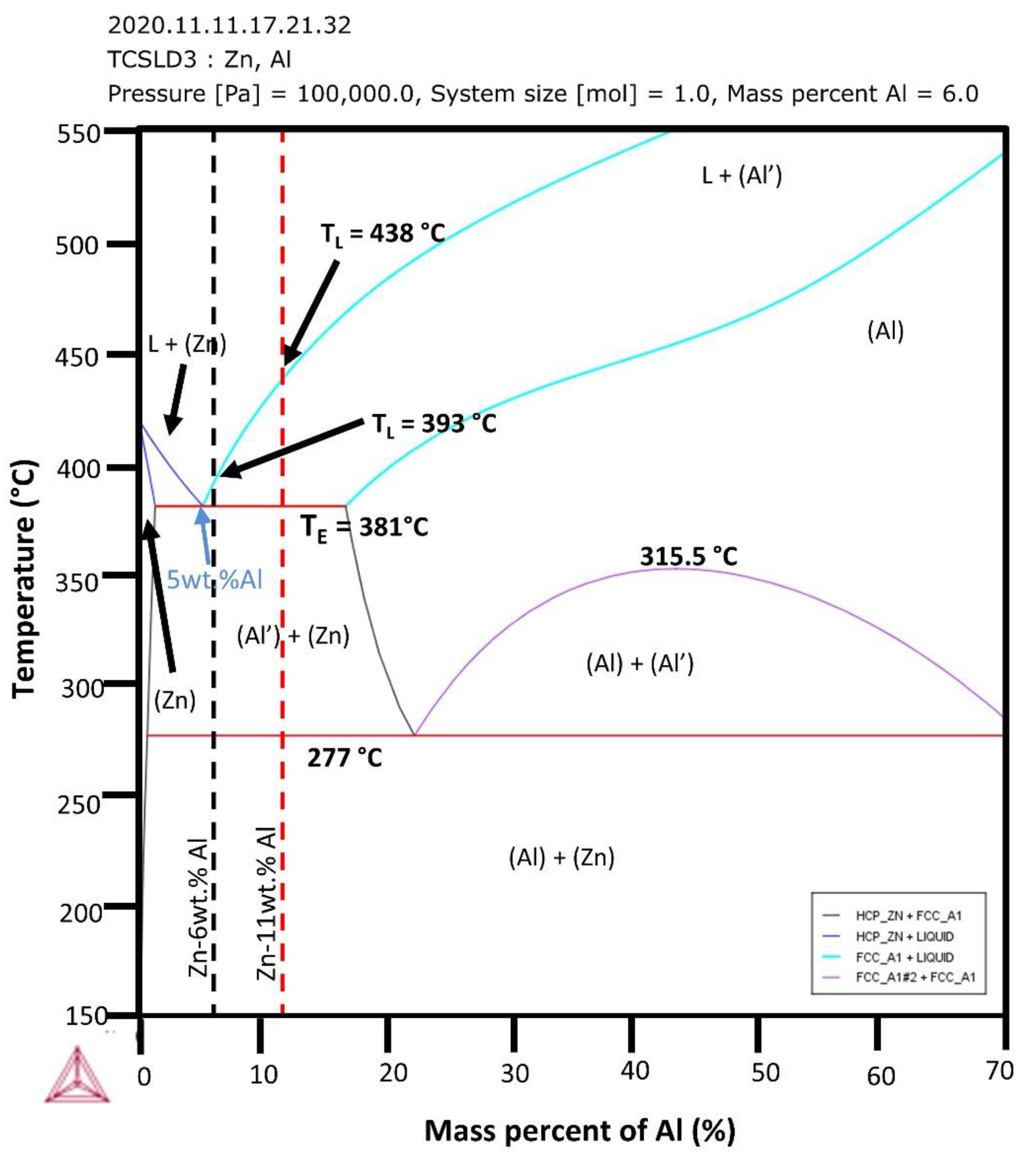
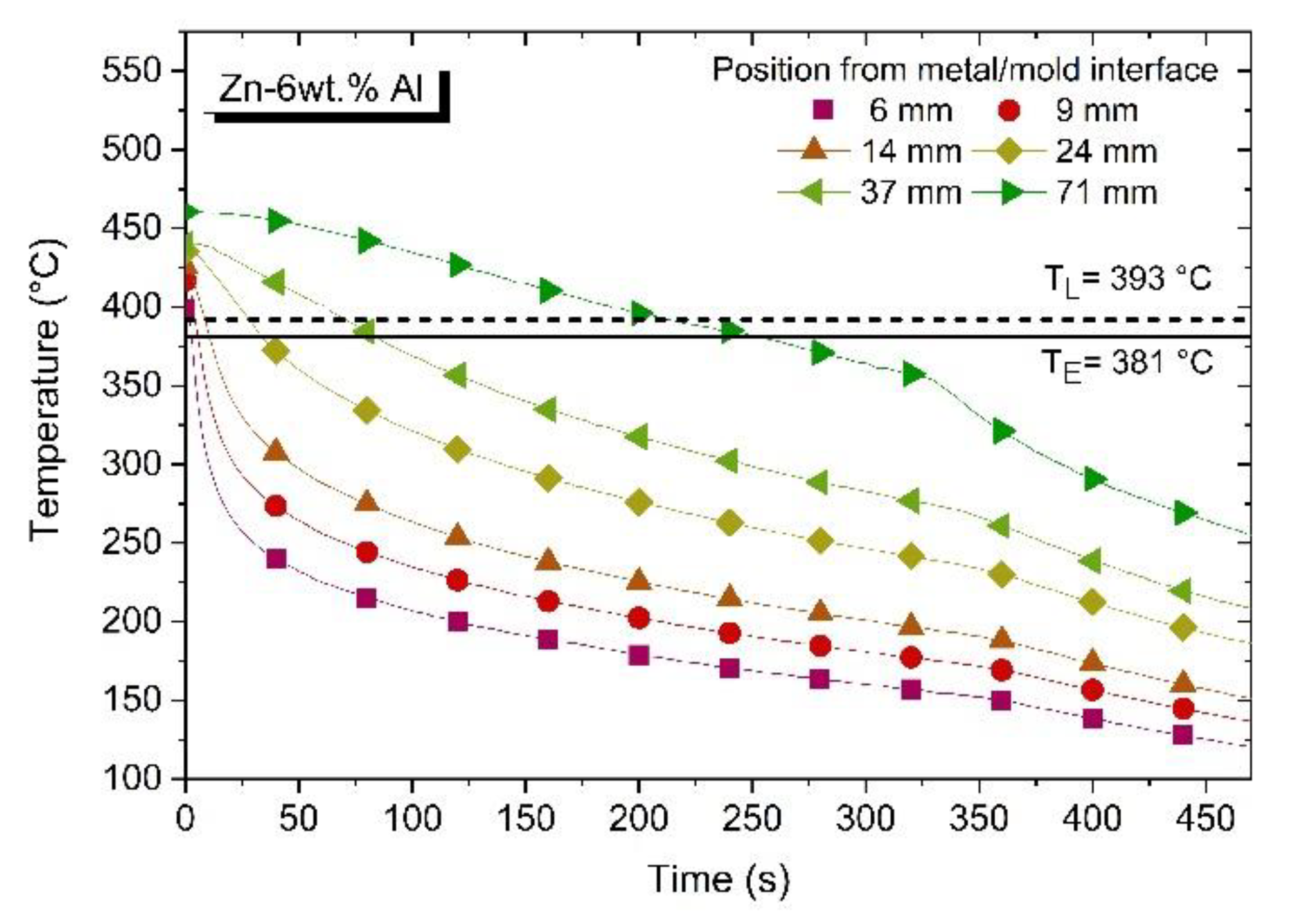
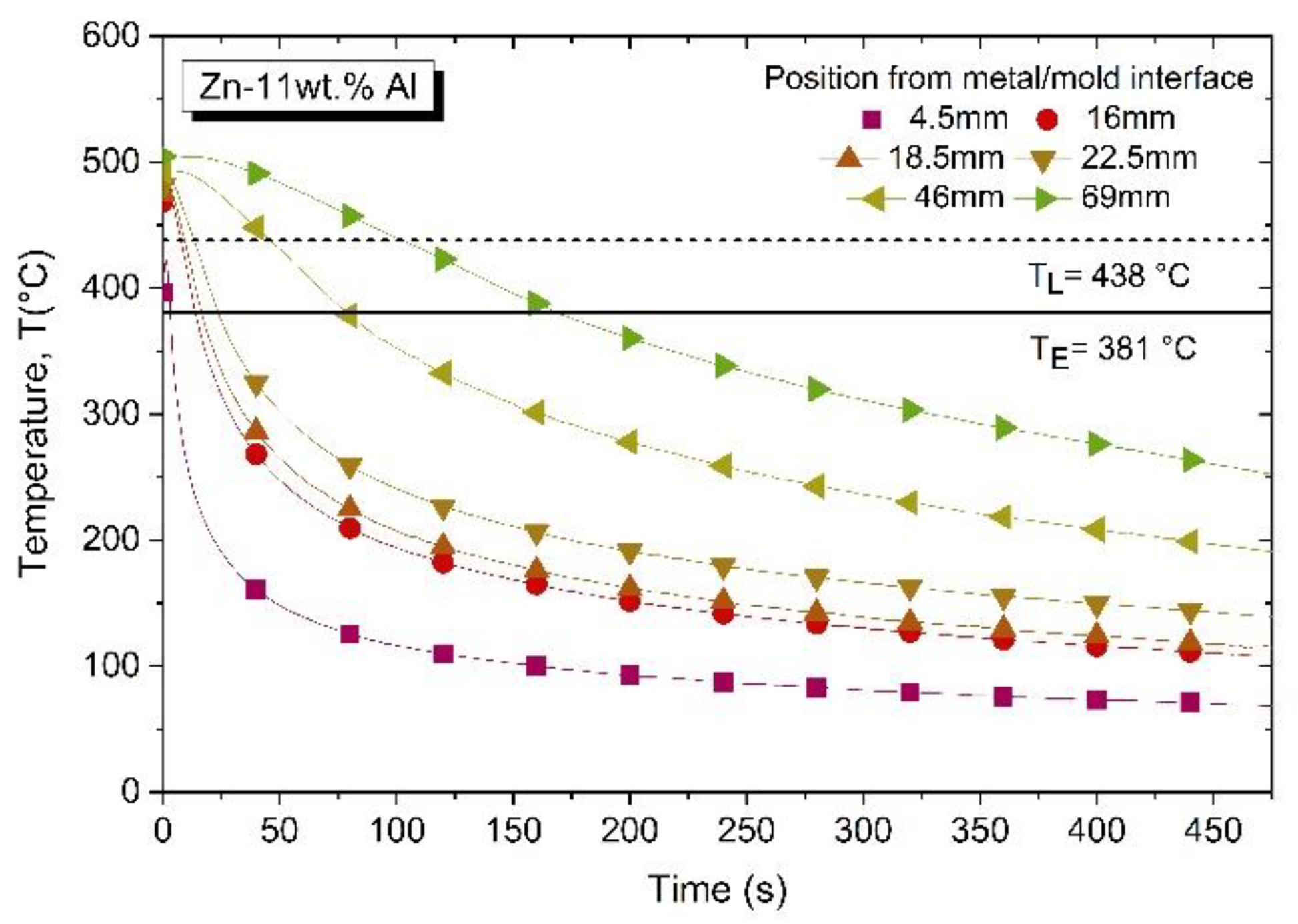
3.1. Solidification Thermal Parameters and Chemical Analysis
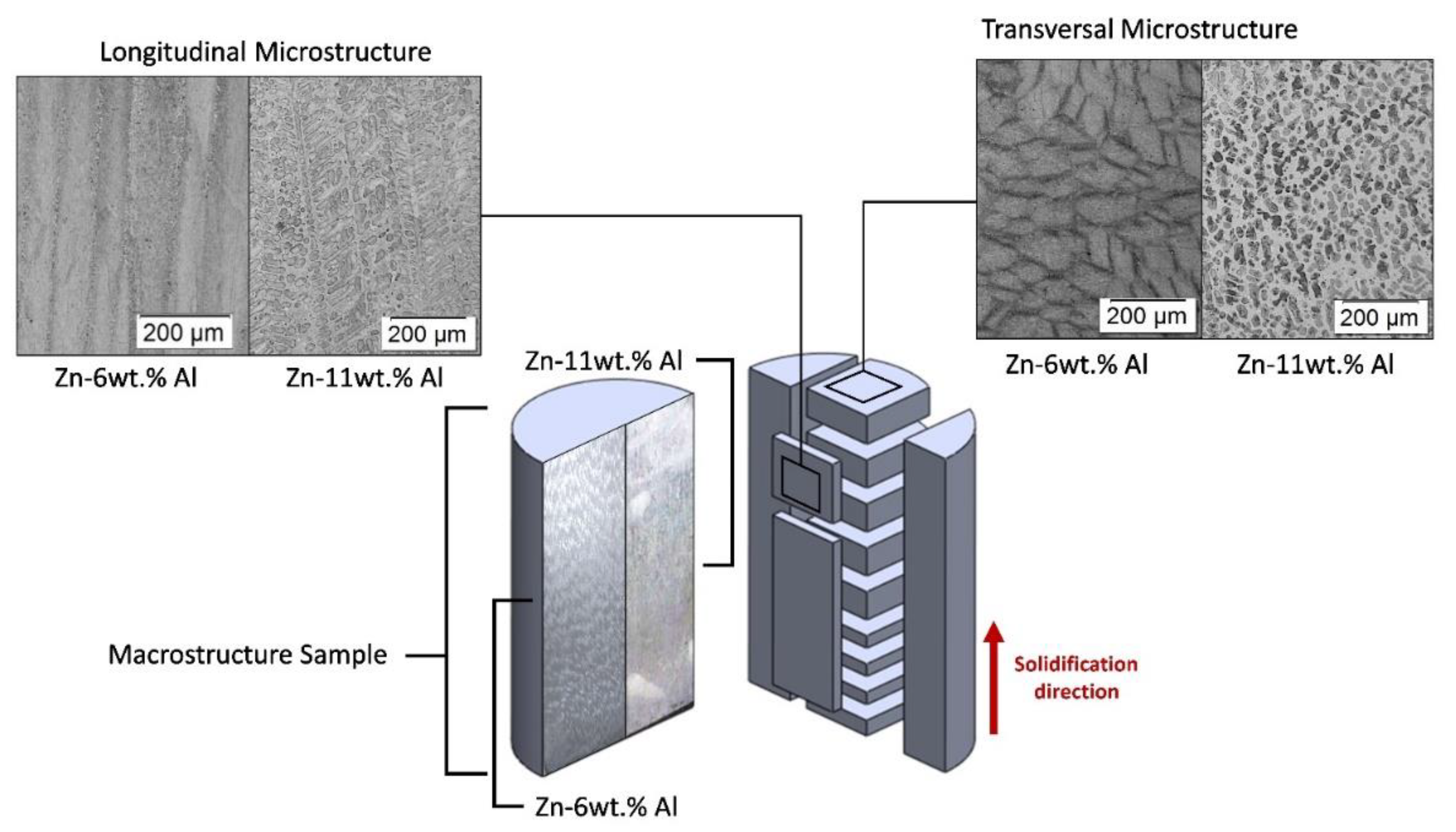
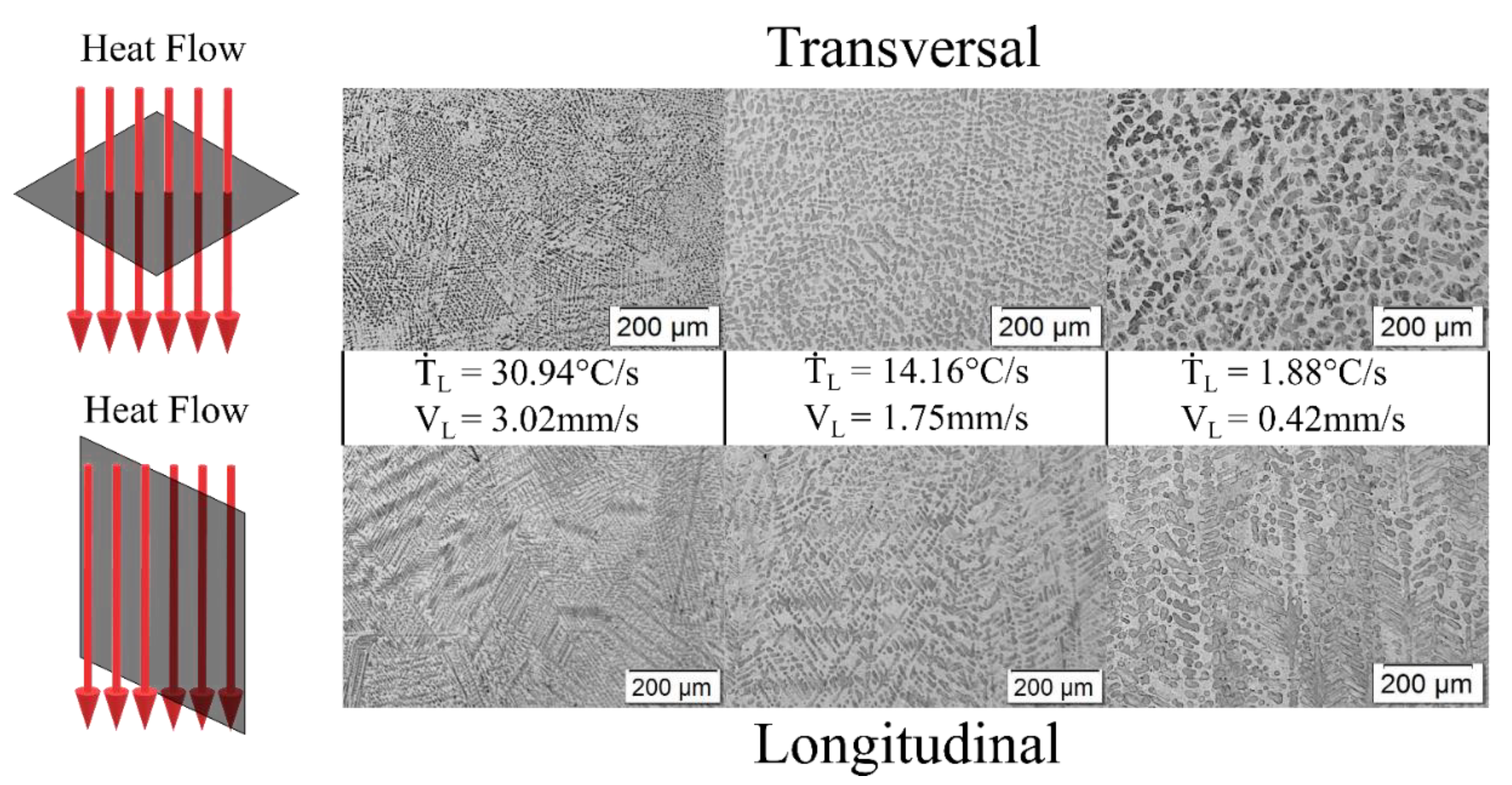
3.2. Microstructural Characterization: Optical Microscopy (OM), Scanning Electron Microscopy (SEM) and X-ray Diffraction (XRD)
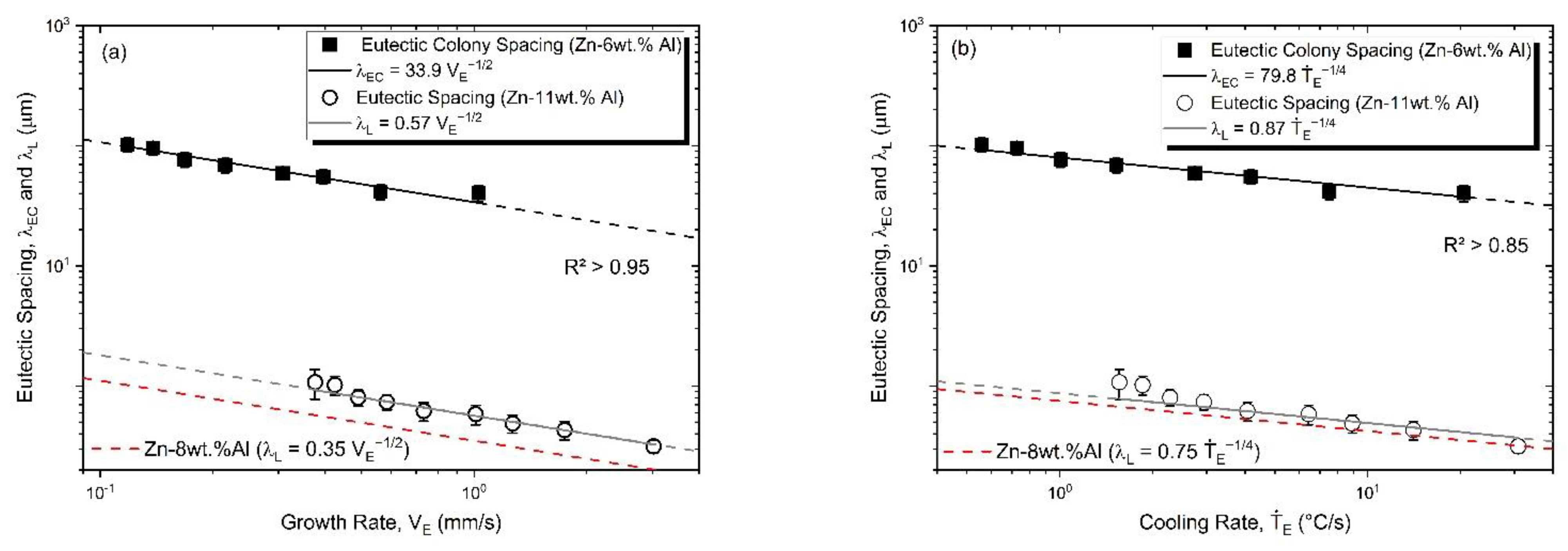
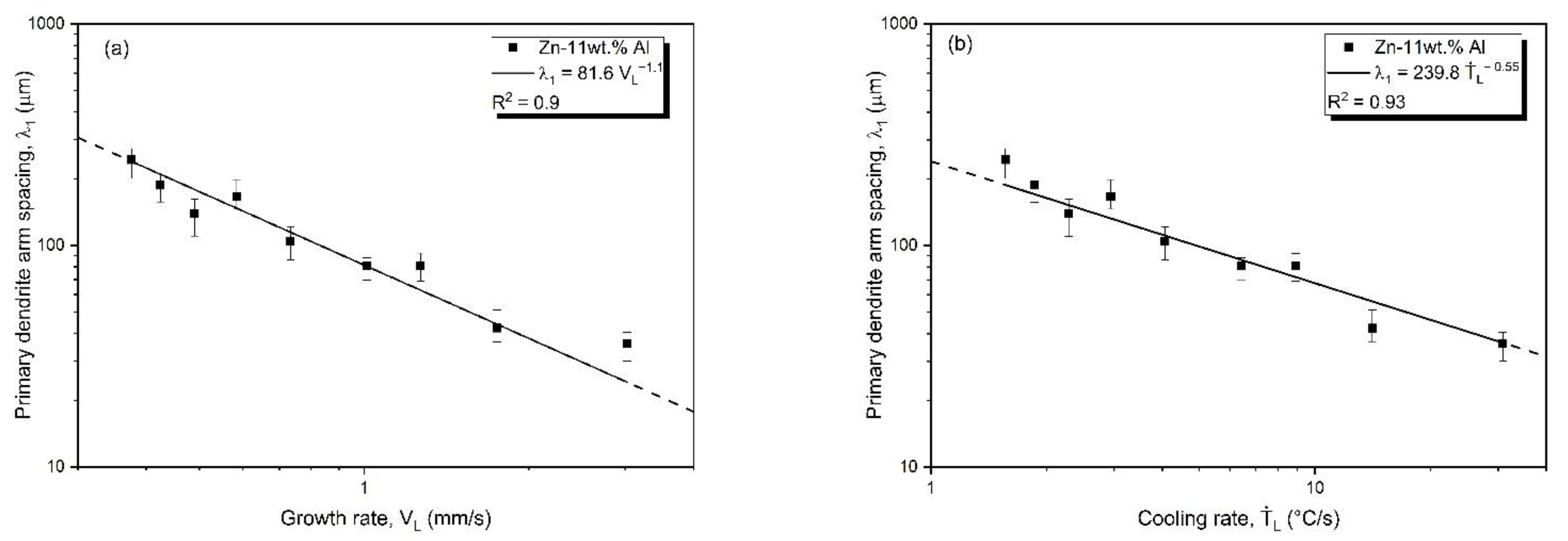
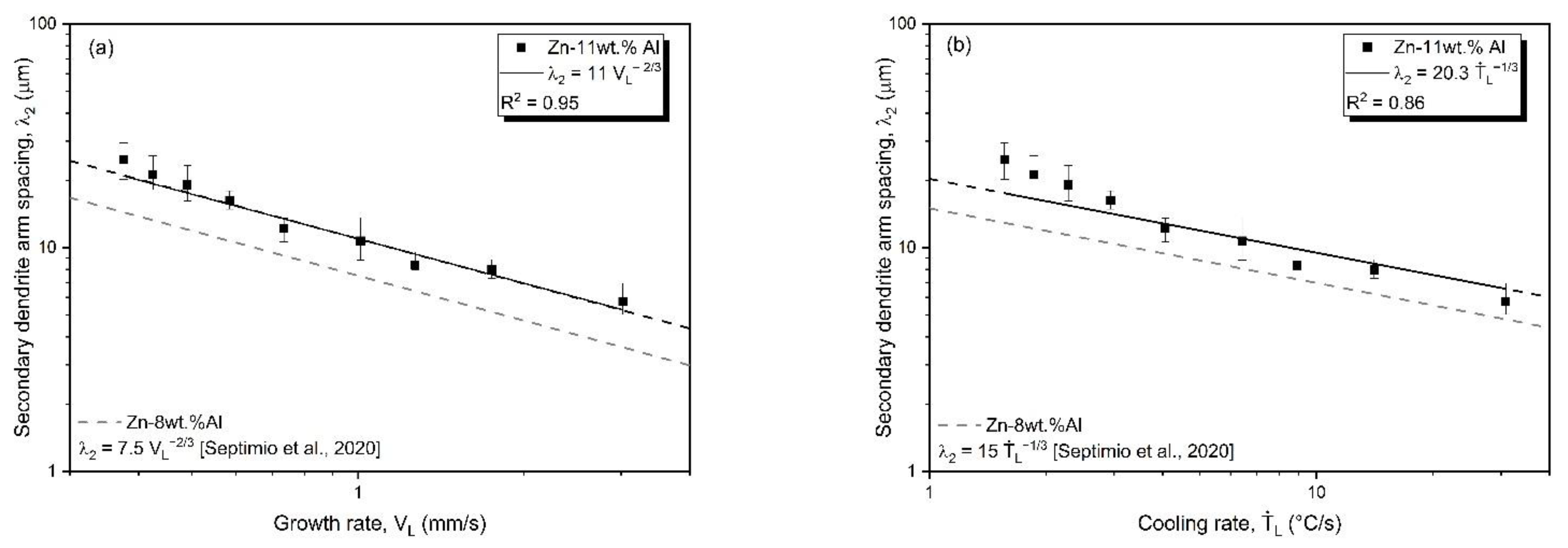
3.3. Microstructural Growth and Correlation with Solidification Thermal Parameters
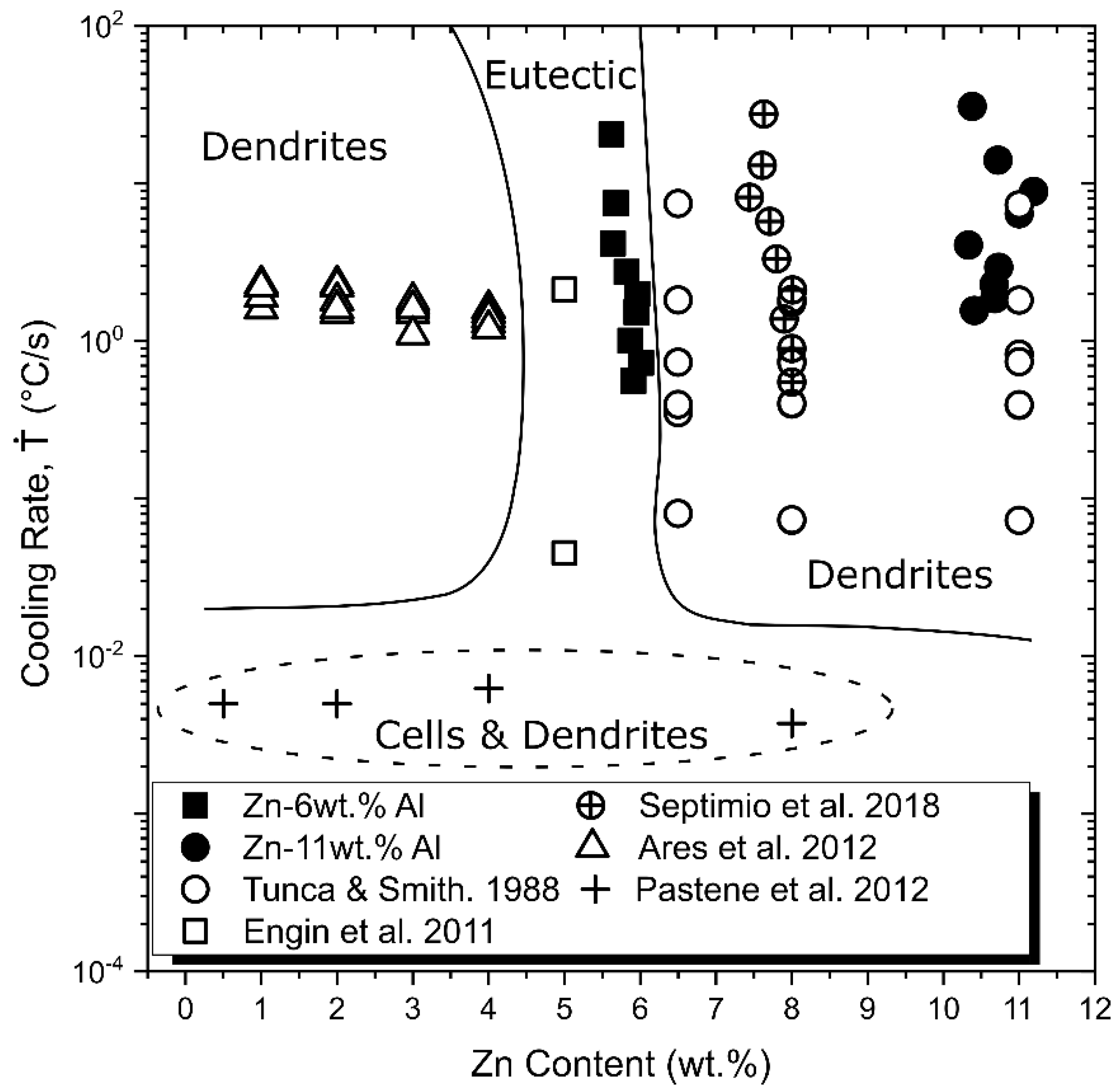
3.4. Coupled Zone
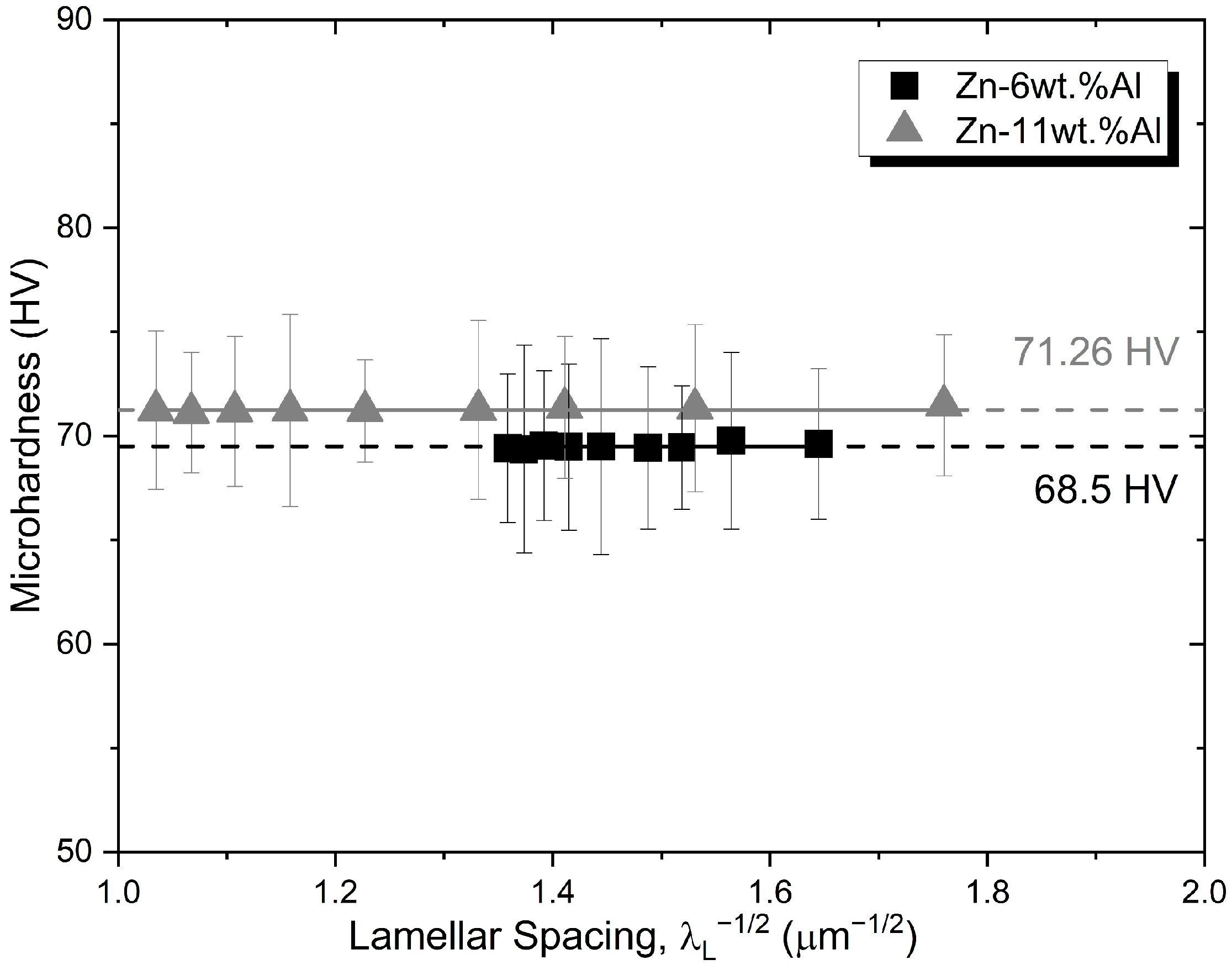
3.5. Vickers Microhardness (HV)
4. Conclusions
- The microstructure of the Zn-6wt.%Al alloy was shown to be characterized by eutectic colonies, constituted by a eutectic mixture of (Zn) and (Al′) phases in the form of lamellae. Since (Al′) is a metastable phase, it undergoes a eutectoid transformation at 277 °C [(Al′)→(Al)→(Zn)]. In contrast, the Zn–11wt.% Al alloy was shown to be characterized by the presence of pro-eutectic (Al′) dendrites and interdendritic regions composed by the eutectic mixture [(Al′) + (Zn)].
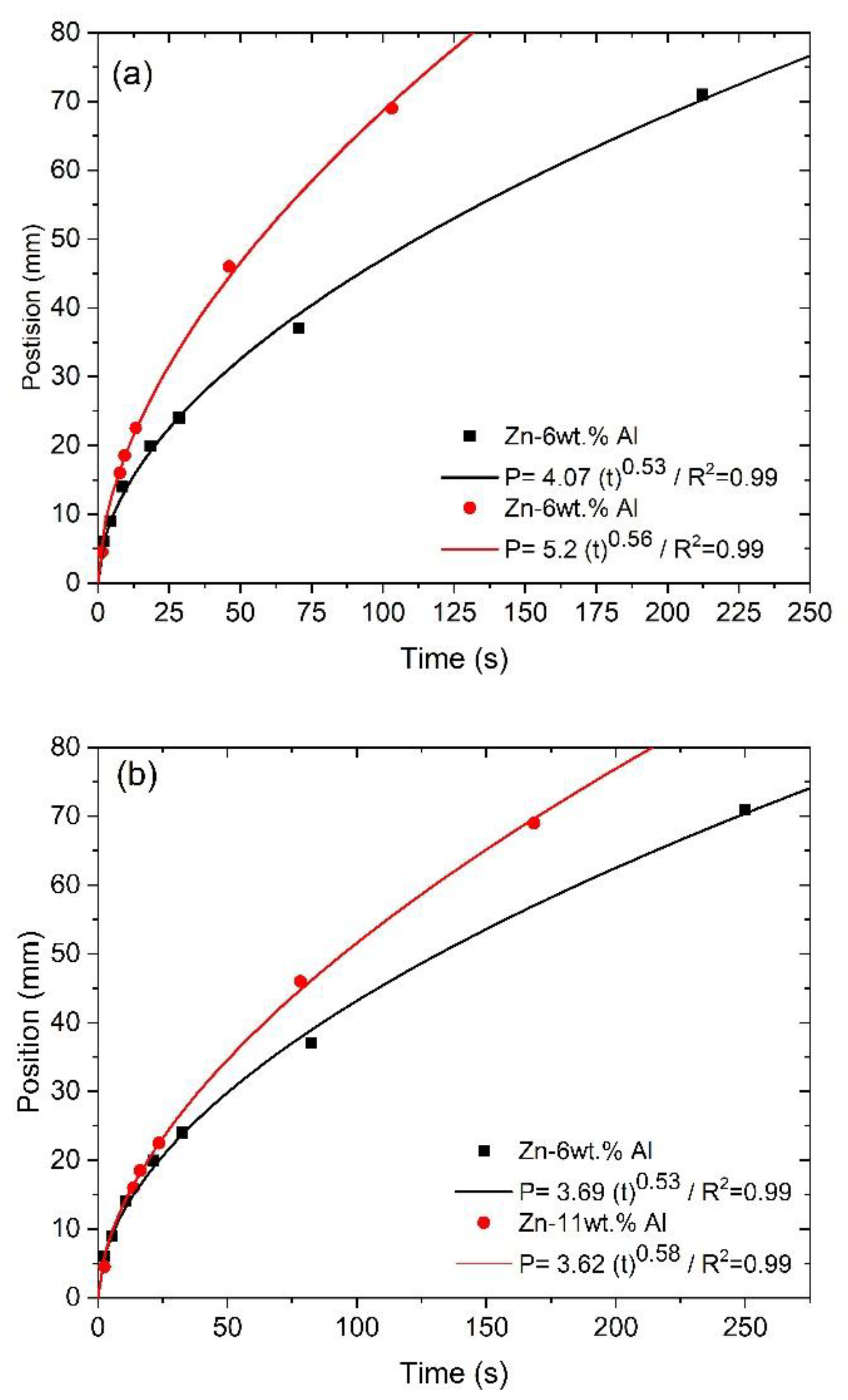
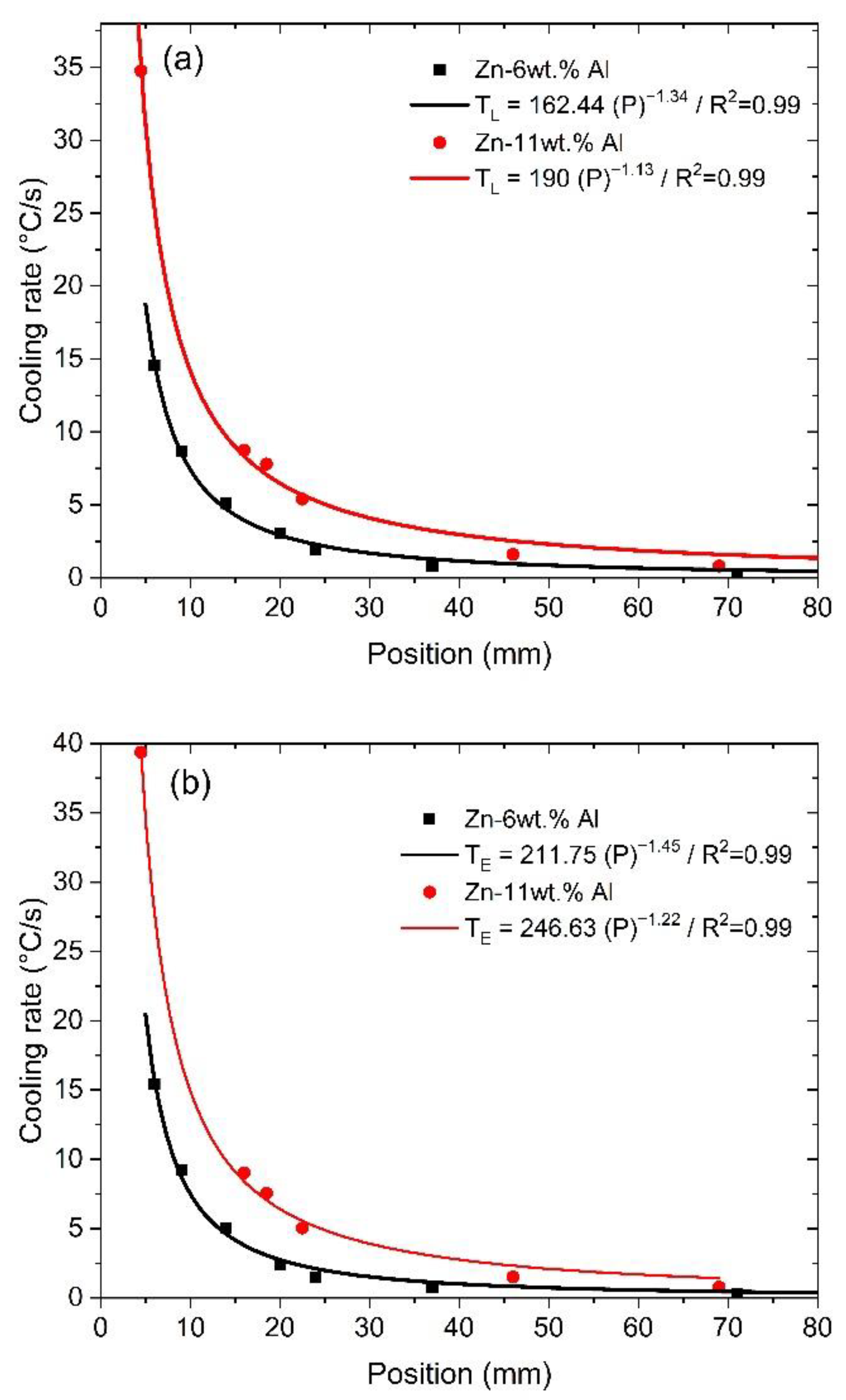
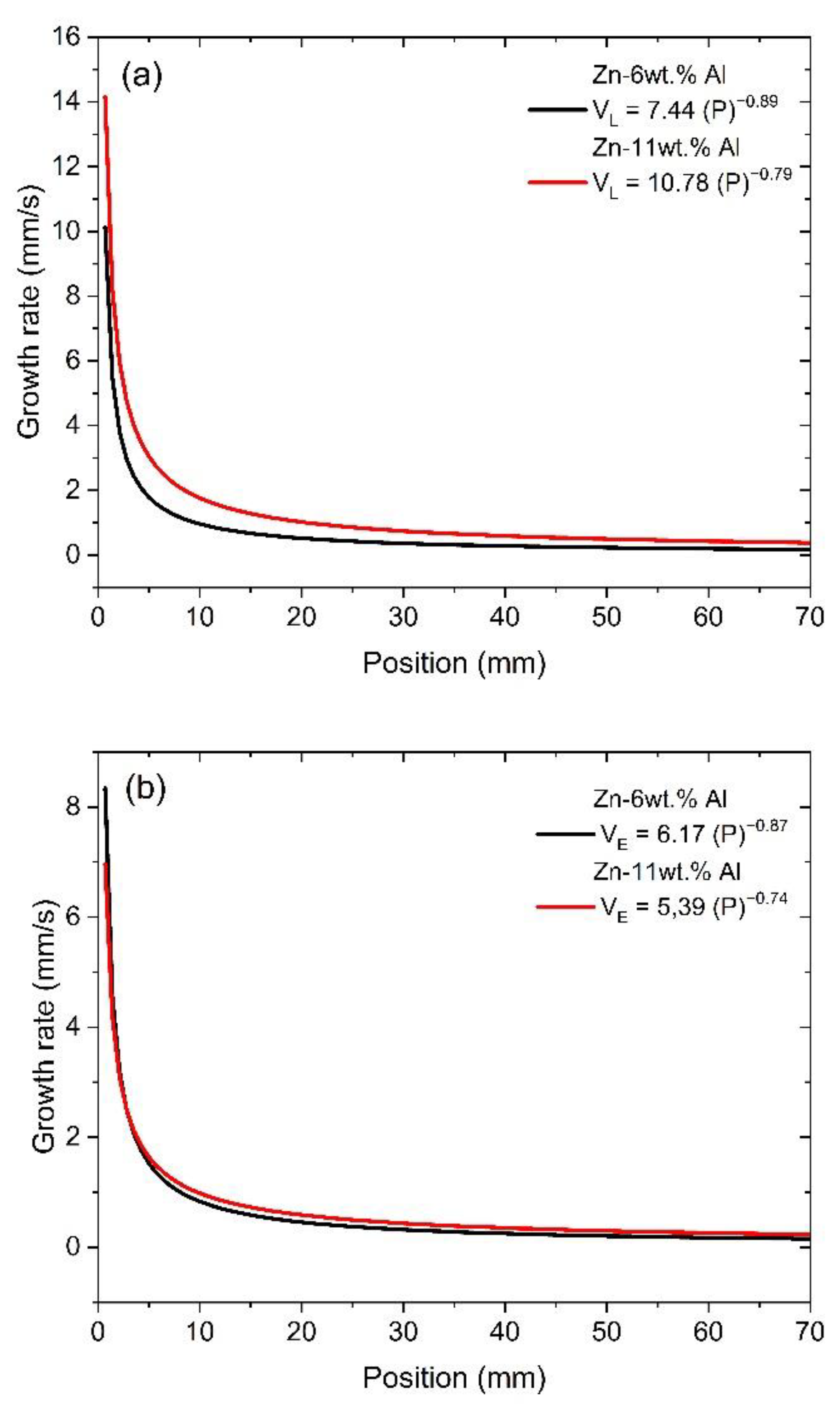
- Correlations between the microstructure and the thermal parameters were established in the form of experimental growth laws relating to eutectic colony spacing (λEC); eutectic lamellae spacing (λL), primary dendritic arm spacing (λ1) and secondary dendritic arm spacing (λ2) to the thermal parameters; liquidus isotherm (ṪL) and eutectic isotherm (ṪE) cooling rates; and liquidus isotherm (VL) and eutectic isotherm (VE) growth rates:
- A diagram exhibiting the coupled zone of Zn–Al alloys as a function of cooling rate (Ṫ) is proposed showing different microstructural morphologies influenced by composition and thermal parameters, that is, the growth rate (V) and the temperature gradient (G), both synthesized by the cooling rate (Ṫ = G.V).
- The microhardness of both Zn-6wt.%Al and Zn-11wt.%Al alloys were shown not to depend on the length scale of the resulting microstructure, with HV being essentially constant. The mean Vickers microhardness of the Zn-11wt.%Al alloy was found to be about 5.5% higher than that of the Zn-6wt.%Al alloy.
- The approach of this research can be useful in alloys of the Zn–Al system with the addition of a third element in order to seek a better understanding of the microstructures solidified under unsteady-state solidification conditions. It is possible, in this way, to raise correlations between mechanical, wear and corrosion properties with microstructural parameters to seek the best control of the processing parameters in others to obtain the final product with specific characteristics.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| DS | directionally solidified |
| EDS | energy dispersive spectrometer |
| XFR | x-ray fluorescence spectrometer |
| XRD | x-ray diffractometry |
| SMSM | solidification microstructural selection map |
| TL | liquidus temperature |
| TE | eutectic temperature |
| λEC | eutectic colony spacing |
| λL | eutectic lamellae spacing |
| λ1 | primary dendritic arm spacing |
| λ2λ2 | secondary dendritic arm spacing |
| Ṫ | cooling rate |
| ṪL | cooling rate of liquidus isotherm |
| ṪE | cooling rate of eutectic isotherm |
| V | growth rate |
| VL | growth rate of liquidus isotherm |
| VL | growth rate of eutectic isotherm |
| GE | gradient profiles ahead the eutectic isotherm |
References
- Zinc and Zinc Alloys. In Metals Handbook Desk Edition; ASM International: Almere, The Netherlands, 2018; pp. 589–597.
- Pola, A.; Tocci, M.; Goodwin, F.E. Review of Microstructures and Properties of Zinc Alloys. Metals 2020, 10, 253. [Google Scholar] [CrossRef] [Green Version]
- Türk, A.; Kurnaz, C.; Şevik, H. Comparison of the Wear Properties of Modified ZA-8 Alloys and Conventional Bearing Bronze. Mater. Des. 2007, 28, 1889–1897. [Google Scholar] [CrossRef]
- Purcek, G.; Altan, B.S.; Miskioglu, I.; Ooi, P.H. Processing of Eutectic Zn-5% Al Alloy by Equal-Channel Angular Pressing. J. Mater. Process. Technol. 2004, 148, 279–287. [Google Scholar] [CrossRef]
- Lachowicz, M.M.; Leśniewski, T.; Lachowicz, M.B.; Jasionowski, R. Tribological Wear of as Cast Zn-4Al Alloy Cooled at Various Rates from the Eutectoid Transformation Temperature. Arch. Foundry Eng. 2020, 20, 108–114. [Google Scholar] [CrossRef]
- Pola, A.; Montesano, L.; Gelfi, M.; la Vecchia, G.M. Comparison of the Sliding Wear of a Novel Zn Alloy with That of Two Commercial Zn Alloys against Bearing Steel and Leaded Brass. Wear 2016, 368–369, 445–452. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Kong, G.; Che, C.S. Corrosion Behavior of Zn-Al, Zn-Mg, and Zn-Mg-Al Coatings in Simulated Concrete Pore Solution. Corrosion 2019, 75, 203–209. [Google Scholar] [CrossRef]
- Marder, A.R. The Metallurgy of Zinc-Coated Steel. Prog. Mater. Sci. 2000, 45, 191–271. [Google Scholar] [CrossRef]
- Li, Z.F.; He, Y.Q.; Cao, G.M.; Tang, J.J.; Zhang, X.J.; Liu, Z.Y. Effects of Al Contents on Microstructure and Properties of Hot-Dip Zn-Al Alloy Coatings on Hydrogen Reduced Hot-Rolled Steel without Acid Pickling. J. Iron Steel Res. Int. 2017, 24, 1032–1040. [Google Scholar] [CrossRef]
- Gancarz, T.; Berent, K.; Skuza, W.; Janik, K. Soldering of Mg Joints Using Zn-Al Solders. Metall. Mater. Trans. A Phys. Metall. Mater. Sci. 2018, 49, 2684–2691. [Google Scholar] [CrossRef] [Green Version]
- Rettenmayr, M.; Lambracht, P.; Kempf, B.; Tschudin, C. Zn-Al Based Alloys as Pb-Free Solders for Die Attach. J. Electron. Mater. 2002, 31, 278–285. [Google Scholar] [CrossRef]
- Kang, N.; Na, H.S.; Kim, S.J.; Kang, C.Y. Alloy Design of Zn-Al-Cu Solder for Ultra High Temperatures. J. Alloys Compd. 2009, 467, 246–250. [Google Scholar] [CrossRef]
- Bazhenov, V.E.; Pashkov, I.N.; Pikunov, M.V.; Cheverikin, V.V.; Anohin, A.A. Interaction of Zn and Zn-4Al, Zn-15Al (Wt-%) Solder Alloys with Aluminium. Mater. Sci. Technol. 2016, 32, 752–759. [Google Scholar] [CrossRef]
- Santos, G.A.; de Moura, C.; Oso, W.R. Design of Mechanical Properties of a Zn27Al Alloy Based on Microstructure Dendritic Array Spacing. Mater. Des. 2007, 28, 2425–2430. [Google Scholar] [CrossRef]
- Osório, W.R.; Garcia, A. Modeling Dendritic Structure and Mechanical Properties of Zn-Al Alloys as a Function of Solidification Conditions. Mater. Sci. Eng. A 2002, 325, 103–111. [Google Scholar] [CrossRef]
- Bertelli, F.; Brito, C.; Ferreira, I.L.; Reinhart, G.; Nguyen-Thi, H.; Mangelinck-Noël, N.; Cheung, N.; Garcia, A. Cooling Thermal Parameters, Microstructure, Segregation and Hardness in Directionally Solidified Al-Sn-(Si;Cu) Alloys. Mater. Des. 2015, 72, 31–42. [Google Scholar] [CrossRef]
- Vida, T.A.; Brito, C.; Lima, T.S.; Spinelli, J.E.; Cheung, N.; Garcia, A. Near-Eutectic Zn-Mg Alloys: Interrelations of Solidification Thermal Parameters, Microstructure Length Scale and Tensile/Corrosion Properties. Curr. Appl. Phys. 2019, 19, 582–598. [Google Scholar] [CrossRef]
- Barnhurst, R.J. Zinc and Zinc Alloys. In Properties and Selection: Nonferrous Alloys and Special-Purpose Materials; ASM International: Almere, The Netherlands, 1990. [Google Scholar]
- Apelian, D.; Paliwal, M.; Herrschaft, D.C. Casting with Zinc Alloys. J. Met. 1981, 12–20. [Google Scholar] [CrossRef]
- Septimio, R.S.; Costa, T.A.; Silva, C.A.P.; Vida, T.A.; de Damborenea, J.; Garcia, A.; Cheung, N. Correlation between Unsteady-State Solidification Thermal Parameters and Microstructural Growth of Zn–8 Mass% Al and Zn–8 Mass% Al–XBi Tribological Alloys. J. Therm. Anal. Calorim. 2020, 139, 1741–1761. [Google Scholar] [CrossRef]
- Murray, J.L. The Al-Zn (Aluminum-Zinc) System. Bull. Alloy Phase Diagr. 1983, 4, 55–73. [Google Scholar] [CrossRef]
- Abou El-Khair, M.T.; Daoud, A.; Ismail, A. Effect of Different Al Contents on the Microstructure, Tensile and Wear Properties of Zn-Based Alloy. Mater. Lett. 2004, 58, 1754–1760. [Google Scholar] [CrossRef]
- Bansod, A.; Patil, A.P.; Deshmukh, B. Microstructural and Corrosion Behavior of Different Aluminum Content on Zinc. In Proceedings of the CORCON 2018, Jaipur, India, 1 December 2018; p. 0MC15. [Google Scholar]
- Osório, W.R.; Santos, C.A.; Quaresma, J.M.V.; Garcia, A. Mechanical Properties as a Function of Thermal Parameters and Microstructure of Zn-Al Castings. J. Mater. Process. Technol. 2003, 143–144, 703–709. [Google Scholar] [CrossRef]
- Porot, P.A.; Spittle, J.A. Structural Variation in Near-Eutectic Zinc-Aluminum Alloys. Metallography 1987, 197, 181–197. [Google Scholar] [CrossRef]
- Kurz, W.; Fisher, D.J. Solidification Microstructure- Processing Maps: Theoryand Application. Adv. Eng. Mater. 2001, 3, 443–452. [Google Scholar] [CrossRef]
- Silva, C.A.P.; Kakitani, R.; Canté, M.V.; Brito, C.; Garcia, A.; Spinelli, J.E.; Cheung, N. Microstructure, Phase Morphology, Eutectic Coupled Zone and Hardness of Al–Co Alloys. Mater. Charact. 2020, 169, 110617. [Google Scholar] [CrossRef]
- Kakitani, R.; Pinto da Silva, C.A.; Silva, B.; Garcia, A.; Cheung, N.; Spinelli, J.E. Local Solidification Thermal Parameters Affecting the Eutectic Extent in Sn-Cu and Sn-Bi Solder Alloys. Solder. Surf. Mt. Technol. 2022, 34, 24–30. [Google Scholar] [CrossRef]
- Rai, P.K.; Rout, D.; Satish Kumar, D.; Sharma, S.; Balachandran, G. Effect of Magnesium on Corrosion Behavior of Hot-Dip Zn-Al-Mg Coating. J. Mater. Eng. Perform. 2021, 30, 4138–4147. [Google Scholar] [CrossRef]
- Xie, Y.; Du, A.; Zhao, X.; Ma, R.; Fan, Y.; Cao, X. Effect of Mg on Fe–Al Interface Structure of Hot–Dip Galvanized Zn–Al–Mg Alloy Coatings. Surf. Coat. Technol. 2018, 337, 313–320. [Google Scholar] [CrossRef]
- Yu, K.C.; Li, J.; Liu, X.; Li, J.G.; Xue, X.H. Microstructure of Hot-Dip Galvanized Zn-Al-Mg Alloy Coating. J. Shanghai Jiaotong Univ. (Sci.) 2012, 17, 663–667. [Google Scholar] [CrossRef]
- Prosek, T.; Hagström, J.; Persson, D.; Fuertes, N.; Lindberg, F.; Chocholatý, O.; Taxén, C.; Šerák, J.; Thierry, D. Effect of the Microstructure of Zn-Al and Zn-Al-Mg Model Alloys on Corrosion Stability. Corros. Sci. 2016, 110, 71–81. [Google Scholar] [CrossRef]
- TCS Solder Alloy Solutions Database (TCSLD3), Thermocalc. 2020. Available online: https://www.engineering-eye.com/THERMOCALC/details/db/pdf/thermo-calc/06/tcsld3-2_extended_info.pdf (accessed on 22 June 2022).
- Gündüz, M.; Çadirli, E. Directional Solidification of Aluminium-Copper Alloys. Mater. Sci. Eng. A 2002, 327, 167–185. [Google Scholar] [CrossRef]
- Spinelli, J.E.; Silva, B.L.; Cheung, N.; Garcia, A. The Use of a Directional Solidification Technique to Investigate the Interrelationship of Thermal Parameters, Microstructure and Microhardness of Bi-Ag Solder Alloys. Mater. Charact. 2014, 96, 115–125. [Google Scholar] [CrossRef]
- Çadirli, E.; Marasli, N.; Bayender, B.; Gündüz, M. Dependency of the Microstructure Parameters on the Solidification Parameters for Camphene. Mater. Res. Bull. 2000, 35, 985–995. [Google Scholar] [CrossRef]
- Kaya, H.; Çadirli, E.; Ülgen, A. Investigation of the Effect of Composition on Microhardness and Determination of Thermo-Physical Properties in the Zn-Cu Alloys. Mater. Des. 2011, 32, 900–906. [Google Scholar] [CrossRef]
- Vida, T.A.; Soares, T.; Septimio, R.S.; Brito, C.C.; Cheung, N.; Garcia, A. Effects of Macrosegregation and Microstructure on the Corrosion Resistance and Hardness of a Directionally Solidified Zn-5.0wt.%Mg Alloy. Mater. Res. 2019, 22, 1–13. [Google Scholar] [CrossRef]
- Dias, M.; Brito, C.; Bertelli, F.; Rocha, O.L.; Garcia, A. Interconnection of Thermal Parameters, Microstructure, Macrosegregation and Microhardness of Unidirectionally Solidified Zn-Rich Zn-Ag Peritectic Alloys. Mater. Des. 2014, 63, 848–855. [Google Scholar] [CrossRef]
- Li, Z.; He, Y.; Cao, G.; Tang, J.; Zhang, X.; Liu, Z. Modifications on Solidification Thermal Parameters, Microstructure and Hardness Induced by Cu Additions to a Hypereutectic Zn–8Al Alloy. J. Iron Steel Res. 2017, 174, 1032–1040. [Google Scholar] [CrossRef]
- Silva, B.L.; Cheung, N.; Garcia, A.; Spinelli, J.E. Evaluation of Solder/Substrate Thermal Conductance and Wetting Angle of Sn-0.7 Wt%Cu-(0-0.1 Wt%Ni) Solder Alloys. Mater. Lett. 2015, 142, 163–167. [Google Scholar] [CrossRef]
- MatWeb. Zinc, Zn. Available online: https://www.matweb.com/search/DataSheet.aspx?MatGUID=8909140a76074049809ad74d536ed606&ckck=1 (accessed on 16 May 2022).
- McAlister, A.; Orr, C. Thermal Conductivities of Aluminum and Zinc Powder Suspensions. J. Chem. Eng. Data 1964, 9, 71–74. [Google Scholar] [CrossRef]
- Kurz, W.; Fisher, D.J. Fundamentals of Solidification, 3rd ed.; Trans Tech Publications: Stafa-Zurich, Switzerland, 1990. [Google Scholar]
- Łent, M.T. Coupled and Competitive Eutectic Growth. Arch. Foundry Eng. 2011, 11, 237–240. [Google Scholar]
- Neikov, O.D. Production of Aluminum Alloy Powders. In Handbook of Non-Ferrous Metal Powders; S. Banerjee, P.M., Ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2007; pp. 481–531. ISBN 9780081005439. [Google Scholar]
- Chapter 3 Solidification, Vitrification, Crystallization and Formation of Quasicrystalline and Nanocrystalline Structures. In Phase Transformations: Examples from Titanium and Zirconium Alloys; Banerjee, S.; Mukhopadhyay, P. (Eds.) Elsevier: Amsterdam, The Netherlands, 2007; Volume 12, pp. 125–255. ISBN 9780080421452. [Google Scholar]
- Ji, Y.; Heo, T.W.; Zhang, F.; Chen, L.Q. Theoretical Assessment on the Phase Transformation Kinetic Pathways of Multi-Component Ti Alloys: Application to Ti-6Al-4V. J. Phase Equilibria Diffus. 2016, 37, 53–64. [Google Scholar] [CrossRef]
- Garcia, A. Solidificação: Fundamentos E Aplicações; Editora Unicamp: Campinas, Brazil, 2007. [Google Scholar]
- Gill, S.C.; Zimmermann, M.; Kurz, W. Laser Resolidification of the A1-A12Cu Eutectic: The Coupled Zone. Acta Metall. Mater. 1992, 40, 2895–2906. [Google Scholar] [CrossRef]
- Abboud, J.; Mazumder, J. Developing of Nano Sized Fibrous Eutectic Silicon in Hypereutectic Al–Si Alloy by Laser Remelting. Sci. Rep. 2020, 10, 12090. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.L.; Li, J.F. The Growth Pattern of Co3Sn2 in Directional Solidification of Co-Sn Hypereutectic Alloy Melts. Metall. Mater. Trans. A Phys. Metall. Mater. Sci. 2020, 51, 6346–6354. [Google Scholar] [CrossRef]
- Jackson, K.A.; Hunt, J.D. Lamellar and Rod Eutectic Growth. Dyn. Curved Front. 1988, 236, 363–376. [Google Scholar] [CrossRef]
- Garcia, A.; Clyne, T.W.; Prates, M. Mathematical Model for the Unidirectional Solidification of Metals: II. Massive Molds. Metall. Trans. B 1979, 10, 85–92. [Google Scholar] [CrossRef]
- Kakitani, R.; Reyes, R.V.; Garcia, A.; Spinelli, J.E.; Cheung, N. Relationship between Spacing of Eutectic Colonies and Tensile Properties of Transient Directionally Solidified Al-Ni Eutectic Alloy. J. Alloys Compd. 2018, 733, 59–68. [Google Scholar] [CrossRef]
- Spinelli, J.E.; Silva, B.L.; Garcia, A. Microstructure, Phases Morphologies and Hardness of a Bi-Ag Eutectic Alloy for High Temperature Soldering Applications. Mater. Des. 2014, 58, 482–490. [Google Scholar] [CrossRef]
- Haghdadi, N.; Phillion, A.B.; Maijer, D.M. Microstructure Characterization and Thermal Analysis of Aluminum Alloy B206 during Solidification. Metall. Mater. Trans. A Phys. Metall. Mater. Sci. 2015, 46, 2073–2081. [Google Scholar] [CrossRef] [Green Version]
- Xavier, M.G.C.; Souza, T.M.G.; Cheung, N.; Garcia, A.; Spinelli, J.E. Effects of Cobalt and Solidification Cooling Rate on Intermetallic Phases and Tensile Properties of a -Cu, -Zn, -Fe Containing Al-Si Alloy. Int. J. Adv. Manuf. Technol. 2020, 107, 717–730. [Google Scholar] [CrossRef]
- Kakitani, R.; Cruz, C.B.; Lima, T.S.; Brito, C.; Garcia, A.; Cheung, N. Transient Directional Solidification of a Eutectic Al–Si–Ni Alloy: Macrostructure, Microstructure, Dendritic Growth and Hardness. Materialia 2019, 7, 100358. [Google Scholar] [CrossRef]
- Xavier, M.G.C.; Cruz, C.B.; Kakitani, R.; Silva, B.L.; Garcia, A.; Cheung, N.; Spinelli, J.E. Directional Solidification of a Sn-0.2Ni Solder Alloy in Water-Cooled Copper and Steel Molds: Related Effects on the Matrix Micromorphology, Nature of Intermetallics and Tensile Properties. J. Alloys Compd. 2017, 723, 1039–1052. [Google Scholar] [CrossRef]
- Silva, B.L.; Cheung, N.; Garcia, A.; Spinelli, J.E. Sn-0.7 Wt%Cu-(XNi) Alloys: Microstructure-Mechanical Properties Correlations with Solder/Substrate Interfacial Heat Transfer Coefficient. J. Alloys Compd. 2015, 632, 274–285. [Google Scholar] [CrossRef]
- Tunca, N.; Smith, R.W. Variation of Dendrite Arm Spacing in AI-Rich Zn-AI off-Eutectic Alloys. J. Mater. Sci. 1988, 23, 111–120. [Google Scholar] [CrossRef]
- Ares, A.E.; Gassa, L.M.; Schvezov, C.E.; Rosenberger, M.R. Corrosion and Wear Resistance of Hypoeutectic Zn-Al Alloys as a Function of Structural Features. Mater. Chem. Phys. 2012, 136, 394–414. [Google Scholar] [CrossRef]
- Pastene, M.P.; Fornaro, O.; Medina, H.O. Study of Directional Solidification of Zn-Al Alloys. Procedia Mater. Sci. 2012, 1, 87–94. [Google Scholar] [CrossRef] [Green Version]
- Engin, S.; Böyük, U.; Kaya, H.; Maraşlı, N. Directional Solidification and Physical Properties Measurements of the Zinc-Aluminum Eutectic Alloy. Int. J. Miner. Metall. Mater. 2011, 18, 659–664. [Google Scholar] [CrossRef]
- Spittle, J.A. Endogenous-Exogenous Freezing Characteristics of Pure and Impure as-Cast Zn-AI Alloys. Met. Sci. 1977, 11, 578–585. [Google Scholar] [CrossRef]
- Hekimoğlu, A.P.; Savaşkan, T. Structure and Mechanical Properties of Zn-(5–25) Al Alloys. Int. J. Mater. Res. 2014, 105, 1084–1089. [Google Scholar] [CrossRef]
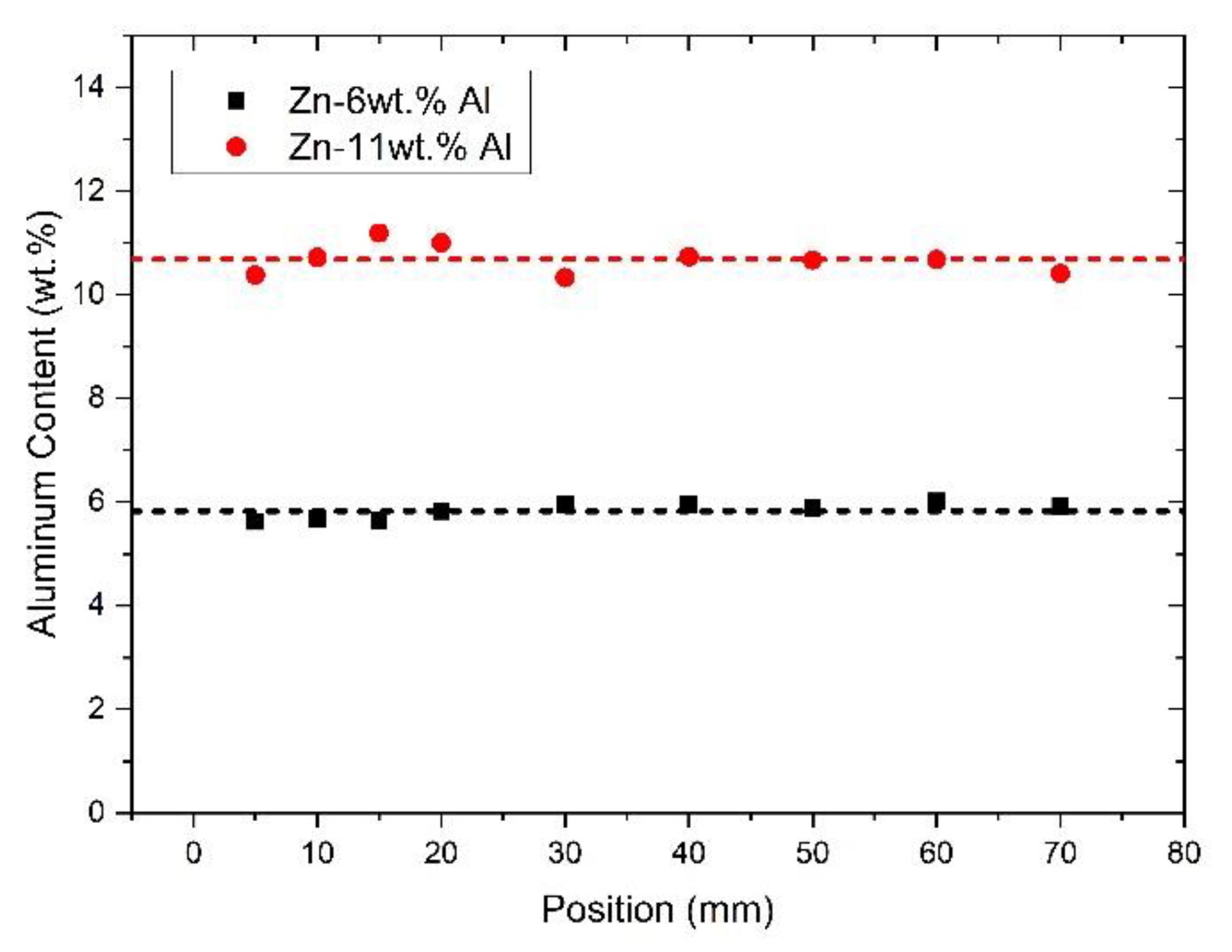
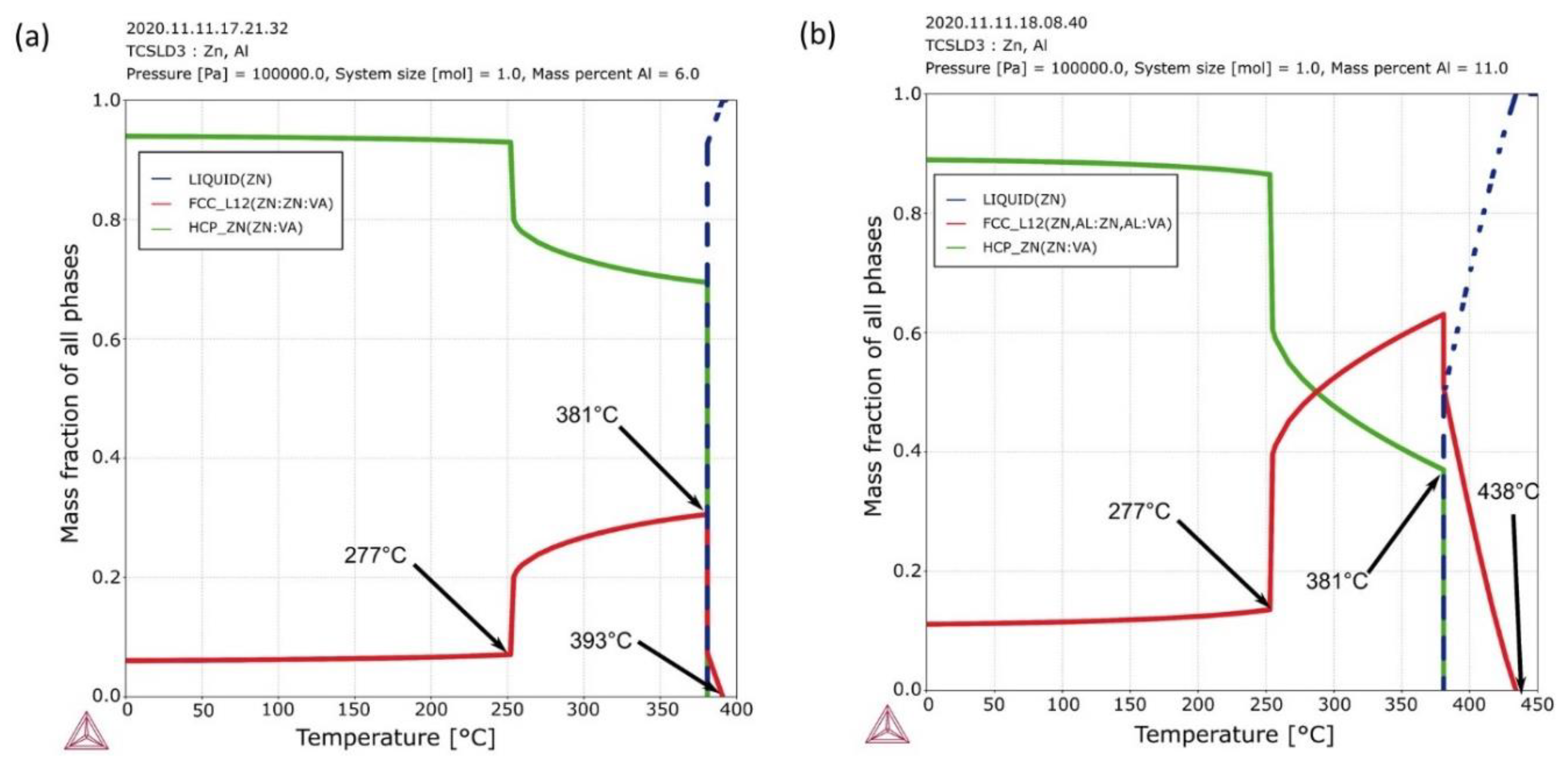

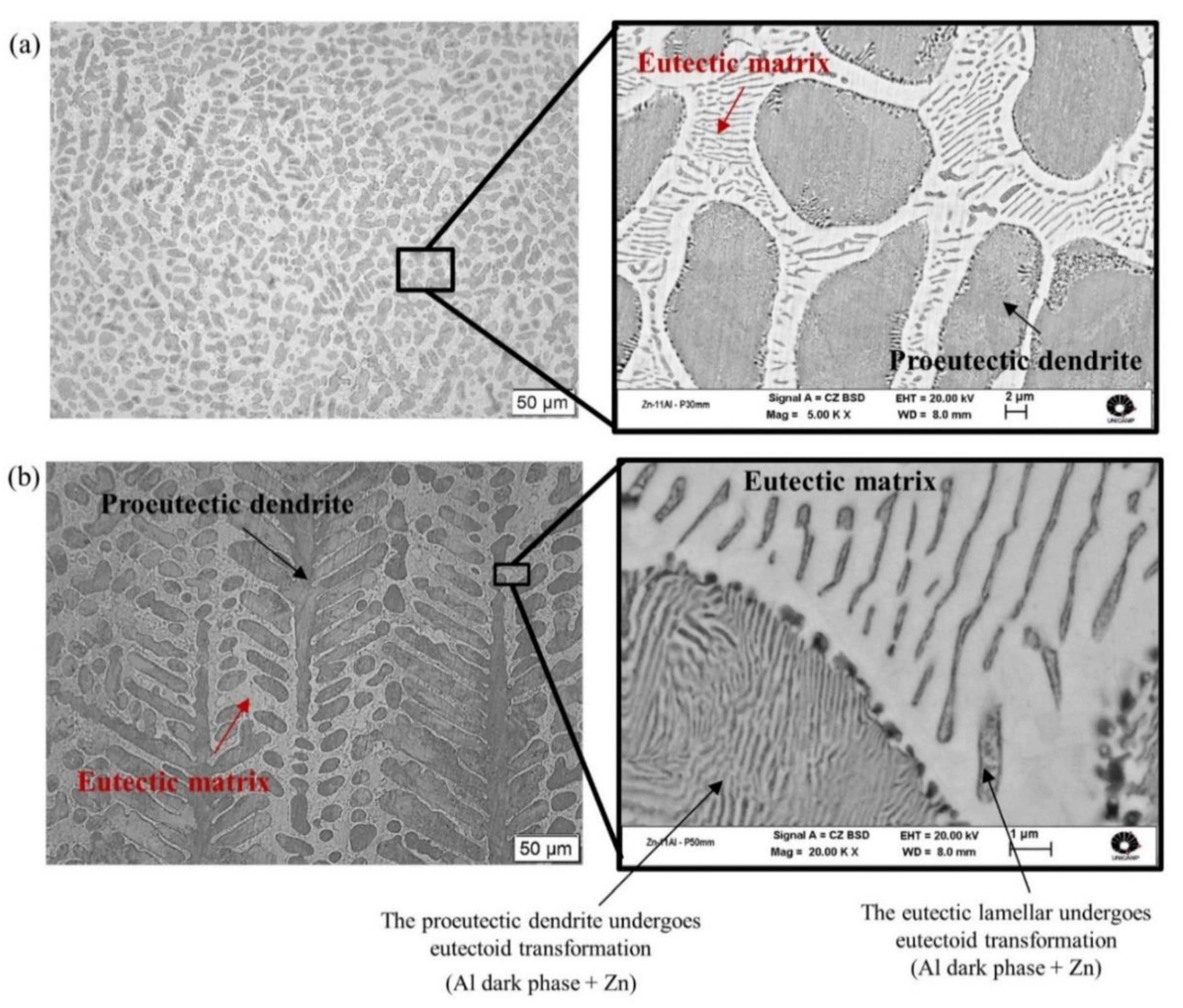
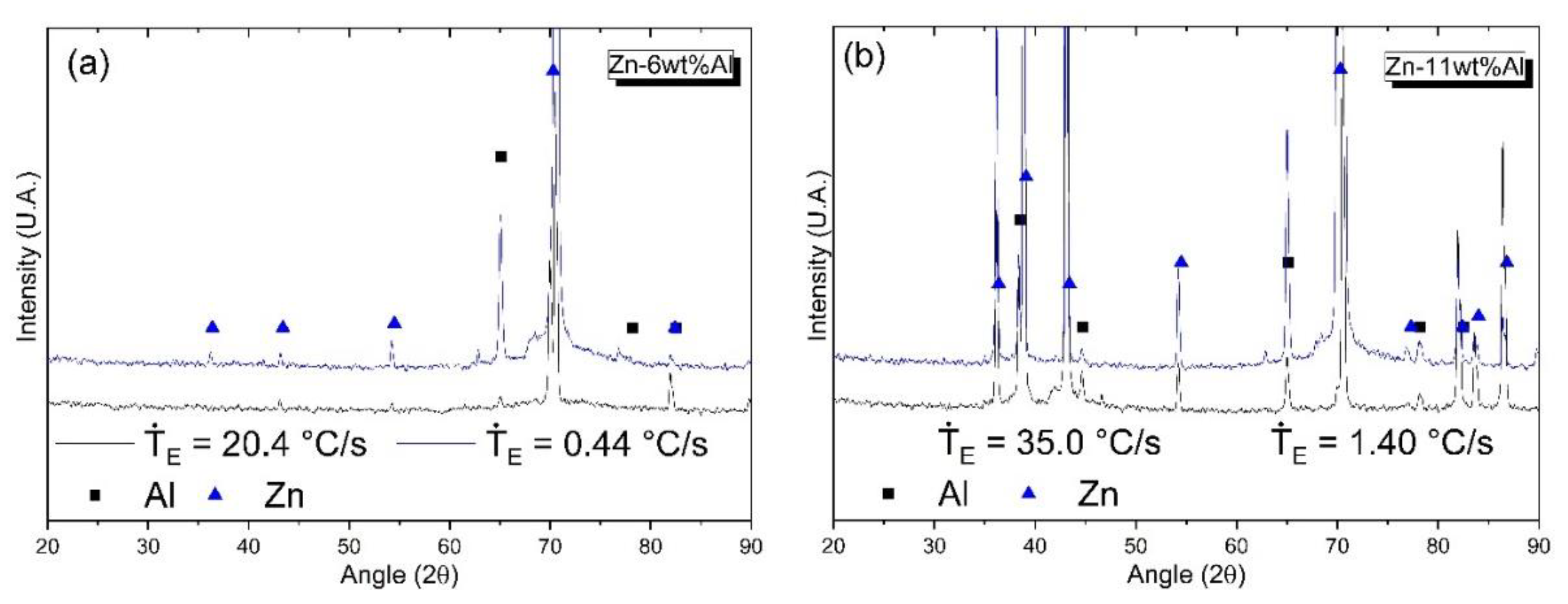

| wt.% | Al | Zn | Fe | Si | Mg | Cu | Pb |
|---|---|---|---|---|---|---|---|
| Al | Balance | 0.01 | 0.03 | 0.03 | 0.01 | 0.01 | - |
| Zn | - | Balance | 0.015 | 0.003 | - | - | 0.012 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Septimio, R.; Silva, C.A.P.; Costa, T.A.; Garcia, A.; Cheung, N. Hypereutectic Zn–Al Alloys: Microstructural Development under Unsteady-State Solidification Conditions, Eutectic Coupled Zone and Hardness. Metals 2022, 12, 1076. https://doi.org/10.3390/met12071076
Septimio R, Silva CAP, Costa TA, Garcia A, Cheung N. Hypereutectic Zn–Al Alloys: Microstructural Development under Unsteady-State Solidification Conditions, Eutectic Coupled Zone and Hardness. Metals. 2022; 12(7):1076. https://doi.org/10.3390/met12071076
Chicago/Turabian StyleSeptimio, Rudimylla, Cássio A. P. Silva, Thiago A. Costa, Amauri Garcia, and Noé Cheung. 2022. "Hypereutectic Zn–Al Alloys: Microstructural Development under Unsteady-State Solidification Conditions, Eutectic Coupled Zone and Hardness" Metals 12, no. 7: 1076. https://doi.org/10.3390/met12071076
APA StyleSeptimio, R., Silva, C. A. P., Costa, T. A., Garcia, A., & Cheung, N. (2022). Hypereutectic Zn–Al Alloys: Microstructural Development under Unsteady-State Solidification Conditions, Eutectic Coupled Zone and Hardness. Metals, 12(7), 1076. https://doi.org/10.3390/met12071076







