Renewable Hydrogen Production Processes for the Off-Gas Valorization in Integrated Steelworks through Hydrogen Intensified Methane and Methanol Syntheses
Abstract
1. Introduction
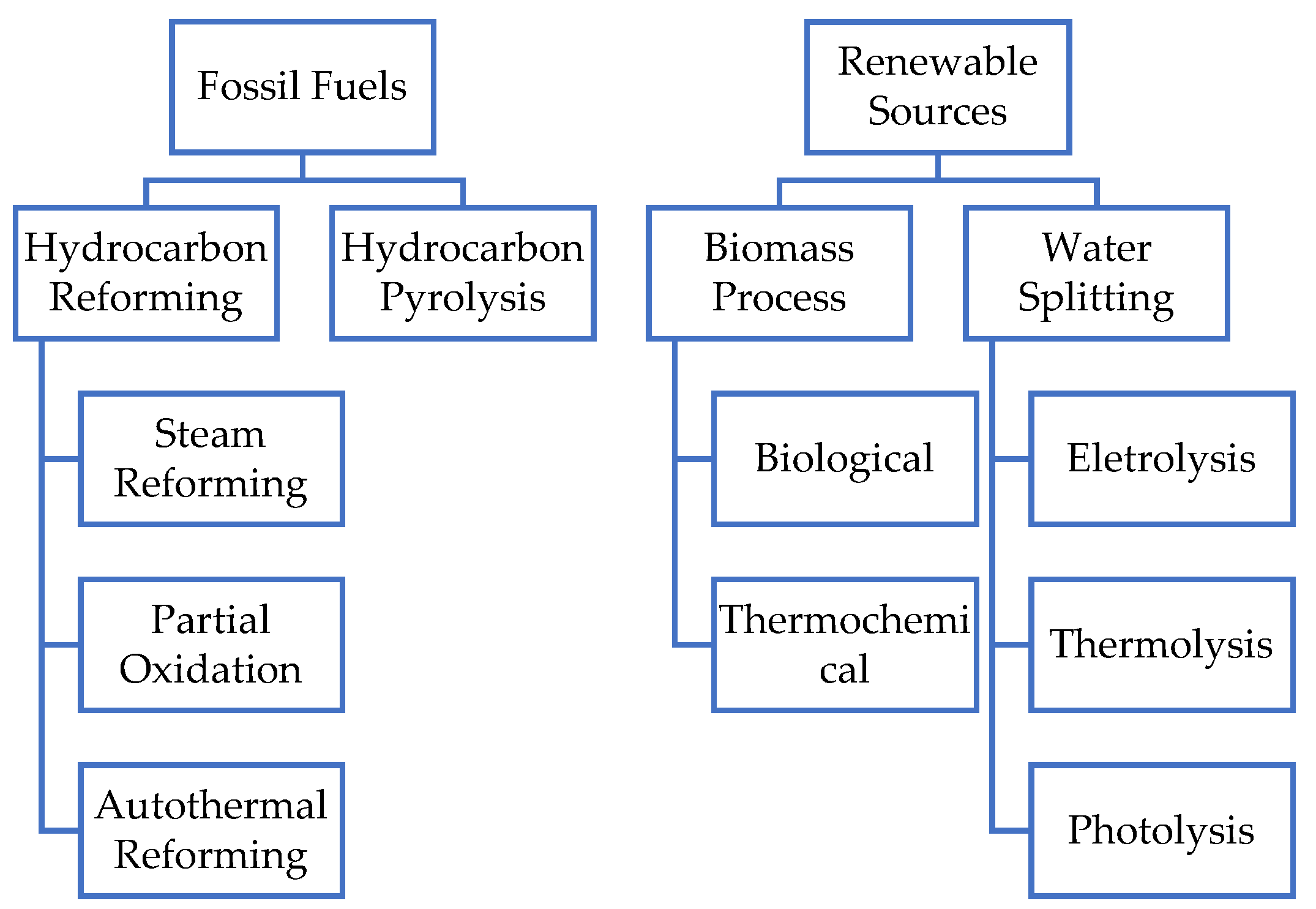
2. Sustainable Hydrogen Production
2.1. Electrolysis
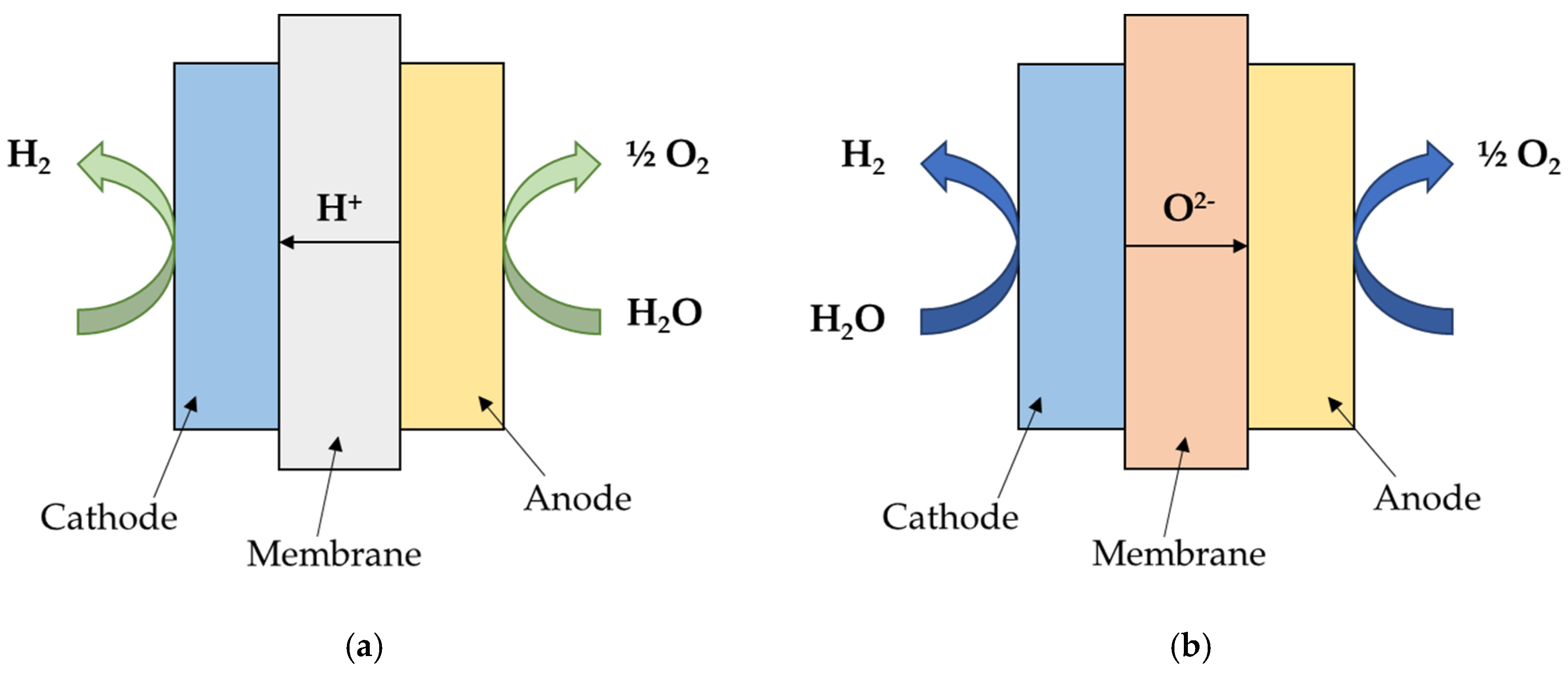
2.1.1. PEM Electrolysis
2.1.2. SOEC Electrolysis
2.1.3. Hydrogen Production from Biomass
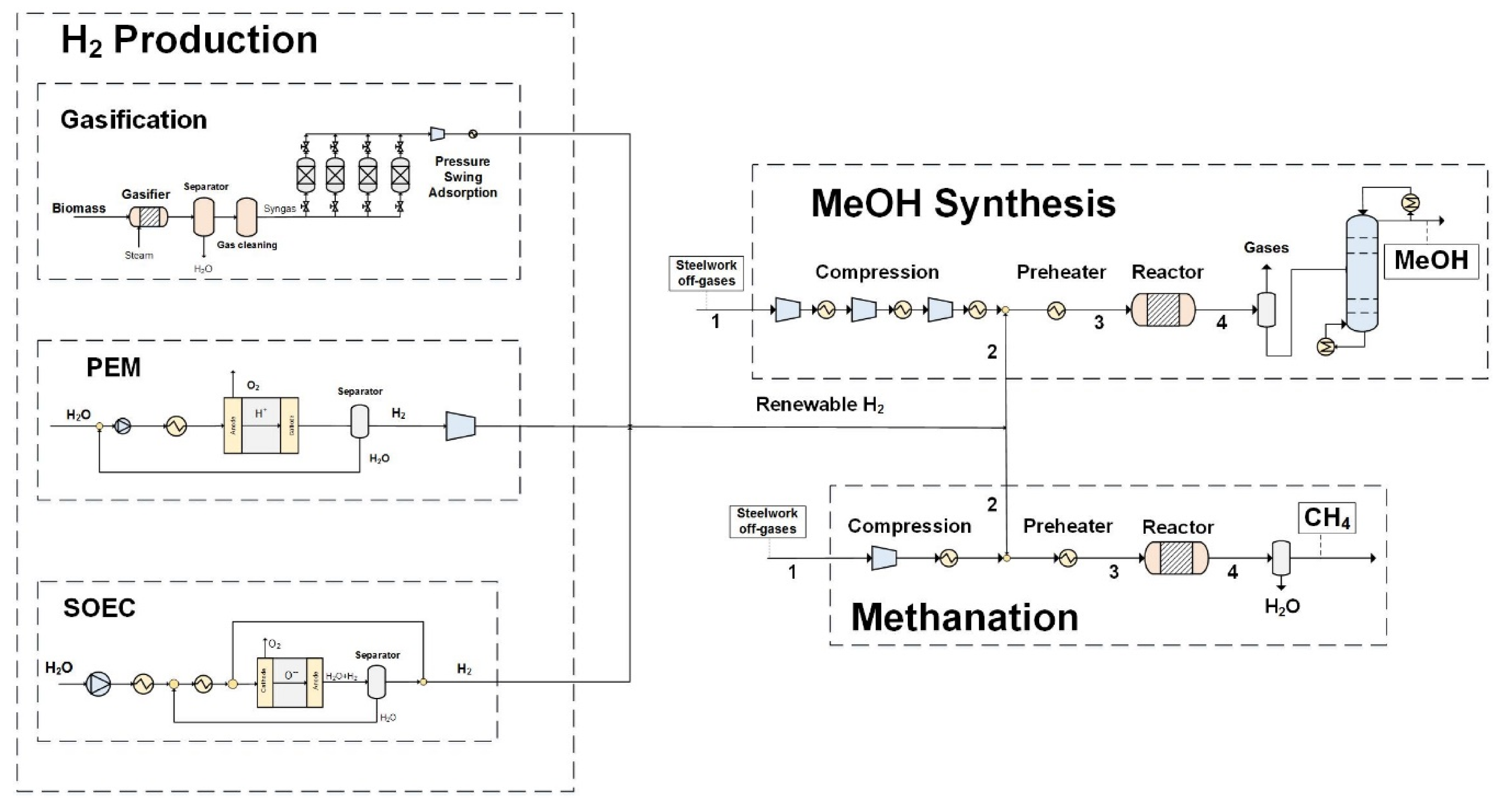
2.2. Integration of Hydrogen Production with Methane and Methanol Synthesis
- BFG: CO 23%, H2 4.5%, CO2 21%, N2 51%;
- BOFG: CO 60.9%, H2 4.3%, CO2 17.2%, N2 15.5%, CH4 0.1%; and
- COG: CO 4.6%, H2 48.9%, CO2 3.2%, N2 3.75%, CH4 21.35%, CxHy 1.9% [16].
3. Materials and Methods
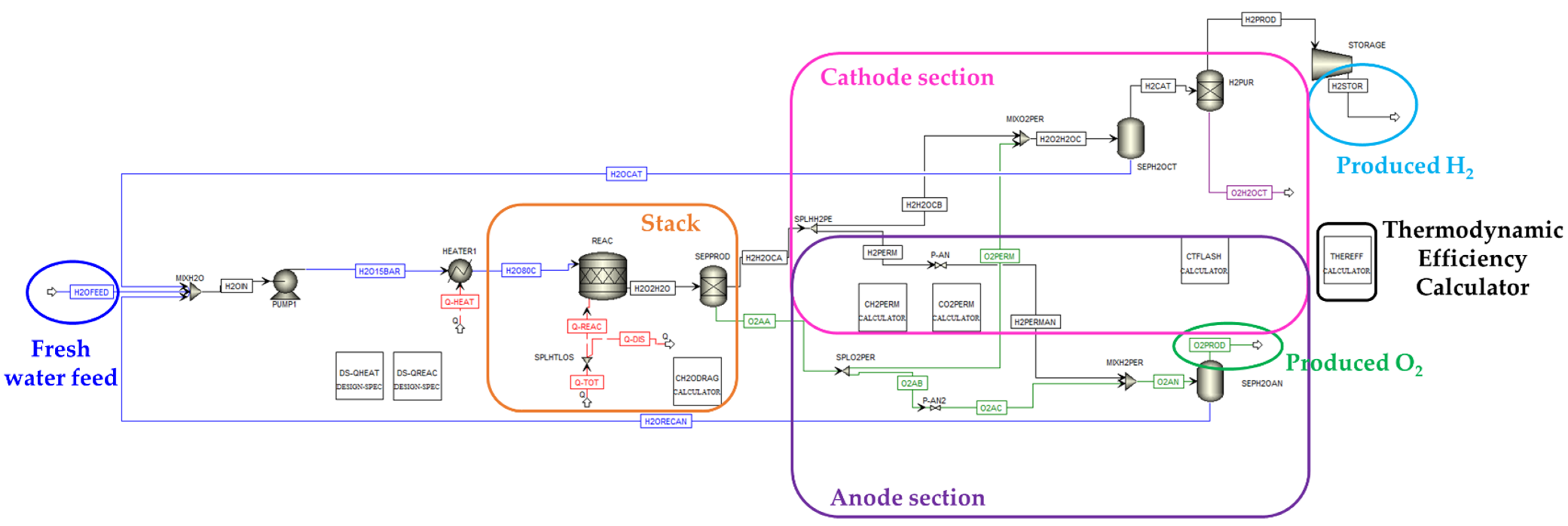
3.1. PEM Electrolysis
- The flash unit “SEPH2OCT” removes the dragged water, that is recovered and recycled at the inlet of the PEM;
- the block “H2PUR” represents the last separation step that purifies the hydrogen stream from the remaining water and from the oxygen traces that is permeated from the anode (the molar fraction composition of stream “H2CAT” before the purification is: H2 = 0.951, H2O = 0.048, O2 = 0.001).
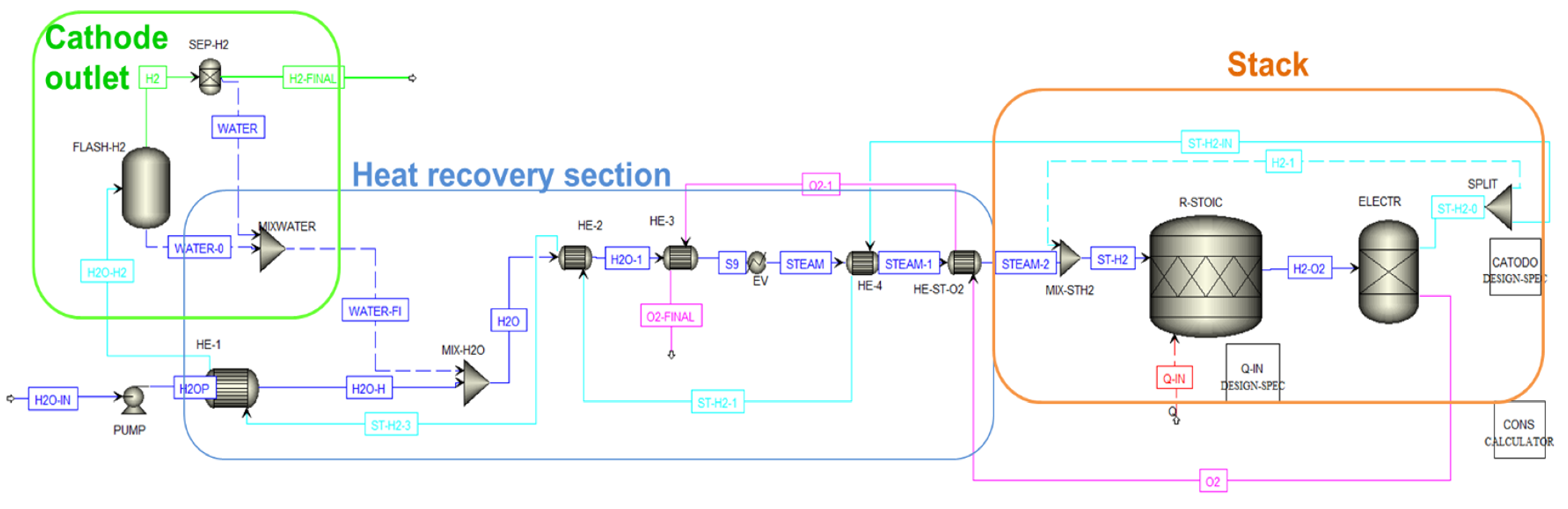
3.2. SOEC Electrolysis
- perfect thermal insulation on the stack is considered and heat losses toward the environment are neglected [54].
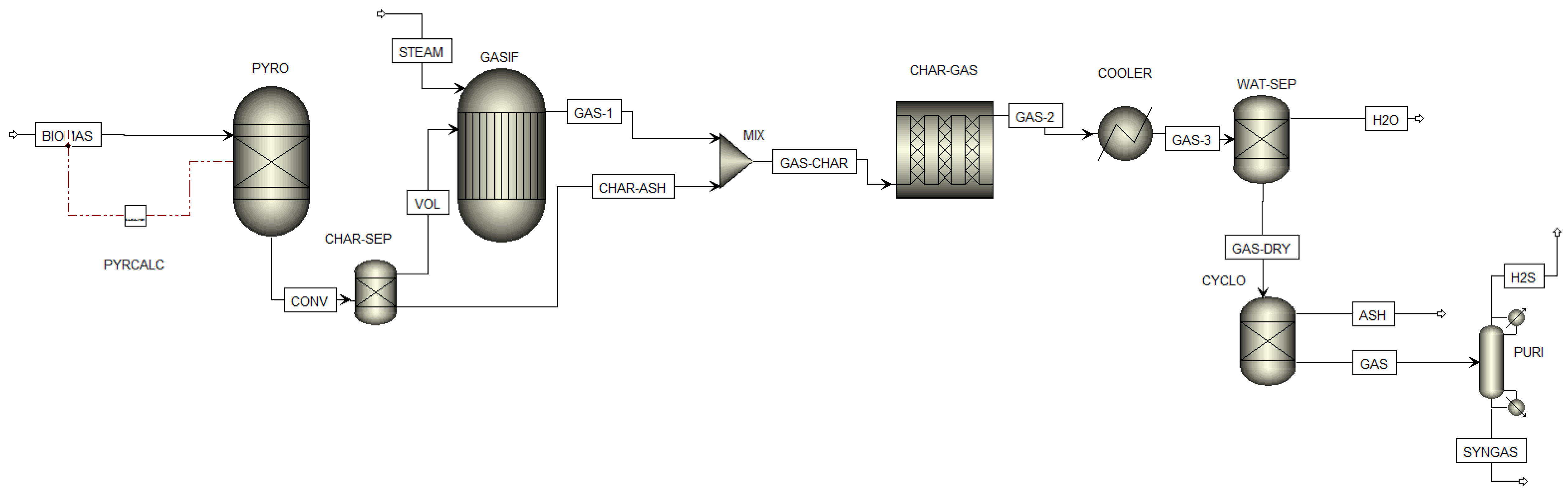
3.3. Biomass Gasification
- The process is considered steady state and isothermal;
- the operations occur at atmospheric pressure (~1 bar);
- the pressure and temperature are considered uniform inside the gasifier;
- the heat losses occurring in the gasifier are neglected;
- the ash and tar formations are neglected: Char has been simplified as pure carbon;
- sulfur (S) is converted to H2S; and
3.4. Integration of Hydrogen in Methanol and Methane Synthesis
4. Results and Discussion
4.1. Hydrogen Production Model Results
- In the case of PEM, 72.20% of the power is required for electrolysis, 26.21% are dissipations, 0.75% is needed for water heating, 0.03% for pumping and 0.81% for H2 compression;
- for SOEC, 84.98% of the power is required from the stack, 15.00% is needed for steam generation and the remaining 0.02% for pumping.
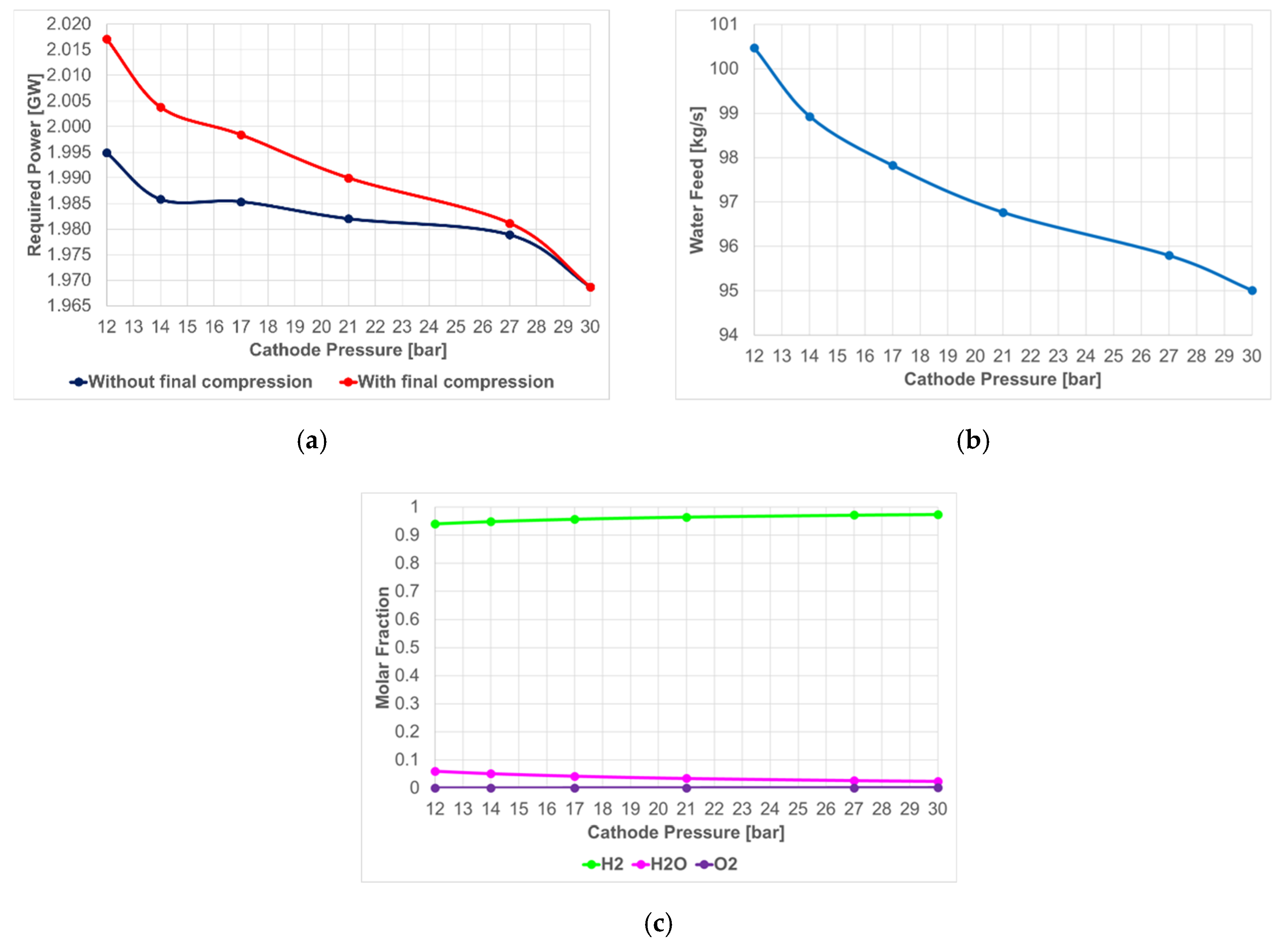
4.1.1. PEM Electrolysis
- Required power (before and after the final compression to obtain H2 stored at 30 bar);
- water feed; and
- composition of gas stream produced in the cathode section before the final purification step.
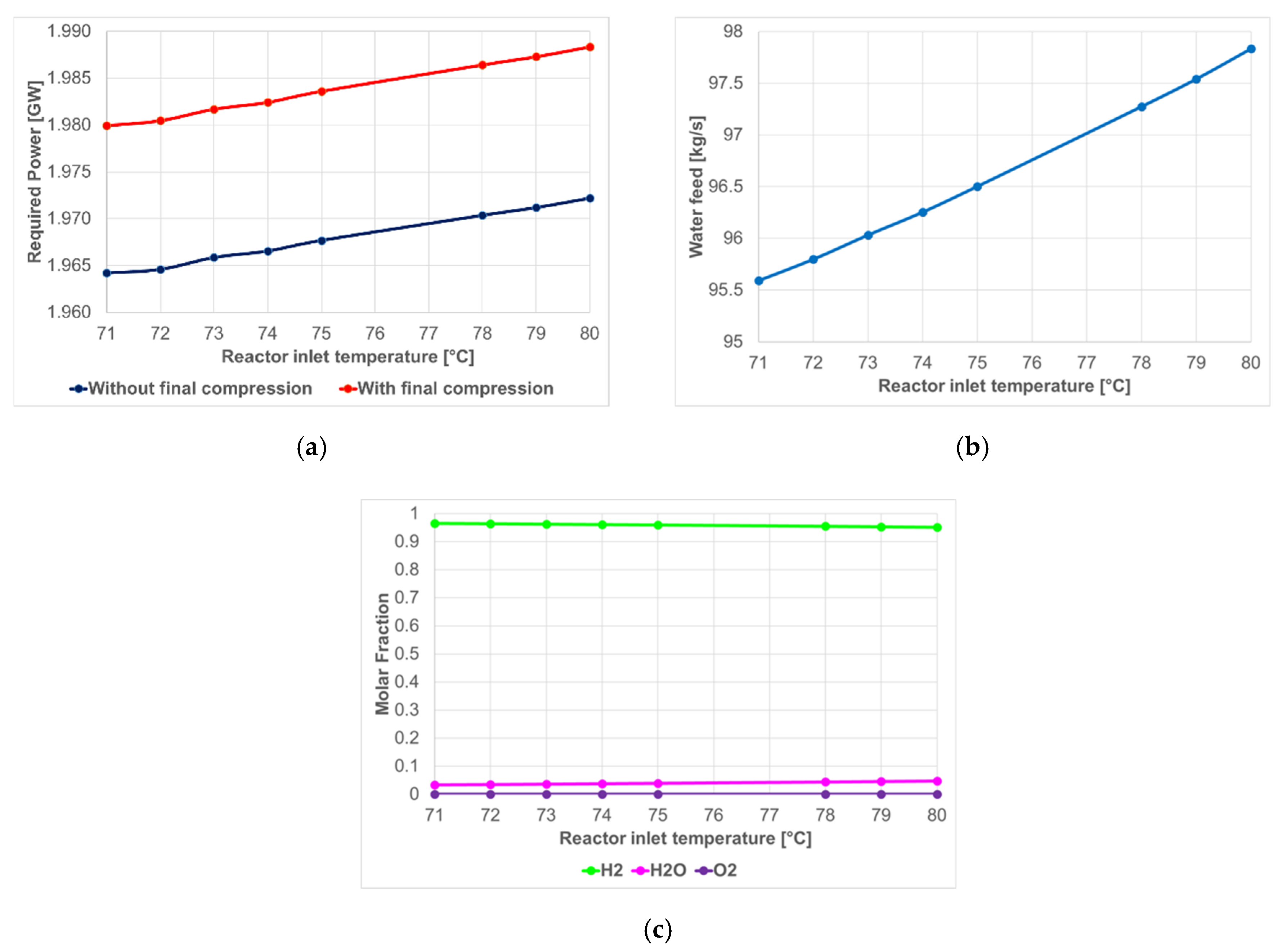
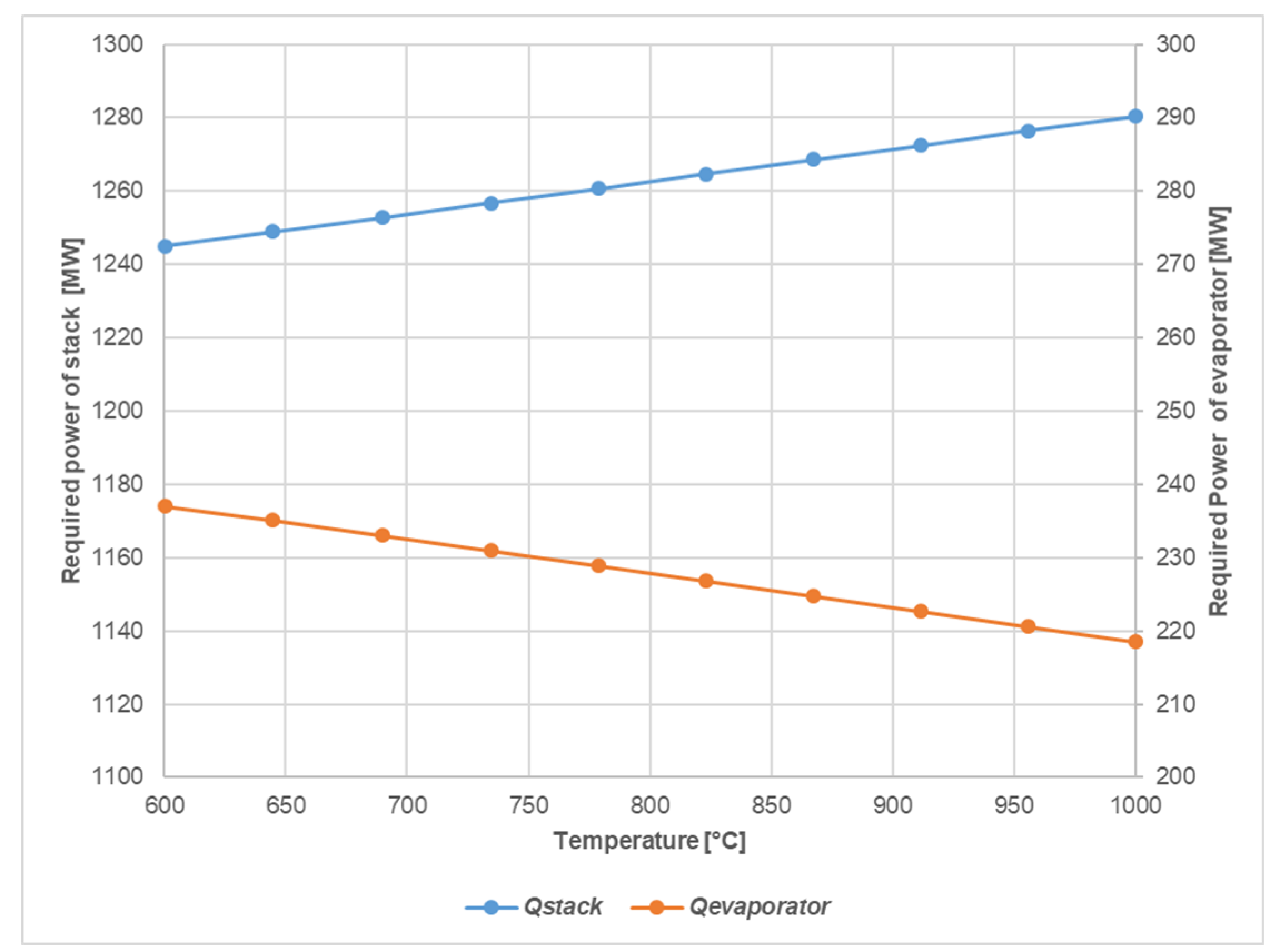
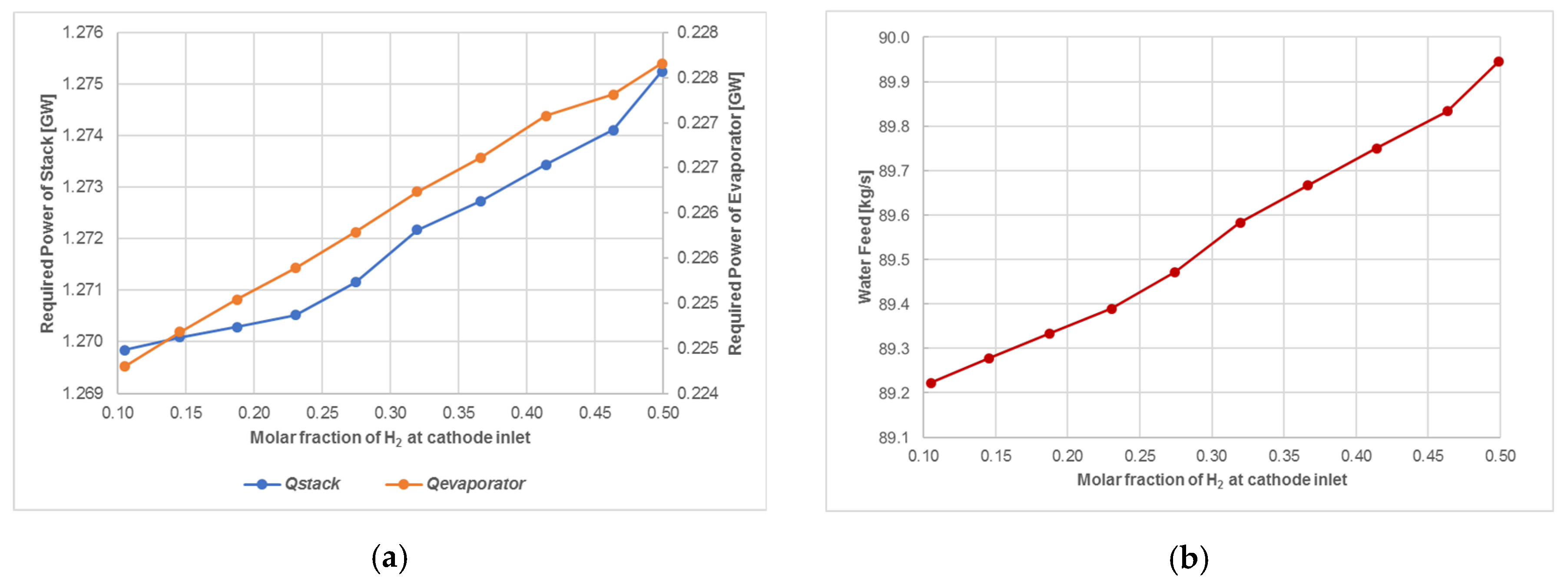
4.1.2. SOEC Electrolysis
4.1.3. Biomass Gasification
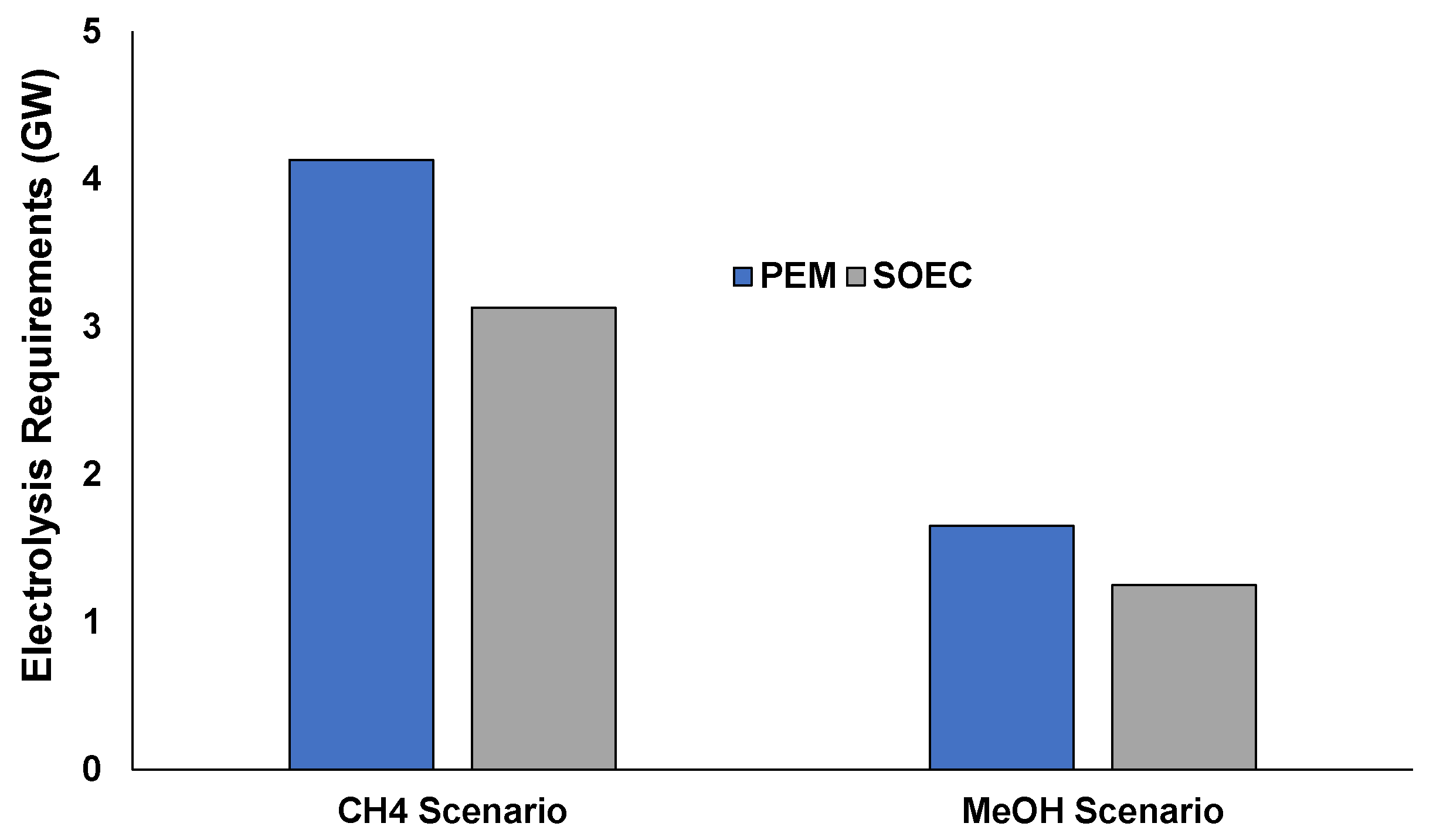
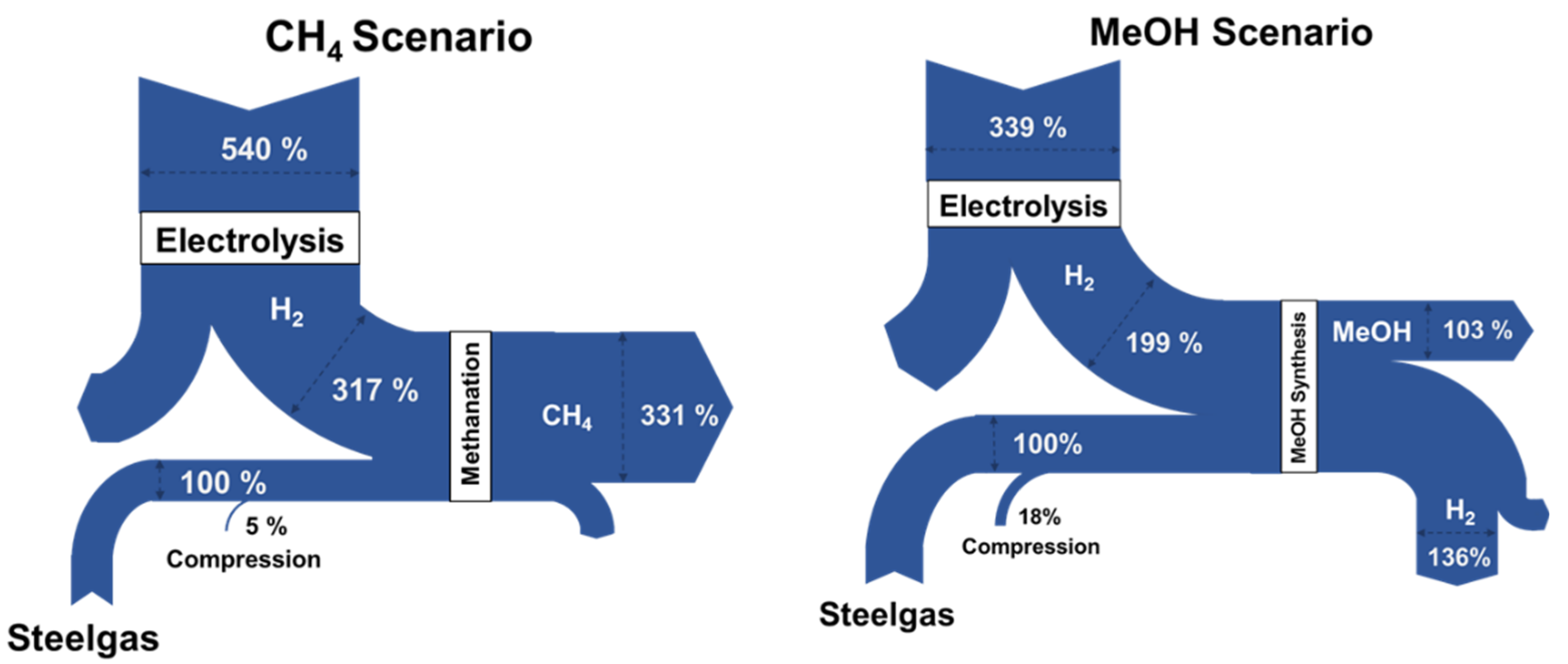
4.2. Integration of Hydrogen in Methanol and Methane Synthesis
- CH4 scenario: 100% exploitation of by-product gases produced in an integrated steelmaking plant for generation of methane: In this scenario the entire available amount of off-gases is used to produce methane (assuming that the vast natural gas market and transporting infrastructure can uptake easily such production). The renewable hydrogen produced either by PEM or SOEC is inserted in basic stoichiometric ratio to produce methane.
- MeOH scenario: Methanol synthesis of a fraction of the by-product gases (~65%) due to the fact that MeOH is a product that has a limited market compared to the natural gas market and infrastructure. Again, the production of H2 is assumed to be coming from either PEM or SOEC.
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sohn, H. Energy Consumption and CO2 Emissions in Ironmaking and Development of a Novel Flash Technology. Metals 2020, 10, 54. [Google Scholar] [CrossRef]
- Development Organisation for Economic Co-Operation (OECD). Energy Efficiency in the Steel Sector: Why It Works Well, But Not Always. 2015. Available online: http://www.oecd.org/sti/ind/Energy-efficiency-steel-sector-1.pdf (accessed on 28 September 2020).
- Baumert, K.; Herzog, T.; Pershing, J. Navigating the Numbers: Greenhouse Gas Data and International Climate Policy. World Resources Institute Report Washington 2005. Available online: www.oecd.org/dataoecd/28/43/36448807.pdf (accessed on 28 July 2020).
- Worldsteel. Steel’s Contribution to a Low Carbon Future: Worldsteel Position Paper. Available online: https://www.worldsteel.org/en/dam/jcr:7ec64bc1-c51c-439b-84b8-94496686b8c6/Position_paper_climate_2020_vfinal.pdf (accessed on 19 June 2020).
- Worldsteel Association. Fact Sheet: Energy Use in the Steel Industry. Available online: https://www.worldsteel.org/en/dam/jcr:f07b864c-908e-4229-9f92-669f1c3abf4c/fact_energy_2019.pdf (accessed on 19 June 2020).
- International Energy Agency. Energy Technologies Perspective 2017: Catalysing Energy Technology Transformations. Available online: https://www.oecd-ilibrary.org/energy/energy-technology-perspectives-2017_energy_tech-2017-en (accessed on 19 June 2020).
- Worldsteel Association. Energy Use in the Steel Industry Report Available Now. Available online: https://www.worldsteel.org/media-centre/press-releases/2015/energy-use-in-the-steel-industry-report-available-now.html (accessed on 22 June 2020).
- American Iron and Steel Institute. A New Roadmap for Transformation of Steelmaking Processes. Saving One Barrel of Oil Per Ton [SOBOT]. Available online: https://steel.org/~/media/Files/AISI/Public%20Policy/saving_one_barrel_oil_per_ton.pdf (accessed on 22 June 2020).
- Ki-Hoon, L. Drivers and Barriers to Energy Efficiency Management for Sustainable Development. Sustain. Dev. 2014, 23, 16–25. [Google Scholar]
- United Nation. Paris Agreement 2015. Available online: https://unfccc.int/files/essential_background/convention/application/pdf/english_paris_agreement.pdf (accessed on 22 June 2020).
- Matino, I.; Colla, V.; Romaniello, L.; Rosito, F.; Portulano, L. Simulation Techniques for an Efficient Use of Resources: An Overview for the Steelmaking Field. In Proceedings of the World Congress on Sustainable Technologies, London, UK, 14–16 December 2015. [Google Scholar]
- Matino, I.; Colla, V.; Colucci, V.; Lamia, P.; Baragiola, S.; Cecca, C.D. Improving Sustainability of Electric Steelworks through Process Simulations. CEt 2016, 52, 763–768. [Google Scholar]
- Florens, F.; Rübbelke, D.; Vögele, S. An analysis of the economic determinants of energy efficiency in the European iron and steel industry. J. Clean. Prod. 2015, 104, 250–263. [Google Scholar]
- Branca, T.; Colla, V.; Algermissen, D.; Granbom, H.; Martini, U.; Morillon, A.; Pietruck, R.; Rosendahl, S. Reuse and Recycling of By-Products in the Steel Sector: Recent Achievements Paving the Way to Circular Economy and Industrial Symbiosis in Europe. Metals 2020, 10, 345. [Google Scholar] [CrossRef]
- Conejo, A.; Birat, J.-P.; Dutta, A. A review of the current environmental challenges of the steel industry and its value chain. J. Environ. Manag. 2020, 259, 109782. [Google Scholar] [CrossRef]
- Remus, R.; Aguado-Monsonet, M.; Roudier, S.; Delgado-Sancho, L. Best Available Techniques (BAT) Reference Document for Iron and Steel Production. Industrial Emissions Directive 2010/75/EU (Integrated Pollution Prevention and Control). 2013. Available online: http://publications.jrc.ec.europa.eu/repository/bitstream/JRC69967/lfna25521enn.pdf (accessed on 30 September 2020).
- Matino, I.; Dettori, S.; Colla, V.; Weber, V.; Salame, S. Forecasting blast furnace gas production and demand through echo state neural network-based models: Pave the way to off-gas optimized management. Appl. Energy 2019, 253, 113578. [Google Scholar] [CrossRef]
- Maddaloni, A.; Matino, R.; Matino, I.; Dettori, S.; Zaccara, A.; Colla, V. A Quadratic Programming Model for the Optimization of Off-Gas Network in Integrated Steelworks. Matériaux Tech. 2020, 107, 5. [Google Scholar] [CrossRef]
- Porzio, G.; Fornai, B.; Amato, A.; Matarese, N.; Vannucci, M.; Chiappelli, L.; Colla, V. Reducing the energy consumption and CO2 emissions of energy intensive industries through decision support systems - An example of application to the steel industry. Appl. Energy 2013, 112, 818–833. [Google Scholar] [CrossRef]
- Maddaloni, A.; Porzio, G.; Nastasi, G.; Colla, V.; Branca, T. Multi-objective optimization applied to retrofit analysis: A case study for the iron and steel industry. Appl. Therm. Eng. 2015, 91, 638–646. [Google Scholar] [CrossRef]
- Zhao, X.; Bai, H.; Lu, X.; Shi, Q.; Han, J. A MILP model concerning the optimisation of penalty factors for the short-term distribution of byproduct gases produced in the iron and steel making process. Appl. Energy 2015, 148, 142–158. [Google Scholar] [CrossRef]
- Kong, H.; Qi, E.; Li, H.; Li, G.; Zhang, X. A MILP model for optimization of by-product gases in the integrated iron and steel plant. Appl. Energy 2009, 87, 2156–2163. [Google Scholar] [CrossRef]
- Porzio, G.; Nastasi, G.; Colla, V.; Vannucci, M.; Branca, T. Comparison of multi-objective optimization techniques applied to off-gas management within an integrated steelwork. Appl. Energy 2014, 136, 1085–1097. [Google Scholar] [CrossRef]
- de Oliveira Junior, V.B.; Pena, J.C.; Salles, J.F. An improved plant-wide multiperiod optimization model of a byproduct gas supply system in the iron and steel-making process. Appl. Energy 2016, 164, 462–474. [Google Scholar] [CrossRef]
- Colla, V.; Matino, I.; Dettori, S.; Petrucciani, A.; Zaccara, A.; Weber, V. Assessing the efficiency of the off-gas network management in integrated steelworks. Mater. Tech. 2019, 107, 104. [Google Scholar] [CrossRef]
- Maruoka, N.; Akiyama, T. Exergy recovery from steelmaking off-gas by latent heat storage for methanol production. Energy 2006, 31, 1632–1642. [Google Scholar] [CrossRef]
- Uribe-Soto, W.; Portha, J.; Commenge, J.; Falk, L. A review of thermochemical processes and technologies to use steelworks off-gases. Renew. Sustain. Energy Rev. 2017, 74, 809–823. [Google Scholar] [CrossRef]
- Dreillarda, M.; Broutina, P.; Huarda, T.; Lettat, A. Application of the DMXTM CO2 Capture Process in Steel Industry. Energy Procedia 2017, 114, 2573–2589. [Google Scholar] [CrossRef]
- Deerberg, G.; Oles, M.; Schlögl, R. The Project Carbon2Chem®. Chem. Ing. Tech. 2018, 90, 1365–1368. [Google Scholar] [CrossRef]
- Mission Innovation Austria. Austria’s Way into the Future of Energy: Strategies and Success Stories. 2019. Available online: http://mission-innovation.net/wp-content/uploads/2019/05/MI-Austrias-Way-Into-the-Future-of-Energy-May-2019.pdf (accessed on 30 September 2020).
- Kim, D.; Han, J. Techno-economic and climate impact analysis of carbon utilization process for methanol production from blast furnace gas over Cu/ZnO/Al2O3 catalyst. Energy 2020, 198, 117355. [Google Scholar] [CrossRef]
- Gao, R.; Zhang, C.; Kwakc, G.; Lee, Y.-J.; Kang, S.C.; Guan, G. Techno-economic evaluation of methanol production using by-product gases from iron and steel works. Energy Convers. Manag. 2020, 213, 112819. [Google Scholar] [CrossRef]
- Shin, S.; Lee, J.-K.; Lee, I.-B. Development and techno-economic study of methanol production from coke-oven gas blended with Linz Donawitz gas. Energy 2020, 200, 117506. [Google Scholar] [CrossRef]
- Nikolaidis, P.; Poullikkas, A. A comparative overview of hydrogen production processes. Renew. Sustain. Energy Rev. 2017, 67, 597–611. [Google Scholar] [CrossRef]
- Hajjaji, N.; Pons, M.-N.; Houas, A.; Renaudin, V. Exergy analysis: An efficient tool for understanding and improving hydrogen production via the steam methane reforming process. Energy Policy 2012, 42, 392–399. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I. Comparative assessment of hydrogen production methods from renewable and non-renewable sources. Int. J. Hydrogen Energy 2014, 39, 1–12. [Google Scholar] [CrossRef]
- Dincer, I.; Acar, C. Review and evaluation of hydrogen production methods for better sustainability. Int. J. Hydrogen Energy 2015, 40, 11094–11111. [Google Scholar] [CrossRef]
- Joshi, A.S.; Dincer, I.; Reddy, B.V. Solar hydrogen production: A comparative performance assessment. Int. J. Hydrogen Energy 2011, 36, 1124–1257. [Google Scholar] [CrossRef]
- Khosravi, A.; Koury, R.; Machado, L.; Pabon, J. Energy, exergy and economic analysis of a hybrid renewable energy with hydrogen storage system. Energy 2018, 148, 1087–1102. [Google Scholar] [CrossRef]
- Bicakova, O.; Straka, P. Production of hydrogen from renewable resources and its effectiveness. Int. J. Hydrogen Energy 2012, 37, 1156–11578. [Google Scholar] [CrossRef]
- Ozbilen, A.; Dincer, I.; Rosen, M.A. Comparative environmental impact and efficiency assessment of selected hydrogen production methods. Environ. Impact Assess. Rev. 2013, 42, 1–9. [Google Scholar] [CrossRef]
- Azwar, M.; Hussain, M.; Abdul-Wahab, A. Development of biohydrogen production by photobiological, fermentation and electrochemical processes: A review. Renew. Sustain. Energy Rev. 2014, 31, 158–173. [Google Scholar] [CrossRef]
- Balat, H.; Kirtay, E. Hydrogen from biomass—Present scenario and future prospects. Int. J. Hydrogen Energy 2010, 35, 7416–7426. [Google Scholar] [CrossRef]
- Kotay, S.M.; Das, D. Biohydrogen as a renewable energy Resource-Prospects and potentials. Int. J. Hydrogen Energy 2008, 33, 258–263. [Google Scholar]
- Cai, Q.; Luna-Ortiz, E.; Adjiman, C.S.; Brandon, N.P. The Effects of Operating Conditions on the Performance of a Solid Oxide Steam Electrolyser: A Moldel-Based Study. Fuel Cells 2010, 10, 1114–1128. [Google Scholar] [CrossRef]
- Amikam, G.; Nativ, P.; Gendel, Y. Chlorine-free alkaline seawater electrolysis for hydrogen production. Int. J. Hydrogen Energy 2018, 43, 6504–6514. [Google Scholar] [CrossRef]
- Sellamia, M.; Loudiyi, K. Electrolytes behaviour during hydrogen production by solar energy. Renew. Sustain. Energy Rev. 2017, 70, 1331–1335. [Google Scholar] [CrossRef]
- Chakik, F.E.; Kaddami, M.; Mikou, M. Effect of operating parameters on hydrogen production by electrolysis of water. Int. J. Hydrogen Energy 2017, 42, 25550–25557. [Google Scholar] [CrossRef]
- Zeng, K.; Zhang, D. Recent progress in alkaline water electrolysis for hydrogen production and applications. Prog. Energy Combust. Sci. 2010, 36, 307–326. [Google Scholar] [CrossRef]
- Bhandari, R.; Trudewind, C.A.; Zapp, P. Life Cycle Assessment of Hydrogen Production via Electrolysis – A review. J. Clean. Prod. 2013, 85, 151–163. [Google Scholar] [CrossRef]
- Vincent, I.; Bessarabov, D. Low cost hydrogen production by anion exchange membrane electrolysis: A review. Renew. Sustain. Energy Rev. 2018, 81, 1690–1704. [Google Scholar] [CrossRef]
- de Araujo, F.H.M.; Braga, L.B.; Colombaroli, T.S.; Pedroso, D.T.; da Silva, M.E.; Silveira, J.; Tapia, L.C.F.; Tuna, C.E.; Vane, L.F.; Vigouroux, R.Z. Sustainable Hydrogen Production Processes, 1st ed.; José, L.S., Ed.; Springer: Sao Paolo, Brazil, 2017. [Google Scholar]
- Ursua, A.; Gandia, L.M.; Sanchis, P. Hydrogen Production from Water Electrolysis: Current Status and Future Trends. Proc. IEEE 2011, 100, 410–426. [Google Scholar] [CrossRef]
- Ferrero, D.; Lanzini, A.; Santarelli, M.; Leone, P. A comparative assessment on hydrogen production from low- and high-temperature electrolysis. Int. J. Hydrogen Energy 2013, 38, 3523–3536. [Google Scholar] [CrossRef]
- Mehmeti, A.; Angelis-Dimakis, A.; Arampatzis, G.S.; McPhail, J.; Ulgiati, S. Life Cycle Assessment and Water Footprint of Hydrogen Production Methods: From Conventional to Emerging Technologies. Enviroments 2018, 5, 24. [Google Scholar] [CrossRef]
- Medina, P.; Santarelli, M. Analysis of water transport in a high pressure PEM electrolyzer. Int. J. Hydrogen Energy 2010, 35, 5173–5186. [Google Scholar] [CrossRef]
- Barbir, F. PEM electrolysis for production of hydrogen from renewable energy sources. J. Sol. Energy 2005, 78, 661–669. [Google Scholar] [CrossRef]
- The World’s Most Efficient and Reliable Electrolyser. Available online: www.nelhydrogen.com (accessed on 15 July 2020).
- Hydrogen Generation HyLYZER® PEM Electrolysis Technology. Available online: www.hydrogenics.com (accessed on 26 May 2020).
- Profit Generators. HOGEN® On-Site Hydrogen Generation Systems. Available online: www.linde-gas.com (accessed on 26 May 2020).
- PEMELECTROLYSERS: Today’s Flexible&Cost Effective Technology. Available online: www.arevah2gen.com (accessed on 26 May 2020).
- Im-orb, K.; Visitdumrongkul, N.; Seabea, D.; Patcharavorachot, Y.; Arpornwichanop, A. Flowsheet-based model and exergy analysis of solid oxide electrolysis cells for clean hydrogen production. J. Clean. Prod. 2018, 170, 1–13. [Google Scholar] [CrossRef]
- KOH, J.; YOON, D.; OH, C.H. Simple Electrolyzer Model Development for High-Temperature Electrolysis System Analysis Using Solid Oxide Electrolysis Cell. J. Nucl. Sci. Technol. 2010, 47, 599–607. [Google Scholar] [CrossRef]
- Rivera-Tinoco, R.; Farran, M.; Bouallou, C.; Aupretre, F.; Valentin, S.; Millet, P.; Ngameni, J. Investigation of power-to-methanol processes coupling electrolytic hydrogen production and catalytic CO2 reduction. Int. J. Hydrogen Energy 2016, 41, 4546–4559. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Hydrogen Production: Biomass Gasification. Available online: https://www.energy.gov/eere/fuelcells/hydrogen-production-biomass-gasification (accessed on 26 July 2020).
- Parthasarathy, P.; Narayanan, K.S. Hydrogen production from steam gasification of biomass: Influence of process parameters on hydrogen yield—A review. J. Renew. Energy 2014, 66, 570–579. [Google Scholar] [CrossRef]
- Neubert, M.; Hauser, A.; Pourhossein, B.; Dillig, M.; Karl, J. Experimental evaluation of a heat pipe cooled structured reactor as part of a two-stage catalytic methanation process in power-to-gas applications. Appl. Energy 2018, 229, 289–298. [Google Scholar] [CrossRef]
- Leimert, J.M.; Neubert, M.; Treiber, P.; Dillig, M.; Karl, J. Combining the Heatpipe Reformer technology with hydrogen-intensified methanation for production of synthetic natural gas. Appl. Energy 2018, 217, 37–46. [Google Scholar] [CrossRef]
- Schildhauer, T.J.; Biollaz, S.M.A. Synthetic Natural Gas from Coal and Dry Biomass, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Koytsoumpa, E.I.; Karellas, S.; Kakaras, E. Modelling of Substitute Natural Gas production via combined gasification and power to fuel. J. Renew. Energy 2019, 135, 1354–1370. [Google Scholar] [CrossRef]
- Thema, M.; Weidlich, T.; Hörl, M.; Bellack, A.; Mörs, F.; Hackl, M.K.F.; Gleich, J.; Stabenau, C.; Huber, H.; Hafenbradl, D.; et al. Biological CO2-Methanation: An Approach to Standardization. Energies 2019, 12, 1670. [Google Scholar] [CrossRef]
- Razzaq, R.; Li, C.; Zhang, S. Coke oven gas: Availability, properties, purification, and utilization in China. Fuel 2013, 113, 287–299. [Google Scholar] [CrossRef]
- Bozzano, G.; Manenti, F. Efficient methanol synthesis: Perspectives, technologies and optimization strategies. Prog. Energy Combust. Sci. 2016, 56, 71–105. [Google Scholar] [CrossRef]
- Schittkowski, J.; Ruland, H.; Laudenschleger, D.; Girod, K.; Kahler, K.; Kaluza, S.; Muhler, M.; Schlogl, R. Methanol Synthesis from Steel Mill Exhaust Gases: Challenges for the Industrial Cu/ZnO/Al2O3 Catalyst. Chem. Ing. Tech. 2018, 90, 1419–1429. [Google Scholar] [CrossRef]
- Graaf, G.H.; Winkelman, J.G.M.; Stamhuis, E.J.; Beenackers, A.A.C.M. Kinetics of the three phase methanol synthesis. Chem. Eng. Sci. 1988, 43, 2161–2168. [Google Scholar] [CrossRef]
- Smolinka, T.; Gunther, M.; Garche, J. NOW-Studie: Stand und Entwicklungspotenzial der Wasserelektrolyse zur Herstellung von Wasserstoff aus Regenerativen Energien; Technical Report; Fraunhofer ISE: Freiburg, Germany, 2011. [Google Scholar]
- PEM Electrolyzer Types. Available online: https://ntrs.nasa.gov/search.jsp?R=20170009573 (accessed on 26 July 2020).
- Houaijia, A.; Breuer, S.; Thomey, D.; Brosig, C.; Säck, J.P.; Roeb, M.; Sattler, C. Solar Hydrogen by High-temperature Electrolysis: Flowsheeting and Experimental Analysis of a Tube-type Receiver Concept for Superheated Steam Production. Energy Procedia 2014, 49, 1960–1969. [Google Scholar] [CrossRef]
- Ioraa, P.; Chiesa, P. High efficiency process for the production of pure oxygen based on solid oxide fuel cell–solid oxide electrolyzer technology. J. Power Sources 2009, 190, 408–416. [Google Scholar] [CrossRef]
- Henke, M.; Hillius, S.; Riedel, M.; Kallo, J.; Friedrich, K.A. Gas recirculation at the hydrogen electrode of solid oxide fuel cell and solid oxide electrolysis cell systems. Fuel Cells 2016, 16, 584–590. [Google Scholar] [CrossRef]
- Bernadet, L.; Moncasi, C.; Torrell, M.; Tarancón, A. High-performing electrolyte-supported symmetrical solid oxide electrolysis cells operating under steam electrolysis and co-electrolysis modes. Int. J. Hydrogen Energy 2020, 45, 14208–14217. [Google Scholar] [CrossRef]
- Udagawa, J.; Aguiar, P.; Brandon, N. Hydrogen production through steam electrolysis: Control strategies for a cathode-supported intermediate temperature solid oxide electrolysis cell. J. Power Sources 2018, 180, 354–364. [Google Scholar] [CrossRef]
- Pala, L.P.R.; Wang, Q.; Kolb, G.; Hessel, V. Steam gasification of biomass with subsequent syngas adjustment using shift reaction for syngas production: An Aspen Plus model. J. Renew. Energy 2017, 101, 484–492. [Google Scholar] [CrossRef]
- Doherty, W.; Reynolds, A.; Kennedy, D. Aspen Plus Simulation of Biomass Gasification in a Steam Blown Dual Fluidised Bed. In Materials and Processes for Energy: Communicating Current Research and Technological Developments; Méndez-Vilas, A., Ed.; Formatex Research Centre: Badajoz, Spain, 2013. [Google Scholar]
- Begum, S.; Rasul, M.; Akbar, D.; Ramzan, N. Performance Analysis of an Integrated Fixed Bed Gasifier Model for Different Biomass Feedstocks. Energies 2013, 6, 6508–6524. [Google Scholar] [CrossRef]
- Nikoo, M.B.; Mahinpey, N. Simulation of biomass gasification in fluidized bed reactor using ASPEN PLUS. Biomass Bioenergy 2008, 32, 1245–1254. [Google Scholar] [CrossRef]
- Matsui, I.; Kunii, D.; Furusawa, T. Study of Fluidized Bed Steam Gasification of Char by Thermogravimetrically Obtained Kinetics. J. Chem. Eng. Jpn. 1985, 18, 105–113. [Google Scholar] [CrossRef]
- Fremaux, S.; Beheshti, S.; Ghassemi, H.; Shahsavan-Markadeh, R. An experimental study on hydrogen-rich gas production via steam gasification of biomass in a research-scale fluidized bed. Energy Convers. Manag. 2015, 91, 427–432. [Google Scholar] [CrossRef]
- Mingyi, L.; Bo, Y.; Jingming, X.; Jing, C. Thermodynamic analysis of the efficiency of high-temperature steam electrolysis system for hydrogen production. J. Power Sources 2008, 177, 493–499. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.H.; Leung, D.Y.C. Energy and exergy analysis of hydrogen production by solid oxide steam electrolyzer plant. Int. J. Hydrogen Energy 2007, 32, 4648–4660. [Google Scholar] [CrossRef]


| Air Gasification | Oxygen Gasification | Steam Gasification | |
|---|---|---|---|
| Product heating value (MJ/Nm3) | Low, 4–6 | Medium, 10–15 | High, 15–20 |
| Products | CO, H2, Water, CO2, HC, Tar, N2 | CO, H2, HCs, CO2 | H2, CO, CO2, CH4, light HCs, tar |
| Average product gas composition | H2 15%, CO 20%, CH4 2%, CO2 15%, N2 48% | H2 40%, CO 40%, CO2 20% | H2 40%, CO 25%, CH4 8%, CO2 25%, N2 2% |
| Reactor temperature (°C) | 900–1100 | 1000–1400 | 700–1200 |
| Cost | Low | High | Medium |
| Reaction Equation | Reaction Enthalpy ΔH0 (kJ/mol) | |
|---|---|---|
| Methane synthesis | CO + 3H2 → CH4 + H2O | −206 |
| CO2 + 4H2 → CH4 + 2H2O | −165 | |
| CO2 + H2 → CO + H2O | +41 | |
| Methanol synthesis | CO + 2H2 → CH3OH | −90 |
| CO2 + 3H2 → CH3OH + H2O | −49 | |
| CO2 + H2 → CO + H2O | +41 |
| Wood Residue → Moisture 5.01 wt.% | |||
|---|---|---|---|
| Proximate Analysis (wt.%, dry) | Ultimate Analysis (wt.%, dry) | ||
| Volatile matter | 81.81 | C | 50.08 |
| Fixed carbon | 17.83 | H | 6.70 |
| Ash | 0.36 | O | 42.51 |
| N | 0.16 | ||
| S | 0.20 | ||
| Cl | - | ||
| Ash | 0.36 | ||
| Reaction Number | Reaction Scheme | Reaction Name | Heat of Reaction ΔH (kJ/mol) |
|---|---|---|---|
| 1 | C + O2 → CO2 | Carbon combustion | −393.0 |
| 2 | C + 0.5 O2 → CO | Carbon partial oxidation | −112.0 |
| 3 | C + CO2 → 2 CO | Boudouard reaction | +172.0 |
| 4 | C + H2O → CO + H2 | Water gas shift reaction | +131.0 |
| 5 | CO + H2O → CO2 + H2 | Water gas shift reaction | −41.0 |
| 6 | C + 2 H2 → CH4 | Methanation of carbon | −74.0 |
| 7 | H2 + 0.5 O2 → H2O | Hydrogen partial combustion | −242.0 |
| 8 | CH4 + H2O → CO + 3 H2 | Steam reforming of methane | +206.0 |
| 9 | H2 + S → H2S | H2S formation | −20.2 |
| H2O Feed (kg/s) | Required Power (MW) | Required Energy (kWh/kg of H2) | H2O Feed (kg/kg of H2) | Thermodynamic Electrolyser Efficiency (%) | |
|---|---|---|---|---|---|
| PEM | 97.8 | 1988.9 | 54.8 | 9.8 | 62.60 |
| SOEC | 89.2 | 1494.5 | 41.6 | 8.9 | 83.50 |
| Biomass Feed (kg/s) | Efficiency (%) | Syngas Molar Composition (%) |
|---|---|---|
| 77.39 | 12.90 | CO2 = 18.4 CH4 = 0.4 H2 = 61.7 CO = 19.4 |
| CH4 Scenario | MeOH Scenario | |
|---|---|---|
| Off-gas feed (kg/s) | 190.3 | 125.3 |
| Feed Compression (MW) | 38.3 | 85.4 |
| H2 Feed (kg/s) | 20.9 | 8.4 |
| H2 Consumption (%) | 98.4 | 37.4 |
| CH4 Product (kg/s) | 50.4 | - |
| CH3OH Product (kg/s) | - | 25.1 |
| Carbon Conversion (%) | 98.6 | 38.8 |
| CO2 Utilization (%) | 96.9 | 5.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaccara, A.; Petrucciani, A.; Matino, I.; Branca, T.A.; Dettori, S.; Iannino, V.; Colla, V.; Bampaou, M.; Panopoulos, K. Renewable Hydrogen Production Processes for the Off-Gas Valorization in Integrated Steelworks through Hydrogen Intensified Methane and Methanol Syntheses. Metals 2020, 10, 1535. https://doi.org/10.3390/met10111535
Zaccara A, Petrucciani A, Matino I, Branca TA, Dettori S, Iannino V, Colla V, Bampaou M, Panopoulos K. Renewable Hydrogen Production Processes for the Off-Gas Valorization in Integrated Steelworks through Hydrogen Intensified Methane and Methanol Syntheses. Metals. 2020; 10(11):1535. https://doi.org/10.3390/met10111535
Chicago/Turabian StyleZaccara, Antonella, Alice Petrucciani, Ismael Matino, Teresa Annunziata Branca, Stefano Dettori, Vincenzo Iannino, Valentina Colla, Michael Bampaou, and Kyriakos Panopoulos. 2020. "Renewable Hydrogen Production Processes for the Off-Gas Valorization in Integrated Steelworks through Hydrogen Intensified Methane and Methanol Syntheses" Metals 10, no. 11: 1535. https://doi.org/10.3390/met10111535
APA StyleZaccara, A., Petrucciani, A., Matino, I., Branca, T. A., Dettori, S., Iannino, V., Colla, V., Bampaou, M., & Panopoulos, K. (2020). Renewable Hydrogen Production Processes for the Off-Gas Valorization in Integrated Steelworks through Hydrogen Intensified Methane and Methanol Syntheses. Metals, 10(11), 1535. https://doi.org/10.3390/met10111535










