Abstract
The formation of palladium hydrides is a well-known phenomenon, observed for both bulk and nanosized samples. The kinetics of hydrogen adsorption/desorption strongly depends on the particle size and shape, as well as the type of support and/or coating of the particles. In addition, the structural properties of hydride phases and their distribution also depend on the particle size. In this work, we report on the in situ characterization of palladium nanocubes coated with HKUST-1 metal-organic framework (Pd@HKUST-1) during desorption of hydrogen by means of synchrotron-based time-resolved X-ray powder diffraction. A slower hydrogen desorption, compared to smaller sized Pd nanoparticles was observed. Rietveld refinement of the time-resolved data revealed the remarkable stability of the lattice parameters of α- and β-hydride phases of palladium during the α- to β- phase transition, denoting the behavior more similar to the bulk materials than nanoparticles. The stability in the crystal sizes for both α- and β-hydride phases during the phase transition indicates that no sub-domains are formed within a single particle during the phase transition.
1. Introduction
Palladium is well-known for its ability for dissociative adsorption and storage of hydrogen [1]. The hydrogen storage capacity and kinetics of hydrogen adsorption and desorption are strongly affected by the particle’s size and shape [2,3,4,5,6,7,8], which is an important parameter for catalytic applications [8,9,10,11,12,13,14]. Bulk palladium materials exhibit a sharp phase transition from α- to β-hydride, resulting in a characteristic plateau in the pressure composition isotherm [1]. For nanostructured samples, the phase transition spans over a wider pressure range [3]. In the previous works, we explained such behavior by a gradual evolution of the cell parameters in the surface region of Pd nanoparticles (NPs) [5,15]. Different theoretical models were suggested as descriptive of the structure of palladium particles during formation and decomposition of hydrides [4,16,17,18,19]. In particular, the core-shell structures were suggested [18].
Metal-organic frameworks (MOFs) represent a new class of porous crystalline materials, built from inorganic cornerstones, connected with each other through organic linkers to three-dimensional structures [20,21]. Extremely high porosity and specific surface area make them promising materials for application in catalysis, gas storage and separation, drug delivery and other fields. For specific applications, MOFs be used as an alternative to conventional supports for stabilization of metal NPs [22,23,24,25,26,27]. HKUST-1 MOF, also known as Cu-BTC, MOF-199, and Basolite C300, represent a porous structure within Fm-3m space group based on copper inorganic cornerstones connected via 1,3,5 benzenetricarboxylate (BTC) linkers [28]. In previous work, we successfully incapsulated palladium particles into the pores of UiO-67 MOF and demonstrated their accessibility to hydrogen and other substrate molecules [29,30,31,32]. Significant enhancement of hydrogen storage capacity and adsorption speed by coating of palladium NPs by HKUST-1 was reported by Li et al [33].
Here, we perform in situ time-resolved X-ray powder diffraction (XRPD) study of the Pd@HKUST-1 material to obtain information about the kinetics of atomic structure of palladium during adsorption and desorption of hydrogen. The synthesized samples were characterized by XRPD and transmission electron microscopy (TEM). Utilization of synchrotron radiation allowed for the collection of high-quality time-resolved data, which revealed the evolution of α- and β-hydride phases of palladium, their corresponding lattice parameters and the crystallite sizes during desorption of hydrogen. The results were compared with supported metal nanoparticles of different size and bulk palladium samples.
2. Materials and Methods
2.1. Synthesis of Pd@HKUST-1
The synthesis was performed following the strategy described in [28]. 22.4 mg of polyvinyl alcohol, 144 mg of ascorbic acid, 720 mg of KBr, and 151.2 mg of K2PdCl4 were dissolved in 26.4 mL of deionized water. The solution was then transferred into 50 mL round-bottomed flask with a reflux condenser and was stirred in a heated bath at 80 °C for 3 h. The resulting mixture was filtered, washed three times by acetone to water 1:1 solution and dried at 40 °C in vacuum. 19.8 mg of copper (II) acetate and 12 mg of H3BTC (benzene-1,3,5-tricarboxylic acid) were dissolved in 12 mL of ethylene and mixed for 48 h together with the obtained palladium particles at room temperature. Finally, the samples were washed twice with ethanol and dried in air at 200 °C. The BET specific surface area for the HKUST-1 one sample without palladium was 1236.4892 m2/g.
2.2. Ex Situ Characterization
TEM images of the synthesized material were taken using Tecnai G2 Spirit TWIN (FEI, Hillsboro, Oregon, MA, USA) transmission electron microscope operated at an accelerating voltage of 80 kV. The obtained images were processed using ImageJ (Version 1.53a, National Institutes of Health, Bethesda, MD, USA) software [34]. For particle size calculation, each visible particle was highlighted by tetragons and an average edge length was calculated as a square root of a tetragon area. The XRPD characterization was performed using D2 PHASER diffractometer (Bruker, Billerica, MA, USA) using Cu Kα1,2 radiation. The patterns were collected in Bragg-Brentano geometry in the 2θ range 5–80° with a 2θ step of 0.02°. Reference HKUST-1 and metallic palladium powder were measured under the same conditions.
2.3. In Situ X-Ray Powder Diffraction
In situ XRPD measurements during absorption/desorption of hydrogen were performed at BM31 beamline [35] of European Synchrotron Radiation Facility (ESRF, Grenoble, France) in Debye-Scherrer geometry. The sample was placed in a 1 mm quartz glass capillary and fixed from both sides by the quartz wool. The remotely controlled gas rig, permanently installed at BM31 beamline, and equipped with fast electric valves, was adopted to add and outgas hydrogen during the experiment. Fast outgassing was performed by opening the electric from the capillary to a pre-evacuated volume (ca. 1 L) connected with a vacuum pump. The time needed for the outgassing was below 1 s, which was tested by monitoring phase transition from hydride to metallic phase in small Pd particles [16], which is considerably faster than the desorption kinetics observed for Pd@HKUST-1. The photon wavelength of λ = 0.505235 Å was selected by Si(111) channel-cut monochromator. CMOS-DEXELA 2D image plate detector were located at 250.14 mm from the sample covering the 2θ angles up to 50° (dmin ~ 0.6 Å). The region below 4° was shielded by the beam-stop, which protected the detector from being damaged by the direct beam. The sample-to-detector distance, detector tilts and the exact photon wavelength were optimized by Rietveld refinement of the reference LaB6 and Si samples. For time-resolved measurements, 5 s acquisition per pattern was adjusted. The two-dimensional (2D) patterns were processed in PyFAI code [36] to obtain I(2θ) diffraction data. Structural analysis was performed by Rietveld refinement of the diffraction data using GSAS II software (Version 4469, General Structure Analysis System-II, Argonne National Laboratory, IL, USA) [37]. The cell parameters of α- and β-hydride phases of palladium, the relative fractions of these phases and isotropic atomic displacement parameters were set as variable parameters during the refinement. The Pseudo-Voight function were used to describe the shape of the Bragg peaks. Additional broadening due to the nanometric particle size was considered and kept as a variable parameter for both phases.
3. Results
3.1. Characterization of the Initial Pd@HKUST-1 Material
TEM images of Pd@HKUST-1 revealed formation of the nanocubes (Figure 1) covered by MOF. Due to big difference in atomic number between Cu and Pd, the scanning electron microscopy images measured in secondary electron and high angle backscattered electron regimes evidence that the cubic particles correspond to palladium (Figure S5). Average edge size of the nanocubes in Pd@HKUST-1 sample of 23 ± 4 nm was obtained by calculation of ca. 300 particles (Figure 2). A fraction of smaller particles (see Figure S4) was visible in some images. These particles were not considered, as they give negligible contribution XRPD signal compared to bigger particles. In addition, smaller particles were reported to rich lower H/Pd values at the same temperature and hydrogen pressure [38], which means that their contribution to the total hydrogen adsorption is also much smaller compared to bigger particles. Comparison of the XRPD profile of Pd@HKUST-1 with those of the reference materials also confirmed the presence of both fcc Pd and HKUST-1 phases in the synthesized material (Figure 3). The refined value of the cell parameter of the palladium nanoparticles is 3.896(1) Å, which is close to the value of 3.8899(1) Å obtained for bulk palladium.

Figure 1.
Representative TEM micrographs of Pd nanocubes (a–c) and Pd@HKUST-1 (d–f).
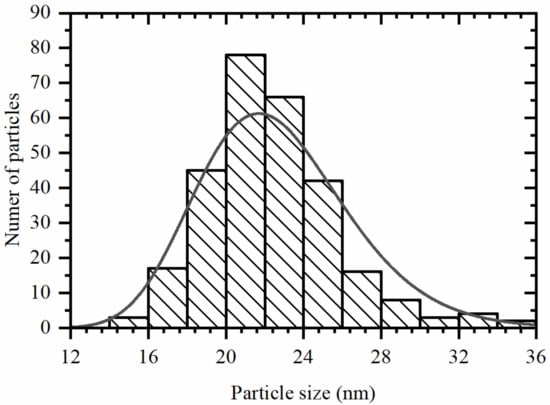
Figure 2.
Particle size distribution calculated from TEM images.
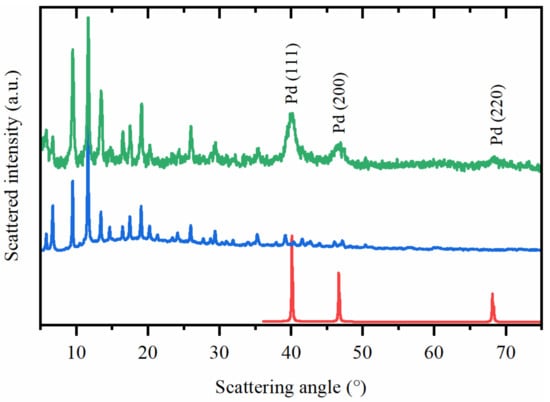
Figure 3.
Ex situ XRPD patterns (Cu Kα radiation) for metallic palladium (red), HKUST-1 (blue) and Pd@HKUST-1 sample (green).
3.2. Evolution of Palladium Structure During Adsorption/Desorption of Hydrogen
To form the hydride phase in palladium NPs, the sample was exposed to 2.1 bar of pure hydrogen at 80 °C. The hydrogen absorption and formation of palladium hydride is an exothermal process which is faster than the desorption of hydrogen. The data that was collected during the formation of palladium hydride are shown in the Supporting Information (Figure S1). Here, we focus on the detailed characterization of the desorption process during fast (<1 s) outgassing from 2.1 bar hydrogen to vacuum. The whole procedure was carried out keeping the sample temperature at 80 °C.
After complete formation of β-hydride phase, the sample was outgassed by opening the electrovalve to the vacuum pump, and the continuous collection of XRPD data with time resolution of ca. 5 s was started. The quality of the data can be appreciated in the Figure S2 of the Supporting Information. The evolution of XRPD profiles is illustrated in Figure 4. The hydrogen desorption from the particles is accompanied by a gradual decrease of the peaks of β-hydride phase (highlighted by dashed vertical lines in Figure 4) with the cell parameter ca. 4.03 Å to α-hydride phase highlighted by dotted vertical lines (cell parameter ca. 3.90 Å).
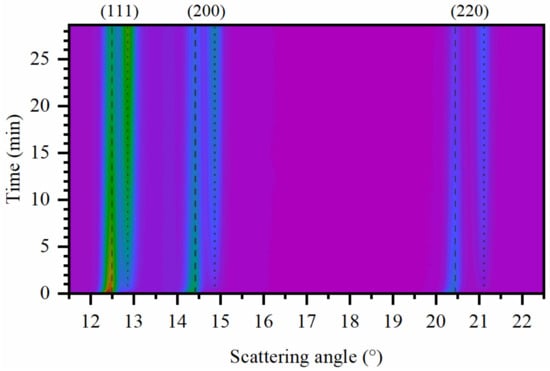
Figure 4.
Evolution of in situ XRPD data collected with 0.505235 Å radiation during H2 desorption at 80 °C after fast outgassing from 2.1 bar hydrogen to vacuum. Vertical dashed and dotted lines highlight the positions of Pd reflections for α- and β-hydride phases respectively. Selected patterns from these series are shown in Figure S2 of the Supporting Information.
Sequential analysis of the whole dataset of XRPD patterns was performed in GSAS II code [37]. The profile parameters of the Pseudo-Voight function (GU, GV, GW, LX, and LY) were initially refined for the reference Si sample to account for the experimental contribution. The additional width observed for Pd reflections in Pd@HKUST-1 was attributed to the nanosized dimensions of the particles. It should be noted, that GW parameter was increased by 9 to account for the additional peak broadening due to a thicker capillary (1.0 mm) used for Pd@HKUST-1 compared to 0.7 mm capillary for Si reference, which gives additional peak width of ca. 0.07° estimated by the Equation (1) [39],
where d is the capillary thickness and r is the sample-to-detector distance.
The sequential Refinement of ca. 300 XRPD patterns illustrated in Figure 4 was run in the reverse order starting from the last pattern where both α- and β-hydride are clearly defined. For each pattern the following set of variables were used: cell parameters aα and aβ of the two phases, the relative fractions of these phases with their sum constrained to 1, isothropic atomic displacement parameters Uiso,α and Uiso,β for both phases, crystallite sizes Dα and Dβ for both phases, and 9-parameter Chebyshev polynomial for the background function, which gives (together with the scale factor) 17 independent parameters. The refined values obtained at each step were used as starting point for the refinement of the next pattern. The first 3 patterns, collected after the beginning of the outgassing procedure, were excluded from the refinement since the α-phase fraction was below the detection limit causing instabilities in the fitting procedure. The main structural parameters obtained from the refinement are shown in Figure 5.
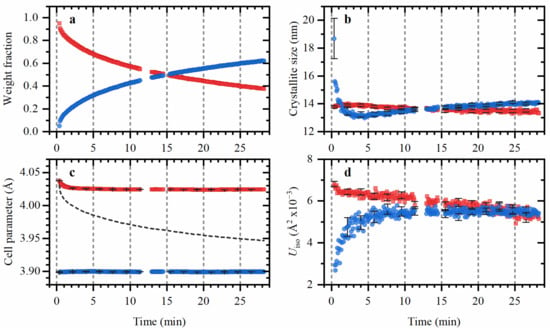
Figure 5.
Fractions (a), crystallite sizes (b), cell parameters (c) and Uiso values (d) for α- (blue circles) and β- (red squares) hydride phases of palladium as function of time after fast outgassing from 2.1 bar hydrogen to vacuum at 80 °C obtained by Rietveld refinement of XRPD data.
4. Discussion
The process of hydrogen desorption from Pd@HKUST-1 is slower than for few-nanometer palladium particles on conventional supports [4,16] and particles confined in the MOF pores [29], which allowed detailed structural characterization along the desorption process. The time needed to decompose 50% of hydride phase was 15 min (Figure 5a). The complete removal of the β-phase was achieved by heating the sample to 120 °C (See Supporting Information, Figure S3). For comparison, 2.6 nm Pd NPs supported on carbon undergo from α- to β-phase in 2 s at room temperature [16], while at higher temperatures, the desorption time was below 1 s. For supported Pd nanoparticles with sized from 1.8 to 5.4 nm, the desorption times from few seconds to minutes were reported with strong dependence on the particle size. The dependence between the desorption time (the authors used the time needed for half of the hydrogen to be adsorbed/desorbed) and the particle size was studied, which suggests further extrapolation to the bigger-sized particles. Considerably slower kinetics than for our Pd@HKUST-1 material, were reported for 4–6 nm particles at room temperature [40], where the time-resolved measurements were performed by measuring X-ray diffraction intensity fixed 2θ-position of metallic Pd (111) reflection, which was calibrated with the cell parameter. Notable, all previous time-resolved measurements were performed “indirectly”, while in this study, high-quality time-resolved X-ray diffraction profiles were collected allowing for a more comprehensive analysis.
Both α- and β-phases are characterized by similar crystallite sizes which are stable along the whole desorption process (Figure 5b), with the only slight deviation for the α-phase in the first minute of desorption, which is attributed to the fact that the fraction of this phase is below 0.1, resulting in the bigger error in determination of its crystallite size and high correlation with other parameters (particularly, with aβ and aα). The stability of the crystallite size indicates that within the limitation of XRPD method there are no sub-domains or core-shell regions formed, which was suggested by theoretical calculations [18,19] and interpretation of the hydrogen desorption kinetics, measured by indirect nanoplasmonic sensing [4] during the hydrogen desorption process. The small fractions of the additional phases and/or amorphous parts of the nanoparticles can escape from XRPD analysis, and are both being possible in the thin surface layer.
Remarkable is the stability of the cell parameters of α- and β-phases (Figure 5c). Such behavior is closer to the bulk materials, than to the nanosized samples, where a variation of cell parameters was observed, resulting in the less defined phase transition [4,5]. The increased values of aβ in the beginning of the outgassing procedure are explained by the fact that in pure β-phase region the cell parameter increases with increasing hydrogen pressure (the starting pressure was 2.1 bar).
Stability of the cell parameters and average crystallite sizes during the phase transition correlates with the sharp plateau observed by Li et al for a similar material [33]. For accurate comparison, it should be noted, that the particle size obtained in this work is bigger than that reported in [33]. However, the time needed for β-hydride phase formation is comparable for the data reported by Li et al in the current work (see Figure S1 of the supporting information). In both cases complete transition to β-hydride occurs within 5 s.
The isotropic atomic displacement parameters are also similar for both phases and do not change significantly during the phase transition. The smaller values observed for α-phase in the first minutes of the hydrogen should be attributed to the small fraction of this phase, which complicates the accurate estimation of the integral peak intensities. A possible reason for such inaccuracy may be the fact that, for a small concentration, the peak shape is not well-defined and the contribution of smaller particles to the Bragg peaks can be partially described by the background function. While, the pseudo-Voight function describes the remaining bigger fraction of particles. This can explain both lower Uiso and bigger particle sizes obtained for the α-phase at the beginning of hydrogen desorption procedure.
5. Conclusions
We performed in situ time-resolved characterization of the phase transition from β- to α-hydride phases in the cubic palladium nanoparticles, with the edge length of ca. 20 nm coated by HKUST-1 MOF, by means of synchrotron-based XRPD. The sequential analysis of the whole experimental dataset provided the structural insights into the process of β-hydride decomposition. In particular, no changes in the crystallite size were observed indicating that each particle is present either, in α- or β-phase, and excluding the core-shell models of phase distribution, or other possibilities of sub-domain formation, within the limitation of XRPD method. A remarkable stability of the cell parameters of α- and β-phase is more similar to that observed for bulk materials, than the nanosized ones, which explains the sharp phase transition previously reported for Pd@HKUST-1 material.
Supplementary Materials
The following are available online at https://www.mdpi.com/2075-4701/10/6/810/s1, Figure S1: In situ XRPD patterns collected with 0.505235 Å radiation during formation of hydride phases Pd@HKUST-1 (from blue to red). The first four spectra are collected within a minute, the last one is after five minutes after sending 2.1 bar hydrogen, Figure S2: XRPD patterns (0.505235 Å radiation) from the first 5 min of the hydrogen desorption from Pd@HKUST-1 reproducing selected data from the whole dataset shown in Figure 4 of the main text. The acquisition time for each pattern was 5 s, Figure S3: XRPD pattern (0.505235 Å radiation) for the Pd@HKUST-1 sample after complete hydrogen desorption at 120 °C, Figure S4: TEM distribution of bigger (black) and smaller (red) particles, Figure S5: SEM image for Pd@HKUST-1 material measured in SE (a) and HA-BSE (b) regimes, Figure S6. XRPD patterns (Cu Kα radiation) for synthesized HKUST-1 (blue) and reference Basolite C300 sample (black).
Author Contributions
Funding acquisition, A.L.B. and A.V.S.; investigation, M.V.K., A.L.B., A.A.S., V.V.B., A.P.B., A.A.G. and A.L.T.; methodology, A.L.B.; project administration, A.V.S.; supervision, V.V.B., A.A.G. and A.V.S.; writing—original draft, A.L.B.; writing—review & editing, M.V.K., A.A.S., A.P.B. and A.A.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Russian Science Foundation Project No. 18-73-00189.
Acknowledgments
A.L.B. acknowledge Russian Science Foundation Project No. 18-73-00189 for funding the research. A.A.S. acknowledge President’s Grant of Russian Federation for Young Scientists (Grant MK-2554.2019.2 to A.L.B., Agreement No. 075-15-2019-1096) for travel support. We acknowledge ESRF for providing the beamtime at BM31 beamliene.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- Manchester, F.D.; San-Martin, A.; Pitre, J.M. The H-Pd (hydrogen-palladium) system. J. Phase Equilib. 1994, 15, 62–83. [Google Scholar] [CrossRef]
- Kishore, S.; Nelson, J.A.; Adair, J.H.; Eklund, P.C. Hydrogen storage in spherical and platelet palladium nanoparticles. J. Alloy. Compd. 2005, 389, 234–242. [Google Scholar] [CrossRef]
- Langhammer, C.; Zhdanov, V.P.; Zoric, I.; Kasemo, B. Size-dependent hysteresis in the formation and decomposition of hydride in metal nanoparticles. Chem. Phys. Lett. 2010, 488, 62–66. [Google Scholar] [CrossRef]
- Langhammer, C.; Zhdanov, V.P.; Zorić, I.; Kasemo, B. Size-dependent kinetics of hydriding and dehydriding of Pd nanoparticles. Phys. Rev. Lett. 2010, 104, 135502. [Google Scholar] [CrossRef] [PubMed]
- Bugaev, A.L.; Guda, A.A.; Lomachenko, K.A.; Shapovalov, V.V.; Lazzarini, A.; Vitillo, J.G.; Bugaev, L.A.; Groppo, E.; Pellegrini, R.; Soldatov, A.V.; et al. Core–shell structure of palladium hydride nanoparticles revealed by combined X-ray absorption spectroscopy and X-ray diffraction. J. Phys. Chem. C 2017, 121, 18202–18213. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Srabionyan, V.V.; Soldatov, A.V.; Bugaev, L.A.; Van Bokhoven, J.A. The role of hydrogen in formation of Pd XANES in Pd-nanoparticles. J. Physics Conf. Ser. 2013, 430, 012028. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Guda, A.A.; Lomachenko, K.A.; Lazzarini, A.; Srabionyan, V.V.; Vitillo, J.G.; Piovano, A.; Groppo, E.; Bugaev, L.A.; Soldatov, A.V.; et al. Hydride phase formation in carbon supported palladium hydride nanoparticles by in situ EXAFS and XRD. J. Physics Conf. Ser. 2016, 712, 012032. [Google Scholar] [CrossRef]
- Tew, M.W.; Miller, J.T.; Van Bokhoven, J.A. Particle size effect of hydride formation and surface hydrogen adsorption of nanosized palladium catalysts: L3 edge vs K edge X-ray absorption spectroscopy. J. Phys. Chem. C 2009, 113, 15140–15147. [Google Scholar] [CrossRef]
- Tew, M.W.; Janousch, M.; Huthwelker, T.; Van Bokhoven, J.A. The roles of carbide and hydride in oxide-supported palladium nanoparticles for alkyne hydrogenation. J. Catal. 2011, 283, 45–54. [Google Scholar] [CrossRef]
- Bugaev, A.; Polyakov, V.; Tereshchenko, A.; Isaeva, A.; Skorynina, A.; Kamyshova, E.; Budnyk, A.; Lastovina, T.; Soldatov, A. Chemical synthesis and characterization of Pd/SiO2: The effect of chemical reagent. Metals 2018, 8, 135. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Guda, A.A.; Pankin, I.A.; Groppo, E.; Pellegrini, R.; Longo, A.; Soldatov, A.V.; Lamberti, C. The role of palladium carbides in the catalytic hydrogenation of ethylene over supported palladium nanoparticles. Catal. Today 2019, 336, 40–44. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Guda, A.A.; Pankin, I.A.; Groppo, E.; Pellegrini, R.; Longo, A.; Soldatov, A.V.; Lamberti, C. Operando X-ray absorption spectra and mass spectrometry data during hydrogenation of ethylene over palladium nanoparticles. Data Brief 2019, 24, 103954. [Google Scholar] [CrossRef]
- Usoltsev, O.A.; Bugaev, A.L.; Guda, A.A.; Guda, S.A.; Soldatov, A.V. Absorption of hydrocarbons on palladium catalysts: From simple models towards machine learning analysis of X-ray absorption spectroscopy data. Top. Catal. 2020. [Google Scholar] [CrossRef]
- Skorynina, A.A.; Tereshchenko, A.A.; Usoltsev, O.A.; Bugaev, A.L.; Lomachenko, K.A.; Guda, A.A.; Groppo, E.; Pellegrini, R.; Lamberti, C.; Soldatov, A. Time-dependent carbide phase formation in palladium nanoparticles. Radiat. Phys. Chem. 2018. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Usoltsev, O.A.; Lazzarini, A.; Lomachenko, K.A.; Guda, A.A.; Pellegrini, R.; Carosso, M.; Vitillo, J.G.; Groppo, E.; Van Bokhoven, J.A.; et al. Time-resolved operando studies of carbon supported Pd nanoparticles under hydrogenation reactions by X-ray diffraction and absorption. Faraday Discuss. 2018, 208, 187–205. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Guda, A.A.; Lomachenko, K.A.; Soldatov, A.V. Kinetics of the atomic structure of palladium nanoparticles during the desorption of hydrogen according to X-ray diffraction. JETP Lett. 2019, 109, 594–599. [Google Scholar] [CrossRef]
- Wadell, C.; Pingel, T.; Olsson, E.; Zoric, I.; Zhdanov, V.P.; Langhammer, C. Thermodynamics of hydride formation and decomposition in supported sub-10 nm Pd nanoparticles of different sizes. Chem. Phys. Lett. 2014, 603, 75–81. [Google Scholar] [CrossRef]
- Zhdanov, V.P.; Kasemo, B. Kinetics of the formation of a new phase in nanoparticles. Chem. Phys. Lett. 2008, 460, 158–161. [Google Scholar] [CrossRef]
- Zhdanov, V.P.; Krozer, A.; Kasemo, B. Kinetics of first-order phase transitions initiated by diffusion of particles from the surface into the bulk. Phys. Rev. B 1993, 47, 11044–11048. [Google Scholar] [CrossRef]
- Butova, V.V.; Soldatov, M.A.; Guda, A.A.; Lomachenko, K.A.; Lamberti, C. Metal-organic frameworks: Structure, properties, methods of synthesis and characterization. Russ. Chem. Rev. 2016, 85, 280–307. [Google Scholar] [CrossRef]
- Zhou, H.C.; Long, J.R.; Yaghi, O.M. Introduction to metal-organic frameworks. Chem. Rev. 2012, 112, 673–674. [Google Scholar] [CrossRef] [PubMed]
- Esken, D.; Zhang, X.; Lebedev, O.I.; Schröder, F.; Fischer, R.A. Pd@MOF-5: Limitations of gas-phase infiltration and solution impregnation of [Zn4O(bdc)3] (MOF-5) with metal–organic palladium precursors for loading with Pd nanoparticles. J. Mater. Chem. 2009, 19. [Google Scholar] [CrossRef]
- Wang, T.; Gao, L.; Hou, J.; Herou, S.J.A.; Griffiths, J.T.; Li, W.; Dong, J.; Gao, S.; Titirici, M.M.; Kumar, R.V.; et al. Rational approach to guest confinement inside MOF cavities for low-temperature catalysis. Nat. Commun. 2019, 10, 1340. [Google Scholar] [CrossRef]
- Proch, S.; Herrmannsdorfer, J.; Kempe, R.; Kern, C.; Jess, A.; Seyfarth, L.; Senker, J. Pt@MOF-177: Synthesis, room-temperature hydrogen storage and oxidation catalysis. Chem. Eur. J. 2008, 14, 8204–8212. [Google Scholar] [CrossRef]
- Braglia, L.; Borfecchia, E.; Lomachenko, K.A.; Bugaev, A.L.; Guda, A.A.; Soldatov, A.V.; Bleken, B.T.L.; Oien-Odegaard, S.; Olsbye, U.; Lillerud, K.P.; et al. Tuning Pt and Cu sites population inside functionalized UiO-67 MOF by controlling activation conditions. Faraday Discuss. 2017, 201, 277–298. [Google Scholar] [CrossRef] [PubMed]
- Braglia, L.; Borfecchia, E.; Maddalena, L.; Øien, S.; Lomachenko, K.A.; Bugaev, A.L.; Bordiga, S.; Soldatov, A.V.; Lillerud, K.P.; Lamberti, C.; et al. Exploring structure and reactivity of Cu sites in functionalized UiO-67 MOFs. Catal. Today 2017, 283, 89–103. [Google Scholar] [CrossRef]
- Braglia, L.; Borfecchia, E.; Martini, A.; Bugaev, A.L.; Soldatov, A.V.; Oien-Odegaard, S.; Lonstad-Bleken, B.T.; Olsbye, U.; Lillerud, K.P.; Lomachenko, K.A. The duality of UiO-67-Pt MOFs: Connecting treatment conditions and encapsulated Pt species by operando XAS. Phys. Chem. Chem. Phys. 2017, 19, 27489–27507. [Google Scholar] [CrossRef]
- Chui, S.S.; Lo, S.M.; Charmant, J.P.; Orpen, A.G.; Williams, I.D. A chemically functionalizable nanoporous material. Science 1999, 283, 1148–1150. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Guda, A.A.; Lomachenko, K.A.; Kamyshova, E.G.; Soldatov, M.A.; Kaur, G.; Øien-Ødegaard, S.; Braglia, L.; Lazzarini, A.; Manzoli, M.; et al. Operando study of palladium nanoparticles inside UiO-67 MOF for catalytic hydrogenation of hydrocarbons. Faraday Discuss. 2018, 208, 287–306. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Skorynina, A.A.; Braglia, L.; Lomachenko, K.A.; Guda, A.; Lazzarini, A.; Bordiga, S.; Olsbye, U.; Lillerud, K.P.; Soldatov, A.V.; et al. Evolution of Pt and Pd species in functionalized UiO-67 metal-organic frameworks. Catal. Today 2019, 336, 33–39. [Google Scholar] [CrossRef]
- Bugaev, A.L.; Skorynina, A.A.; Kamyshova, E.G.; Lomachenko, K.A.; Guda, A.A.; Soldatov, A.V.; Lamberti, C. In situ X-ray absorption spectroscopy data during formation of active Pt- and Pd-sites in functionalized UiO-67 metal-organic frameworks. Data Brief 2019, 25, 104280. [Google Scholar] [CrossRef] [PubMed]
- Kamyshova, E.G.; Skorynina, A.A.; Bugaev, A.L.; Lamberti, C.; Soldatov, A.V. Formation and growth of Pd nanoparticles in UiO-67 MOF by in situ EXAFS. Radiat. Phys. Chem. 2019. [Google Scholar] [CrossRef]
- Li, G.; Kobayashi, H.; Taylor, J.M.; Ikeda, R.; Kubota, Y.; Kato, K.; Takata, M.; Yamamoto, T.; Toh, S.; Matsumura, S.; et al. Hydrogen storage in Pd nanocrystals covered with a metal-organic framework. Nat. Mater. 2014, 13, 802–806. [Google Scholar] [CrossRef] [PubMed]
- Collins, T. ImageJ for microscopy. Biotechniques 2007, 43, S25–S30. [Google Scholar] [CrossRef]
- Van Beek, W.; Safonova, O.V.; Wiker, G.; Emerich, H. SNBL, a dedicated beamline for combinedin situ X-ray diffraction, X-ray absorption and Raman scattering experiments. Phase Transit. 2011, 84, 726–732. [Google Scholar] [CrossRef]
- Kieffer, J.; Wright, J.P. PyFAI: A Python library for high performance azimuthal integration on GPU. Powder Diffr. 2013, 28, S339–S350. [Google Scholar] [CrossRef]
- Toby, B.H.; Von Dreele, R.B. GSAS-II: The genesis of a modern open-source all purpose crystallography software package. J. Appl. Crystallogr. 2013, 46, 544–549. [Google Scholar] [CrossRef]
- Berube, V.; Radtke, G.; Dresselhaus, M.; Chen, G. Size effects on the hydrogen storage properties of nanostructured metal hydrides: A review. Int. J. Energy Res. 2007, 31, 637–663. [Google Scholar] [CrossRef]
- Karen, P.; Woodward, P.M. Liquid-mix disorder in crystalline solids: ScMnO3. J. Solid State Chem. 1998, 141, 78–88. [Google Scholar] [CrossRef]
- Vogel, W.; He, W.; Huang, Q.-H.; Zou, Z.; Zhang, X.-G.; Yang, H. Palladium nanoparticles “breathe” hydrogen; a surgical view with X-ray diffraction. Int. J. Hydrogen Energy 2010, 35, 8609–8620. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).