On the Adsorption of Cerium(III) Using Multiwalled Carbon Nanotubes
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Estimation of the Isoelectronic Points
3.2. Adsorption Using Nonoxidized Multiwalled Carbon Nanotubes (MWCNTs)
3.2.1. Influence of the Stirring Speed
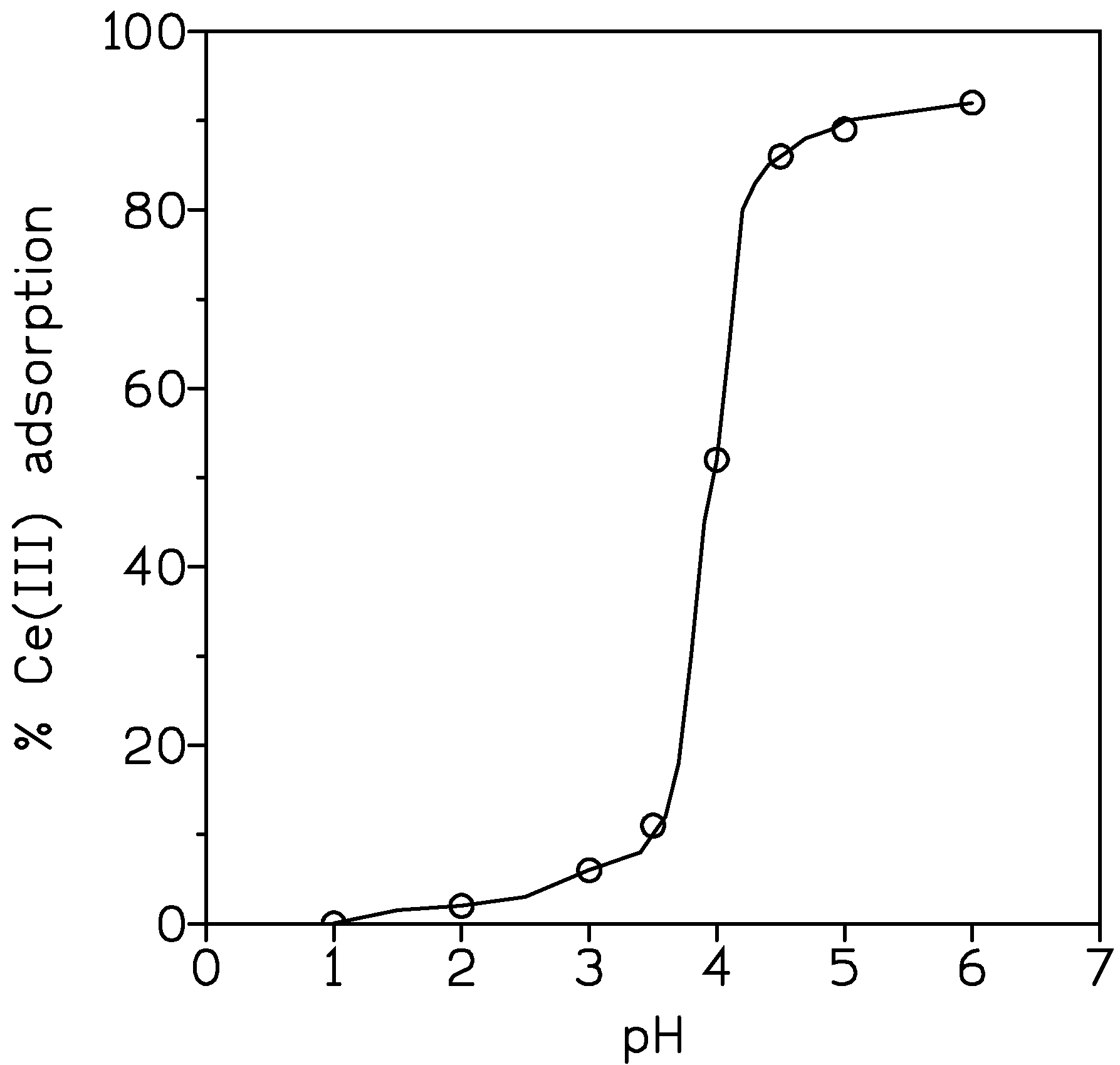
3.2.2. Influence of Aqueous pH
3.2.3. Effect of the Initial Metal Concentration on Cerium Uptake
3.2.4. Effect of the Dosage Absorbent on Cerium Adsorption
3.3. Adsorption Using Oxidized Multiwalled Carbon Nanotubes (ox-MWCNTs)
3.3.1. Influence of the Stirring Speed
3.3.2. Influence of the Initial Cerium Concentration
3.3.3. Influence of the pH
3.3.4. Influence of the of the Adsorbent Dosage on the Adsorption of Cerium
3.4. Desorption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Alguacil, F.J.; Alcaraz, L.; García-Díaz, I.; López, F.A. Removal of Pb2+ in wastewater via adsorption onto an activated carbon produced from winemaking waste. Metals 2018, 8, 697. [Google Scholar] [CrossRef]
- Abbas, A.; Al-Amer, A.M.; Laoui, T.; Al-Marri, M.J.; Nasser, M.S.; Khraisheh, M.; Atieh, M.A. Heavy metal removal from aqueous solution by advanced carbon nanotubes: Critical review of adsorption applications. Sep. Purif. Technol. 2016, 157, 141–161. [Google Scholar] [CrossRef]
- Hu, Y.; Florek, J.; Larivière, D.; Fontaine, F.-G.; Kleitz, F. Recent advances in the separation of rare earth elements using mesoporous hybrid materials. Chem. Rec. 2018, 18, 1261–1276. [Google Scholar] [CrossRef] [PubMed]
- Alguacil, F.J.; Garcia-Diaz, I.; Lopez, F.; Rodriguez, O. Removal of Cr(VI) and Au(III) from aqueous streams by the use of carbon nanoadsorption technology. Desalin. Water Treat. 2017, 63, 351–356. [Google Scholar] [CrossRef]
- García-Díaz, I.; López, F.A.; Alguacil, F.J. Carbon nanofibers: A new adsorbent for copper removal from wastewater. Metals 2018, 8, 914. [Google Scholar] [CrossRef]
- Egbosiuba, T.C.; Abdulkareem, A.S.; Kovo, A.S.; Afolabi, E.A.; Tijani, J.O.; Roos, W.D. Enhanced adsorption of As(V) and Mn(VII) from industrial wastewater using multi-walled carbon nanotubes and carboxylated multi-walled carbon nanotubes. Chemosphere 2020, 254, 126780. [Google Scholar] [CrossRef]
- Roccaro, P.; Sgroi, M.; Vagliasindi, F.G.A. Removal of xenobiotic compounds from wastewater for environment protection: Treatment processes and costs. Chem. Eng. Trans. 2013, 32, 505–510. [Google Scholar]
- Guo, L.; Liu, Y.; Dou, J.; Huang, Q.; Lei, Y.; Chen, J.; Wen, Y.; Li, Y.; Zhang, X.; Wei, Y. Highly efficient removal of Eu3+ ions using carbon nanotubes-based polymer composites synthesized from the combination of Diels-Alder and multicomponent reactions. J. Mol. Liq. 2020, 308, 112964. [Google Scholar] [CrossRef]
- Ramasamy, D.L.; Puhakka, V.; Doshi, B.; Iftekhar, S.; Sillanpää, M. Fabrication of carbon nanotubes reinforced silica composites with improved rare earth elements adsorption performance. Chem. Eng. J. 2019, 365, 291–304. [Google Scholar] [CrossRef]
- Gambogi, J. Rare Earths. Mineral Commodity Summaries 2020; USA Geological Survey: Reston, VA, USA, 2020.
- Omodara, L.; Pitkäaho, S.; Turpeinen, E.-M.; Saavalainen, P.; Oravisjärvi, K.; Keiski, R.L. Recycling and substitution of light rare earth elements, cerium, lanthanum, neodymium, and praseodymium from end-of-life applications-A review. J. Clean. Prod. 2019, 236, 117573. [Google Scholar] [CrossRef]
- Jowitt, S.M.; Werner, T.T.; Weng, Z.; Mudd, G.M. Recycling of the rare earth elements. Curr. Opin. Green Sustain. Chem. 2018, 13, 1–7. [Google Scholar] [CrossRef]
- Gao, J.; Si, Z.; Xu, Y.; Liu, L.; Zhang, Y.; Wu, X.; Ran, R.; Weng, D. Pd–Ag@CeO2 catalyst of core–shell structure for low temperature oxidation of toluene under visible light irradiation. J. Phys. Chem. C 2019, 123, 1761–1769. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, T.C.; He, H.; Ouyang, L.; Yuan, S. A stearic Acid/CeO2 bilayer coating on AZ31B magnesium alloy with superhydrophobic and self-cleaning properties for corrosion inhibition. J. Alloys Compd. 2020, 834, 155210. [Google Scholar] [CrossRef]
- Nurhasanah, I.; Gunawan, V.; Sutanto, H. Cerium oxide nanoparticles application for rapid adsorptive removal of tetracycline in water. J. Environ. Chem. Eng. 2020, 8, 103613. [Google Scholar] [CrossRef]
- Rivoira, L.P.; Valles, V.A.; Martínez, M.L.; Sa-ngasaeng, Y.; Jongpatiwut, S.; Beltramone, A.R. Catalytic oxidation of sulfur compounds over Ce-SBA-15 and Ce-Zr-SBA-15. Catal. Today 2019, in press. [Google Scholar] [CrossRef]
- Jacinto, J.; Henriques, B.; Duarte, A.C.; Vale, C.; Pereira, E. Removal and recovery of Critical Rare Elements from contaminated waters by living Gracilaria gracilis. J. Hazard. Mater. 2018, 344, 531–538. [Google Scholar] [CrossRef]
- Pujar, M.S.; Hunagund, S.M.; Barretto, D.A.; Desai, V.R.; Patil, S.; Vootla, S.K.; Sidarai, A.H. Synthesis of cerium-oxide NPs and their surface morphology effect on biological activities. Bull. Mater. Sci. 2019, 43. [Google Scholar] [CrossRef]
- Wang, Y.; Xie, X.; Chen, X.; Huang, C.; Yang, S. Biochar-loaded Ce3+-enriched ultra-fine ceria nanoparticles for phosphate adsorption. J. Hazard. Mater. 2020, 396, 122626. [Google Scholar] [CrossRef]
- Shi, Y.; Zhao, Y.; Cao, M.; Chen, H.; Hu, Z.; Chen, X.; Zhang, Z.; Liu, Q. Dense Ce3+ doped Lu3A15O12 ceramic scintillators with low sintering adds: Doping content effect, luminescence characterization and proton irradiation hardness. J. Lumin. 2020, 225, 117336. [Google Scholar] [CrossRef]
- Silva, V.D.; Ferreira, L.S.; Araújo, A.J.M.; Simões, T.A.; Grilo, J.P.F.; Tahir, M.; Medeiros, E.S.; Macedo, D.A. Ni and Ce oxide-based hollow fibers as battery-like electrodes. J. Alloys Compd. 2020, 830, 154633. [Google Scholar] [CrossRef]
- Duan, J.; Tang, W.; Wang, R.; Tang, X.; Li, J.; Tang, M.; Li, P. Inhibited voltage decay and enhanced electrochemical performance of the Li-rich layered Li1.2Mn0.54Ni0.13Co0.13O2 cathode material by CeAlOδ surface coating modification. Appl. Surf. Sci. 2020, 521, 146504. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z.; Li, J.; Niu, B.; Yang, K.; Kang, F. A novel surface-heterostructured Li1.2Mn0.54Ni0.13Co0.13O2@Ce0.8Sn0.2O2−σ cathode material for Li-ion batteries with improved initial irreversible capacity loss. J. Mater. Chem. A 2018, 6, 13883–13893. [Google Scholar] [CrossRef]
- EU Report on Critical Raw Materials and the Circular Economy-Publications Office of the EU. Available online: https://publications.europa.eu/en/publication-detail/-/publication/d1be1b43-e18f-11e8-b690-01aa75ed71a1/language-en/format-PDF/source-80004733 (accessed on 18 February 2019).
- Parisi, F. Adsorption and separation of crystal violet, cerium(III) and lead(II) by means of a multi-step strategy based on k10-montmorillonite. Minerals 2020, 10, 466. [Google Scholar] [CrossRef]
- Han, Q.; Du, M.; Guan, Y.; Luo, G.; Zhang, Z.; Li, T.; Ji, Y. Removal of simulated radioactive cerium (III) based on innovative magnetic trioctylamine-polystyrene composite microspheres. Chem. Phys. Lett. 2020, 741, 137092. [Google Scholar] [CrossRef]
- Wójcik, G. Sorption behaviors of light lanthanides(III) (La(III), Ce(III), Pr(III), Nd(III)) and Cr(III) using nitrolite. Materials 2020, 13, 2256. [Google Scholar] [CrossRef]
- Jiménez-Reyes, M.; Almazán-Sánchez, P.T.; Solache-Ríos, M. Behaviour of cerium(III) in the presence of components of soils and its humate complex. Environ. Technol. 2020, 1–9, in press. [Google Scholar] [CrossRef]
- Parisi, F.; Lazzara, G.; Merli, M.; Milioto, S.; Princivalle, F.; Sciascia, L. Simultaneous removal and recovery of metal ions and dyes from wastewater through montmorillonite clay mineral. Nanomaterials 2019, 9, 1699. [Google Scholar] [CrossRef]
- Zhao, L.; Azhar, M.R.; Li, X.; Duan, X.; Sun, H.; Wang, S.; Fang, X. Adsorption of cerium (III) by HKUST-1 metal-organic framework from aqueous solution. J. Colloid Interface Sci. 2019, 542, 421–428. [Google Scholar] [CrossRef]
- Bouchmila, I.; Bejaoui Kefi, B.; Souissi, R.; Abdellaoui, M. Purification, characterization and application of cherty rocks as sorbent for separation and preconcentration of rare earths. J. Mater. Res. Technol. 2019, 8, 2910–2923. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Hubicki, Z.; Fila, D. Recovery of rare earth elements from acidic solutions using macroporous ion exchangers. Sep. Sci. Technol. 2019, 54, 2059–2076. [Google Scholar] [CrossRef]
- Fila, D.; Hubicki, Z.; Kołodyńska, D. Recovery of metals from waste nickel-metal hydride batteries using multifunctional Diphonix resin. Adsorption 2019, 25, 367–382. [Google Scholar] [CrossRef]
- Alguacil, F.J.; Lopez, F.A.; Rodriguez, O.; Martinez-Ramirez, S.; Garcia-Diaz, I. Sorption of indium (III) onto carbon nanotubes. Ecotoxicol. Environ. Saf. 2016, 130, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Alguacil, F.J. Adsorption of gold(I) and gold(III) using multiwalled carbon nanotubes. Appl. Sci. 2018, 8, 2264. [Google Scholar] [CrossRef]
- Alguacil, F.J.; Lopez, F.A. Removal of Cr(VI) from waters by multi-walled carbon nanotubes: Optimization and kinetic investigations. In Water and Wastewater Treatment; Eyvaz, M., Ed.; IntechOpen: London, UK, 2019; Chapter 4. [Google Scholar]
- Alguacil, F.J.; Lopez, F.A. On the active adsorption of chromium(III) from alkaline solutions using multiwalled carbon nanotubes. Appl. Sci. 2020, 10, 36. [Google Scholar] [CrossRef]
- Alguacil, F.J.; Garcia-Diaz, I.; Escudero Baquero, E.; Rodriguez Largo, O.; Lopez, F.A. Oxidized and non-oxidized multiwalled carbon nanotubes as materials for adsorption of lanthanum(III) aqueous solutions. Metals 2020, 10, 765. [Google Scholar] [CrossRef]
- Bayazit, G.; Tastan, B.E.; Gui, U.D. Biosorption, isotherm and kinetic properties of common textile dye by Phormidium animale. Glob. NEST J. 2020, 22, 1–7. [Google Scholar]
- Hubbe, M.A.; Azizian, S.; Douven, S. Implications of apparent pseudo-second-order adsorption kinetics onto cellulosic materials: A review. Bioresource 2019, 14, 7582–7626. [Google Scholar]
- Seema, K.M.; Mamba, B.B.; Njuguna, J.; Bakhtizin, R.Z.; Mishra, A.K. Removal of lead (II) from aqueous waste using (CD-PCL-TiO2) bio-nanocomposites. Int. J. Biol. Macromol. 2018, 109, 136–142. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Le, T.T.N.; Le, V.T.; Dao, M.U.; Nguyen, Q.V.; Vu, T.T.; Nguyen, M.H.; Tran, D.L.; Le, H.S. Preparation of magneticgraphene oxide/chitosan composite beads for effective removal of heavy metals and dyesfrom aqueous solutions. Chem. Eng. Commun. 2019, 206, 1337–1352. [Google Scholar] [CrossRef]
- Ippolito, N.M.; Maffei, G.; Medici, F.; Piga, L. Adsorption and regeneration of fluoride ion on a high alumina content bauxite. Chem. Eng. Trans. 2016, 47, 217–222. [Google Scholar]
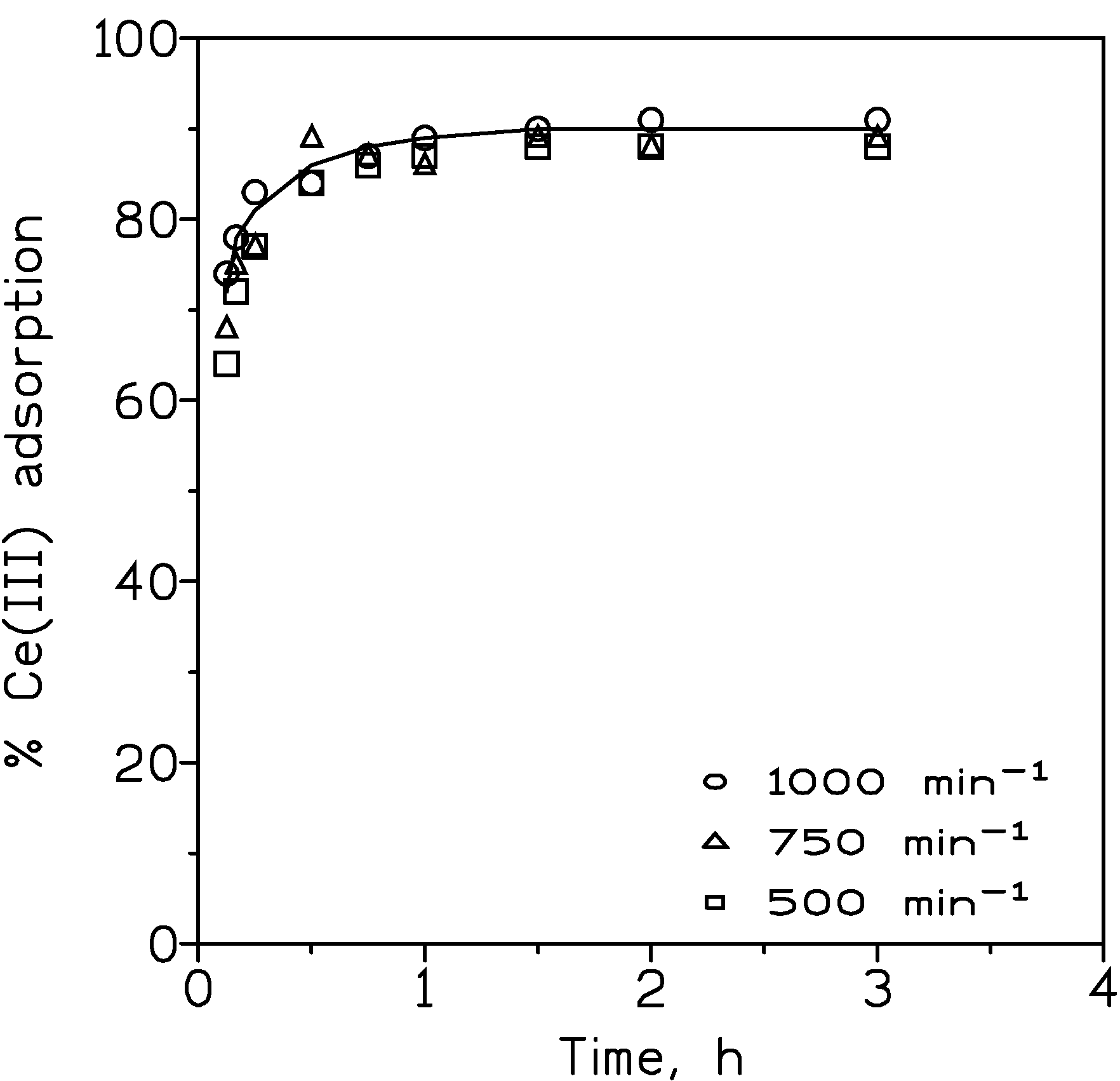
| Stirring Speed, min−1 | Ce(III) Adsorption, % |
|---|---|
| 250 | 90 |
| 500 | 99 |
| 750 | 98 |
| 1000 | 96 |
| pH | Ce(III) Adsorption, % |
|---|---|
| 1 | 1 |
| 2 | 10 |
| 3 | 29 |
| 4 | 78 |
| 5 | 85 |
| 6 | 99 |
| [Ce]aq,0, g/L | Ce(III) Adsorption, % |
|---|---|
| 0.01 | 99 |
| 0.02 | 78 |
| 0.03 | 77 |
| 0.04 | 76 |
| 0.06 | 52 |
| 0.08 | 38 |
| Nanotubes Dosage, g/L | Ce(III) Adsorption, % |
|---|---|
| 1 | 0.5 |
| 2 | 28 |
| 3 | 99 |
| 4 | 99 |
| [Ce]aq,0, g/L | Ce(III) Adsorption, % |
|---|---|
| 0.01 | 92 |
| 0.02 | 92 |
| 0.03 | 77 |
| 0.04 | 61 |
| 0.06 | 39 |
| 0.08 | 30 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alguacil, F.J.; García-Díaz, I.; Escudero Baquero, E.; Rodríguez Largo, O.; López, F.A. On the Adsorption of Cerium(III) Using Multiwalled Carbon Nanotubes. Metals 2020, 10, 1057. https://doi.org/10.3390/met10081057
Alguacil FJ, García-Díaz I, Escudero Baquero E, Rodríguez Largo O, López FA. On the Adsorption of Cerium(III) Using Multiwalled Carbon Nanotubes. Metals. 2020; 10(8):1057. https://doi.org/10.3390/met10081057
Chicago/Turabian StyleAlguacil, Francisco José, Irene García-Díaz, Esther Escudero Baquero, Olga Rodríguez Largo, and Félix Antonio López. 2020. "On the Adsorption of Cerium(III) Using Multiwalled Carbon Nanotubes" Metals 10, no. 8: 1057. https://doi.org/10.3390/met10081057
APA StyleAlguacil, F. J., García-Díaz, I., Escudero Baquero, E., Rodríguez Largo, O., & López, F. A. (2020). On the Adsorption of Cerium(III) Using Multiwalled Carbon Nanotubes. Metals, 10(8), 1057. https://doi.org/10.3390/met10081057








