Sustainable Recovery of Secondary and Critical Raw Materials from Classified Mining Residues Using Mycorrhizal-Assisted Phytoextraction
Abstract
1. Introduction
- New sensitive technologies (such as remote sensing technologies) for mapping and classification of mining wastes containing minerals and metals;
- New solutions for chemical, physical and morphological characterization;
- An eco-innovative methodology for converting mining waste into resource: mycorrhizal-assisted phytoextraction (MAP) of mining wastes, and the consequent accumulation of secondary and critical raw materials (SRMs and CRMs, respectively) in biomass tissues;
- Recovery of SRMs and CRMs from biomass through hydrometallurgy and/or electrochemical methods. The leaching extraction is selective and allows the recovery of all the contained elements because specific reagents are used for each of them, applying innovative processes (e.g., thiourea process, thiosulfate process, etc.). The metals are purified to a high degree of purity by electrochemical methods.

2. Materials and Methods
2.1. Fe and Mn Mine Wastes
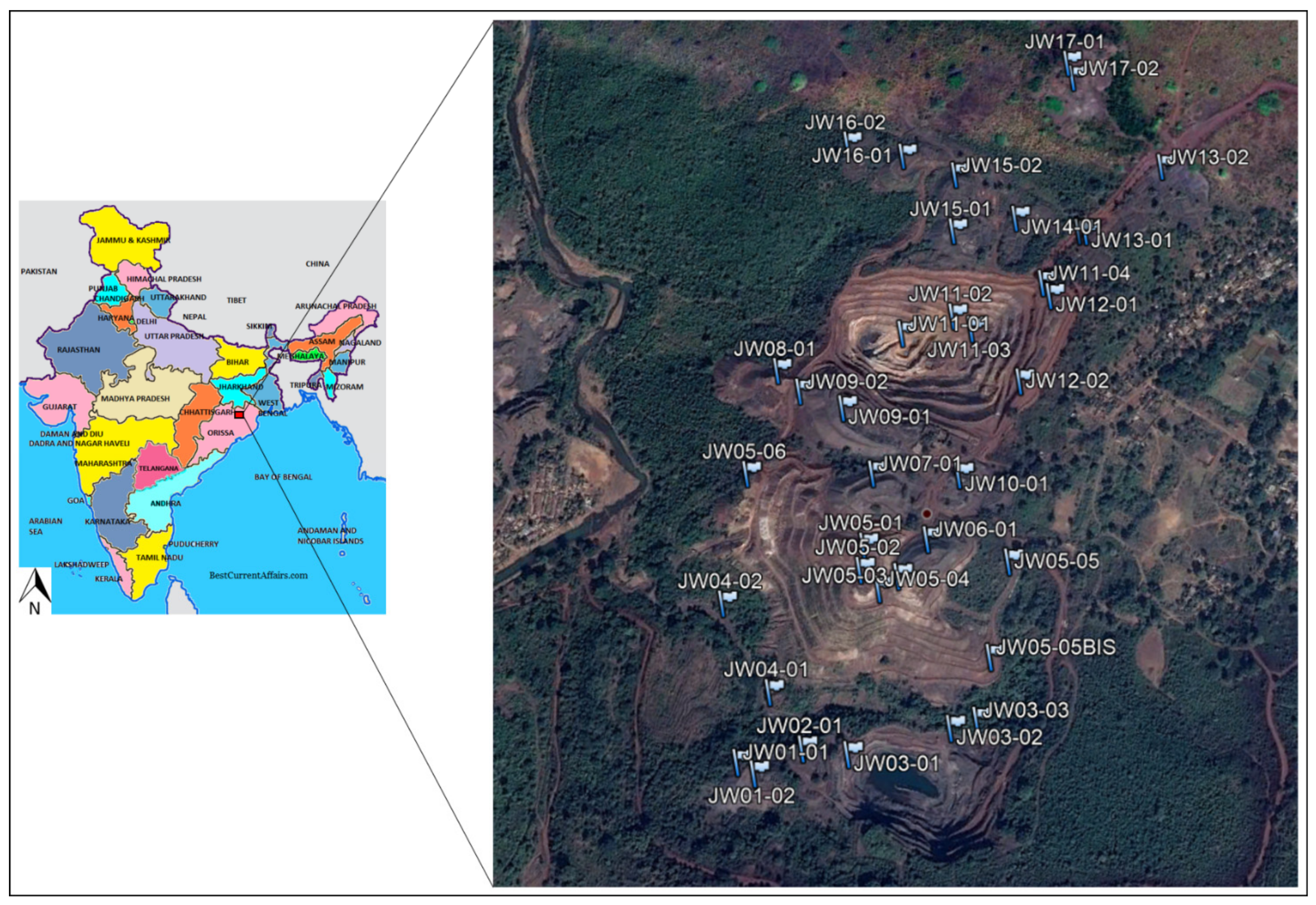
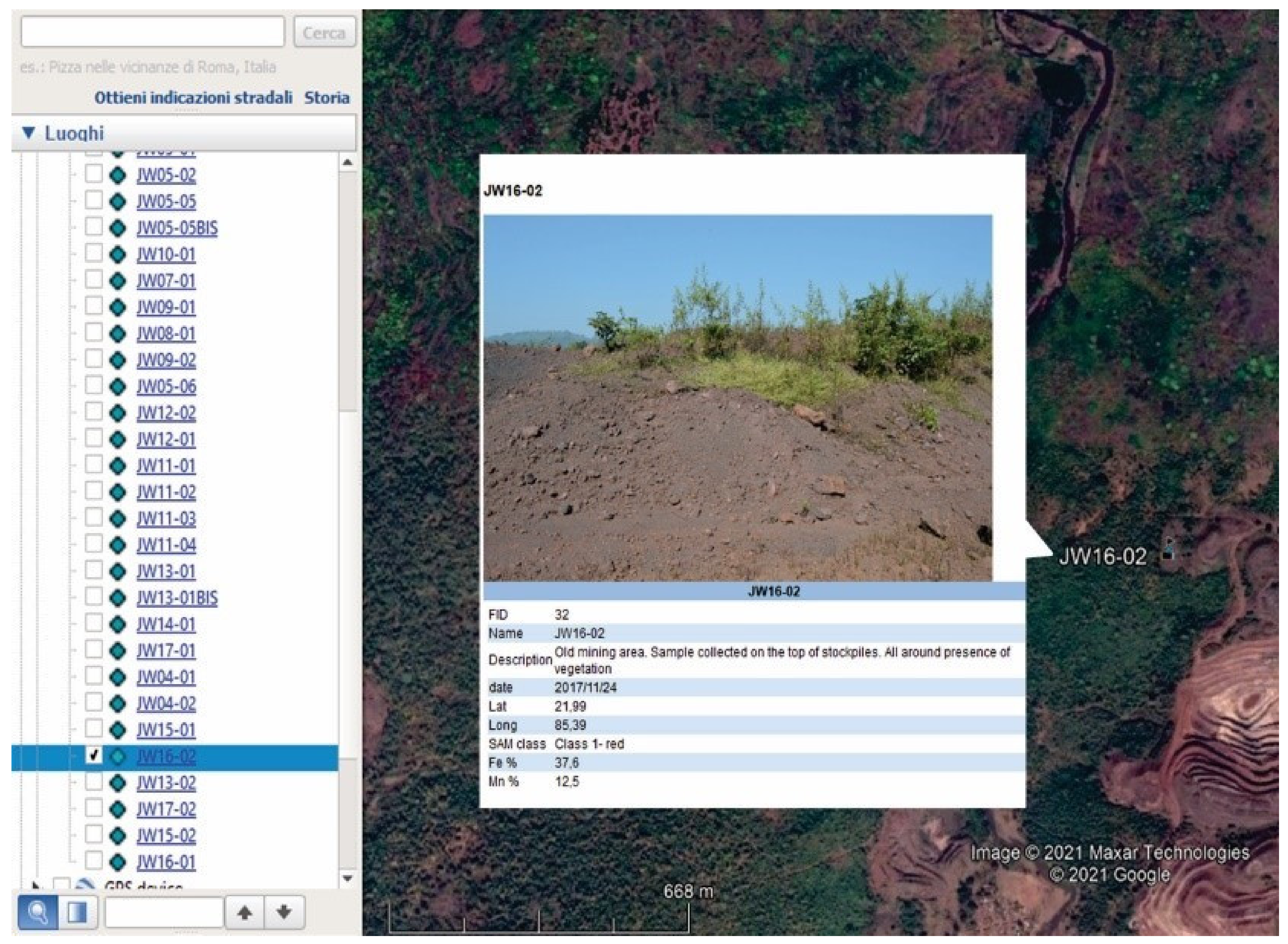
2.2. The Joda West Iron and Manganese Mine Geodatabase
2.3. MAP Test at TRL-2 Scale
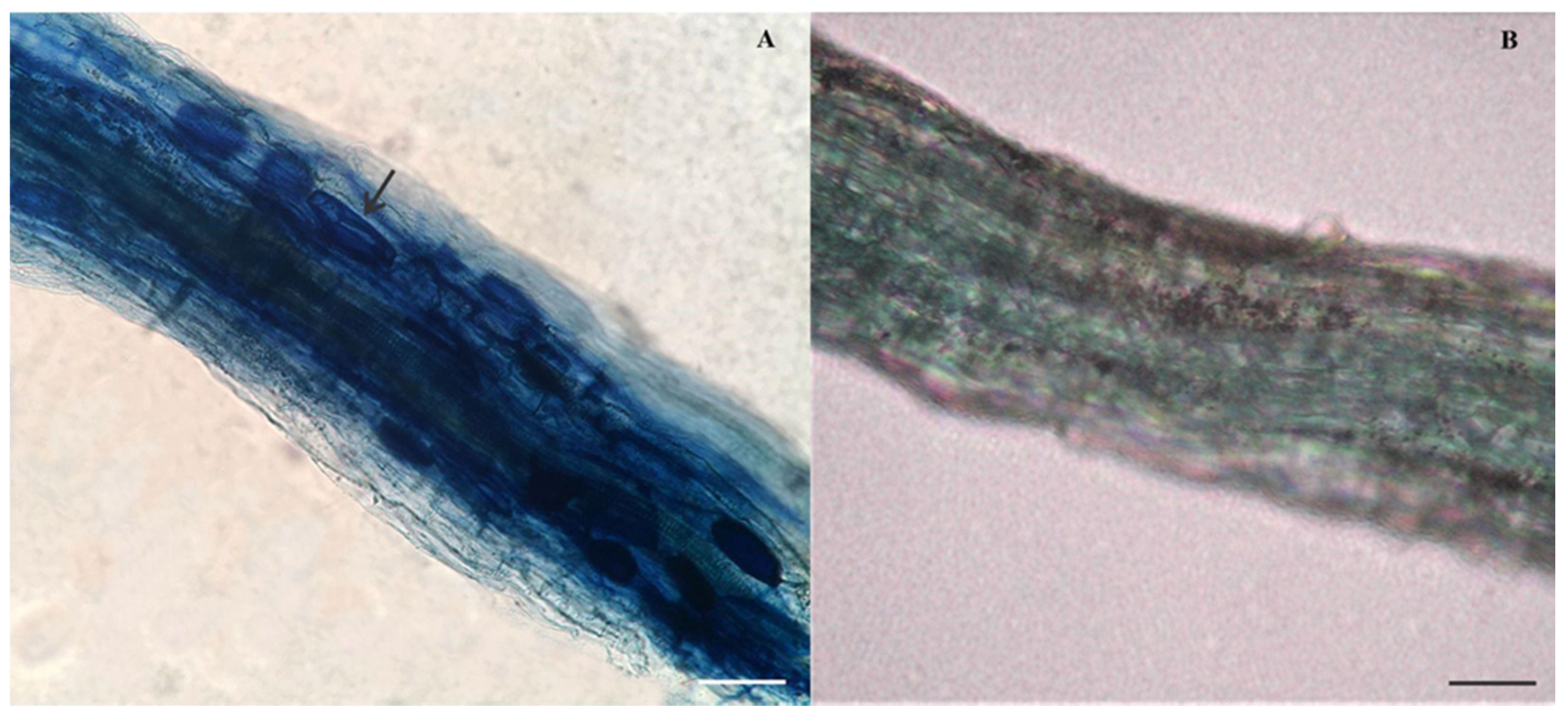
2.3.1. Plant Material and AM Fungal Strain
2.3.2. TRL-2 Experimental Setup
- MA1–4+. Four pots filled with CS and VA in a 1:1 (v/v) ratio, completing to 500 mL and 125 mL granular PS. Considering the CS only, 300–500 ppm ZnSO4 were added. At least three sunflower seeds were sown and inoculated with a piece of 4-month old monoxenic culture (containing approximately 300 spores, mycelia and colonized roots).
- MA1–4−. Four pots filled with mixed CS and VA in a 1:1 (v/v) ratio, completing to 500 mL and 125 mL PS. Considering the CS only, 300–500 ppm ZnSO4 were added. At least three sunflower seeds were sown in each pot.
- B1–4+. Four pots filled with 500 mL FCT and 125 mL PS. At least three sunflower seeds were sown and inoculated with a piece of 4-month old monoxenic culture (containing approximately 300 spores, mycelia and colonized roots).
- B1–4−. Four pots filled with 500 mL FCT and 125 mL PS. At least three sunflower seeds were planted in each pot.
2.3.3. Harvest and Sample Analyses
2.3.4. TXRF Analysis
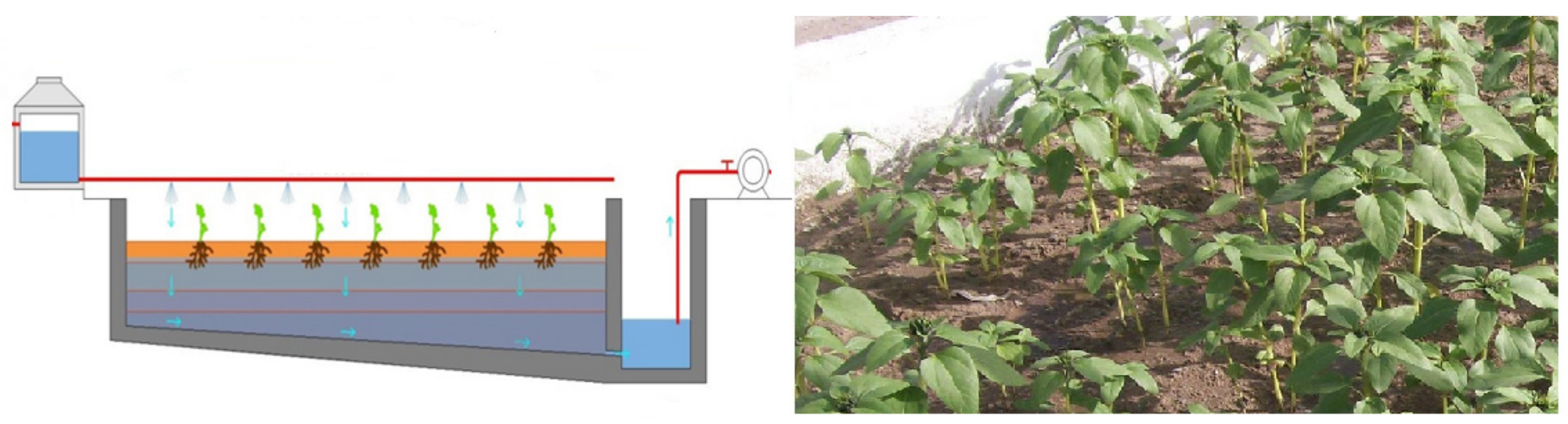
2.4. Bioextracting Potential (BP) in VDM
2.5. Calculations and Statistical Analyses
2.5.1. Bioconcentration Coefficients and Translocation Factor
- Cp(S,R): concentration of element in the aerial (shoots, S) or radicular (roots, R) plant tissue (ppm)
- Csoil: concentration of element in soil (ppm)
- Cp(S): concentration of element in the aerial plant tissue (shoots; ppm)
- Cp(R): concentration of element in the radicular plant tissue (roots; ppm)
2.5.2. Bioextracting Potential (BP)
- Wtot(S,R): Total weight (g) of aerial (shoots, S) and radical (roots, R) plant tissue.
- Cp(S,R): concentration (ppm) of element in the aerial (shoots, S) or radicular (roots, R) plant tissue.
3. Results
MAP Test at TRL-2 Scale
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Treatment | N | Mn (%) | P (ppm) | As (ppm) | Ga (ppm) | Zn (ppm) | Ti (%) | Cr (ppm) | Ni (ppm) | Cu (ppm) | Rb (ppm) | Fe (%) | Sr (ppm) | Al (%) | Ba (%) | K (%) | S (ppm) | Ca (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B− | 4 | 0.068 (0.011) | 2275 (170.8) | 7.050 (2.106) | 5.125 (0.378) | 213.8 (7.411) | 0.135 (0.006) | 87.25 (10.72) | 43.50 (3.317) | 110.5 (3.697) | 206.8 (48.03) | 1.475 (0.096) | 1081 (114.3) | 1.525 (0.250) | 0.043 (0.006) | 1.350 (0.100) | 3450 (264.6) | 5.500 (0.316) |
| B+ | 4 | 0.062 (0.002) | 2125 (50.00) | 8.775 (0.818) | 5.000 (0.683) | 210.8 (18.79) | 0.128 (0.005) | 86.50 (18.23) | 42.25 (1.500) | 108.0 (5.354) | 211.5 (28.21) | 1.425 (0.050) | 1100 (115.5) | 1.363 (0.048) | 0.043 (0.004) | 1.325 (0.096) | 3525 (221.7) | 5.325 (0.126) |
| Sample Type | Biomass (g/sp) | M+ | M− |
|---|---|---|---|
| Contaminated | Shoots | 0.24 a (0.03) | 0.24 a (0.02) |
| Roots | 0.02 b (0.01) | 0.02 b (0.004) | |
| Blank | Shoots | 0.35 c (0.01) | 0.23 a (0.02) |
| Roots | 0.11 d (0.001) | 0.02 b (0.002) |
References
- Guglietta, D.; Belardi, G.; Cappai, G.; Casentini, B.; Godeas, A.; Milia, S.; Passeri, D.; Salvatori, R.; Scotti, A.; Silvani, V.; et al. Toward a multidisciplinary strategy for the classification and reuse of iron and manganese mining wastes. Chem. J. Mold. 2020, 15, 21–30. [Google Scholar] [CrossRef]
- Mathieux, F.; Ardente, F.; Bobba, S.; Nuss, P.; Blengini, G.; Alves Dias, P.; Blagoeva, D.; Torres De Matos, C.; Wittmer, D.; Pavel, C.; et al. Critical Raw Materials and the Circular Economy–Background Report; Publications Office of the European Union: Bruxelles, Belgium, 2017. [Google Scholar] [CrossRef]
- Glick, B.R. Phytoremediation: Synergistic use of plants and bacteria to clean up the environment. Biotechnol. Adv. 2003, 21, 383–393. [Google Scholar] [CrossRef]
- Ker, K.; Charest, C. Nickel remediation by AM colonized sunflower. Mycorrhiza 2010, 20, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Kastori, P.; Petrovic, N.; Petrovic, M. Effects of lead on water relations, proline concentration and nitrate reductase activity in sunflower plants. Acta Agron. Hungar. 1996, 44, 21–28. [Google Scholar]
- Turgut, C.; Pepe, M.K.; Cutright, T.J. The effect of EDTA and citric acid on phytoremediation of Cd, Cr, and Ni from soil using Helianthus annuus. Environ. Pollut. 2004, 131, 147–154. [Google Scholar] [CrossRef]
- Davies, F.T.; Puryear, J.D.; Newton, R.J.; Egilla, J.N.; Saraiva Grossi, J.A. Mycorrhizal fungi increase chromium uptake by sunflower plants: Influence on tissue mineral concentration, growth, and gas exchange. J. Plant. Nutr. 2002, 25, 2389–2407. [Google Scholar] [CrossRef]
- Scotti, A.; Silvani, V.; Cerioni, J.; Visciglia, M.; Benavidez, M.; Godeas, A.A. Pilot testing of a bioremediation system of water and soils contaminated with heavy metals: Vegetable Depuration Module. Int. J. Phytoremediation 2019, 21, 899–907. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: New York, NY, USA, 2008. [Google Scholar]
- Lenoir, I.; Fontaine, J.; Lounès-Hadj Sahraoui, A. Arbuscular mycorrhizal fungal responses to abiotic stresses: A review. Phytochemistry 2016, 123, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Gohre, V.; Paszkowski, U. Contribution of the arbuscular mycorrhizal symbiosis to heavy metal phytoremediation. Planta 2006, 223, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liang, Y.; Han, X.; Chiu, T.Y.; Ghosh, A.; Chen, H.; Tang, M. The roles of arbuscular mycorrhizal fungi (AMF) in phytoremediation and tree-herb interactions in Pb contaminated soil. Sci. Rep. 2016, 6, 20469. [Google Scholar] [CrossRef]
- Rivelli, A.R.; De Maria, S.; Puschenreiter, M.; Gherbin, P. Accumulation of cadmium, zinc, and copper by Helianthus annuus L.: Impact on plant growth and uptake of nutritional elements. Int. J. Phytoremediation 2012, 14, 320–334. [Google Scholar] [CrossRef] [PubMed]
- Scotti, A.; Godeas, A.; Silvani, V. Sistema Biorremediador Para Tratamiento de Suelo y/o Aguas Contaminadas. Argentina Patent 130100620, 22 October 2014. [Google Scholar]
- Scotti, A.; Milia, S.; Silvani, V.; Cappai, G.; Guglietta, D.; Trapasso, F.; Belardi, G.; Salvatori, R.; Tempesta, E.; Passeri, D.; et al. Development of an integrated multidisciplinary strategy for gallium, iron and manganese recovery from mining residues in a context of circular economy. In Proceedings of the International Center of Earth Sciences Conference, Mendoza, Argentina, 23 November 2020; Martín, G., Dino, F., Eds.; CNEA: Buenos Aires City, Argentina, 2021; pp. 898–910. [Google Scholar]
- Kadlec, R.H.; Knight, R.L.; Vymazal, J.; Brix, H.; Cooper, P. Constructed Wetlands for Pollution Control; IWA Publishing: London, UK, 2000. [Google Scholar]
- Mena, J.; Rodríguez, L.; Núñez, J.; Fernández, F.J.; Villaseñor, J. Design of horizontal and vertical subsurface flow constructed wetlands treating industrial wastewaters. WIT Trans. Ecol. Environ. 2008, 111, 555–564. [Google Scholar]
- Madera-Parra, C.A. Treatment of landfill leachate by polyculture constructed wetlands planted with native plants. Ing. Comptet. 2016, 18, 183–192. [Google Scholar]
- Ibañez, J. Niveles de madurez de la tecnología [Technology readiness levels: TRLS]: Una introducción. Econ. Indus. 2014, 393, 165–171. [Google Scholar]
- Silvani, V.A.; Fernández Bidondo, L.; Bompadre, M.J.; Pérgola, M.; Bompadre, A.; Fracchia, S.; Godeas, A.M. Growth dynamics of geographically different arbuscular mycorrhizal fungal isolates belonging to the ‘Rhizophagus clade’ under monoxenic conditions. Mycologia 2014, 106, 963–975. [Google Scholar] [CrossRef]
- Bompadre, M.J.; Silvani, V.A.; Fernández Bidondo, L.; Ríos de Molina, M.C.; Colombo, R.; Pardo, A.G.; Godeas, A. Arbuscular mycorrhizal fungi alleviate oxidative stress in pomegranate plants growing under different irrigation conditions. Botany 2014, 92, 187–193. [Google Scholar] [CrossRef]
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–160. [Google Scholar] [CrossRef]
- Giovannetti, M.; Mosse, M. An evaluation of techniques for measuring vesicular arbuscular infection in roots. New Phytol. 1980, 84, 589–600. [Google Scholar] [CrossRef]
- Cornejo, P.; Pérez-Tienda, J.; Meier, S.; Valderas, A.; Borie, F.; Azcón-Aguilar, C.; Ferrol, N. Copper compartmentalization in spores as a survival strategy of arbuscular mycorrhizal fungi in Cu-polluted environments. Soil Biol. Biochem. 2013, 57, 925–928. [Google Scholar] [CrossRef]
- Gonzalez-Chavez, C.; D’Haen, J.; Vangronsveld, J.; Dodd, J.C. Copper sorption and accumulation by the extraradical mycelium of different Glomus spp. (arbuscular mycorrhizal fungi) isolated from the same polluted soil. Plant Soil 2020, 240, 287–297. [Google Scholar] [CrossRef]
- Yruela, I. Copper in plants. Braz. J. Plant Physiol. 2005, 17, 145–156. [Google Scholar] [CrossRef]
- Gonzalez-Chavez, M.; Carrillo-Gonzalez, R.; Wright, S.F.; Nichols, K.A. The role of glomalin, a protein produced by arbuscular mycorrhizal fungi, in sequestering potentially toxic elements. Environ. Pollut. 2004, 130, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Christie, P.; Li, X. A modified glass bead compartment cultivation system for studies on nutrient and trace metal uptake by arbuscular mycorrhiza. Chemosphere 2001, 42, 185–192. [Google Scholar] [CrossRef]
- Xu, L.J.; Hao, Z.P.; Xie, W.; Li, F.; Chen, B.D. Transmembrane H + and Ca 2+ fluxes through extraradical hyphae of arbuscular mycorrhizal fungi in response to drought stress. Chin. J. Plant. Ecol. 2018, 42, 764–773. [Google Scholar]
- Allen, J.W.; Shachar-Hill, Y. Sulfur Transfer through an Arbuscular Mycorrhiza. Plant. Physiol. 2009, 149, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Muhammad, A.; Sultan, S.; Muhammad, A.; Naeem, A.; Sher, M.S.; Allah, W.; Ali, R.; Babar, H.B. Optimizing sulfur for improving salt tolerance of sunflower (Helianthus annuus L.). Soil Environ. 2019, 38, 222–233. [Google Scholar] [CrossRef]
- Ducic, T.; Polle, A. Transport and detoxification of manganese and copper in plants. Braz. J. Plant. Physiol. 2005, 17, 103–112. [Google Scholar] [CrossRef]
- Lyu, S.; Wei, X.; Chen, J.; Wang, C.; Wang, X.; Pan, D. Titanium as a Beneficial Element for Crop Production. Front. Plant. Sci. 2017, 8, 597. [Google Scholar] [CrossRef] [PubMed]
- European Commission, COM 2020/474 Final. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52020DC0474 (accessed on 21 July 2021).
- Correa, A.; Cruz, C.; Pérez-Tienda, J.; Ferrol, N. Shedding light onto nutrient responses of arbuscular mycorrhizal plants: Nutrient interactions may lead to unpredicted outcomes of the symbiosis. Plant. Sci. 2014, 221–222, 29–41. [Google Scholar] [CrossRef]
- Ferrol, N.; Tamayo, E.; Vargas, P. The heavy metal paradox in arbuscular mycorrhizas: From mechanisms to biotechnological applications. J. Experim. Bot. 2016, 67, 6253–6265. [Google Scholar] [CrossRef]
- Garcia, K.; Zimmermann, S.D. The role of mycorrhizal associations in plant potassium nutrition. Front. Plant Sci. 2014, 5, 337. [Google Scholar] [CrossRef] [PubMed]
- Olsson, P.A.; Hammer, E.C.; Pallon, J.; van Aarle, I.M.; Wallander, H. Elemental composition in vesicles of an arbuscular mycorrhizal fungus, as revealed by PIXE analysis. Fungal Biol. 2011, 115, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, C.V.; Casper, B.B. Lateral root function and root overlap among mycorrhizal and non mycorrhizal herbs in Florida shrubland, measured using rubidium as a nutrient analog. Am. J. Bot. 2002, 89, 1289–1294. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Lilleskov, E. Arsenic Uptake and Phytoremediation Potential by Arbuscular Mycorrhizal Fungi. In Mycorrhizal fungi: Use in Sustainable Agriculture and Land Restoration; Solaiman, Z., Abbot, L.K., Varma, A., Eds.; Springer Nature: Berlin/Heidelberg, Germany, 2014; Volume 41, pp. 259–275. [Google Scholar]
- Imran, A.; Chaudhry, M.N.; Masood, K.R.; Iqbal, N. Response of sunflower (Helianthus annuus L.) to arsenic stress: Accumulation and portioning in different plant parts. Soil Environ. 2015, 34, 44–50. [Google Scholar]
- Burger, A.; Lichtscheidl, I. Strontium in the environment: Review about reactions of plants towards stable and radioactive strontium isotopes. Sci. Total Environ. 2019, 653, 1458–1512. [Google Scholar] [CrossRef]
- Hanaka, A.; Dresler, S.; Wójciak-Kosior, M.; Strzemski, M.; Kováčik, J.; Latalski, M.; Zawiślak, G.; Sowa, I. The Impact of Long- and Short-Term Strontium Treatment on Metabolites and Minerals in Glycine max. Molecules 2019, 24, 3825. [Google Scholar] [CrossRef] [PubMed]
- Jensen, H.; Gaw, S.; Lehto, N.J.; Hassall, L.; Robinson, B.H. The mobility and plant uptake of gallium and indium, two emerging contaminants associated with electronic waste and other sources. Chemosphere 2018, 209, 675–684. [Google Scholar] [CrossRef]
- Yu, X.Z.; Feng, X.H.; Feng, Y.X. Phytotoxicity and Transport of Gallium (Ga) in Rice Seedlings for 2-Day of Exposure. Bull. Environ. Contam. Toxicol. 2015, 95, 122–125. [Google Scholar] [CrossRef]
- Seguel, A.; Cumming, J.R.; Klugh-Stewart, K.; Cornejo, P.; Borie, F. The role of arbuscular mycorrhizas in decreasing aluminium phytotoxicity in acidic soils: A review. Mycorrhiza 2013, 23, 167–183. [Google Scholar] [CrossRef]
- Coscione, A.R.; Berton, R.S. Barium extraction potential by mustard, sunflower and castor bean. Sci. Agric. 2009, 66, 59–63. [Google Scholar] [CrossRef][Green Version]
- Ubaldini, S.; Guglietta, D.; Vegliò, F.; Giuliano, V. Valorization of Mining Waste by Application of Innovative Thiosulphate Leaching for Gold Recovery. Metals 2019, 9, 274. [Google Scholar] [CrossRef]

| Sample Type | Sample ID | N | Ga (ppm) | P (ppm) | Ti (ppm) | Sr (ppm) | Ba (ppm) |
|---|---|---|---|---|---|---|---|
| Contaminated | MA− | 4 | 12.88 (0.572) | 731 (32.8) | 2575 (94.65) | 110 (3.47) | 2550 (86.6) |
| MA+ | 4 | 20.13 (1.994) | 711 (15.14) | 2675 (25) | 112(4.94) | 2600 (40.8) | |
| Cp(S)+ | 2 | 31.50 (9.596) | 3803 (209.1) | 9,5 (0.5) | 410 (172) | 7.85 (0.15) | |
| Cp(S)− | 2 | 13.70 (1.616) | 2878 (158.6) | 16 (1) | 254 (9.60) | 11.5 (0.5) | |
| Cp(R)+ | 2 | 25.55 (3.889) | 1228 (35.86) | 446 (24.24) | 79(4.04) | 98.5 (1.51) | |
| Cp(R)− | 2 | 22.00 (1.010) | 880 (27.27) | 947.5 (53) | 138 (7.07) | 163.5 (6.5) | |
| Blank | B+ | 4 | 5.00 (0.34) | 2125 (25.00) | 1275 (25) | 1100 (57.7) | 431.25 (21.29) |
| Cp(S)+ | 2 | 3.02 (0.75) | 3583 (200.1) | 9 (3) | 229 (1.01) | 8.0 (1.0) | |
| Cp(R)+ | 2 | 3.00 (0.10) | 1412 (39.39) | 509 (11.11) | 106 (5.56) | 28.5 (1.51) |
| Sample Type | Sample ID | N | Mn (%) | Fe (%) | As (ppm) | Zn (ppm) | Cr (ppm) | Ni (ppm) | Cu (ppm) | Rb (ppm) | Al (ppm) | K (%) | S (ppm) | Ca (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Contaminated | MA− | 4 | 4.93 (0.13) | 11.2 (0.26) | 12.5 (0.59) | 1500 (108) | 110 (7.67) | 67.0 (2.24) | 60.3 (1.65) | 78.3 (1.49) | 62,500 (1040.8) | 2.30 (0.04) | 1195 (113.1 | 1.12 (0.10) |
| MA+ | 4 | 5.55 (0.03) | 12.2 (0.06) | 23.2 (1.24) | 1475 (25.0) | 113.3 (2.93) | 72.3 (1.49) | 64.5 (2.53) | 80.5 (1.76) | 64,750 (250) | 2.20 (0.04) | 1125 (125) | 1.05 (0.04) | |
| Cp(S)+ | 2 | 0.052 (0.003) | 0.03 (0.001) | 4.05 (0.05) | 1328 (37.9) | 2.80 (0.20) | 1.80 (0.10) | 16.5 (0.51) | 116 (4.04) | 0.95 (0.05) | 7.70 (0.40) | 5945 (162.1) | 2.25 (0.15) | |
| Cp(S)− | 2 | 0.05 (0.003) | 0.03 (0.002) | 1.50 (0.10) | 1410 (39.4) | 0.65 (0.05) | 1.00 (0.10) | 14.0 (1.01) | 102 (2.53) | 0.93 (0.07) | 7.10 (0.20) | 6189 (162.1) | 2.06 (0.06) | |
| Cp(R)+ | 2 | 0.455 (0.024) | 1.19 (0.06) | 6.50 (0.505) | 1162 (32.3) | 29.5 (1.52) | 38.0 (2.02) | 17.5 (0.51) | 12.5 (0.51) | 5550 (2272.4) | 4.65 (0.15) | 16,885 (672.3) | 0.99 (0.02) | |
| Cp(R)− | 2 | 0.74 (0.04) | 2.13 (0.13) | 6.50 (0.505) | 1717 (92.4) | 10.00 (1.01) | 14.0 (10.0) | 36.0 (2.02) | 109 (3.54) | 21,375 (419.2) | 5.20 (0.15) | 13,772 (307.6) | 2.46 (0.08) | |
| Blank | B+ | 4 | 0.06 (0.001) | 1.43 (0.03) | 8.78 (0.41) | 210.8 (9.4) | 86.50 (9.1) | 42.3 (0.75) | 108 (2.68) | 212 (14.1) | 13,625 (239.3) | 1.33 (0.05) | 3525 (110.9) | 5.33 (0.06) |
| Cp(S)+ | 2 | 0.007 (0.0006) | 0.01 (0.001) | 1.65 (0.25) | 54.50 (4.6) | 3.45 (1.26) | 2.80 (1.31) | 15.0 (0.00) | 76.0 (1.01) | 8.9 (0.04) | 5.40 (0.10) | 2045 (49.5) | 1.86 (0.11) | |
| Cp(R)+ | 2 | 0.03 (0.002) | 0.22 (0.01) | 3.55 (0.02) | 166.5 (4.6) | 12.0 (1.01) | 6.45 (0.25) | 42.5 (2.53) | 33.0 (2.02) | 13,900 (303,04) | 1.06 (0.03) | 1725 (29.3) | 4.32 (0.09) |
| Sample ID | Parameter | Mn | Ga | Fe | P | As | Zn | Ti | Sr | Cr | Ni | Cu | Rb | Al | Ba | K | S | Ca |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MA+ | BCS+ | 0.009 (0.001) | 1.565 (0.632) | 0.002 (0.0001) | 5.349 (0.408) | 0.175 (0.012) | 0.900 (0.046) | 0.004 (0.0002) | 3.653 (3.356) | 0.025 (0.002) | 0.025 (0.002) | 0.256 (0.018) | 1.441 (0.082) | 0.000 (0.000) | 0.003 (0.0001) | 3.500 (0.247) | 5.284 (0.731) | 2.138 (0.233) |
| BCR+ | 0.082 (0.005) | 1.270 (0.319) | 0.098 (0.006) | 1.726 (0.087) | 0.281 (0.162) | 0.788 (0.035) | 0.167 (0.011) | 0.704 (0.388) | 0.261 (0.020) | 0.526 (0.039) | 0.271 (0.019) | 0.155 (0.010) | 0.086 (0.035) | 0.038 (0.001) | 2.114 (0.107) | 15.008 (2.265) | 0.936 (0.053) | |
| TF+ | 0.114 (0.013) | 1.233 (0.808) | 0.023 (0.003) | 3.098 (0.393) | 0.623 (0.401) | 1.142 (0.064) | 0.021 (0.002) | 5.190 (3.973) | 0.095 (0.012) | 0.047 (0.005) | 0.943 (0.056) | 9.280 (0.698) | 0.0002 0.0001 | 0.080 (0.003) | 1.656 (0.141) | 0.352 (0.024) | 2.284 (0.189) | |
| MA− | BCS− | 0.011 (0.001) | 1.064 (0.173) | 0.003 (0.0002) | 3.937 (0.394) | 0.125 (0.014) | 0.940 (0.100) | 0.006 (0.001) | 2.310 (0.161) | 0.006 (0.001) | 0.015 (0.002) | 0.232 (0.023) | 1.297 (0.057) | 0.000 (0.000) | 0.005 (0.0004) | 3.087 (0.143) | 5.181 (0.626) | 1.835 (0.224) |
| BCR− | 0.150 (0.012) | 1.709 (0.154) | 0.190 (0.016) | 1.204 (0.091) | 0.522 (0.065) | 1.144 (0.144) | 0.368 (0.034) | 1.257 (0.104) | 0.091 (0.016) | 0.209 (0.022) | 0.598 (0.050) | 1.387 (0.072) | 0.342 (0.012) | 0.064 (0.048) | 2.283 (0.106) | 11.529 (1.349) | 2.187 (0.270) | |
| TF− | 0.073 (0.008) | 0.623 (0.157) | 0.014 (0.002) | 3.271 (0.576) | 0.231 (0.034) | 0.821 (0.067) | 0.017 (0.002) | 1.837 (0.164) | 0.065 (0.012) | 0.071 (0.012) | 0.389 (0.050) | 0.936 (0.054) | 0.000 (0.000) | 0.070 (0.006) | 1.352 (0.078) | 0.449 (0.022) | 0.839 (0.051) |
| Parameter | Mn | Ga | Fe | P | As | Zn | Ti | Cr | Ni | Cu | Rb | Sr | Al | Ba | K | S | Ca |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BCS | (−) | (=) | (−) | (+) | (+) | (=) | (−) | (+) | (+) | (=) | (+) | (=) | (=) | (−) | (+) | (=) | (=) |
| BCR | (−) | (=) | (−) | (+) | (−) | (−) | (−) | (+) | (+) | (−) | (−) | (−) | (−) | (−) | (=) | (+) | (−) |
| TF | (+) | (=) | (+) | (=) | (+) | (+) | (+) | (+) | (−) | (+) | (+) | (+) | (+) | (+) | (+) | (−) | (+) |
| Parameter | Mn (ppm) | Fe (ppm) | Ga (ppm) | P (ppm) | As (ppm) | Zn (ppm) | Ti (ppm) | Cr (ppm) | Ni (ppm) | Cu (ppm) | Rb (ppm) | Sr (ppm) | Al (ppm) | Ba (ppm) | K (ppm) | S (ppm) | Ca (ppm) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cp(S)+ | 520 (42) | 270,5 (20,5) | 31.5 (13,4) | 3803 (292) | 4.05 (0.07) | 1328 (53) | 9.5 (0.7) | 2.8 (0.3) | 1.8 (0.1) | 16.5 (0.7) | 116 (5.66) | 410 (240) | 0.95 (0.07) | 7.85 (0.21) | 77,000 (5657) | 5944.5 (227) | 22,500 (2121) |
| Cp(R)+ | 4550 (343) | 11,888 (898) | 25.5 (5,4) | 1228 (50) | 6.5 (0.7) | 1162 (45) | 446 (34) | 29.5 (2.1) | 38 (2.8) | 17.5 (0.71) | 12.5 (0.71) | 79 (5.7) | 5550 (3181) | 98.5 (2.12) | 46,500 (2121) | 16,885 (941) | 9850 (212) |
| Extracting g/VDM * | 34.82 | 60.01 | 1.017 | 114.7 | 0.145 | 43.22 | 2.199 | 0.208 | 0.216 | 0.550 | 3.392 | 12.14 | 23.99 | 0.6515 | 2417 | 243.9 | 690.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scotti, A.; Milia, S.; Silvani, V.; Cappai, G.; Guglietta, D.; Trapasso, F.; Tempesta, E.; Passeri, D.; Godeas, A.; Gómez, M.; et al. Sustainable Recovery of Secondary and Critical Raw Materials from Classified Mining Residues Using Mycorrhizal-Assisted Phytoextraction. Metals 2021, 11, 1163. https://doi.org/10.3390/met11081163
Scotti A, Milia S, Silvani V, Cappai G, Guglietta D, Trapasso F, Tempesta E, Passeri D, Godeas A, Gómez M, et al. Sustainable Recovery of Secondary and Critical Raw Materials from Classified Mining Residues Using Mycorrhizal-Assisted Phytoextraction. Metals. 2021; 11(8):1163. https://doi.org/10.3390/met11081163
Chicago/Turabian StyleScotti, Adalgisa, Stefano Milia, Vanesa Silvani, Giovanna Cappai, Daniela Guglietta, Francesca Trapasso, Emanuela Tempesta, Daniele Passeri, Alicia Godeas, Martín Gómez, and et al. 2021. "Sustainable Recovery of Secondary and Critical Raw Materials from Classified Mining Residues Using Mycorrhizal-Assisted Phytoextraction" Metals 11, no. 8: 1163. https://doi.org/10.3390/met11081163
APA StyleScotti, A., Milia, S., Silvani, V., Cappai, G., Guglietta, D., Trapasso, F., Tempesta, E., Passeri, D., Godeas, A., Gómez, M., & Ubaldini, S. (2021). Sustainable Recovery of Secondary and Critical Raw Materials from Classified Mining Residues Using Mycorrhizal-Assisted Phytoextraction. Metals, 11(8), 1163. https://doi.org/10.3390/met11081163










