Corrosion Behavior of Passivated Martensitic and Semi-Austenitic Precipitation Hardening Stainless Steel
Abstract
:1. Introduction
2. Experimental Methodology
2.1. Materials
2.2. Passivation Treatment
2.3. Microstructural Characterization
2.4. Corrosion Test
2.5. XPS Characterization
3. Results and Discussion
3.1. OM-SEM Microstructural
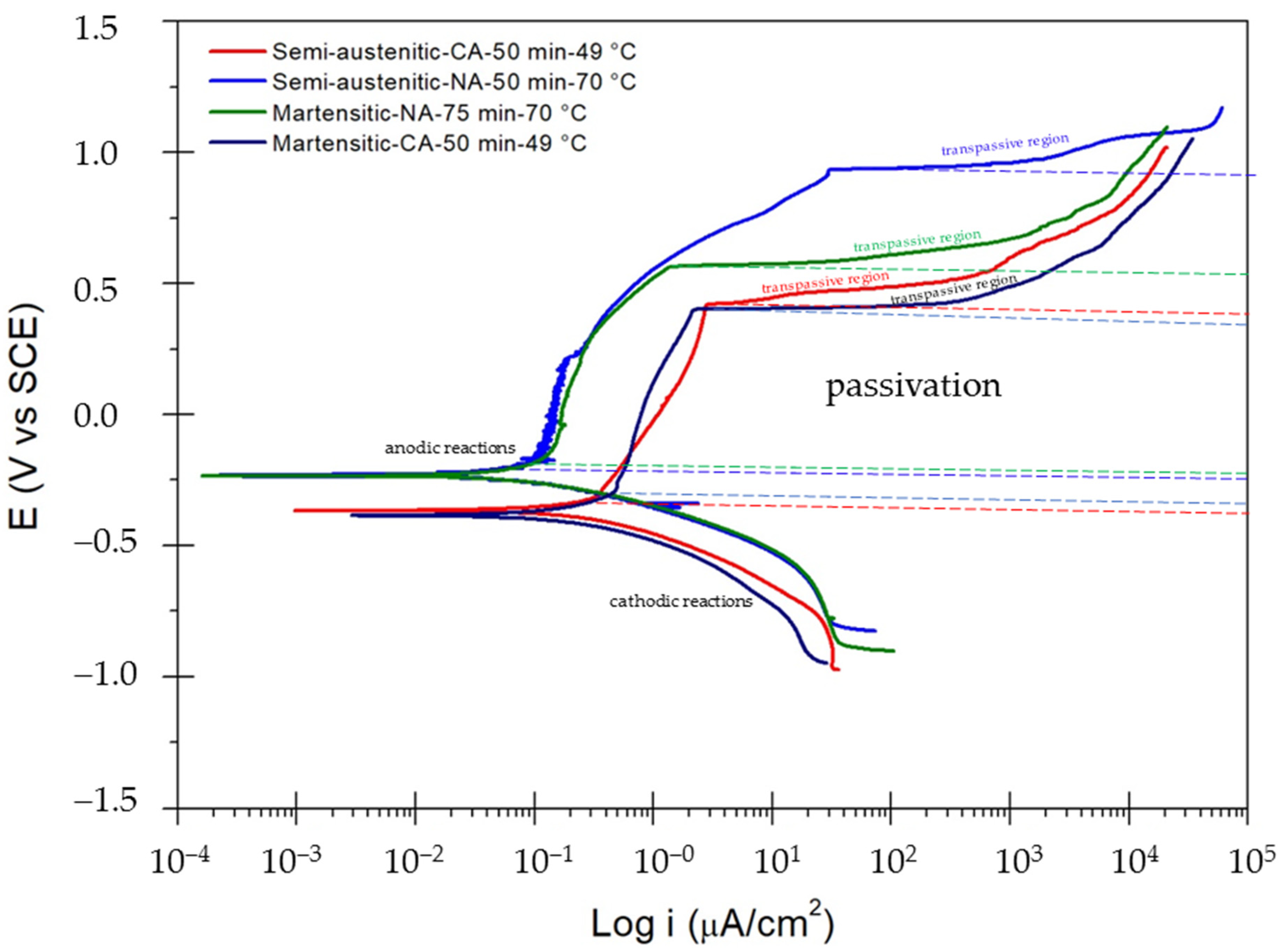
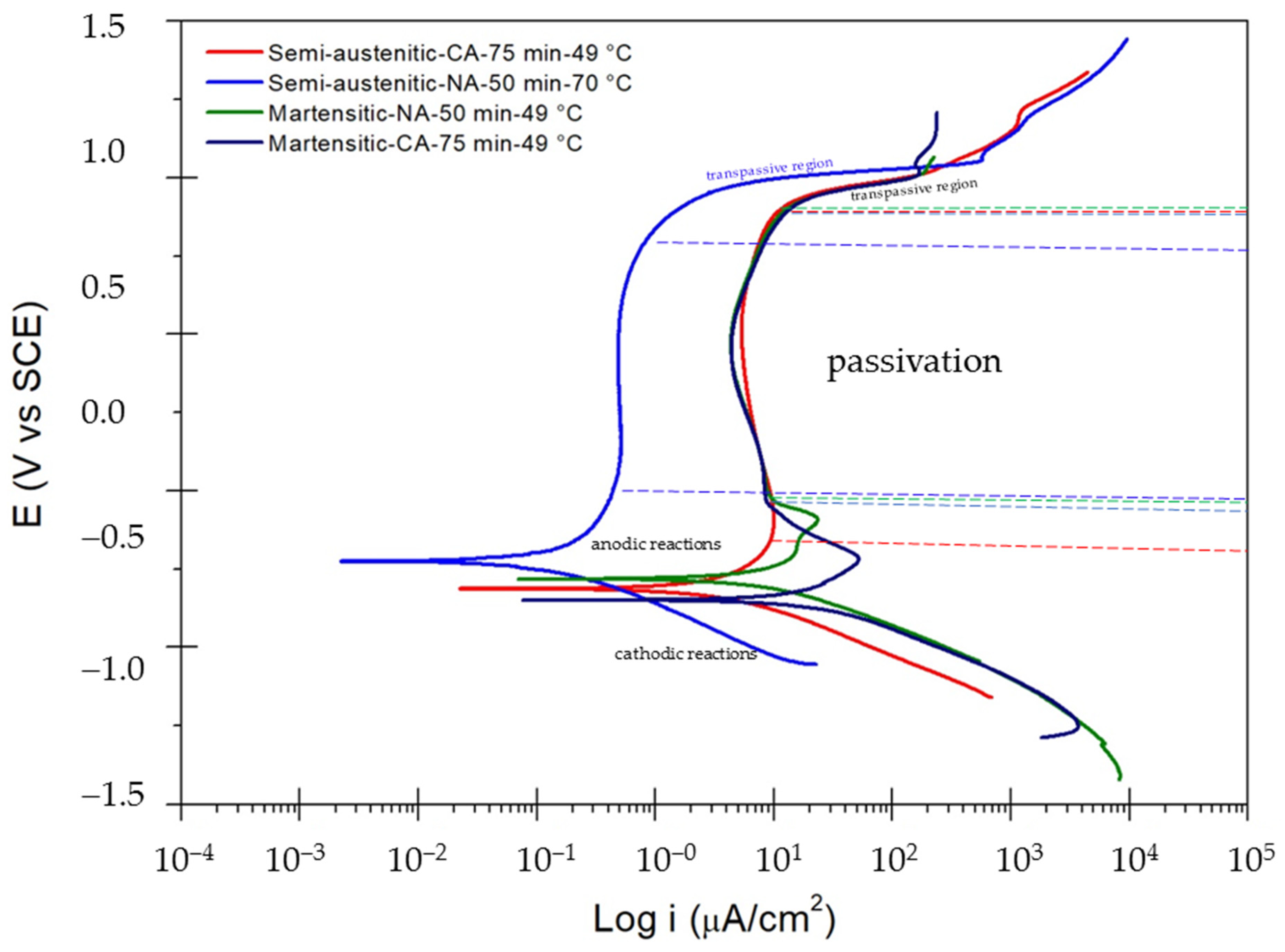
3.2. Corrosion Test
Potentiodynamic Polarization
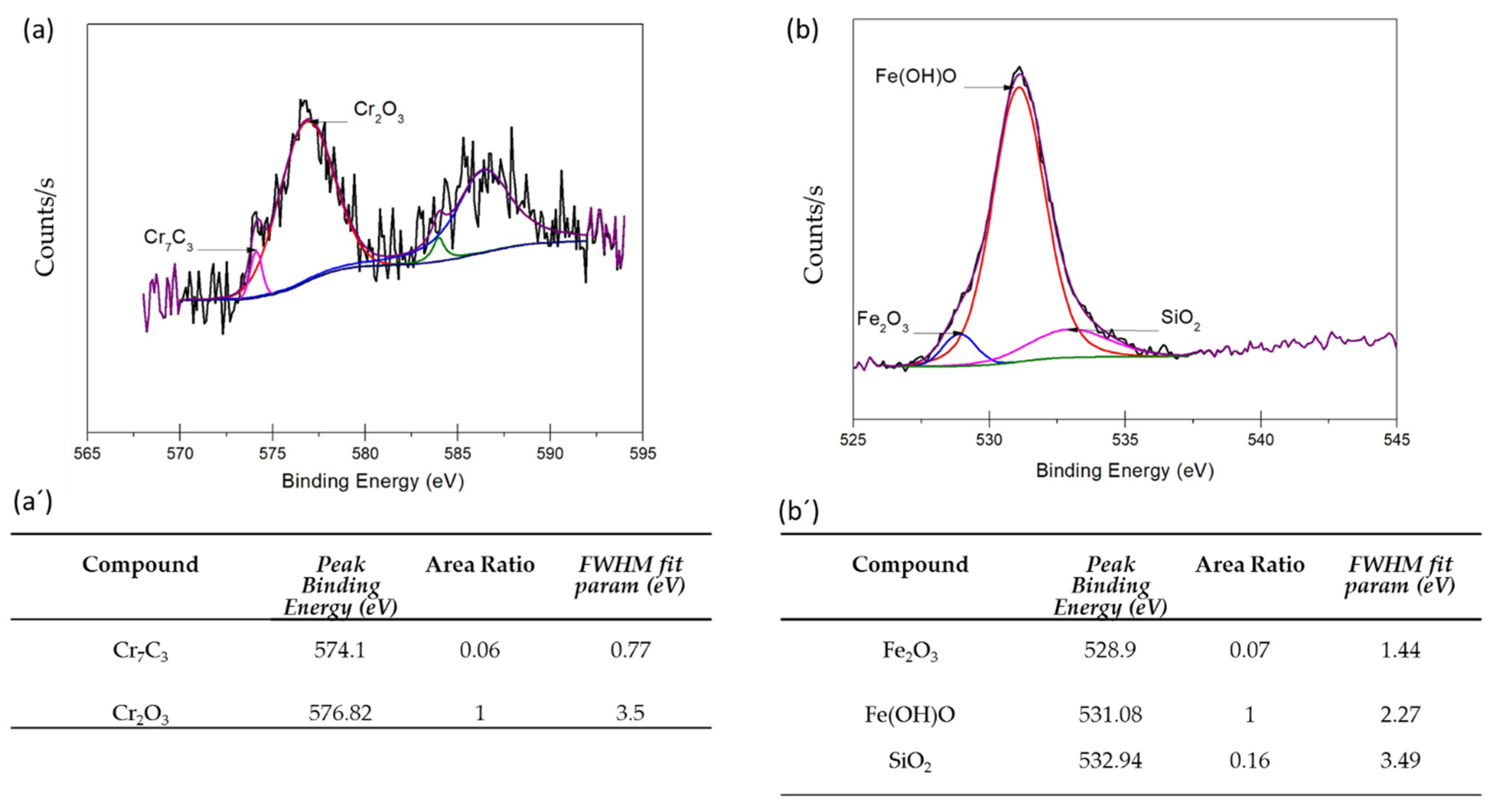
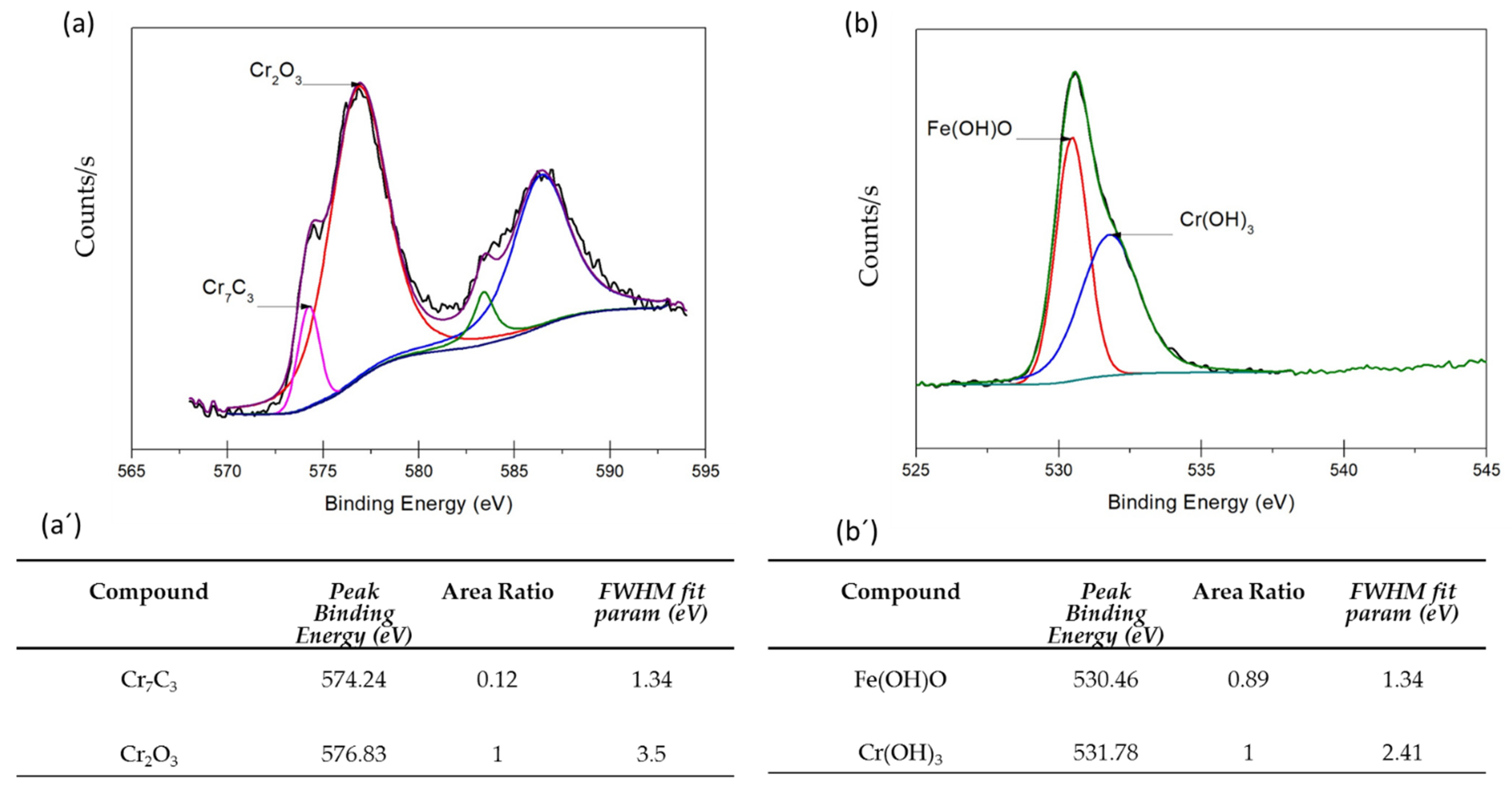
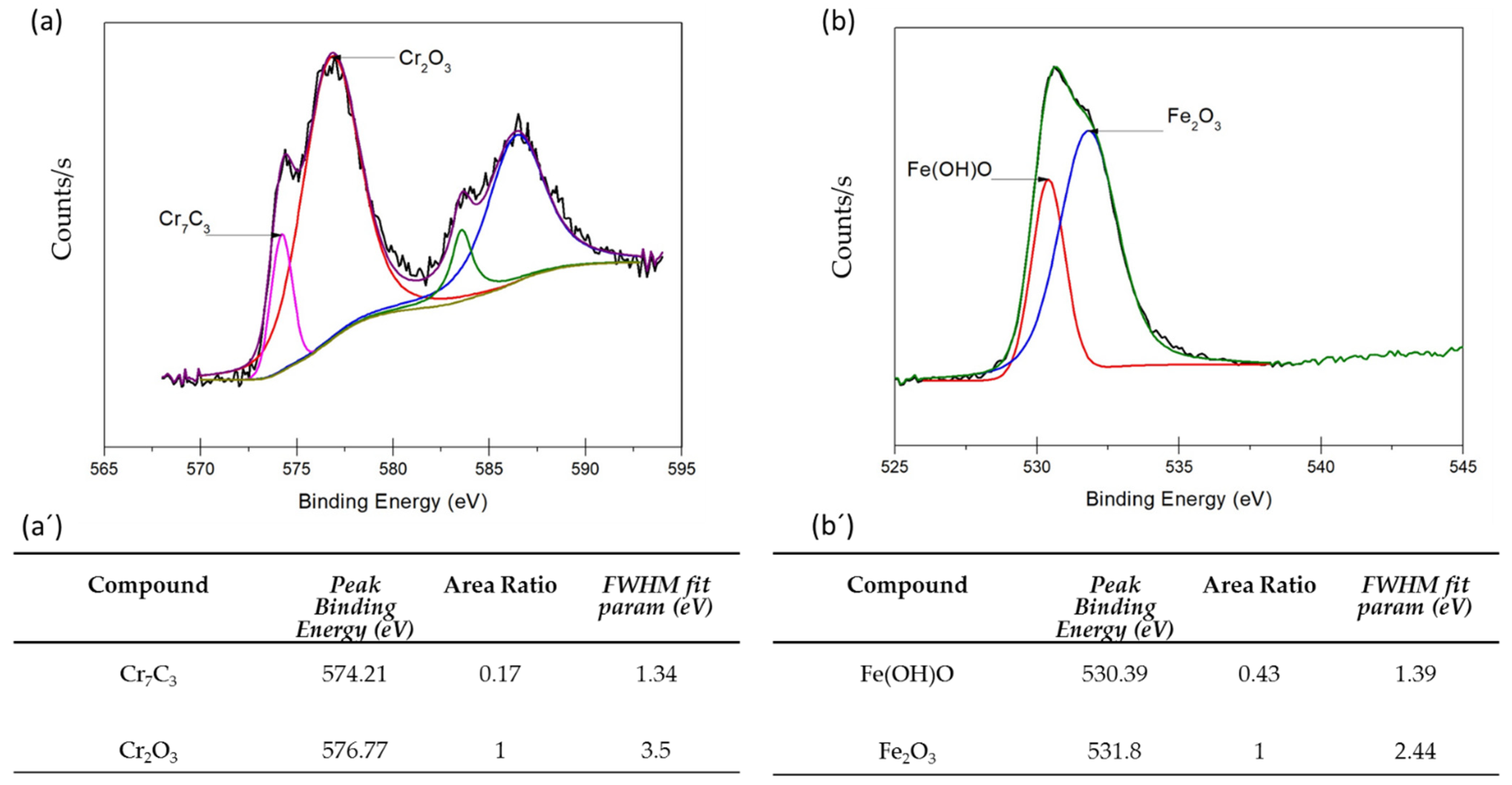
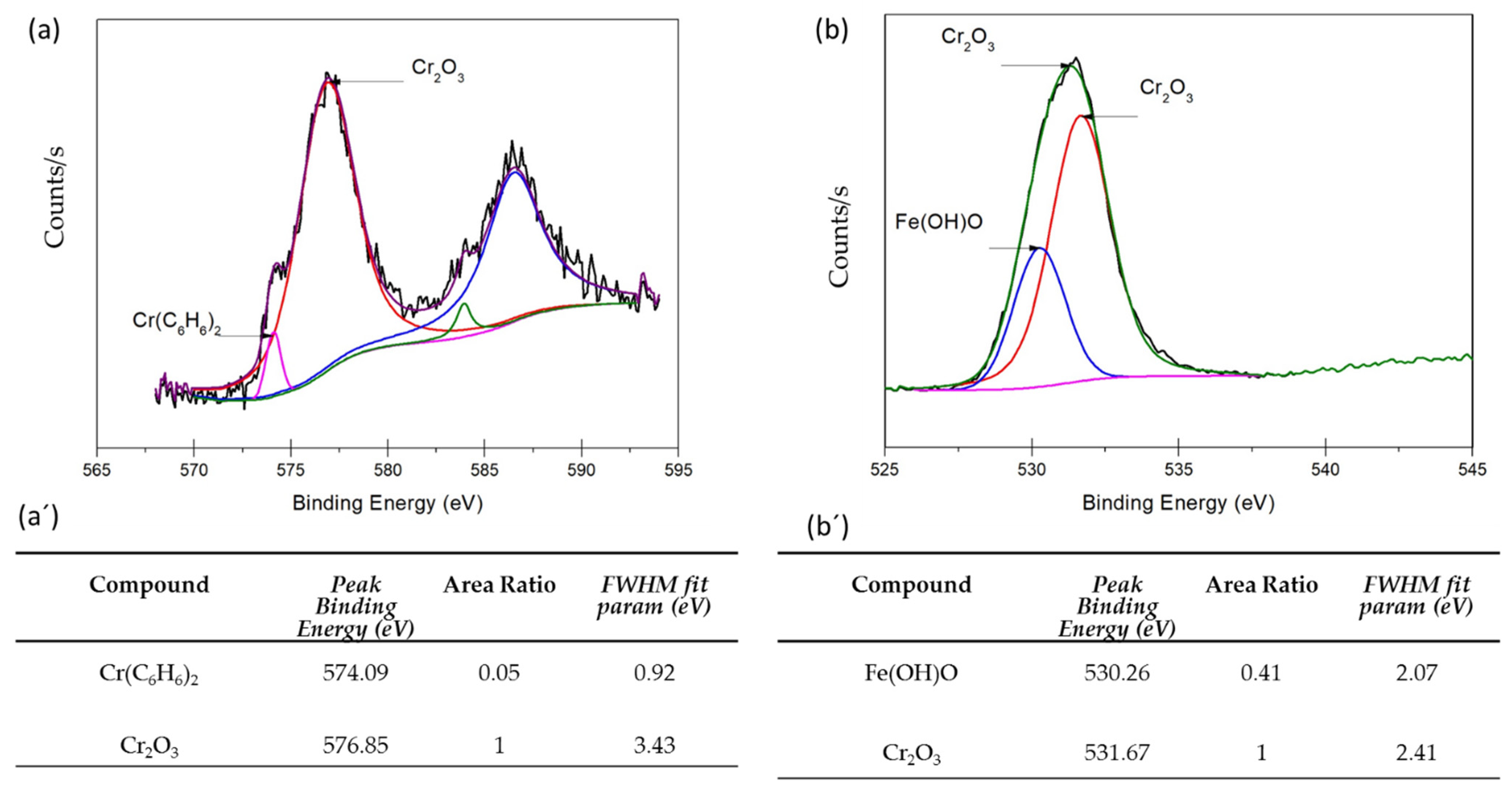
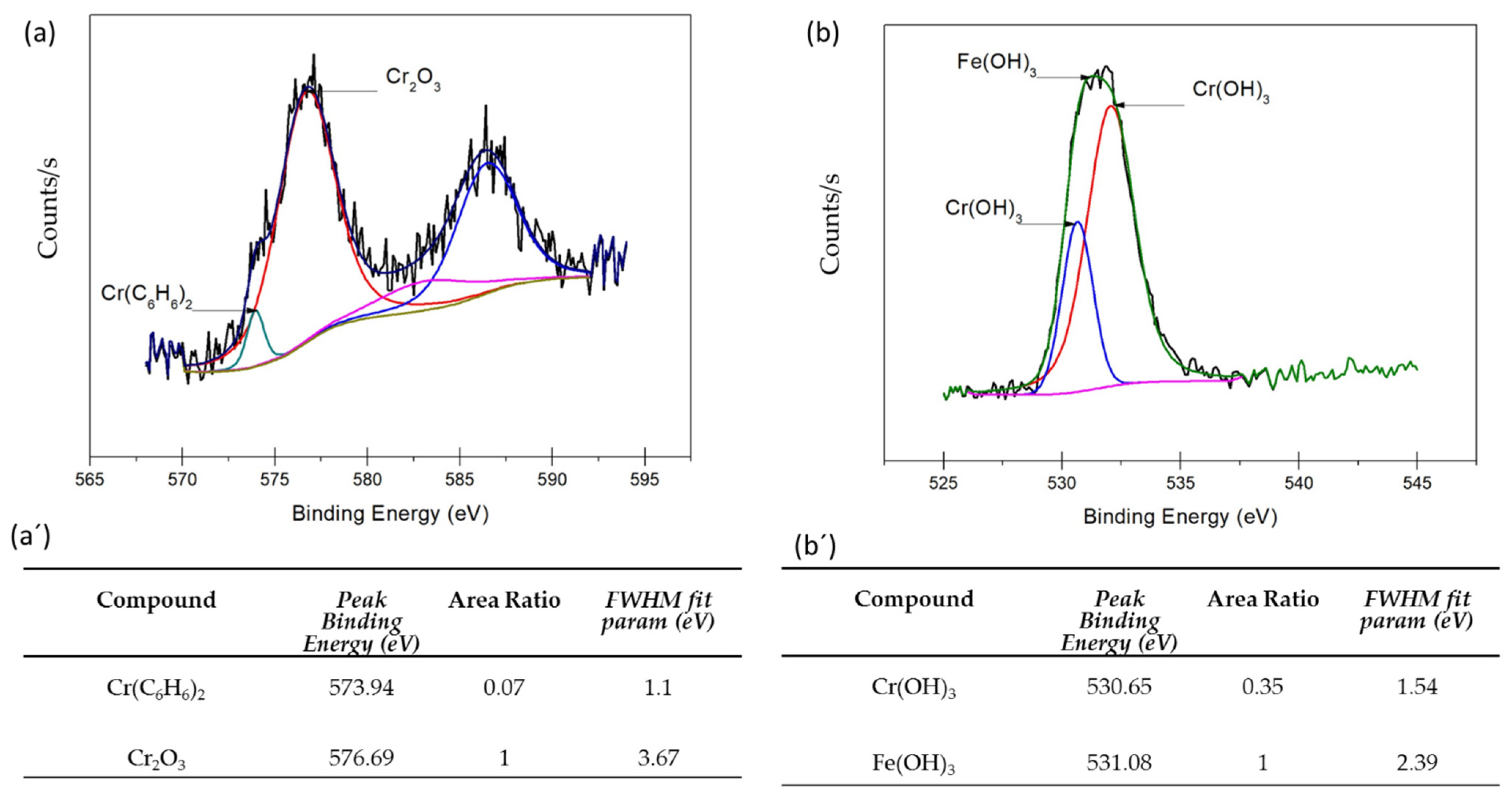
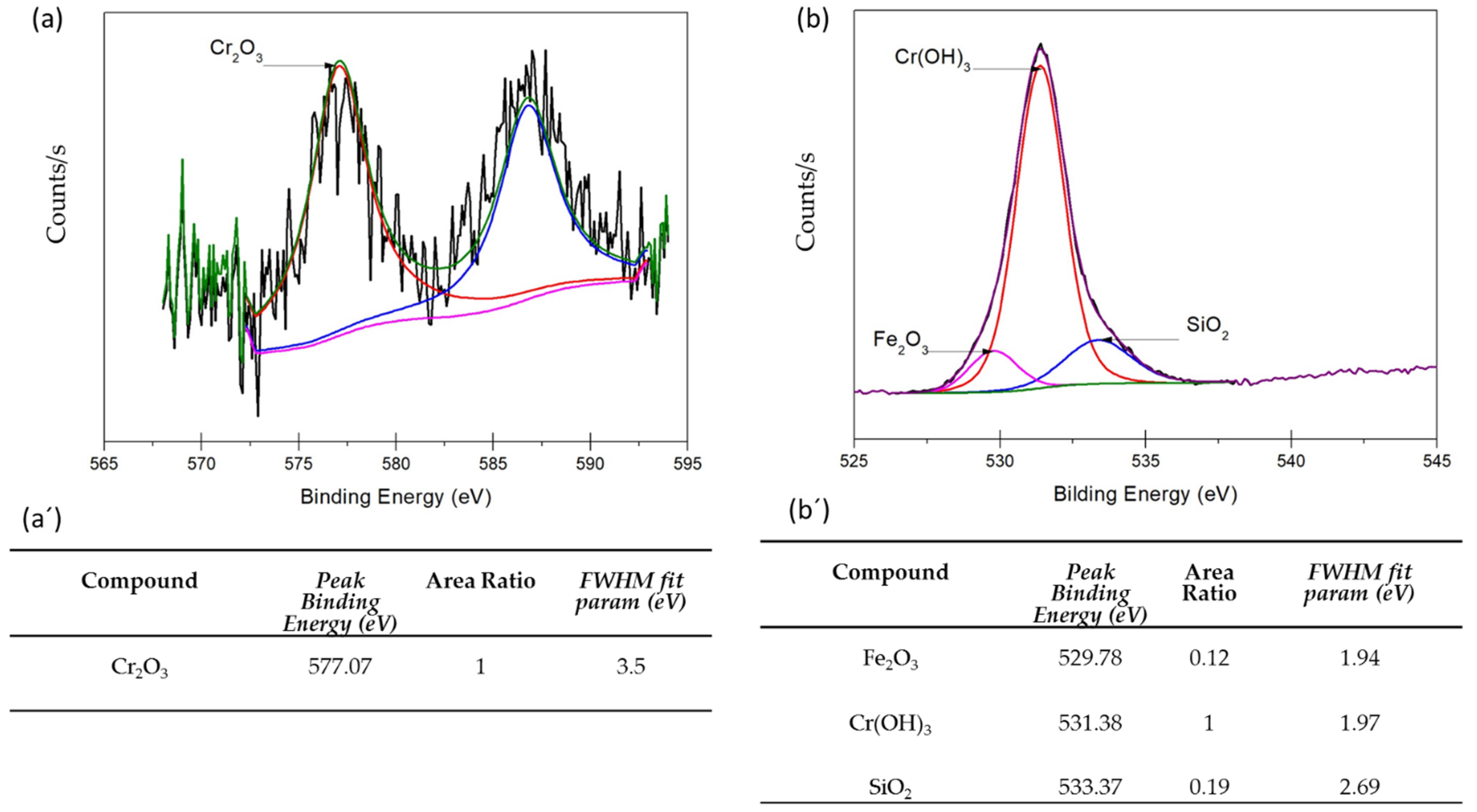
3.3. XPS Analysis
4. Conclusions
- OM-SEM characterization indicated that the martensitic PHSS presented a microstructure with a martensitic (α’) phase and a semi-austenitic PHSS containing a microstructure of austenite (γ) and delta (δ) ferrite phases, respectively. Based on the values obtained from PREN, the semi-austenitic PHSS (25.37) presented a higher corrosion resistance than the martensitic PHSS (17.26).
- Potentiodynamic polarization results indicated that martensitic and semi-austenitic steels passivated in nitric acid showed lower corrosion resistance values (in the order of ×10−4 mm/yr).
- Nitric acid passivation made the surface susceptible to localized corrosion. The potentiodynamic polarization curves for PHSS immersed in 5 wt.% NaCl solution indicated that the passivation showed a trend since it is not fully defined.
- Despite having a well-defined passivation layer, passive samples studied in H2SO4 solution presented an increase in corrosion kinetics.
- Passivation current, ipass, was found to have higher values for samples exposed to sulfuric acid (from 0.506 up to 9.837 µA·cm−2).
- XPS analysis determined that the different chemical species on the surface film of martensitic and semi-austenitic PHSS in this work included Cr2O3 and Fe(OH)O. Passive films contained iron and chromium oxide and hydroxide.
- The samples passivated at 70 °C are the ones that presented the best results independently of the steel, passivating solution, and time.
- The citric acid passivation process on PHSS could be a green alternative to the currently employed nitric acid passivation process.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gialanella, S.; Malandruccolo, A. Aerospace Alloys. In Topics in Mining, Metallurgy and Materials Engineering; Bergmann, C.P., Ed.; Springer: Cham, Switzerland, 2020; ISSN 2364-3307. [Google Scholar] [CrossRef]
- Mouritz, P.A. Introduction to Aerospace Materials; Woodhead Publishing: Cambridge, UK, 2012. [Google Scholar]
- Gloria, A.; Montanari, R.; Richetta, M.; Varone, A. Alloys for aeronautic applications: State of the art and perspectives. Metals 2019, 9, 662. [Google Scholar] [CrossRef] [Green Version]
- Cabral-Miramontes, J.A.; Barceinas-Sánchez, J.D.O.; Poblano-Salas, C.A.; Pedraza-Basulto, G.K.; Nieves-Mendoza, D.; Zambrano-Robledo, P.C.; Almeraya-Calderón, F.; Chacón-Nava, J.G. Corrosion behavior of AISI 409Nb stainless steel manufactured by powder metallurgy exposed in H2SO4 and NaCl solutions. Int. J. Electrochem. Sci. 2013, 8, 564–577. [Google Scholar]
- Lara-Banda, M.; Gaona-Tiburcio, C.; Zambrano-Robledo, P.; Delgado, -E.M.; Cabral-Miramontes, J.A.; Nieves-Mendoza, D.; Maldonado-Bandala, E.; Estupiñan-López, F.; Chacón-Nava, J.G.; Almeraya-Calderón, F. Alternative to nitric acid passivation of 15-5 and 17-4PH stainless steel using electrochemical techniques. Materials 2020, 13, 2836. [Google Scholar] [CrossRef] [PubMed]
- Lara Banda, M.; Gaona-Tiburcio, C.; Zambrano-Robledo, P.; Cabral, M.J.A.; Estupinan, F.; Baltazar-Zamora, M.A.; Almeraya-Calderon, F. Corrosion Behaviour of 304 Austenitic, 15–5PH and 17-4PH Passive Stainless Steels in acid solutions. Int. J. Electrochem. Sci. 2018, 13, 10314–10324. [Google Scholar] [CrossRef]
- Lopes, J.C. Material selection for aeronautical structural application. Sci. Technol. Adv. Mat. 2008, 20, 78–82. [Google Scholar]
- Lo, K.H.; Shek, C.H.; Lai, J.K.L. Recent developments in stainless steels. Mater. Sci. Eng. R 2009, 65, 39–104. [Google Scholar] [CrossRef]
- Mortezaei, S.; Arabi, H.; Seyedein, H.; Momeny, A.; Soltanalinezhad, M. Investigation on Microstructure Evolution of a Semi-Austenitic Stainless Steel Through Hot Deformation. Iran. J. Mater. Sci. Eng. 2020, 17, 60–90. [Google Scholar] [CrossRef]
- Ha, H.Y.; Jang, J.H.; Lee, T.H.; Won, C.; Lee, C.H.; Moon, J.; Lee, C.G. Investigation of the localized corrosion and passive behavior of type 304 stainless steels with 0.2–1.8 wt% B. Materials 2018, 11, 2097. [Google Scholar] [CrossRef] [Green Version]
- Abdelshehid, M.; Mahmodieh, K.; Mori, K.; Chen, L.; Stoyanov, P.; Davlantes, D.; Foyos, J.; Ogren, J.; Clark, R., Jr.; Es-Said, O.S. On the correlation between fracture toughness and precipitation hardening heat treatments in 15-5PH stainless steel. Eng. Fail. Anal. 2007, 14, 626–631. [Google Scholar] [CrossRef]
- Esfandiari, M.; Dong, H. The corrosion and corrosion–wear behaviour of plasma nitrided 17-4PH precipitation hardening stainless steel. Surf. Coat. Technol. 2007, 202, 466–478. [Google Scholar] [CrossRef]
- Dong, H.; Esfandiari, M.; Li, X.Y. On the microstructure and phase identification of plasma nitrided 17-4PH precipitation hardening stainless steel. Surf. Coat. Technol. 2008, 202, 2969–2975. [Google Scholar] [CrossRef]
- Hsiao, C.N.; Chiou, C.S.; Yang, J.R. Aging reactions in a 17-4 PH stainless steel. Mater. Chem. Phys. 2002, 74, 134–142. [Google Scholar] [CrossRef]
- Gladman, T. Precipitation hardening in metals. Mater. Sci. Technol. 1999, 15, 30–36. [Google Scholar] [CrossRef]
- Schade, C.; Stears, P.; Lawley, A.; Doherty, R. Precipitation Hardening PM Stainless Steels. Int. J. Powder Metall. 2006, 1, 51–59. [Google Scholar]
- Cobb, H.M. The History of Stainless Steel; ASM International: Geauga, OH, USA, 2010. [Google Scholar]
- Farrar, J. The Alloy Tree—A Guide to Low-Alloy Steels, Stainless Steels and Nickel-Base Alloys; Woodhead Publishing Limited and CRC Press: Sawston, UK, 2004. [Google Scholar]
- Baltazar, M.A.; Bastidas, D.M.; Santiago, G.; Mendoza, J.M.; Gaona, C.; Bastidas, J.M.; Almeraya, F. Effect of silica fume and fly ash admixtures on the corrosion behavior of AISI 304 embedded in concrete exposed in 3.5% NaCl solution. Materials 2019, 12, 4007. [Google Scholar] [CrossRef] [Green Version]
- Schmuki, P. From bacon to barriers: A review on the passivity of metal and alloys. J. Solid. State Electr. 2002, 6, 145–164. [Google Scholar] [CrossRef]
- ASTM A967-17; Standard Specification for Chemical Passivation Treatments for Stainless Steel Parts. ASTM International: West Conshohocken, PA, USA, 1999.
- Shibata, T. Stochastic studies of passivity breakdown. Corros. Sci. 1990, 31, 413–423. [Google Scholar] [CrossRef]
- Ashassi-Sorkhabi, H.; Seifzadeh, D.; Raghibi-Boroujeni, M. Analysis of electrochemical noise data in both time and frequency domains to evaluate the effect of ZnO nanopowder addition on the corrosion protection performance of epoxy coatings. Arab. J. Chem. 2016, 9, S1320–S1327. [Google Scholar] [CrossRef] [Green Version]
- Isselin, J.; Kasada, R.; Kimura, A. Effects of aluminum on the corrosion behavior of 16% Cr ODS ferritic steels in a nitric acid solution. J. Nucl. Sci. Techmol. 2011, 48, 169–171. [Google Scholar] [CrossRef]
- Martínez-Villafañe, A.; Almeraya-Calderón, M.F.; Gaona-Tiburcio, C.; Gonzalez-Rodriguez, J.G.; Porcayo-Calderón, J. High-Temperature Degradation and Protection of Ferritic and Austenitic Steels in Steam Generators. J. Mater. Eng. Perform. 1998, 7, 108–113. [Google Scholar] [CrossRef]
- Lewis, P.; Kolody, M. Alternative to Nitric Acid Passivation of Stainless Steel Alloys. In Technology Evaluation for Environmental Risk Mitigation Compendium, Proceedings of the NASA Technology Evaluation for Environmental Risk Mitigation Principal Center (TEERM); Department od Defense (DoD) and NASA, Kennedy Space Center: Merritt Island, FL, USA, 2008. [Google Scholar]
- Yasensky, D.; Reali, J.; Larson, C.; Carl, C. Citric acid passivation of stainless steel. In Proceedings of the Aircraft Airworthiness and Sustainment Conference, San Diego, CA, USA, 18–21 April 2009. [Google Scholar]
- Gaydos, S.P. Passivation of aerospace stainless parts with citric acid solutions. Plat. Surf. Finish. 2003, 90, 20–25. [Google Scholar]
- Suresh, G.U.; Kamachi, M.S. Electrochemical Noise Analysis of Pitting Corrosion of Type 304L Stainless Steel. Corrosion 2014, 70, 283–293. [Google Scholar] [CrossRef]
- Montoya-Rangel, M.; de Garza-Montes, O.N.; Gaona-Tiburcio, C.; Colás, R.; Cabral-Miramontes, J.; Nieves-Mendoza, D.; Maldonado-Bandala, E.; Chacón-Nava, J.; Almeraya-Calderón, F. Electrochemical Noise Measurements of Advanced High-Strength Steels in Different Solutions. Metals 2020, 10, 1232. [Google Scholar] [CrossRef]
- Bragaglia, M.; Cherubini, V.; Cacciotti, I.; Rinaldi, M.; Mori, S.; Soltani, P.; Nanni, F.; Kaciulis, S.; Montesperelli, G. Citric Acid Aerospace Stainless Steel Passivation: A Green Approach. In Proceedings of the CEAS Aerospace Europe Conference 2015, Delft, The Netherlands, 7–11 September 2015. [Google Scholar]
- Marcelin, S.; Pébèrea, N.; Régnierb, S. Electrochemical characterisation of a martensitic stainless steel in a neutral chloride solution. Electrochim. Acta 2013, 87, 32–40. [Google Scholar] [CrossRef] [Green Version]
- El-Taib Heakal, F.; Ghoneim, A.; Fekry, A. Stability of spontaneous passive films on high strength Mo-containing stainless steels in aqueous solutions. J. Appl. Electrochem. 2007, 37, 405–413. [Google Scholar] [CrossRef]
- Ameer, M.A.; Fekry, A.M.; El-TaibHeakal, F. Electrochemical behaviour of passive films on molybdenum-containing austenitic stainless steels in aqueous solutions. Electrochim. Acta 2007, 50, 43–49. [Google Scholar] [CrossRef]
- El-Taib Heakal, F.; Ameer, M.A.; El-Aziz, A.M.; Fekry, A.M. Electrochemical behavior of Mo-containing austenitic stainless steel in buffer solutions. Mater. Sci. Eng. Technol. 2004, 35, 407–413. [Google Scholar] [CrossRef]
- Tseng, K.H.; Chen, Y.C.; Chen, Y.C. Micro Plasma Arc Welding of AM 350 Precipitation Hardening Alloys. Appl. Mech. Mater. 2012, 121, 2681–2685. [Google Scholar] [CrossRef]
- Shaikh, H.; George, G.; Khatak, H.S. Failure analysis of an AM 350 steel bellows. Eng. Fail. Anal. 2001, 8, 571–576. [Google Scholar] [CrossRef]
- Fakić, B.; Ćubela, D. Review of the Development of Research in the Design of Semi Austenitic Stainless Steel 17-7PH. In Proceedings of the 17th International Research/Expert Conference ”Trends in the Development of Machinery and Associated Technology” TMT 2013, Istanbul, Turkey, 10–11 September 2013; pp. 113–116. [Google Scholar]
- Warren, J.; Wei, D.Y. The low cycle fatigue behavior of the controlled transformation stainless steel alloy AM355 at 121, 204 and 315 °C. Mater. Sci. Eng. A 2008, 475, 148–156. [Google Scholar] [CrossRef]
- Favor, R.J.; Deel, O.L.; Achbach, W.P. Design Information on AM-350 Stainless Steel for Aircraft and Missiles; Defense Metals Information Center, Battelle Memorial Institute: Columbus, OH, USA, 1961; Volume 156. [Google Scholar]
- Kumar, T.S.; Ramanujam, R.; Vignesh, M.; Rohith, D.; Manoj, V.; Sankar, P.H. Comparative machining studies on custom 450 alloy with TiCN, TiAlN coated and uncoated carbide tools using Taguchi-Fuzzy logic approach. Mater. Res. Express 2019, 6, 066411. [Google Scholar] [CrossRef]
- Bocchetta, P.; Chen, L.-Y.; Tardelli, J.D.C.; Reis, A.C.D.; Almeraya-Calderón, F.; Leo, P. Passive layers and corrosion resistance of biomedical Ti-6Al-4V and β-Ti alloys. Coatings 2021, 11, 487. [Google Scholar] [CrossRef]
- Lin, C.K.; Tsai, W.J. Corrosion fatigue behaviour of a 15Cr-6Ni precipitation-hardening stainless steel in different tempers. Fatigue Fract. Eng. Mater. Struct. 2000, 23, 489–497. [Google Scholar] [CrossRef]
- Mollapour, Y.; Poursaeidi, E. Experimental and numerical analysis of Pitting Corrosion in CUSTOM 450 Stainless Steel. Eng. Fail. Anal. 2021, 128, 105589. [Google Scholar] [CrossRef]
- Lin, C.K.; Chu, C.C. Mean stress effects on low-cycle fatigue for a precipitation-hardening martensitic stainless steel in different tempers. Fatigue Fract. Eng. Mater. Struct. 2000, 23, 545–553. [Google Scholar] [CrossRef]
- Samaniego-Gámez, O.; Almeraya-Calderón, F.; Chacón-Nava, J.; Maldonado-Bandala, E.; Nieves-Mendoza, D.; Flores-De los Rios, J.P.; Jáquez-Muñoz, J.M.; Delgado, A.D.; Gaona-Tiburcio, C. Corrosion Behavior of Passivated CUSTOM450 and AM350 Stainless Steels For Aeronautical Applications. Metals 2022, 12, 666. [Google Scholar] [CrossRef]
- AM 350 Technical Data. Available online: https://www.hightempmetals.com/techdata/hitempAM350data.php (accessed on 30 April 2022).
- Custom 450® Stainless Steel. Available online: https://www.ulbrich.com/uploads/data-sheets/Custom-450-Stainless-Steel-Wire-UNS-S45000.pdf (accessed on 30 April 2022).
- ASTM E3-95; Standard Practice for Preparation of Metallographic Specimens. ASTM International: West Conshohocken, PA, USA, 1995.
- ASTM A380-17; Standard Practice for Cleaning, Descaling and Passivation of Stainless-Steel Parts, Equipment, and Systems. ASTM International: West Conshohocken, PA, USA, 1999.
- Olguin Coca, F.J.; Loya Tello, M.U.; Gaona-Tiburcio, C.; Romero, J.A.; Martínez-Villafañe, A.; Maldonado, B.E.; Almeraya-Calderón, F. Corrosion Fatigue of Road Bridges: A review. Int. J. Electrochem. Sci. 2011, 6, 3438–3451. [Google Scholar]
- Samaniego-Gámez, P.; Almeraya-Calderón, F.; Martin, U.; Ress, J.; Gaona-Tiburcio, C.; Silva-Vidaurri, L.; Cabral-Miramontes, J.; Bastidas, J.M.; Chacón-Nava, J.G.; Bastidas, D.M. Efecto del tratamiento de sellado en el comportamiento frente a corrosión de la aleación anodizada de aluminio-litio AA2099. Rev. Met. 2020, 56, e180. [Google Scholar] [CrossRef]
- ASTM G5-11; Standard Reference Test Method for Making Potentiostatic and Potentiodynamic Anodic Polarization Measurements. ASTM International: West Conshohocken, PA, USA, 2011.
- Gupta, A.; Bhargava, A.K.; Tewari, R.; Tiwari, A.N. TEM studies of boron-modified 17Cr-7Ni precipitation-hardenable stainless steel via rapid solidification route. Metal. Mater. Trans. A 2013, 44, 4248–4256. [Google Scholar] [CrossRef]
- Xu, X.L.; Yu, Z.W. Metallurgical analysis on a bending failed pump-shaft made of 17-7PH precipitation-hardening stainless steel. J. Mater. Process. Technol. 2008, 198, 254–259. [Google Scholar] [CrossRef]
- Abedi, H.R.; Hanzaki, A.Z.; Haghdadi, N.; Hodgson, P.D. Substructure induced twinning in low density steel. Scr. Mater. 2017, 128, 69–73. [Google Scholar] [CrossRef]
- The British Stainless Steel Association 2022. Available online: https://bssa.org.uk/bssa_articles/calculation-of-pren/accesseddate (accessed on 2 June 2022).
- Schweitzer, P. Fundamentals of Metallic Corrosion: Atmospheric and Media Corrosion of Metals; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2007; ISBN 10:0-8493-8243-2. [Google Scholar]
- Tafel, J. Über die Polarisation bei kathodischer Wasserstoffentwicklung. Z. Phys. Chem. 1905, 50, 641–712. [Google Scholar] [CrossRef]
- Pellegrini-Cervantes, M.J.; Almeraya-Calderon, F.; Borunda-Terrazas, A.; Bautista-Margulis, R.G.; Chacón-Nava, J.G.; Fajardo-San-Miguel, G.; Almaral-Sanchez, J.L.; Barrios-Durstewitz, C.; Martinez-Villafañe, A. Corrosion Resistance, Porosity and Strength of lended Portland Cement Mortar Containing Rice Husk Ash and Nano-SiO2. Int. J. Electrochem. Sci. 2013, 8, 10697–10710. [Google Scholar]
- Butler, J.A.V. Studies in heterogeneous equilibria. Part II.—The kinetic interpretation on the Nernst theory of electromotive force. Trans. Faraday Soc. 1924, 19, 729–733. [Google Scholar] [CrossRef]
- Jaquez-Muñoz, J.; Gaona-Tiburcio, C.; Lira-Martinez, A.; Zambrano-Robledo, P.; Maldonado-Bandala, E.; Samaniego-Gamez, O.; Nieves-Mendoza, D.; Olguin-Coca, J.; Estupiñan-Lopez, F.; Almeraya-Calderon, F. Susceptibility to pitting corrosion of Ti-CP2, Ti-6Al-2Sn-4Zr-2Mo, and Ti-6Al-4V alloys for aeronautical applications. Metals 2021, 11, 1002. [Google Scholar] [CrossRef]
- Zeng, Y.; Kang, L.; Wu, Y.; Wan, S.; Liao, B.; Li, N.; Guo, X. Melamine modified carbon dots as high effective corrosion inhibitor for Q235 carbon steel in neutral 3.5 wt% NaCl solution. J. Mol. Liq. 2022, 349, 118108. [Google Scholar] [CrossRef]
- Wan, S.; Zhan, T.; Chen, H.; Liao, B.; Guo, X. Kapok leaves extract and synergistic iodide as novel effective corrosion inhibitors for Q235 carbon steel in H2SO4 medium. Ind. Crops Prod. 2022, 178, 114649. [Google Scholar] [CrossRef]
- Buchanan, A.R.; Stansbury, E.E. Handbook of Environmental Degradation of Materials (Second Edition) Chapter 4—Electrochemical Corrosion; Elsevier Inc.: Amsterdam, The Netherlands, 2012; Volume 1, pp. 87–125. [Google Scholar] [CrossRef]
- Cheng, M.; Chaofang, D.; Zhongyu, C.; Kui, X. A comparative study of primary and secondary passive films formed on AM355 stainless steel in 0.1 M NaOH. Appl. Surf. Sci. 2018, 427, 763–773. [Google Scholar] [CrossRef]
- Betova, I.; Bojinov, M.; Laitinen, T.; Mäkelä, K.; Pohjanne, P.; Saario, T. The transpassive dissolution mechanism of highly alloyed stainless steels: I. Experimental results and modelling procedure. Corros. Sci. 2002, 44, 2675–2697. [Google Scholar] [CrossRef]
- Ye, W.; Li, Y.; Wang, F. The improvement of the corrosion resistance of 309stainless steel in the transpassive region by nano-crystallization. Electrochim. Acta 2009, 54, 1339–1349. [Google Scholar] [CrossRef]
- Gaona-Tiburcio, C.; Almeraya-Calderón, F.; Chacon-Nava, J.; Matutes-Aquino, A.M.; Martinez-Villafañe, A. Electrochemical response of permanent magnets in different solutions. J. Alloys Compd. 2004, 369, 78–80. [Google Scholar] [CrossRef]
- Cabral-Miramontes, J.A.; Bastidas, M.D.; Baltazar, M.A.; Zambrano-Robledo, P.; Bastidas, J.M.; Almeraya-Calderón, F.; Gaona-Tiburcio, C. Corrosion behavior of Zn-TiO2 and Zn-ZnO Electrodeposited Coatings in 3.5% NaCl solution. Int. J. Electrochem. Sci. 2019, 14, 4226–4239. [Google Scholar] [CrossRef]
- Huang, J.; Wu, X.; Han, E.H. Electrochemical properties and growth mechanism of passive films on Alloy 690 in high-temperature alkaline environments. Corros. Sci. 2010, 52, 3444–3452. [Google Scholar] [CrossRef]
- Calinski, C.; Strehblow, H.H. ISS depth profiles of the passive layer on Fe/Cr alloys. J. Electrochem. Soc. 1989, 36, 1328–1331. [Google Scholar] [CrossRef]
- Gaona-Tiburcio, C.; Aguilar, L.M.R.; Zambrano-Robledo, P.; Estupiñán-López, F.; Cabral-Miramontes, J.A.; Nieves-Mendoza, D.; Castillo-González, E.; Almeraya-Calderón, F. Electrochemical Noise Analysis of Nickel Based Superalloys in Acid Solutions. Int. J. Electrochem. Sci. 2014, 9, 523–533. [Google Scholar]
- Corral-Higuera, R.; Arredondo-Rea, P.; Neri-Flores, M.A.; Gómez-Soberón, J.M.; Almaral-Sánchez, J.L.; Castorena-González, J.C.; Almeraya-Calderón, F. Chloride ion penetrability and corrosion behavior of steel in concrete with sustainability characteristics. Int. J. Electrochem. Sci. 2011, 6, 958–970. [Google Scholar]
- Núñez-Jaquez, R.E.; Buelna-Rodríguez, J.E.; Barrios-Durstewitz, C.P.; Gaona-Tiburcio, C.; Almeraya-Calderón, F. Corrosion of modified concrete with sugar cane bagasse ash. Int. J. Corros. 2012, 2012, 451864. [Google Scholar] [CrossRef] [Green Version]
- Fattah-alhosseini, A.; Saatchi, A.; Golozar, M.A.; Raeissi, K. The transpassive dissolution mechanism of 316L stainless steel. Electrochim. Acta 2009, 54, 3645–3650. [Google Scholar] [CrossRef]
- Macdonald, D.; Urquidi-Macdonald, M. Deterministic models for passivity breakdown. Corros. Sci. 1990, 31, 425–430. [Google Scholar] [CrossRef]
- Macdonald, D.D. Point defect model for the passive state. J. Electrochem. Soc. 1992, 139, 3434–3449. [Google Scholar] [CrossRef]
- Cui, Z.; Wang, L.; Ni, H.; Hao, W.; Man, C.; Chen, S.; Li, X. Influence of temperature on the electrochemical and passivation behavior of 2507 super duplex stainless steel in simulated desulfurized flue gas condensates. Corros. Sci. 2017, 118, 31–48. [Google Scholar] [CrossRef]
- Bojinov, M.; Betova, I.; Fabricius, G.; Laitinen, T.; Saario, T. The stability of the passive state of iron–chromium alloys in sulphuric acid solution. Corros. Sci. 1999, 41, 1557–1584. [Google Scholar] [CrossRef]
- Bojinov, M.; Fabricius, G.; Kinnunen, P.; Laitinen, T.; Mäkelä, K.; Saario, T.; Sundholm, G. The mechanism of transpassive dissolution of Ni–Cr alloys in sulphate solutions. Electrochem. Acta 2000, 45, 2791–2802. [Google Scholar] [CrossRef]
- Maldonado-Bandala, E.E.; Jiménez- Quero, V.; Olguin-Coca, F.; Lizarraga, M.L.G.; Baltazar-Zamora, M.A.; Ortiz-C, A.; Almeraya, C.F.; Zambrano, R.P.; Gaona-Tiburcio, C. Electrochemical Characterization of Modified Concretes with Sugar Cane Bagasse Ash. Int. J. Electrochem. Sci. 2011, 6, 4915–4926. [Google Scholar]
- Radhakrishnamurty, P.; Adaikkalam, P. pH-potential diagrams at elevated temperatures for the chromium/water systems. Corros. Sci. 1982, 22, 753–773. [Google Scholar] [CrossRef]
- Fattah-alhosseini, A.; Taheri Shoja, S.; Heydari Zebardast, B.; Mohamadian Samim, P. An Electrochemical Impedance Spectroscopic Study of the Passive State on AISI 304 Stainless Steel. Int. J. Electrochem. 2011, 2011, 8. [Google Scholar] [CrossRef] [Green Version]
- Castro, B.E.; Vilche, R.J. Investigation of passive layers on iron and iron-chromium alloys by electrochemical impedance spectroscopy. Electrochim. Acta 1993, 38, 1567–1572. [Google Scholar] [CrossRef]
- Martinez-Villafane, A.; Chacon-Nava, J.G.; Gaona-Tiburcio, C.; Almeraya-Calderon, F.; Dominguez-Patino, G.; Gonzalez-Rodriguez, J.G. Oxidation performance of a Fe–13Cr alloy with additions of rare earth elements. Mater. Sci. Eng. A 2003, 363, 15–19. [Google Scholar] [CrossRef]
- Cabral Miramontes, J.A.; Barceinas Sánchez, J.D.O.; Almeraya Calderón, F.; Martínez Villafañe, A.; Chacón Nava, J.G. Effect of Boron Additions on Sintering and Densification of a Ferritic Stainless Steel. J. Mater. Eng. Perform. 2010, 19, 880–884. [Google Scholar] [CrossRef]
- Hryniewicz, T.; Rokosz, K.; Rokicki, R. Electrochemical and XPS studies of AISI 316L stainless steel after electropolishing in a magnetic field. Corros. Sci. 2008, 50, 2676–2681. [Google Scholar] [CrossRef]
- Hermas, A.A. XPS analysis of the passive film formed on austenitic stainless steel coated with conductive polymer. Corros. Sci. 2008, 50, 2498–2505. [Google Scholar] [CrossRef]
- Zhidkov, I.S.; Kukharenko, A.I.; Makarov, A.V.; Savrai, R.A.; Gavrilov, N.V.; Cholakh, S.O.; Kurmaev, E.Z. XPS characterization of surface layers of stainless steel nitrided in electron beam plasma at low temperature. Surf. Coat. Technol 2020, 386, 125492. [Google Scholar] [CrossRef]
- Natarajan, R.; Palaniswamy, N.; Natesan, M.; Muralidharan, V.S. XPS analysis of passive film on stainless steel. Corros. J. 2009, 2, 114–124. [Google Scholar] [CrossRef] [Green Version]
- Jung, R.H.; Tsuchiya, H.; Fujimoto, S. XPS characterization of passive films formed on Type 304 stainless steel in humid atmosphere. Corros. Sci. 2012, 58, 62–68. [Google Scholar] [CrossRef]
- Mesquita, T.J.; Chauveau, E.; Mantel, M.; Nogueira, R.P. A XPS study of the Mo effect on passivation behaviors for highly controlled stainless steels in neutral and alkaline conditions. Appl. Surf. Sci. 2013, 270, 90–97. [Google Scholar] [CrossRef]
- Macdonald, D.D. The history of the Point Defect Model for the passive state: A brief review of film growth aspects. Electrochim. Acta 2011, 56, 1761–1772. [Google Scholar] [CrossRef]
- Jinlong, L.; Tongxiang, L.; Chen, W.; Limin, D. Comparison of corrosion properties of passive films formed on coarse grained and ultrafine grained AISI 2205 duplex stainless steels. J. Electroanal. Chem. 2015, 757, 263–269. [Google Scholar] [CrossRef]
- Feng, Z.; Cheng, X.; Dong, C.; Xu, L.; Li, X. Passivity of 316L stainless steel in borate buffer solution studied by Mott–Schottky analysis, atomic absorption spectrometry and X-ray photoelectron spectroscopy. Corros. Sci. 2010, 52, 3646–3653. [Google Scholar] [CrossRef]



| PHSS | Elements | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cr | Ni | Mo | Mn | Cu | Ti | Nb | N | Si | S | C | Fe | |
| Martensitic | 14.0–16.0 | 5.0–7.0 | 0.50–1.0 | 1.00 | 1.25–1.75 | 0.90–1.40 | 0.5–0.75 | ≤0.1 | 1.00 | 0.030 | ≤0.05 | Balance |
| Semi-austenitic | 16.0–17.0 | 4.0–5.0 | 2.50–3.25 | 0.50–1.25 | – | – | – | 0.07–0.13 | ≤0.50 | 0.030 | 0.07–0.11 | Balance |
| PHSS | Passivation Baths | Time (min) | Temperature (°C) | Nomenclature Samples * |
|---|---|---|---|---|
| semi-austenitic | C6H8O7 | 50 | 49 | semi-austenitic-CA-75 min-49 °C |
| HNO3 | 50 | 70 | semi-austenitic-NA–50 min-70 °C | |
| HNO3 | 50 | 70 | semi-austenitic-NA–50 min-70 °C | |
| C6H8O7 | 75 | 49 | semi-austenitic-CA–75 min-49 °C | |
| martensitic | HNO3 | 75 | 70 | martensitic-NA–75 min–70 °C |
| HNO3 | 50 | 49 | martensitic-NA–50 min–49 °C | |
| C6H8O7 | 50 | 49 | martensitic-CA–50 min–49 °C | |
| C6H8O7 | 75 | 49 | martensitic-CA-75 min-49 °C |
| PHSS | Cr | Mo | N | PREN |
|---|---|---|---|---|
| Martensitic | 14.0–16.0 | 0.50–1.0 | ≤0.1 | 17.26 |
| Semi-austenitic | 16.0–17.0 | 2.50–3.25 | 0.07–0.13 | 25.37 |
| Sample | Ecorr (Volts) | Epitt (Volts) | icorr (µA·cm−2) | ipass (µA·cm−2) | Range Passive (Volts) | CR (mm/yr) |
|---|---|---|---|---|---|---|
| Semi-austenitic-CA–50 min–49 °C | −0.365 | 0.422 | 7.82 × 10−1 | 0.352 | 0.736 | 1.26 × 10−3 |
| Semi-austenitic-NA–50 min–70 °C | −0.234 | 0.934 | 1.58 × 10−1 | 0.082 | 1.119 | 2.55 × 10−4 |
| Martensitic-NA–75 min–70 °C | −0.234 | 0.565 | 1.37 × 10−1 | 0.119 | 0.740 | 2.25 × 10−4 |
| Martensitic-CA–50 min–49 °C | −0.384 | 0.401 | 3.44 × 10−1 | 0.480 | 0.688 | 5.65 × 10−3 |
| Sample | Ecorr (Volts) | Epitt (Volts) | icorr (µA·cm−2) | ipass (µA·cm−2) | Range passive (Volts) | CR (mm/yr) |
|---|---|---|---|---|---|---|
| Semi-austenitic-CA–75 min–49 °C | −0.314 | 0.875 | 0.7 × 10−1 | 9.837 | 1.055 | 1.13 × 10−2 |
| Semi-austenitic-NA–50 min–70 °C | −0.226 | 0.766 | 2.24 × 10−1 | 0.506 | 0.692 | 3.62 × 10−4 |
| Martensitic-NA–50 min–49 °C | −0.282 | 0.898 | 2.25 × 10−1 | 8.529 | 0.899 | 3.69 × 10−2 |
| Martensitic-CA–75 min–49 °C | −0.349 | 0.857 | 5.37 × 10−1 | 8.193 | 0.880 | 8.82 × 10−2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almeraya-Calderón, F.; Samaniego-Gámez, O.; Maldonado-Bandala, E.; Nieves-Mendoza, D.; Olguín-Coca, J.; Jáquez-Muñoz, J.M.; Cabral-Miramontes, J.; Flores-De los Rios, J.P.; Bautista-Margulis, R.G.; Gaona-Tiburcio, C. Corrosion Behavior of Passivated Martensitic and Semi-Austenitic Precipitation Hardening Stainless Steel. Metals 2022, 12, 1033. https://doi.org/10.3390/met12061033
Almeraya-Calderón F, Samaniego-Gámez O, Maldonado-Bandala E, Nieves-Mendoza D, Olguín-Coca J, Jáquez-Muñoz JM, Cabral-Miramontes J, Flores-De los Rios JP, Bautista-Margulis RG, Gaona-Tiburcio C. Corrosion Behavior of Passivated Martensitic and Semi-Austenitic Precipitation Hardening Stainless Steel. Metals. 2022; 12(6):1033. https://doi.org/10.3390/met12061033
Chicago/Turabian StyleAlmeraya-Calderón, Facundo, Oliver Samaniego-Gámez, Erick Maldonado-Bandala, Demetrio Nieves-Mendoza, Javier Olguín-Coca, Jesús Manuel Jáquez-Muñoz, José Cabral-Miramontes, Juan Pablo Flores-De los Rios, Raul German Bautista-Margulis, and Citlalli Gaona-Tiburcio. 2022. "Corrosion Behavior of Passivated Martensitic and Semi-Austenitic Precipitation Hardening Stainless Steel" Metals 12, no. 6: 1033. https://doi.org/10.3390/met12061033
APA StyleAlmeraya-Calderón, F., Samaniego-Gámez, O., Maldonado-Bandala, E., Nieves-Mendoza, D., Olguín-Coca, J., Jáquez-Muñoz, J. M., Cabral-Miramontes, J., Flores-De los Rios, J. P., Bautista-Margulis, R. G., & Gaona-Tiburcio, C. (2022). Corrosion Behavior of Passivated Martensitic and Semi-Austenitic Precipitation Hardening Stainless Steel. Metals, 12(6), 1033. https://doi.org/10.3390/met12061033