Preparation of a Nickel Layer with Bell-Mouthed Macropores via the Dual-Template Method
Abstract
:1. Introduction
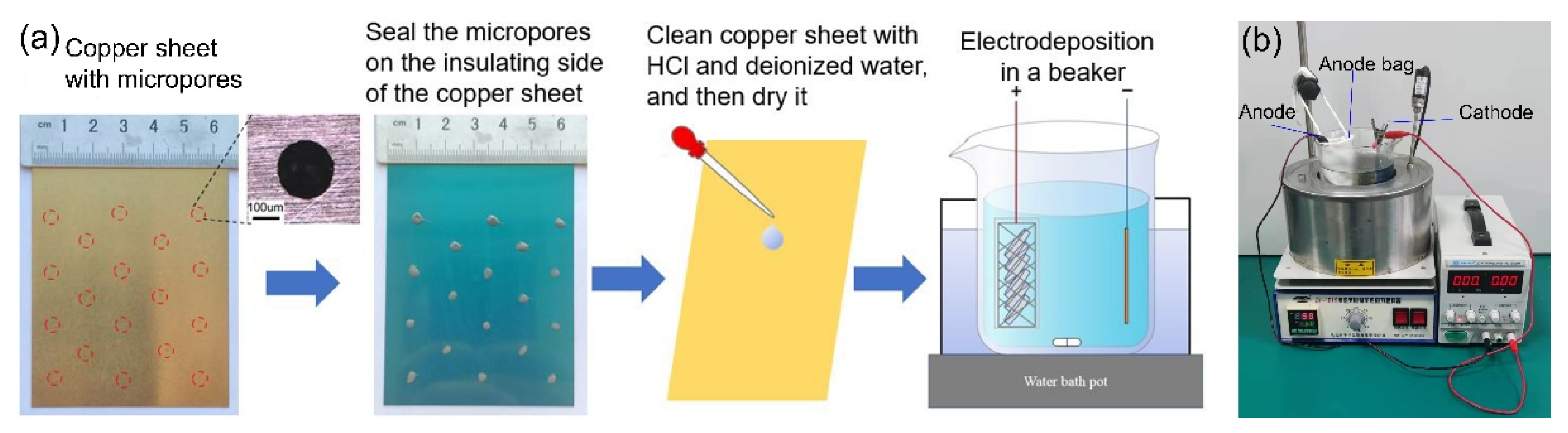
2. Materials and Methods
3. Results and Discussion
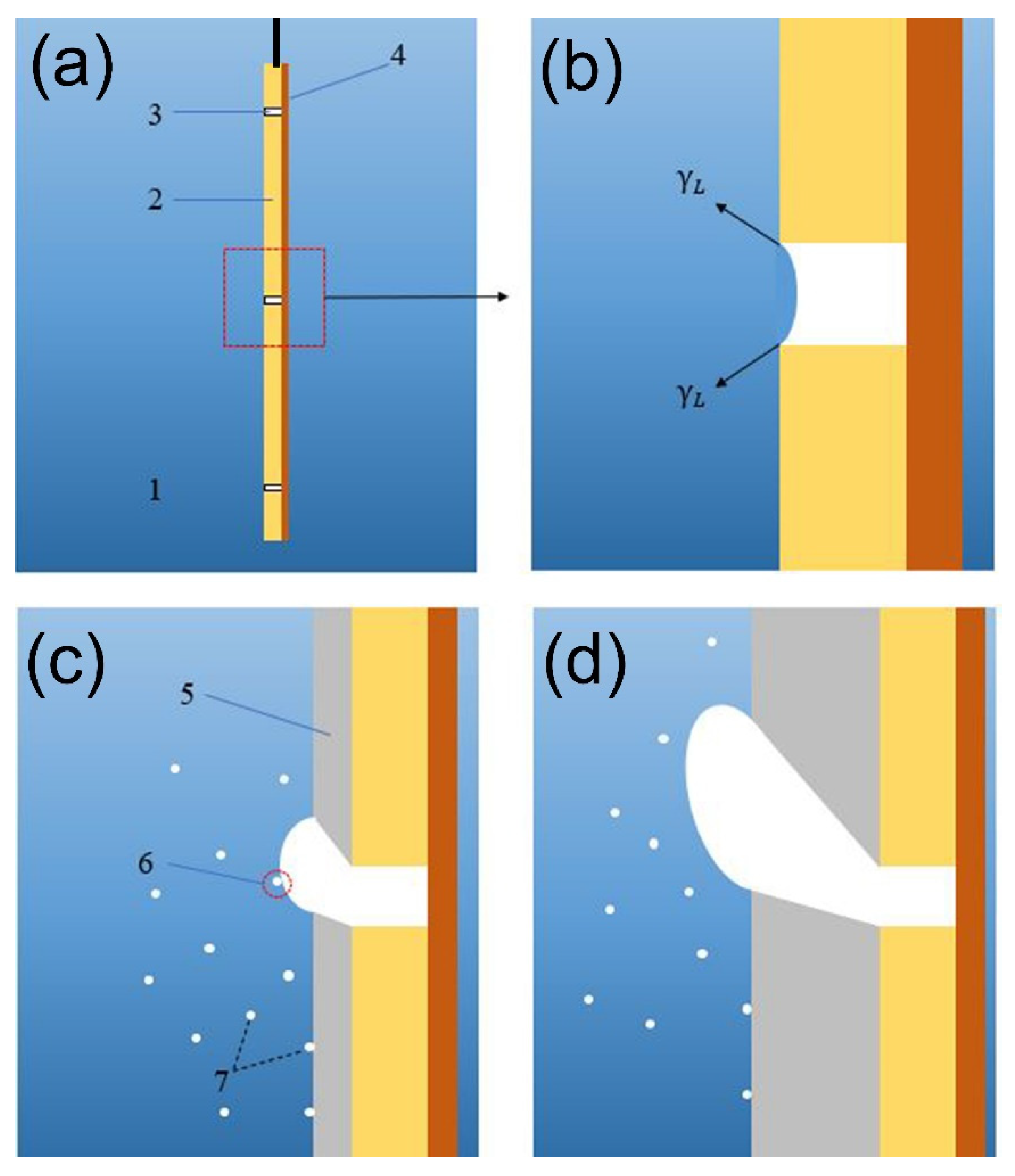
3.1. Analysis of the Formation of Bell-Mouthed Macropores in Deposition
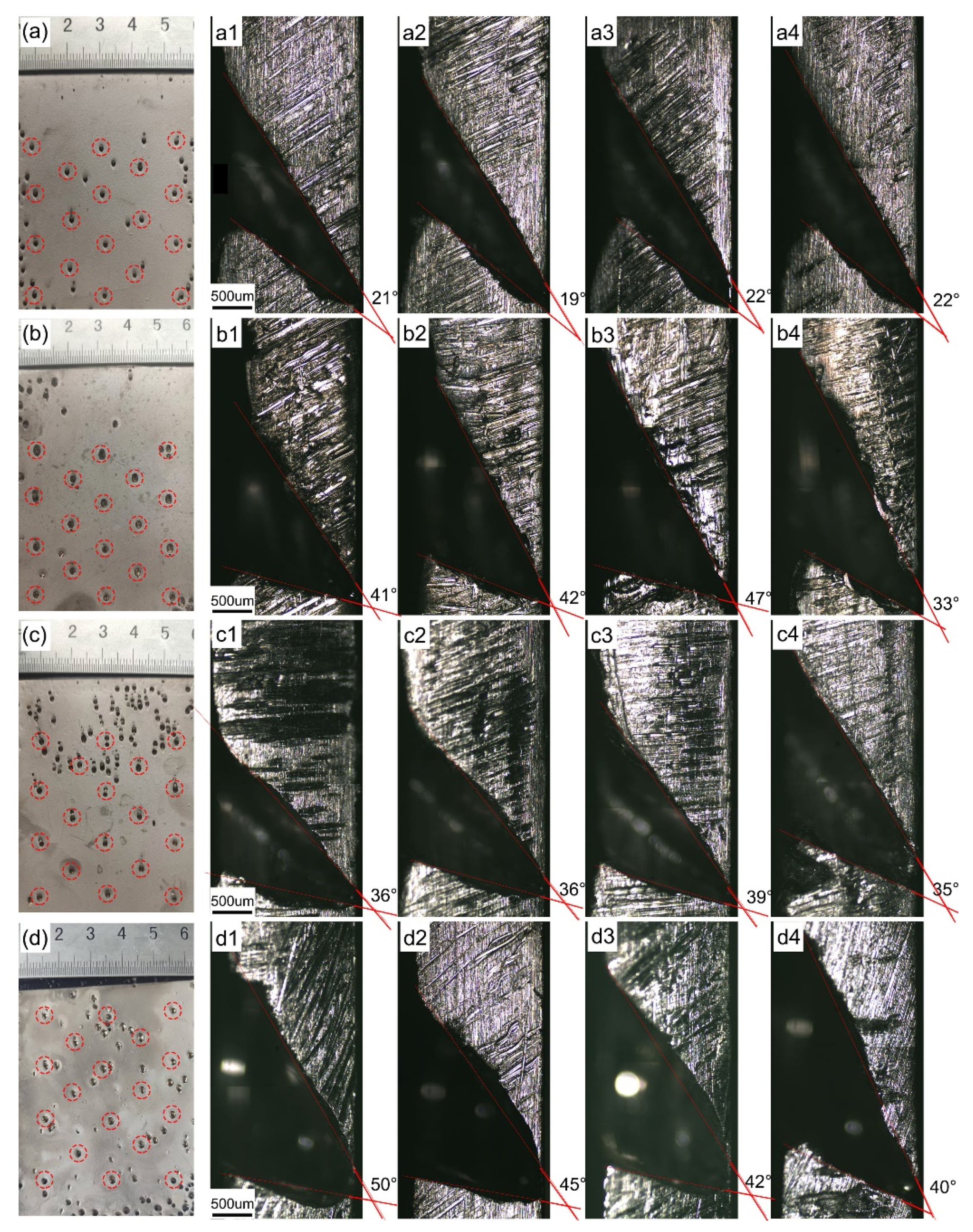
3.2. Effect of Stirring Speed of the Solution
3.3. Effect of Current Density
4. Conclusions
- The surface tension of the solution is a decisive factor in obtaining the target nickel layer. When the content of SDS in the base bath is 0.0050 g/L ( = 50.2 mN/m), the desired bubble template can be formed and there are fewer bubbles attached to other areas on the substrate. The bubble template is relatively static due to the hydrogen bubbles staying attached to the surface of the substrate, and the bubbles grow slowly. The micropores on the substrate provide a preferential condition for the formation of the bell-mouthed macropores in the deposition and make the positions of the bell-mouthed macropores controllable.
- The sizes of the bell-mouthed macropores can be adjusted by varying the stirring speed of the solution and the current density. As the solution stirring speed increases from 160 to 480 r/min, the angle range of the bell-mouthed macropore cross-sectional profile is increased from 21.0° to 44.3°. In addition, the angle range of the bell-mouthed macropores cross-sectional profile is increased from 39.3° to 46.3° with the current density increasing from 1 to 2.5 A/dm2.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Unit adhesion energy | |
| Adhesion energy; the work required in detaching the bubble from the electrode | |
| Area of interface between bubble and deposition | |
| Surface tension of the H2–liquid interface | |
| Contact angle of the bubble at the cathode surface | |
| DHBT | Dynamic hydrogen bubble template |
| SDS | Sodium dodecyl sulfate |
References
- Parlett, C.M.A.; Wilson, K.; Lee, A.F. Hierarchical porous materials: Catalytic applications. Chem. Soc. Rev. 2013, 42, 3876–3893. [Google Scholar] [CrossRef] [PubMed]
- Walcarius, A. Template-directed porous electrodes in electroanalysis. Anal. Bioanal. Chem. 2010, 396, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fu, Z.-Y.; Su, B.-L. Hierarchically Structured Porous Materials for Energy Conversion and Storage. Adv. Funct. Mater. 2012, 22, 4634–4667. [Google Scholar] [CrossRef]
- Li, X.-M.; Reinhoudt, D.; Crego-Calama, M. What do we need for a superhydrophobic surface? A review on the recent progress in the preparation of superhydrophobic surfaces. Chem. Soc. Rev. 2007, 36, 1350–1368. [Google Scholar] [CrossRef]
- Marozzi, C.; Chialvo, A. Development of electrode morphologies of interest in electrocatalysis. Part 1: Electrodeposited porous nickel electrodes. Electrochim. Acta 2000, 45, 2111–2120. [Google Scholar] [CrossRef]
- Yang, F.; Cheng, K.; Xue, X.; Yin, J.; Wang, G.; Cao, D. Three-dimensional porous Ni film electrodeposited on Ni foam: High performance and low-cost catalytic electrode for H2O2 electrooxidation in KOH solution. Electrochim. Acta 2013, 107, 194–199. [Google Scholar] [CrossRef]
- Yu, X.; Wang, M.; Wang, Z.; Gong, X.; Guo, Z. The structure evolution mechanism of electrodeposited porous Ni films on NH4Cl concentration. Appl. Surf. Sci. 2016, 360, 502–509. [Google Scholar] [CrossRef]
- Plowman, B.J.; Jones, L.A.; Bhargava, S.K. Building with bubbles: The formation of high surface area honeycomb-like films via hydrogen bubble templated electrodeposition. Chem. Commun. 2015, 51, 4331–4346. [Google Scholar] [CrossRef]
- Cherevko, S.; Xing, X.; Chung, C.-H. Electrodeposition of three-dimensional porous silver foams. Electrochem. Commun. 2010, 12, 467–470. [Google Scholar] [CrossRef]
- Wang, M.; Yu, X.; Wang, Z.; Gong, X.; Guo, Z.; Dai, L. Hierarchically 3D porous films electrochemically constructed on gas–liquid–solid three-phase interface for energy application. J. Mater. Chem. A 2017, 5, 9488–9513. [Google Scholar] [CrossRef]
- Tawfik, M.; Diez, F. On the relation between onset of bubble nucleation and gas supersaturation concentration. Electrochim. Acta 2014, 146, 792–797. [Google Scholar] [CrossRef]
- Vogt, H. The actual current density of gas-evolving electrodes—Notes on the bubble coverage. Electrochim. Acta 2012, 78, 183–187. [Google Scholar] [CrossRef]
- Cherevko, S.; Chung, C.-H. Impact of key deposition parameters on the morphology of silver foams prepared by dynamic hydrogen template deposition. Electrochim. Acta 2010, 55, 6383–6390. [Google Scholar] [CrossRef]
- Nørskov, J.K.; Bligaard, T.; Logadottir, A.; Kitchin, J.R.; Chen, J.G.; Pandelov, S.; Stimming, U. Trends in the Exchange Current for Hydrogen Evolution. J. Electrochem. Soc. 2005, 152, J23–J26. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.; Guo, X.; Zeng, J.; Nie, L.; Dong, J.; Peng, L.; Ding, W. Influence of sodium dodecyl sulphate on the surface morphology and infrared emissivity of porous Ni film. Infrared Phys. Technol. 2018, 93, 162–170. [Google Scholar] [CrossRef]
- Kristof, P.; Pritzker, M. Effect of electrolyte composition on the dynamics of hydrogen gas bubble evolution at copper microelectrodes. J. Appl. Electrochem. 1997, 27, 255–265. [Google Scholar] [CrossRef]
- Najdovski, I.; O’Mullane, A.P. The effect of electrode material on the electrochemical formation of porous copper surfaces using hydrogen bubble templating. J. Electroanal. Chem. 2014, 722–723, 95–101. [Google Scholar] [CrossRef] [Green Version]
- Nikolic, N.; Popov, K.I.; Pavlović, L.J.; Pavlović, M.G. Phenomenology of a formation of a honeycomb-like structure during copper electrodeposition. J. Solid State Electrochem. 2006, 11, 667–675. [Google Scholar] [CrossRef]
- Zhang, W.; Ding, C.; Wang, A.; Zeng, Y. 3-D Network Pore Structures in Copper Foams by Electrodeposition and Hydrogen Bubble Templating Mechanism. J. Electrochem. Soc. 2015, 162, D365–D370. [Google Scholar] [CrossRef]
- Li, Y.; Jia, W.-Z.; Song, Y.-Y.; Xia, X.-H. Superhydrophobicity of 3D Porous Copper Films Prepared Using the Hydrogen Bubble Dynamic Template. Chem. Mater. 2007, 19, 5758–5764. [Google Scholar] [CrossRef]
- Qiu, H.; Lu, L.; Xue, L.; Huang, X. Facile electrochemical preparation of three-dimensional porous Cu films by potential perturbation. Electrochem. Acta 2010, 55, 6081–6087. [Google Scholar] [CrossRef]
- Shin, H.-C.; Dong, J.; Liu, M. Nanoporous Structures Prepared by an Electrochemical Deposition Process. Adv. Mater. 2003, 15, 1610–1614. [Google Scholar] [CrossRef]
- Zhang, H.; Ye, Y.; Shen, R.; Ru, C.; Hu, Y. Effect of Bubble Behavior on the Morphology of Foamed Porous Copper Prepared via Electrodeposition. J. Electrochem. Soc. 2013, 160, D441–D445. [Google Scholar] [CrossRef]
- Davis, R.F.; Wolfe, K.M.; France, W.G. Effect of Wetting Agents on Electrodeposition of Nickel. Ind. Eng. Chem. 1941, 33, 1546–1548. [Google Scholar] [CrossRef]
- Luo, H.X.; Huang, X.L.; Mei, Y.X. Causes and Solutions of Pinholes Fault on the Nickel Coating. Hydraul. Pneum. Seals 2014, 3, 62–63. [Google Scholar] [CrossRef]
- Yu, Q.Z.; Guo, Z.Q.; Ye, J.S. Influence of Surface State on Coating Quality. Corros. Prot. 2016, 37. [Google Scholar] [CrossRef]
- Tsuru, Y.; Nomura, M.; Foulkes, F. Effects of boric acid on hydrogen evolution and internal stress in films deposited from a nickel sulfamate bath. J. Appl. Electrochem. 2002, 32, 629–634. [Google Scholar] [CrossRef]
- Fengmei, Z.S.L.Y.Y. Efection of Current Density on the Nickel Coating of Nickel Sulfamate. Shandong Chem. Ind. 2011, 40, 55–57. [Google Scholar] [CrossRef]
- Giurlani, W.; Zangari, G.; Gambinossi, F.; Passaponti, M.; Salvietti, E.; Di Benedetto, F.; Caporali, S.; Innocenti, M. Electroplating for Decorative Applications: Recent Trends in Research and Development. Coatings 2018, 8, 260. [Google Scholar] [CrossRef] [Green Version]
- Fernández, D.; Maurer, P.; Martine, M.; Coey, J.M.D.; Möbius, M. Bubble Formation at a Gas-Evolving Microelectrode. Langmuir 2014, 30, 13065–13074. [Google Scholar] [CrossRef]
- Jelinek, T.W. Galvanisches Verzinken, Eugen G; Leuze Verlag Karlstra: Bad Saulgau, Germany, 1982. [Google Scholar]



| SDS (g/L) | 0.0020 | 0.0040 | 0.0050 | 0.0060 | 0.0080 | 0.0100 |
| Surface tension (mN/m) | 60.0 ± 0.3 | 53.4 ± 0.2 | 50.2 ± 0.2 | 47.4 ± 0.3 | 46.7 ± 0.1 | 46.0 ± 0.1 |
| Stirring speed (r/min) | 240 | 320 | 400 | 480 |
| Average angle of macropores profile (°) | 21.0 ± 1.4 | 39.5 ± 4.3 | 36.5 ± 1.7 | 44.3 ± 4.3 |
| Current Density (A/dm2) | 1 | 1.5 | 2 | 2.5 |
| Average angle of macropores profile (°) | 39.2 ± 3.3 | 39.5 ± 4.3 | 44 ± 5.4 | 46.3 ± 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, R.; Yao, W.; Qian, G.; Dou, Y. Preparation of a Nickel Layer with Bell-Mouthed Macropores via the Dual-Template Method. Metals 2021, 11, 1894. https://doi.org/10.3390/met11121894
Yang R, Yao W, Qian G, Dou Y. Preparation of a Nickel Layer with Bell-Mouthed Macropores via the Dual-Template Method. Metals. 2021; 11(12):1894. https://doi.org/10.3390/met11121894
Chicago/Turabian StyleYang, Ruishan, Weiguo Yao, Guangguang Qian, and Yanli Dou. 2021. "Preparation of a Nickel Layer with Bell-Mouthed Macropores via the Dual-Template Method" Metals 11, no. 12: 1894. https://doi.org/10.3390/met11121894
APA StyleYang, R., Yao, W., Qian, G., & Dou, Y. (2021). Preparation of a Nickel Layer with Bell-Mouthed Macropores via the Dual-Template Method. Metals, 11(12), 1894. https://doi.org/10.3390/met11121894





