Leaching of Pure Chalcocite in a Chloride Media Using Waste Water at High Temperature
Abstract
1. Introduction
2. Materials and Methods
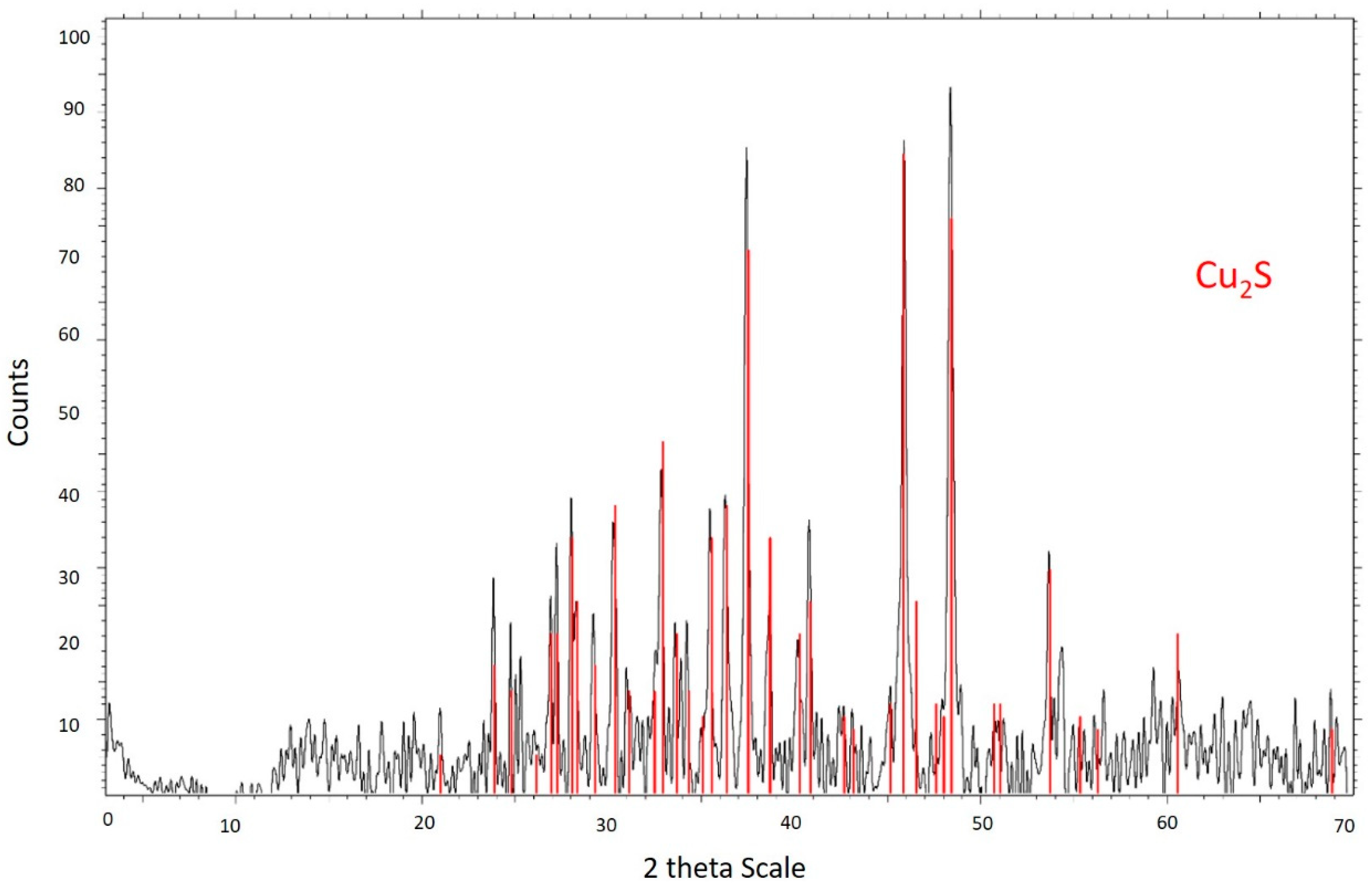
2.1. Chalcocite
2.2. Reagents and Leaching Tests
2.3. Experimental Design
3. Results
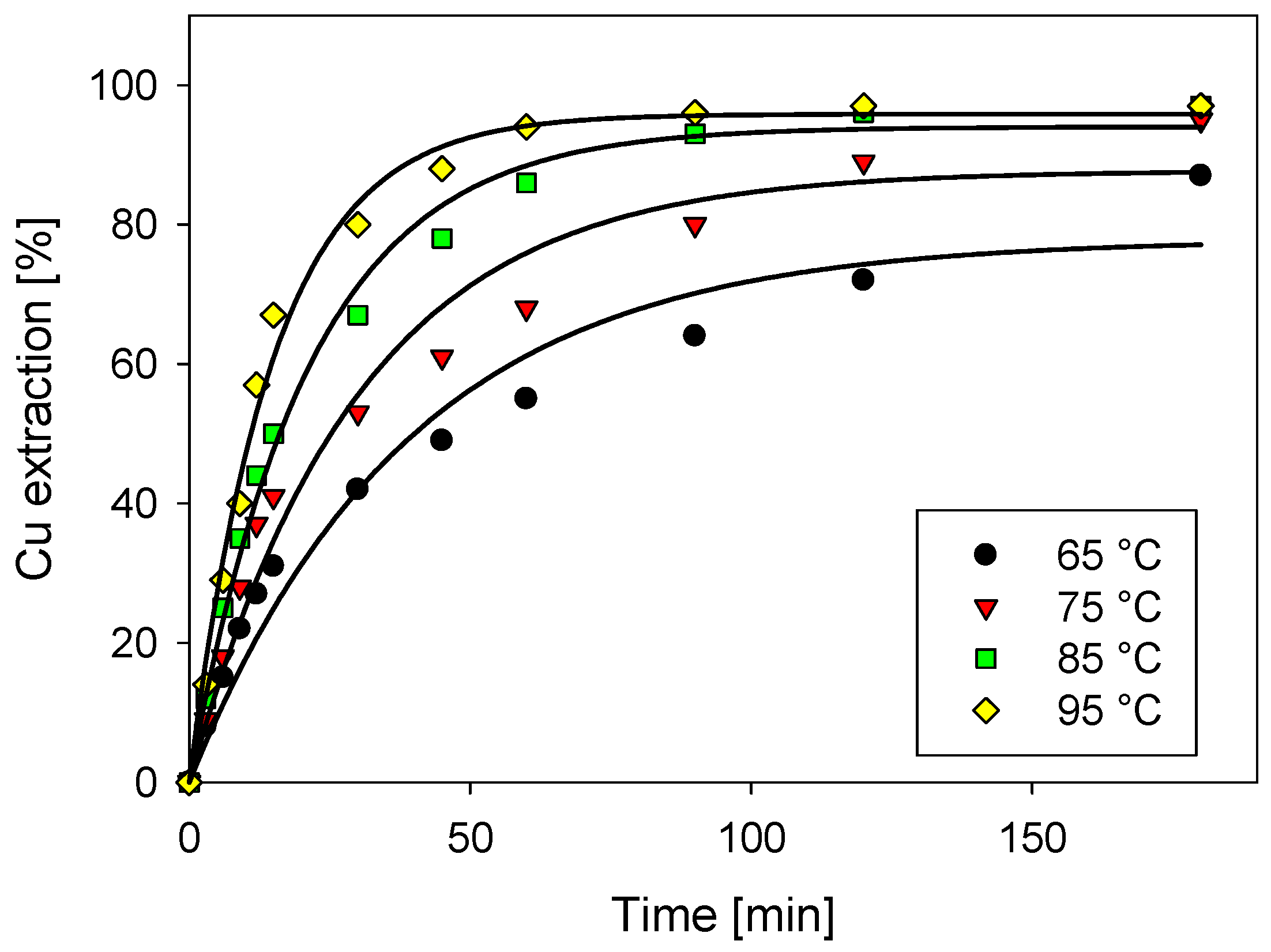
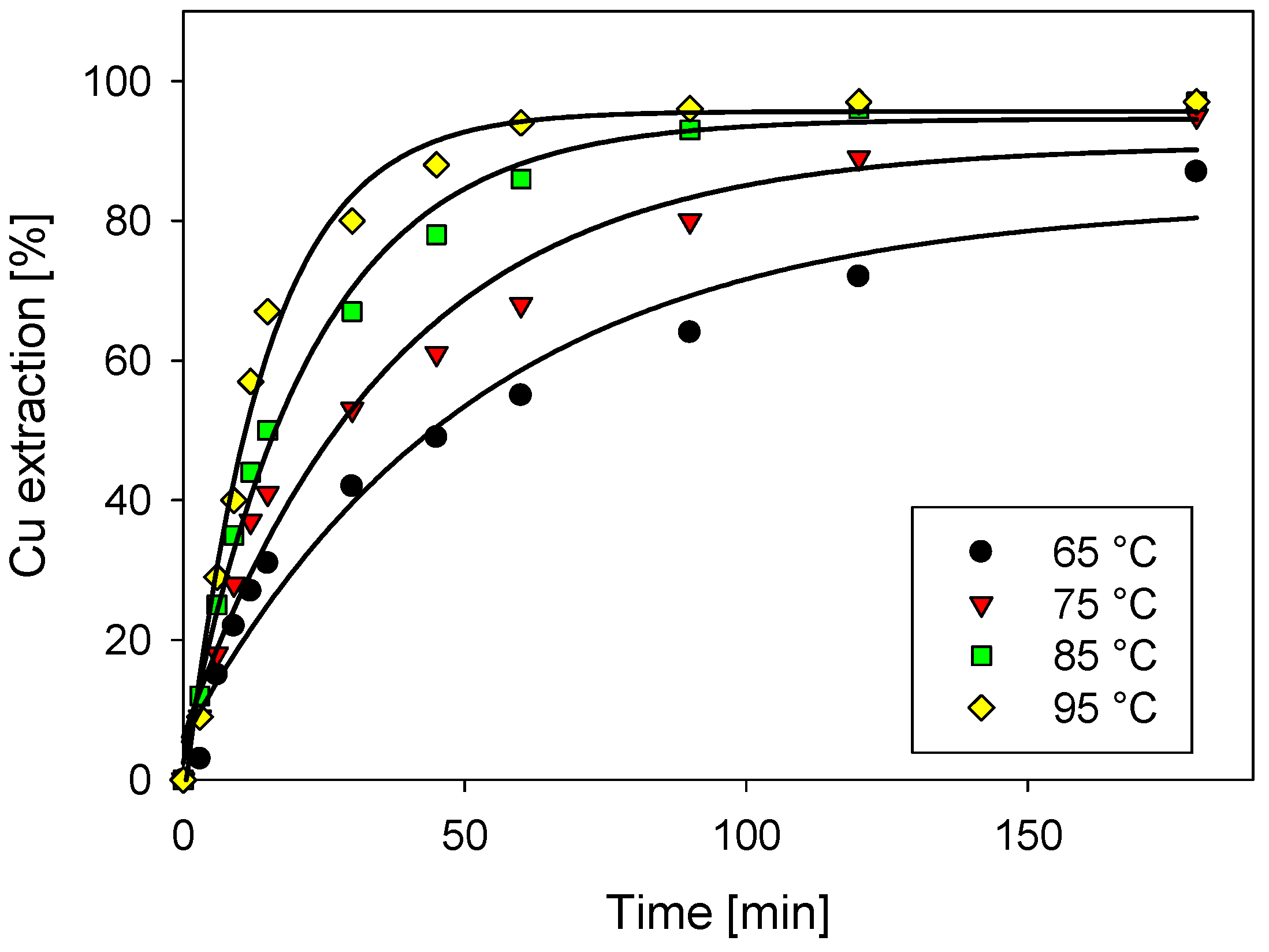
3.1. Effect of Temperature in Copper Extraction
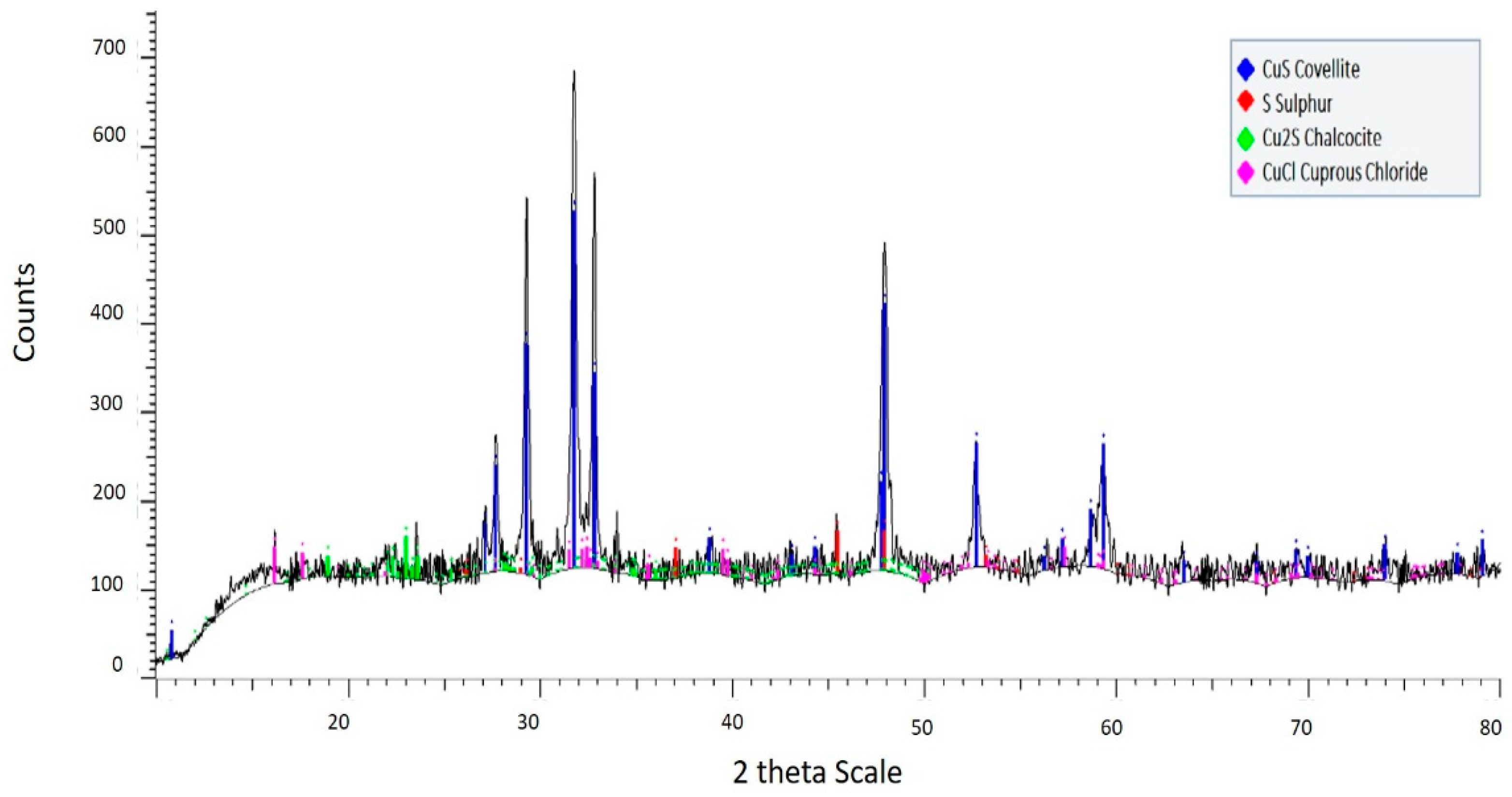
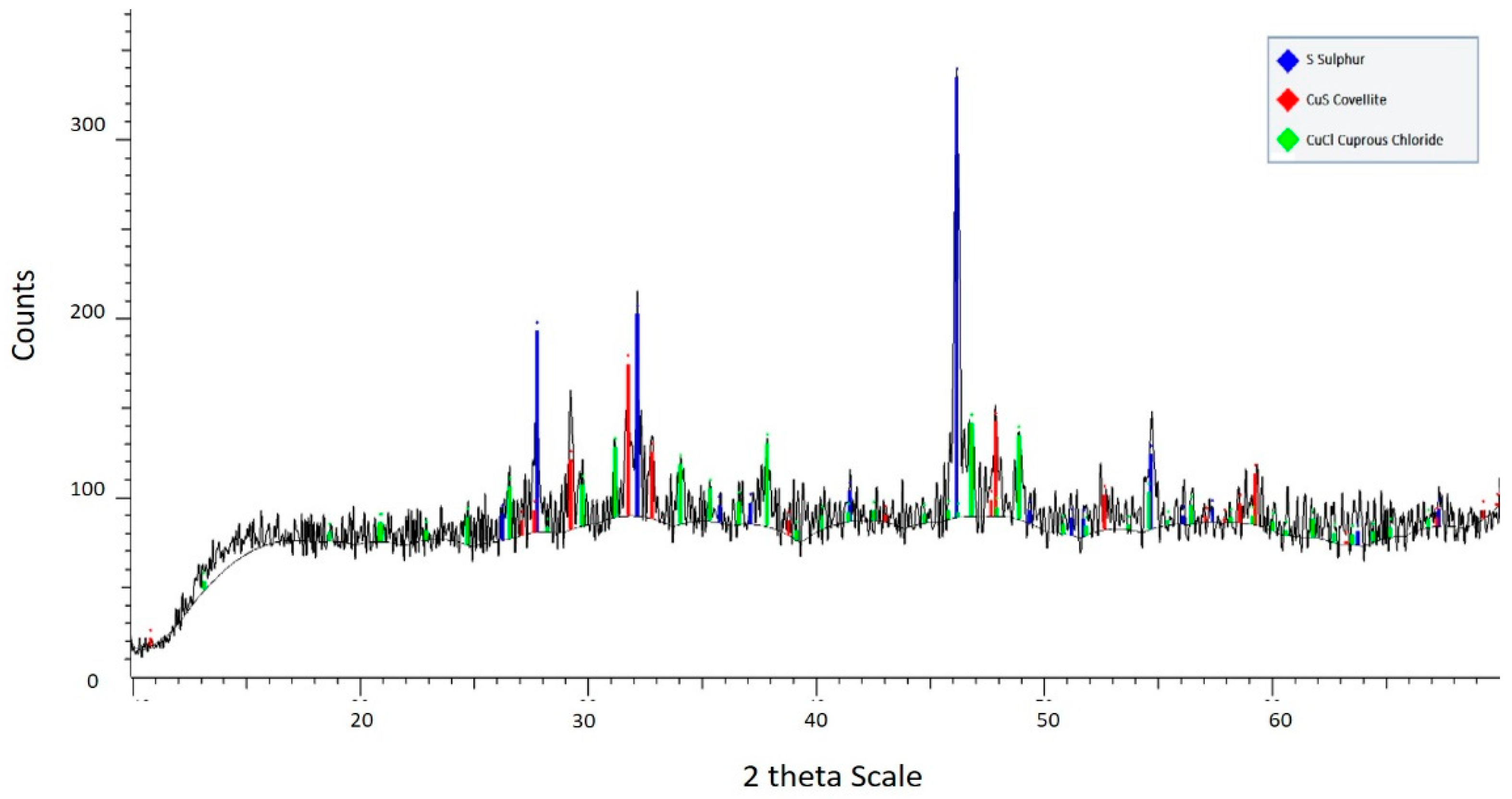
3.2. Residue Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pérez, K.; Toro, N.; Campos, E.; González, J.; Jeldres, R.I.; Nazer, A.; Rodriguez, M.H. Extraction of Mn from Black Copper Using Iron Oxides from Tailings and Fe2+ as Reducing Agents in Acid Medium. Metals 2019, 9, 1112. [Google Scholar] [CrossRef]
- Beiza, L.; Quezada, V.; Melo, E.; Valenzuela, G. Electrochemical behaviour of chalcopyrite in chloride solutions. Metals 2019, 9, 67. [Google Scholar] [CrossRef]
- Velásquez Yévenes, L. The Kinetics of the Dissolution of Chalcopyrite in Chloride Media. Ph.D. Thesis, Murdoch University, Perth, Australia, March 2009. [Google Scholar]
- Consumo De Agua En La Mineria Del Cobre. Available online: http://www.cochilco.cl/descargas/estudios/informes/agua/CONSUMO-DE-AGUA-EN-LA-MINERIA-DEL-COBRE-2011.pdf (accessed on 11 December 2019).
- Proyección De Consumo De Agua En La Minería Del Cobre 2018–2029. Available online: https://www.cochilco.cl/Mercado de Metales/Proyección de la producción esperada de cobre 2018–2029 Vfinal.pdf (accessed on 11 December 2019).
- Hernández, P.C.; Taboada, M.E.; Herreros, O.O.; Torres, C.M.; Ghorbani, Y. Chalcopyrite dissolution using seawater-based acidic media in the presence of oxidants. Hydrometallurgy 2015, 157, 325–332. [Google Scholar] [CrossRef]
- Watling, H.R.; Shiers, D.W.; Li, J.; Chapman, N.M.; Douglas, G.B. Effect of water quality on the leaching of a low-grade copper sulfide ore. Miner. Eng. 2014, 58, 39–51. [Google Scholar] [CrossRef]
- Torres, C.M.; Taboada, M.E.; Graber, T.A.; Herreros, O.O.; Ghorbani, Y.; Watling, H.R. The effect of seawater based media on copper dissolution from low-grade copper ore. Miner. Eng. 2015, 71, 139–145. [Google Scholar] [CrossRef]
- Mu, Y.; Peng, Y. The effect of saline water on copper activation of pyrite in chalcopyrite flotation. Miner. Eng. 2019, 131, 336–341. [Google Scholar] [CrossRef]
- Jeldres, R.I.; Forbes, L.; Cisternas, L.A. Effect of Seawater on Sulfide Ore Flotation: A Review. Miner. Process. Extr. Metall. Rev. 2016, 37, 369–384. [Google Scholar] [CrossRef]
- Niu, X.; Ruan, R.; Tan, Q.; Jia, Y.; Sun, H. Study on the second stage of chalcocite leaching in column with redox potential control and its implications. Hydrometallurgy 2015, 155, 141–152. [Google Scholar] [CrossRef]
- Petersen, J.; Dixon, D. Principles, mechanisms and dynamics of chalcocite heap bioleaching. Microb. Process. Met. Sulfides 2007, 193–218. [Google Scholar]
- Miki, H.; Nicol, M.; Velásquez-Yévenes, L. The kinetics of dissolution of synthetic covellite, chalcocite and digenite in dilute chloride solutions at ambient temperatures. Hydrometallurgy 2011, 105, 321–327. [Google Scholar] [CrossRef]
- Ruan, R.; Zou, G.; Zhong, S.; Wu, Z.; Chan, B.; Wang, D. Why Zijinshan copper bioheapleaching plant works efficiently at low microbial activity-Study on leaching kinetics of copper sulfides and its implications. Miner. Eng. 2013, 48, 36–43. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Lawson, F. The kinetics of leaching chalcocite in acidic oxygenated sulphate-chloride solutions. Hydrometallurgy 1991, 27, 249–268. [Google Scholar] [CrossRef]
- Herreros, O.; Viñals, J. Leaching of sulfide copper ore in a NaCl-H2SO4-O2 media with acid pre-treatment. Hydrometallurgy 2007, 89, 260–268. [Google Scholar] [CrossRef]
- Ruiz, M.C.; Abarzúa, E.; Padilla, R. Oxygen pressure leaching of white metal. Hydrometallurgy 2007, 86, 131–139. [Google Scholar] [CrossRef]
- Toro, N.; Pérez, K.; Saldaña, M.; Jeldres, R.I.; Jeldres, M.; Cánovas, M. Dissolution of pure chalcopyrite with manganese nodules and waste water. J. Mater. Res. Technol. 2019, 9, 798–805. [Google Scholar] [CrossRef]
- Toro, N.; Briceño, W.; Pérez, K.; Cánovas, M.; Trigueros, E.; Sepúlveda, R.; Hernández, P. Leaching of Pure Chalcocite in a Chloride Media Using Sea Water and Waste Water. Metals 2019, 9, 780. [Google Scholar] [CrossRef]
- Nicol, M.; Basson, P. The anodic behaviour of covellite in chloride solutions. Hydrometallurgy 2017, 172, 60–68. [Google Scholar] [CrossRef]
- Dutrizac, J.E. The leaching of sulphide minerals in chloride media. Hydrometallurgy 1992, 29, 1–45. [Google Scholar] [CrossRef]
- Senanayake, G. Chloride assisted leaching of chalcocite by oxygenated sulphuric acid via Cu(II)-OH-Cl. Miner. Eng. 2007, 20, 1075–1088. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Quezada-Reyes, V. Influence of seawater and discard brine on the dissolution of copper ore and copper concentrate. Hydrometallurgy 2018, 180, 88–95. [Google Scholar] [CrossRef]
- Quezada, V.; Velásquez, L.; Roca, A.; Benavente, O.; Melo, E.; Keith, B. Effect of curing time on the dissolution of a secondary copper sulphide ore using alternative water resources. IOP Conf. Ser. Mater. Sci. Eng. 2018, 427, 26–29. [Google Scholar] [CrossRef]
- Herreros, O. Dissolution kinetics of copper, white metal and natural chalcocite in Cl2-Cl y media. Metals 1999. [Google Scholar] [CrossRef]
- Fisher, W.W.; Flores, F.A.; Henderson, J.A. Comparison of chalcocite dissolution in the oxygenated, aqueous sulfate and chloride systems. Miner. Eng. 1992, 5, 817–834. [Google Scholar] [CrossRef]

| Investigation | Leaching Agent | Temperature Range (°C) | Reference | Max Cu Extraction (%) |
|---|---|---|---|---|
| The kinetics of leaching chalcocite (synthetic) in acidic oxygenated sulphate-chloride solutions | NaCl, H2SO4, HCl, HNO3 and Fe3+ | 65–94 | [15] | 97 |
| Leaching of pure chalcocite in a chloride media using sea water and waste water | NaCl, H2SO4 and Cl- from waste water | 25 | [19] | 68 |
| Leaching of sulfide copper ore in a NaCl–H2SO4–O2 media with acid pre-treatment | NaCl and H2SO4 | 20 | [16] | 78 |
| The kinetics of dissolution of synthetic covellite, chalcocite and digenite in dilute chloride solutions at ambient temperatures | HCl, Cu2+ and Fe3+ | 35 | [13] | 95 |
| Component | Cu | S |
| Mass (%) | 79.83 | 20.17 |
| Compound | Concentration (g/L) |
|---|---|
| Fluorine (F−) | 0.01 |
| Calcium (Ca2+) | 0.80 |
| Magnesium (Mg2+) | 2.65 |
| Bicarbonate (HCO3-) | 1.10 |
| Chloride (Cl-) | 39.16 |
| Calcium carbonate (CaCO3) | 13.00 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez, K.; I. Jeldres, R.; Nieto, S.; Salinas-Rodríguez, E.; Robles, P.; Quezada, V.; Hernández-Ávila, J.; Toro, N. Leaching of Pure Chalcocite in a Chloride Media Using Waste Water at High Temperature. Metals 2020, 10, 384. https://doi.org/10.3390/met10030384
Pérez K, I. Jeldres R, Nieto S, Salinas-Rodríguez E, Robles P, Quezada V, Hernández-Ávila J, Toro N. Leaching of Pure Chalcocite in a Chloride Media Using Waste Water at High Temperature. Metals. 2020; 10(3):384. https://doi.org/10.3390/met10030384
Chicago/Turabian StylePérez, Kevin, Ricardo I. Jeldres, Steven Nieto, Eleazar Salinas-Rodríguez, Pedro Robles, Víctor Quezada, Juan Hernández-Ávila, and Norman Toro. 2020. "Leaching of Pure Chalcocite in a Chloride Media Using Waste Water at High Temperature" Metals 10, no. 3: 384. https://doi.org/10.3390/met10030384
APA StylePérez, K., I. Jeldres, R., Nieto, S., Salinas-Rodríguez, E., Robles, P., Quezada, V., Hernández-Ávila, J., & Toro, N. (2020). Leaching of Pure Chalcocite in a Chloride Media Using Waste Water at High Temperature. Metals, 10(3), 384. https://doi.org/10.3390/met10030384









