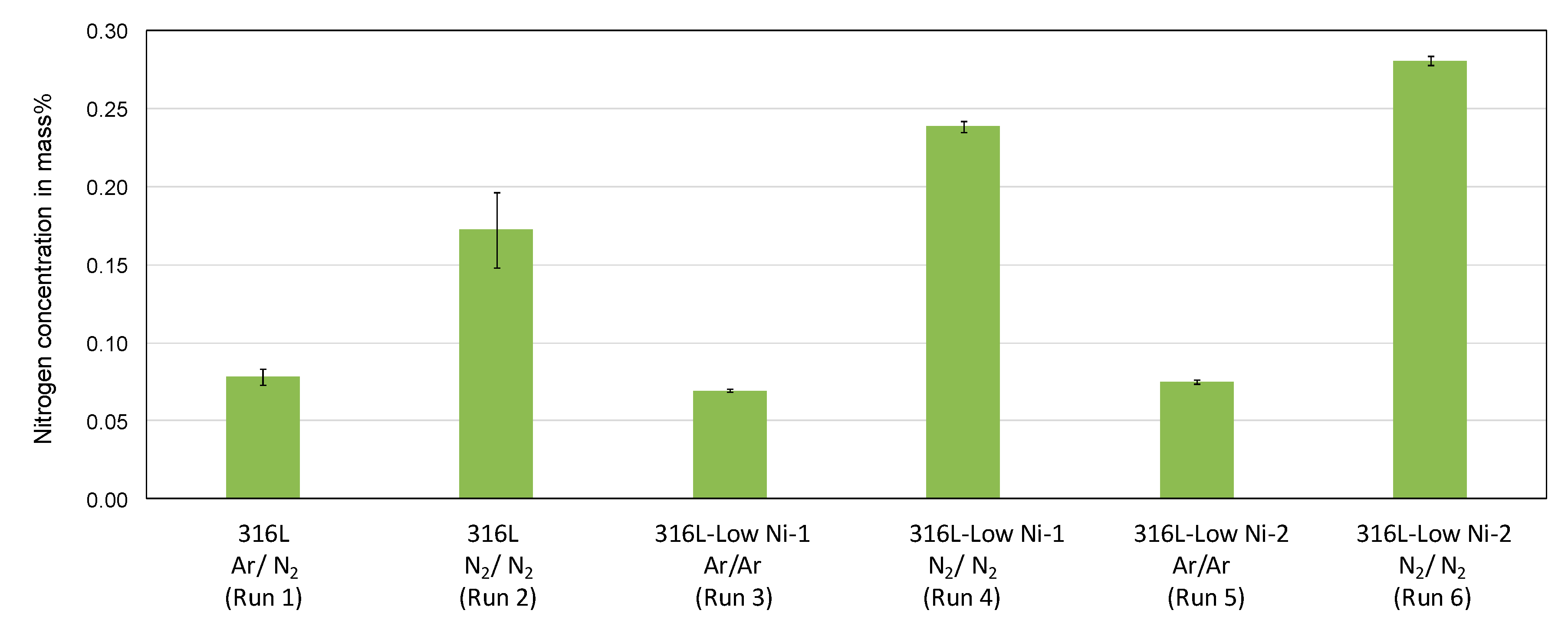3. Results and Discussion
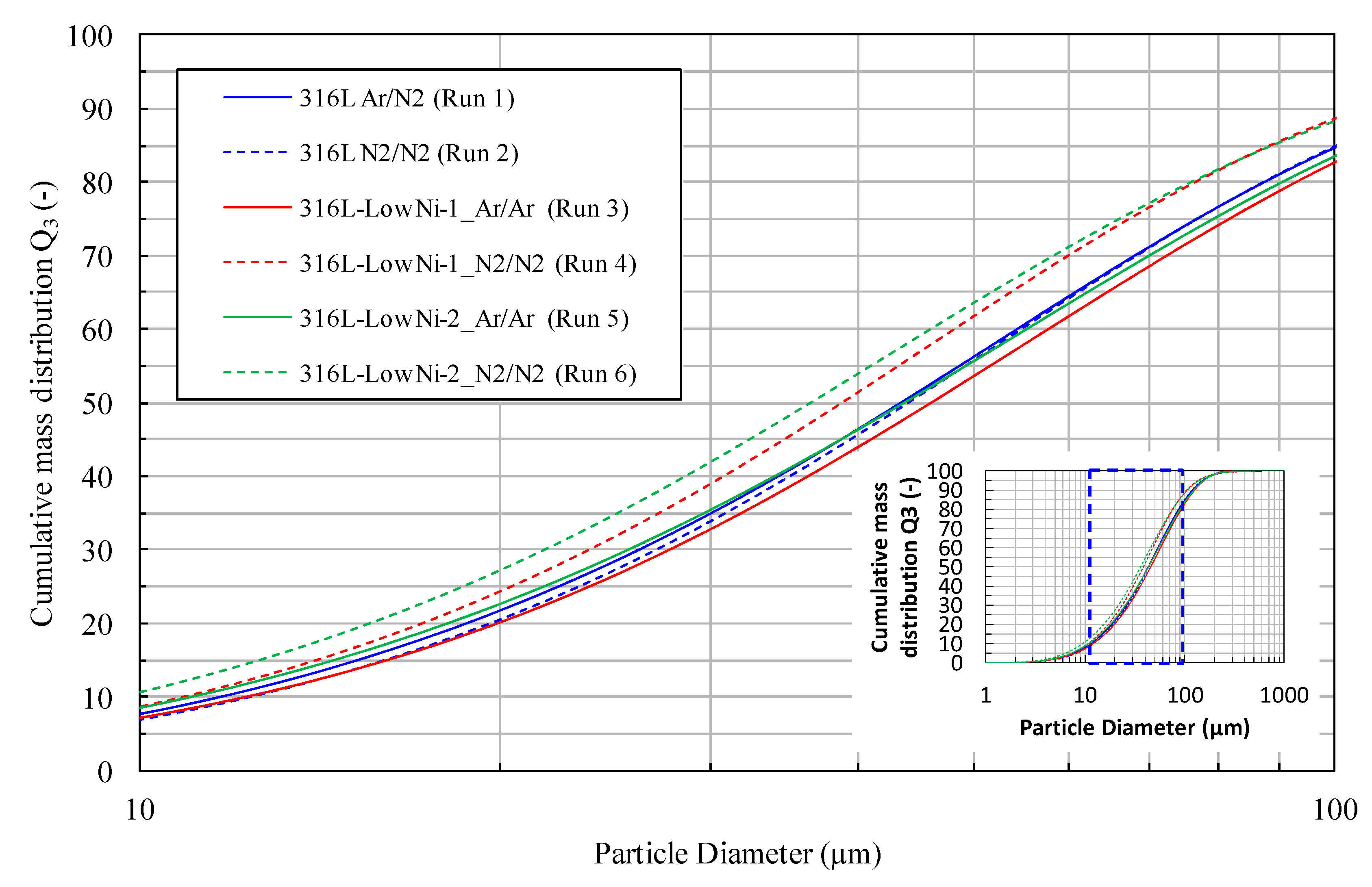
The cumulative mass distributions (Q3) of the particles are shown in
Figure 3. The particle size distribution of the steel powders seems to be unaffected by the alloy composition and the melting atmosphere. However, in
Figure 3, the particle size distribution of the 316L-Low Ni-1 (Run 4) and 316L-Low Ni-2 powder (Run 6) shifts to the left, indicating that finer particles are produced. The median diameter d50 is 39–41 µm for the two powders, while it is 46–49 µm for the other powders. A possible reason for this deviation is as follows: the pouring temperature of the melts in experiment run 4 and 6 is 40 to 50 °C higher than that of the other experiments, resulting in a lower viscosity and surface tension of the melts, and thereby producing a smaller atomized droplet size. The gas-atomized particles typically show a spherical shape and smooth surface, with a few observed satellite particles (see
Figure 4).
A variety of shape parameters such as circularity, convexity, solidity, and aspect ratio are employed by the image analysis system Morphologi G3 to characterize the particle shape [
29]. It is unlikely that a single shape parameter can perfectly characterize particles in all applications. For example, an application concerned with perfectly spherical particles could use circularity to measure any deviation. However, circularity would not be appropriate in an application where both ‘spikey’ and elliptical particles are present in the sample. Since the gas atomized particles are generally in spherical shape, only circularity of the steel powders is presented in this paper. According to the definition by the Morphologi G3 [
29], Circularity is the ratio of the circumference of the projected/equivalent area of a particle to the actual perimeter of the particle:
Circularity quantifies how close the shape is to a perfect circle. A perfect circle has circularity of 1.0, while a very narrow, elongated particle has circularity close to 0.
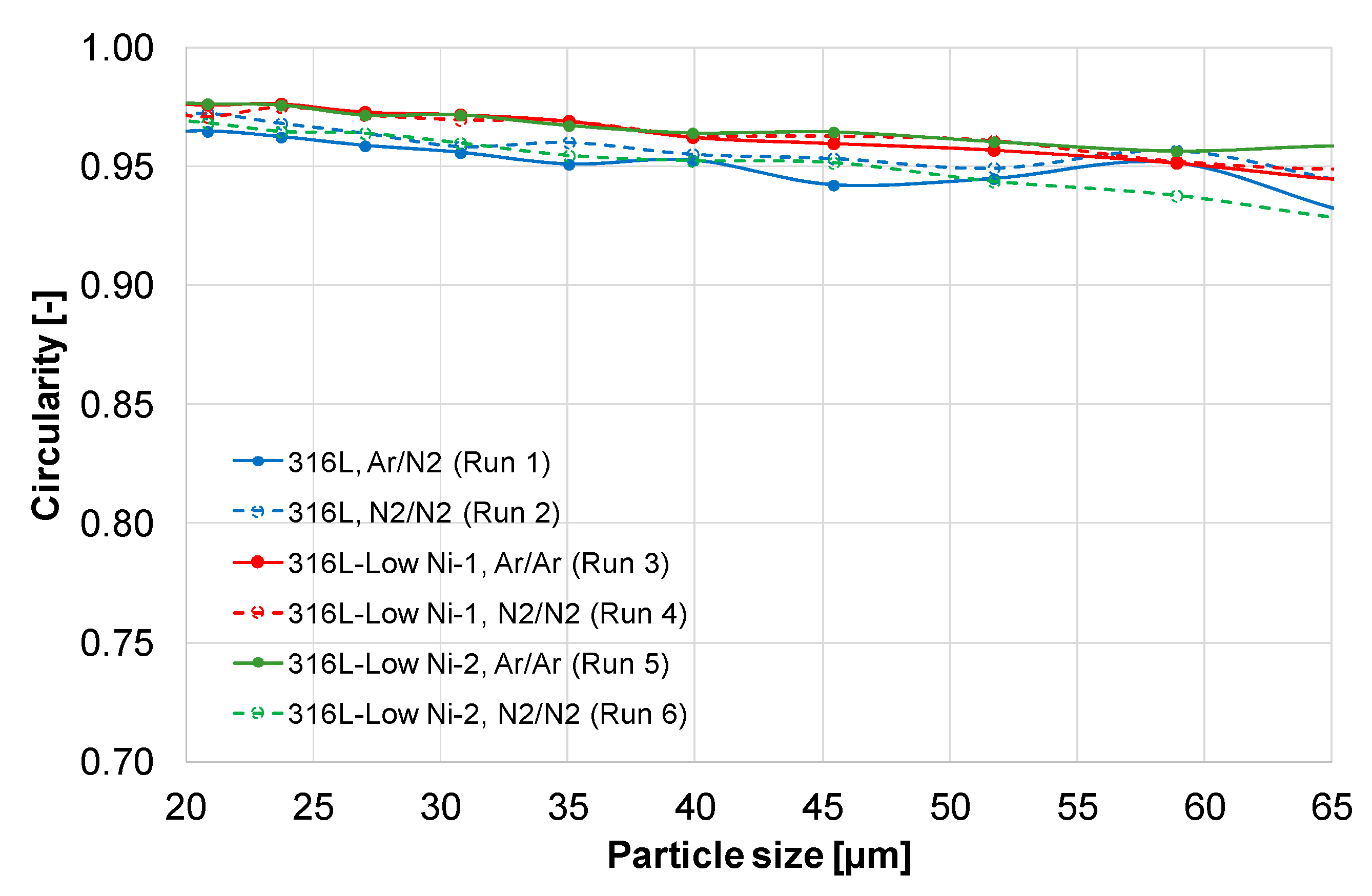
The shape parameter circularity of the various steel powder particles is presented in
Figure 5. The circularity of the powder particles (size 20–63 µm) is in a range of 0.93 to 0.97, with a higher circularity for the smaller particles. For the large particles with a size of 200 µm the circularity is about 0.90. The small particles are cooled and solidified faster than the large particles, and it is less possible for the collided particles to be embedded in the small particles that are completely solidified. The modified 316L powders show a higher level of circularity than the 316L powders, possibly due to lower viscosity and higher surface tension of the melts. In general, a high circularity means good flowability of the powder. The spherical particles have less contact surface area, therefore the friction and interlocking between the particles is lower than that of the particles in irregular shape [
28,
30].
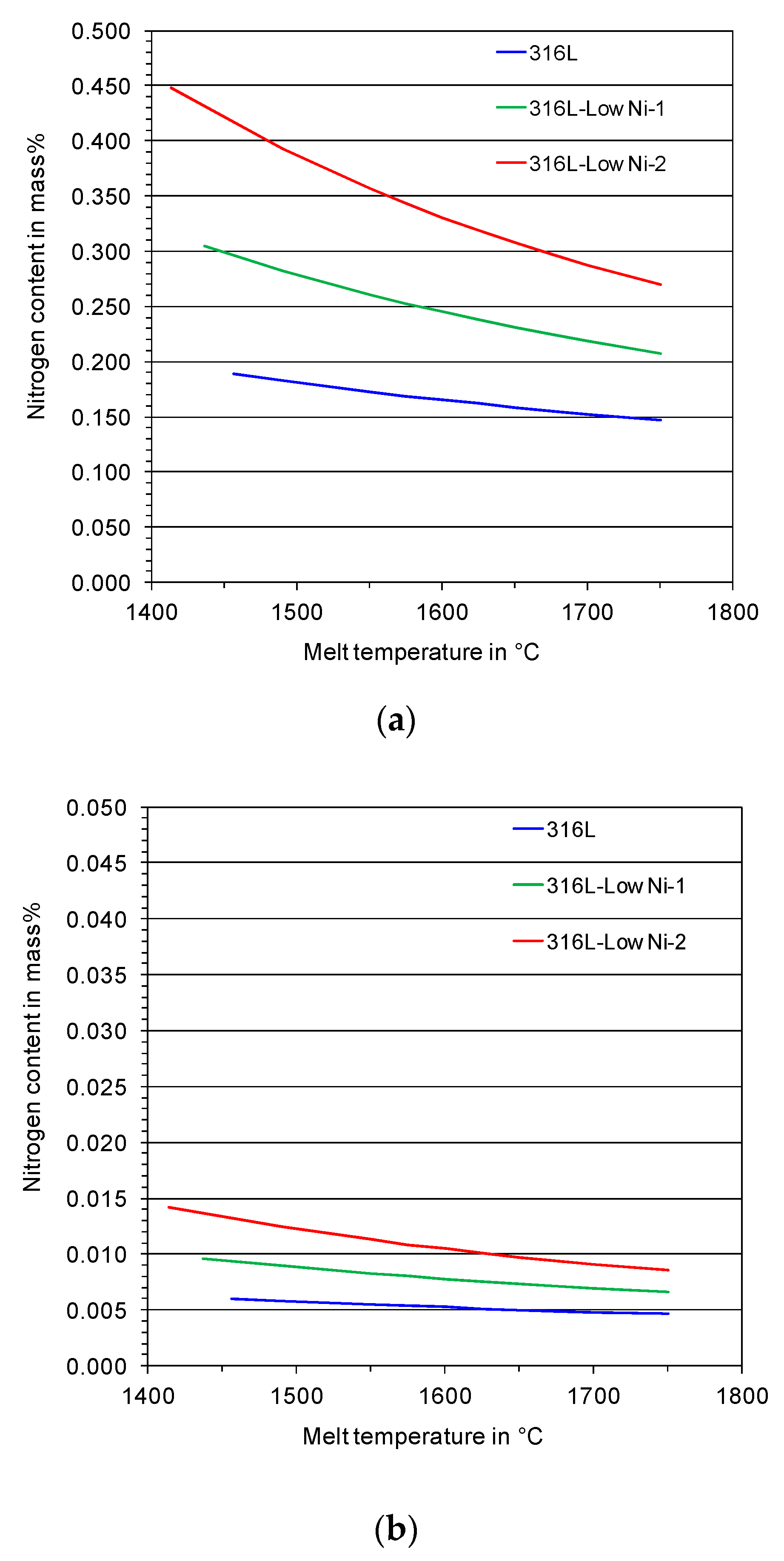
The nitrogen content in the feedstock (316L and modified 316L prepared by inductive melting and casting) is 200–300 ppm. It is increased in the atomized particles (see
Figure 6). When melting under an argon atmosphere, the nitrogen content is 600–800 ppm, independent of the chemical composition of the stainless steels. Under a nitrogen atmosphere, the nitrogen pick-up is significantly increased. The addition of manganese in the austenitic stainless steel also contributes significantly to the pick-up of nitrogen in the steel.
The measured nitrogen content in the powders is very close to the calculated values as shown in
Section 2.1. This means that almost all of the dissolved nitrogen in the steel melts remains in the atomized powders. The solidification of the steel melts possibly with primary δ-ferrite, which has a very low solubility for nitrogen [
20], did not result in significant nitrogen outgassing due to rapid solidification of the atomized droplets.
After mechanical sieving, the powder fraction of 20–63 µm is investigated further. This powder fraction accounts for approximately 45 percent of the total atomized powder. The flow time of this powder is 15–16 s for a 50 g powder sample through a Hall flow funnel (outlet diameter 2.5 mm). The good flowability of the powder is attributed to the spherical shape of the particles and very few satellite particles. These satellite particles are formed when the finer solidified particles stick to the molten or semi-molten surface of the coarser ones as a result of the in-flight collisions. There are very few satellites in the powder due to the use of the anti-satellite system in the gas atomization. The apparent density and the tap density of the powder is 4.5 g/cm3 and 5.0 g/cm3, respectively. These powder particles have been successfully processed by means of laser powder bed fusion, which will be reported in the future.
The particle morphology of the gas-atomized powder is shown in
Figure 7. Typical dendritic structure can be observed at the particle surface. It is also clearly seen in the metallographic sections of the particles processed under a nitrogen atmosphere (see
Figure 8). However, the dendrites are not so apparent in the particles processed under argon melting atmosphere. It appears that the chemical etchant used erodes the dendrites more significantly in the interior region than at the boundaries of the dendrites. This difference between the etching effects correlates to the different nitrogen concentrations in the powders. The addition of nitrogen to the stainless steel promotes a more homogeneous distribution of chromium in the matrix, and therefore an improved corrosion resistance [
2,
7,
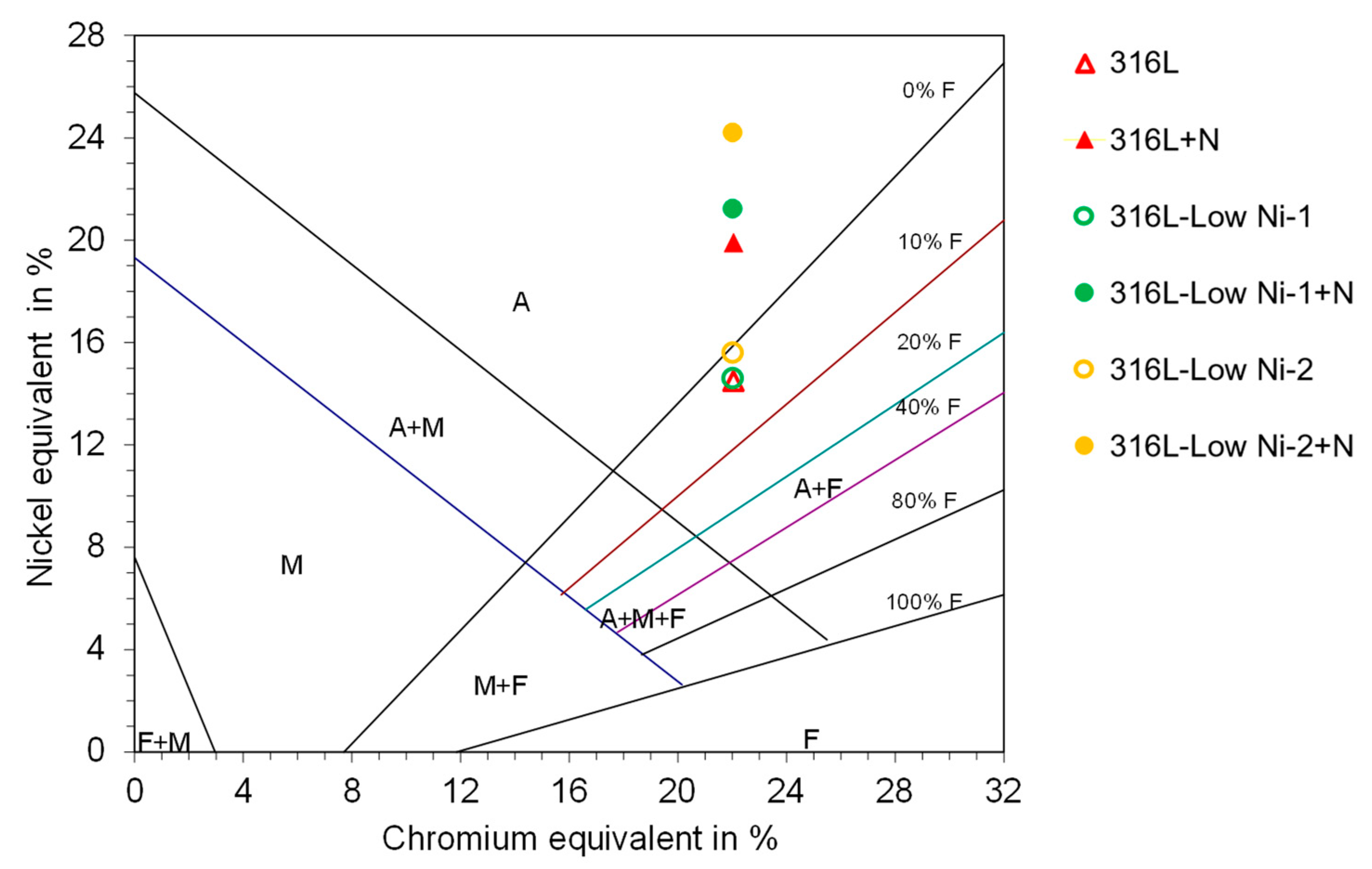
8]. According to the Schaeffler diagram, the 316L and the modified 316L alloys are composed of austenite and small fraction of ferrite (less than 5%). With addition of nitrogen, the alloys are essentially austenite. From the micrographs it is difficult to distinguish the ferrite from the austenite.
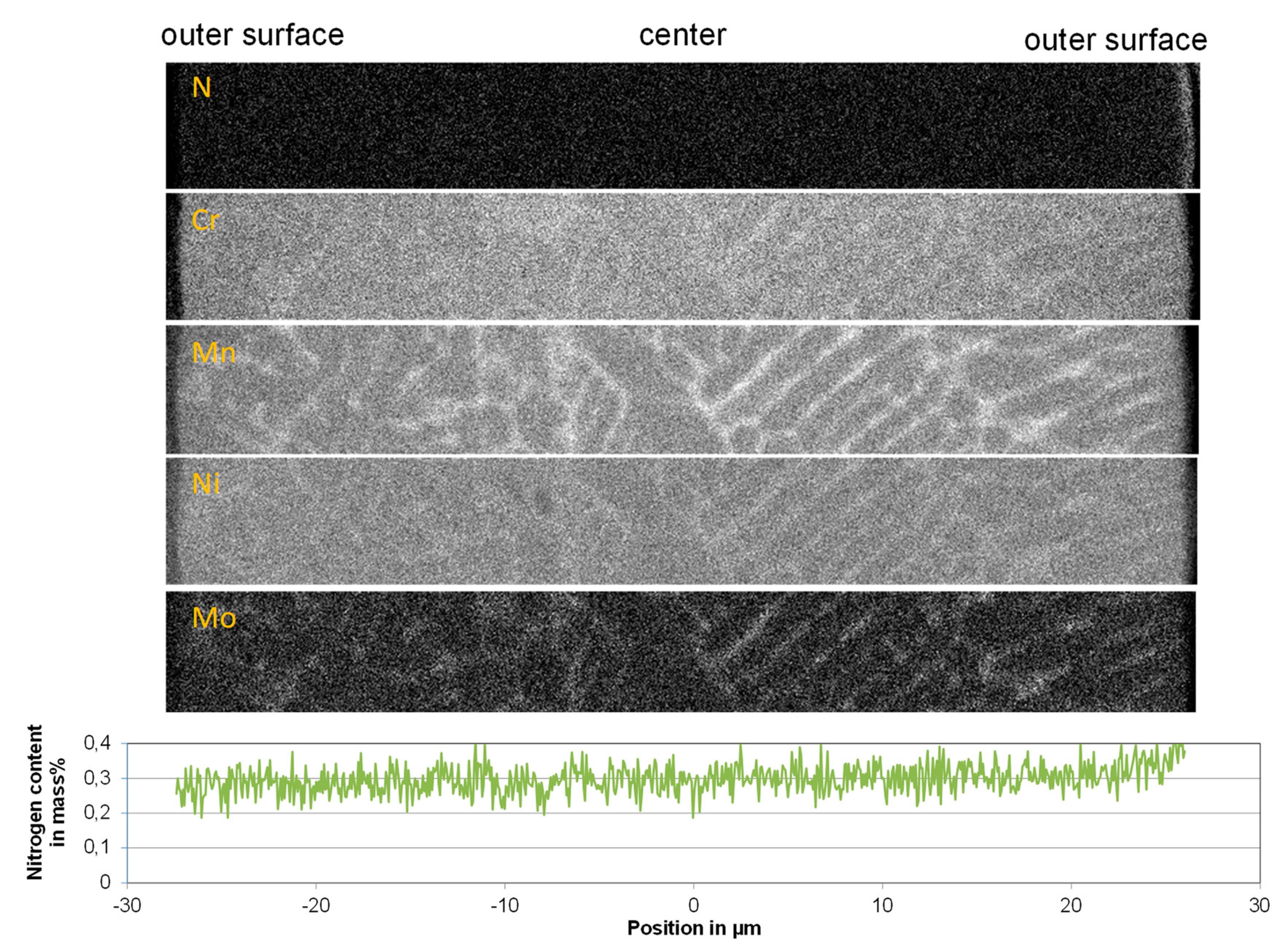
Element mapping (EPMA) on the cross section of the particles show that the nitrogen distribution in the particles is relatively homogeneous. Between the outer surface and the core of the particles, no nitrogen concentration gradient is observed, as represented in
Figure 9. The average nitrogen concentration of the 316L-Low Ni-2 powder particles processed under a nitrogen atmosphere is about 0.3 mass %, which is in good agreement with the measured results in
Figure 6. Slight segregation of chromium is seen by element mapping, while the segregation of the alloying elements manganese, nickel, and molybdenum in the dendrite structure of the particle is more significant. Since the segregation occurs in micrometer scale, it could have a very limited effect on the laser additive manufactured parts.
Author Contributions
Conceptualization, A.S. and V.U.; methodology, C.C.; formal analysis, C.C.; investigation, C.C.; resources, V.U.; writing—original draft preparation, C.C.; writing—review and editing, A.S., V.U. and H.-W.Z.; supervision, V.U. and A.S.; project administration, V.U. and A.S.; funding acquisition, H.-W.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Deutsche Forschungsgemeinschaft (DFG), grant number Zo140/22-1.
Acknowledgments
We acknowledge for the cooperation and valuable discussion with the groups of Theisen (Ruhr University Bochum) and Walther (TU Dortmund) in the collaboration project.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Yang, K.; Ren, Y. Nickel-free austenitic stainless steels for medical applications. Sci. Technol. Adv. Mater. 2010, 11, 014105. [Google Scholar] [CrossRef] [PubMed]
- Gavriljuk, V.G.; Shanina, B.D.; Berns, H. On the correlation between electron structure and short range atomic order in iron-based alloys. Acta Mater. 2000, 48, 3879–3893. [Google Scholar] [CrossRef]
- Levey, P.R.; van Bennekom, A. A mechanistic study of the effects of nitrogen on the corrosion properties of stainless steels. Corrosion 1995, 51, 911–921. [Google Scholar] [CrossRef]
- Baba, H.; Kodama, T.; Katada, Y. Role of nitrogen on the corrosion behavior of austenitic stainless steels. Corros. Sci. 2002, 44, 2393–2407. [Google Scholar] [CrossRef]
- Jargelius-Pettersson, R.F.A. Electrochemical investigation of the influence of nitrogen alloying on pitting corrosion of austenitic stainless steels. Corros. Sci. 1999, 41, 1639–1664. [Google Scholar] [CrossRef]
- Richardson, T.J.; Cottis, B.R.A.; Lindsay, R.; Lyon, S.; Scantlebury, D.J.D.; Stott, H.; Graham, M. Shreir’s Corrosion; Elsevier: Burlington, MA, USA, 2009. [Google Scholar]
- Berns, H.; Gavriljuk, V.; Riedner, S. High Interstitial Stainless Austenitic Steels; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Gavriljuk, V.G.; Berns, H. High Nitrogen Steels: Structure, Properties, Manufacture, Applications; Springer: Berlin, Germany, 1999. [Google Scholar]
- Stein, G.; Hucklenbroich, I. Manufacturing and Applications of High Nitrogen Steels. Mater. Manuf. Process. 2004, 19, 7–17. [Google Scholar] [CrossRef]
- Zitelli, C.; Folgarait, P.; Schino, A.D. Laser Powder Bed Fusion of Stainless Steel Grades: A Review. Metals 2019, 9, 731. [Google Scholar] [CrossRef]
- Zhai, Y.; Lados, D.A.; LaGoy, J.L. Additive Manufacturing: Making Imagination the Major Limitation. JOM 2014, 66, 808–816. [Google Scholar] [CrossRef]
- European Powder Metallurgy Association (EPMA)—Introduction to Additive Manufacturing Technology (Brochure). Available online: https://www.epma.com/epma-free-publications/product/introduction-toadditive-manufacturing-brochure (accessed on 3 August 2018).
- Horn, T.J.; Harrysson, O.L.A. Overview of Current Additive Manufacturing Technologies and Selected Applications. Sci. Prog. 2012, 95, 255–282. [Google Scholar] [CrossRef]
- Glasschroeder, J.; Prager, E.; Zaeh, M.F. Powder-bed-based 3D-printing of function integrated parts. Rapid Prototyp. J. 2015, 21, 207–215. [Google Scholar] [CrossRef]
- Sanín Pérez, P. A Study of Additive Manufacturing Applied to the Design and Production of LED Luminaires; Politecnico di Milano: Milano, Italy, 2013. [Google Scholar]
- Tang, Y.; Yang, S.; Zhao, Y.F. Sustainable Design for Additive Manufacturing through Functionality Integration and Part Consolidation. In Handbook of Sustainability in Additive Manufacturing; Muthu, S.S., Savalani, M.M., Eds.; Springer: Singapore, 2016; pp. 101–144. [Google Scholar]
- Bartolomeu, F.; Buciumeanu, M.; Pinto, E.; Alves, N.; Carvalho, O.; Silva, F.S.; Miranda, G. 316L stainless steel mechanical and tribological behavior—A comparison between selective laser melting, hot pressing and conventional casting. Addit. Manuf. 2017, 16, 81–89. [Google Scholar] [CrossRef]
- Capek, J.; Machova, M.; Fousova, M.; Kubásek, J.; Vojteˇch, D.; Fojt, J.; Jablonská, E.; Lipov, J.; Ruml, T. Highly porous, low elastic modulus 316L stainless steel scaffold prepared by selective laser melting. Mater. Sci. Eng. C 2016, 69, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zhou, Y.; Gu, R.; Zhang, X.; Quach, W.; Yan, M. A Comprehensive Study of Steel Powders (316L, H13, P20 and 18Ni300) for Their Selective Laser Melting Additive Manufacturing. Metals 2019, 9, 86. [Google Scholar] [CrossRef]
- Feichtinger, H.; Stein, G. Melting of High Nitrogen Steels. Mater. Sci. Forum 1999, 318–320, 261–270. [Google Scholar] [CrossRef]
- Pridantsev, M.V.; Levin, F.L. Effect of manganese on the structure and properties of nonmagnetic stainless steels. Met. Sci. Heat Treat. 1966, 7, 786–789. [Google Scholar] [CrossRef]
- Boes, J.; Röttger, A.; Becker, L.; Theisen, W. Processing of gas-nitrided AISI 316L steel powder by laser powder bed fusion—Microstructure and properties. Addit. Manuf. 2019, 30, 100836. [Google Scholar] [CrossRef]
- Medovar, B.I.; Saenko, V.Y.; Grigorenko, G.M.; Pomarin, Y.M.; Kumysh, V.I. Arc Slag Remelting of Steel and Alloys; Cambridge International Science. Publishing Ltd.: Cambridge, UK, 1996; p. 47. [Google Scholar]
- Guiraldenq, P.; Duparc, O.H. The genesis of the Schaeffler diagram in the history of stainless steel. Metall. Res. Technol. 2017, 114, 613. [Google Scholar] [CrossRef]
- De Long, W.T. A Modified Phase Diagram for Stainless Steel Weld Metals. Met. Prog. 1960, 77, 98–100. [Google Scholar]
- Schwenck, D.; Ellendt, N.; Fischer-Bühner, J.; Hofmann, P.; Uhlenwinkel, V. A novel convergent–divergent annular nozzle design for close-coupled atomization. Powder Metall. 2017, 60, 198–207. [Google Scholar] [CrossRef]
- Ciftci, N.; Ellendt, N.; Soares Barreto, E.; Mädler, L.; Uhlenwinkel, V. Increasing the amorphous yield of {(Fe0.6Co0.4)0.75B0.2Si0.05}96Nb4 powders by hot gas atomization. Adv Powder Technol. 2018, 29, 380–385. [Google Scholar] [CrossRef]
- Mostaghimi, F.; Fischer-Bühner, J.; Heemann, L.; Hofmann, P.; von Hehl, A.; Uhlenwinkel, V. Anti-satellite system for the improvement of powder quality in additive manufacturing using a FeMnAlSi alloy. In Proceedings of the Euro PM2018, Bilbao, Spain, 14–17 October 2018. Session 22. [Google Scholar]
- Malvern Instruments Ltd. Morphologi G3 User Manual, MAN0410-07-EN-00; Malvern Instruments Ltd.: Malvern, UK, 2015. [Google Scholar]
- Fereiduni, E.; Ghasemi, A.; Elbestawi, M. Characterization of Composite Powder Feedstock from Powder Bed Fusion Additive Manufacturing Perspective. Materials 2019, 12, 3673. [Google Scholar] [CrossRef] [PubMed]
Figure 1.
Nitrogen solubility in the austenitic stainless steels at (a) 0.1 MPa N2 and (b) 100 Pa N2, calculated according to Equations (1) and (2).
Figure 2.
The position of the 316L steel and the modified 316L steels in Schaeffler diagram (A: austenite, F: ferrite, M: martensite).
Figure 3.
Particle size distributions of the gas atomized 316L and modified 316L powders. The nomenclature used in the diagram, for instance Ar/N2, indicates that the first element corresponds to the melting atmosphere and the second to the atomization gas.
Figure 4.
SEM images of gas atomized 316L and modified 316L steel powders (particle size < 200 µm): (a) 316L Ar/N2 (Run 1), (b) 316L N2/N2 (Run 2), (c) 316L-Low Ni-1 Ar/Ar (Run 3), (d) 316L-Low Ni-1 N2/N2 (Run 4), (e) 316L-Low Ni-2 Ar/Ar (Run 5), and (f) 316L-Low Ni-2 N2/N2 (Run 6).
Figure 5.
Circularity of the gas atomized 316L and modified 316L steel powders.
Figure 6.
Concentration of N in gas atomized 316L and modified 316L steel powders, measured by means of carrier hot gas extraction.
Figure 7.
SEM images of gas-atomized powder (particle size 20–63 µm): (a) 316L-Low Ni-1 Ar/Ar (Run 3), (b) 316L-Low Ni-1 N2/N2 (Run 4), (c) 316L-Low Ni-2 Ar/Ar (Run 5), and (d) 316L-Low Ni-2 N2/N2 (Run 6).
Figure 8.
Optical micrographs of gas-atomized powder (particle size 20–63 µm): (a) 316L-Low Ni-1 Ar/Ar (Run 3), (b) 316L-Low Ni-1 N2/N2 (Run 4), (c) 316L-Low Ni-2 Ar/Ar (Run 5), and (d) 316L-Low Ni-2 N2/N2 (Run 6). The samples were etched with V2A etchant at 60 °C for 30 s.
Figure 9.
Element mapping (EPMA) on the cross section of a 316L-Low Ni-2 N2/N2 (Run 6) particle from the powder faction of 20–63 µm and the nitrogen concentration line scan on the cross section of the particle. In total, 100 lines scans were conducted on the particle at a distance of 0.0698 µm; the mean values are plotted in the diagram.
Table 1.
Chemical composition of the raw material 316L and modified 316L (in mass %).
| Alloy | C | Cr | Ni | Mo | Mn | Si | Fe |
|---|
| 316L | 0.02 | 18.4 | 13.7 | 1.8 | 0.4 | 0.7 | bal. |
| 316L-Low Ni-1 | 0.02 | 18.4 | 11.0 | 1.8 | 6.0 | 0.7 | bal. |
| 316L-Low Ni-2 | 0.02 | 18.4 | 9.0 | 1.8 | 12.0 | 0.7 | bal. |
Table 2.
Gas atomization parameters of 316L and modified 316L steel powder.
Run Number
(Exp. ID-Number) | Unit | Run 1
(PA7-159) | Run 2
(PA7-161) | Run 3
(PA7-233) | Run 4
(PA7-217) | Run 5
(PA7-234) | Run 6
(PA7-218) |
|---|
| Material | - | 316L | 316L | 316L-Low Ni-1 | 316L-Low Ni-1 | 316L-Low Ni-2 | 316L-Low Ni-2 |
| Atomization gas | - | N2 | N2 | Ar | N2 | Ar | N2 |
| Melting atmosphere | - | Ar | N | Ar | N2 | Ar | N2 |
| Delivery tube diameter | mm | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
| Pouring temperature | °C | 1650 | 1650 | 1650 | 1690 | 1650 | 1700 |
| Atomization gas pressure | MPa | 1.6 | 1.6 | 1.6 | 1.6 | 1.6 | 1.6 |
| Atomization gas flow rate | kg/h | 590 | 590 | 775 | 590 | 775 | 590 |
| Melt flow rate | kg/h | - | 297 | 268 | 289 | 268 | 288 |
| GMR | - | - | 1.99 | 2.89 | 2.05 | 2.89 | 2.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).