Reductive Smelting of Neutralized Red Mud for Iron Recovery and Produced Pig Iron for Heat-Resistant Castings
Abstract
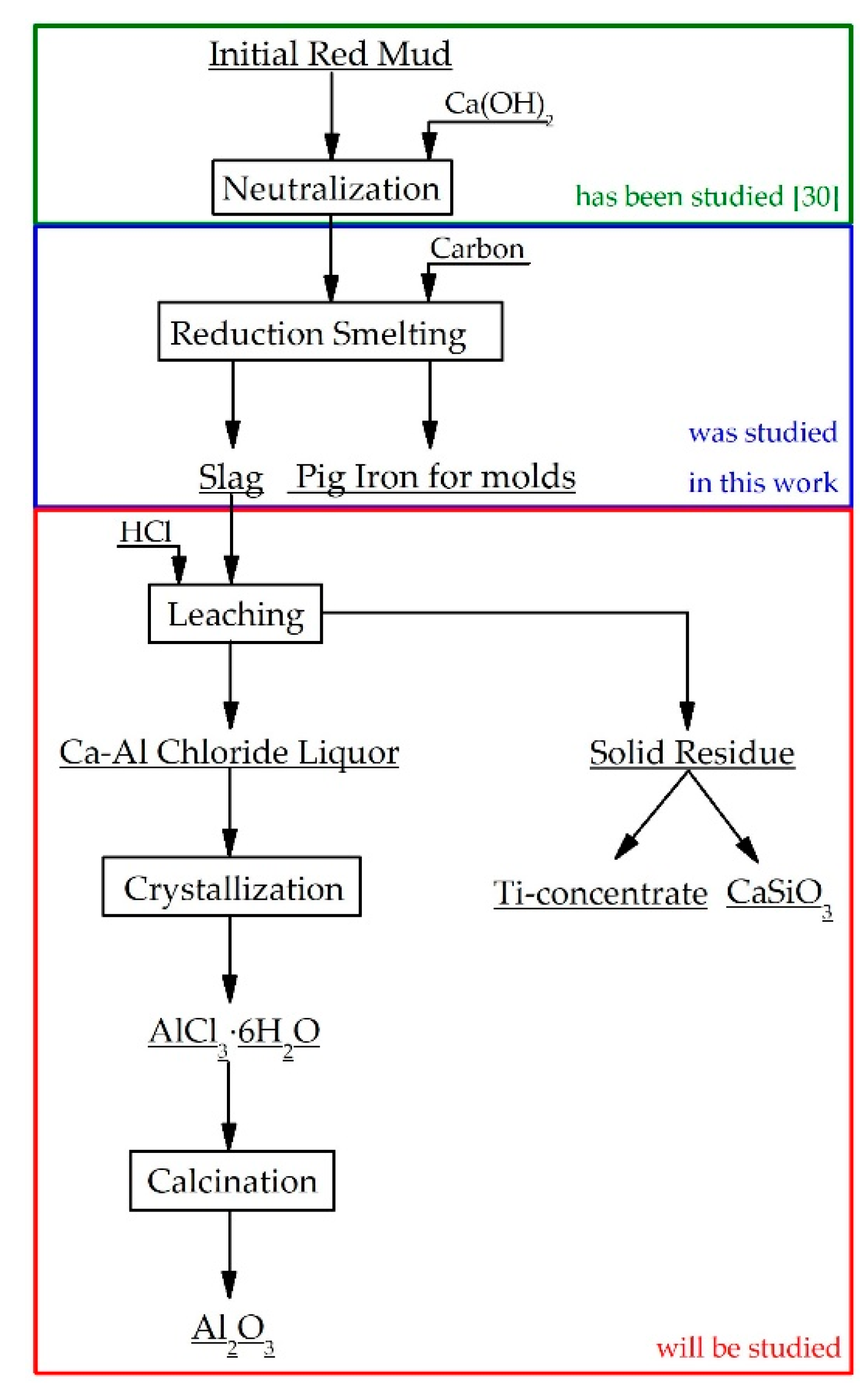
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Thermodynamic Modeling
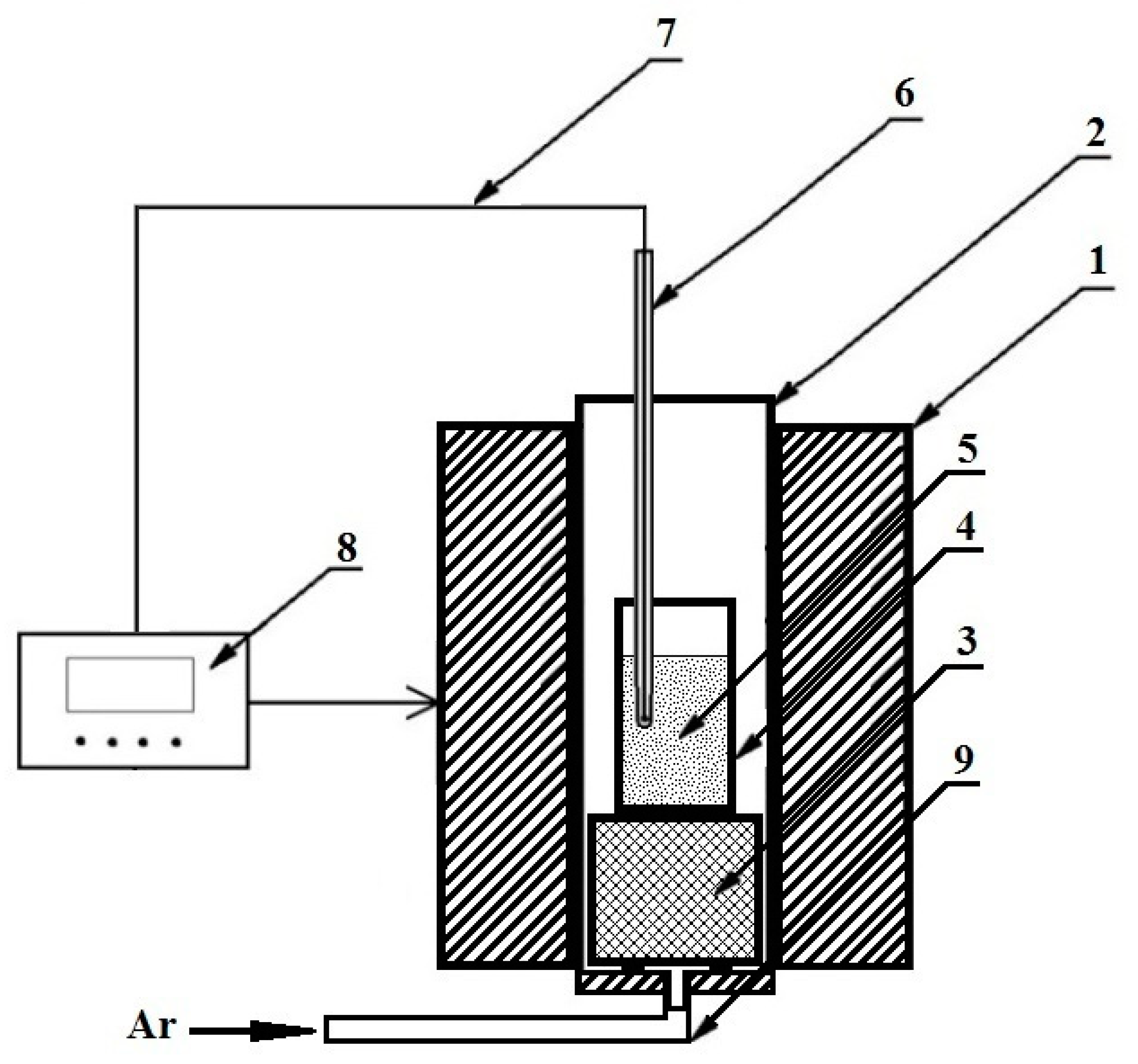
2.3. Reductive Smelting of Red Mud
3. Results
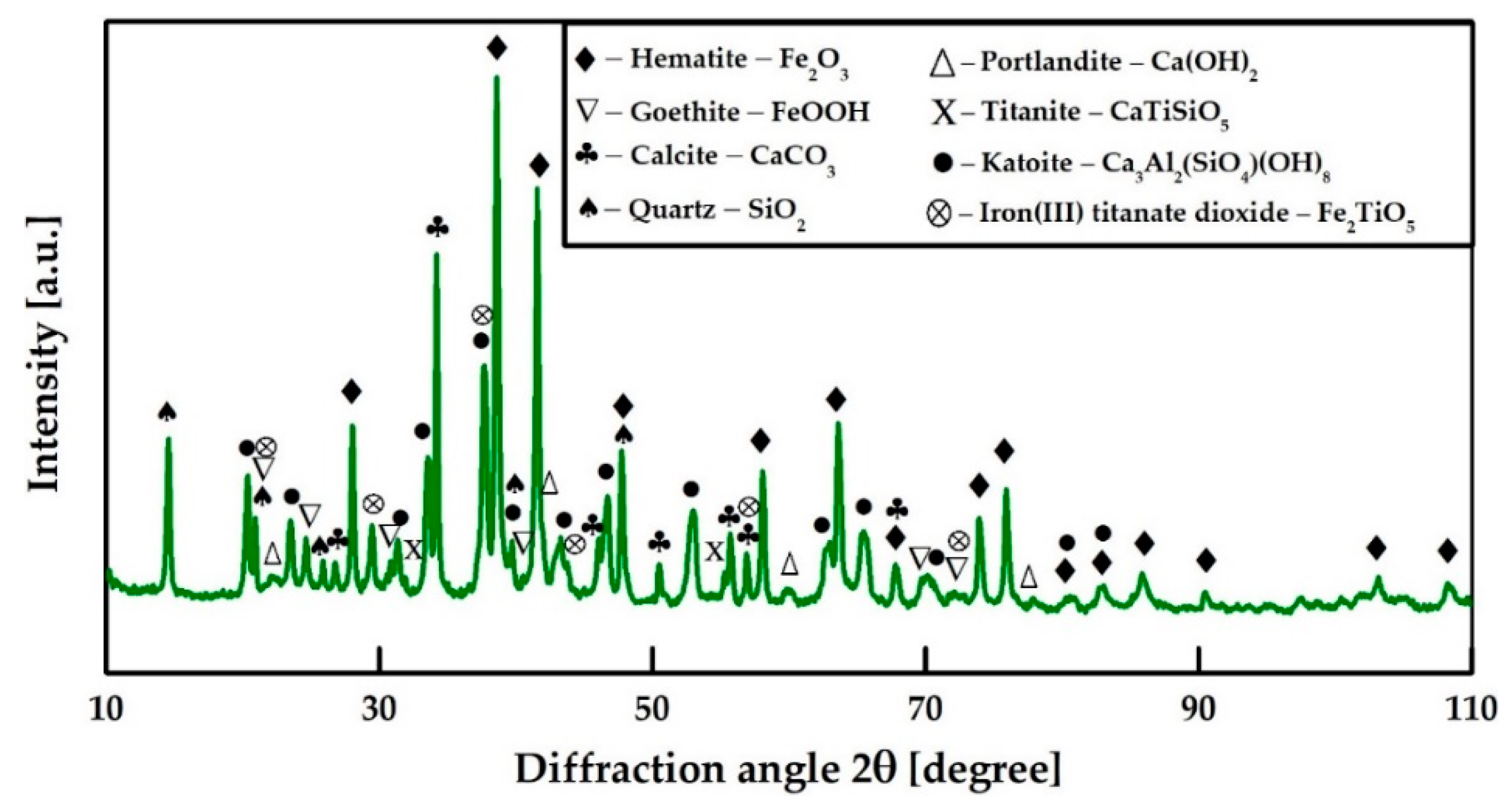
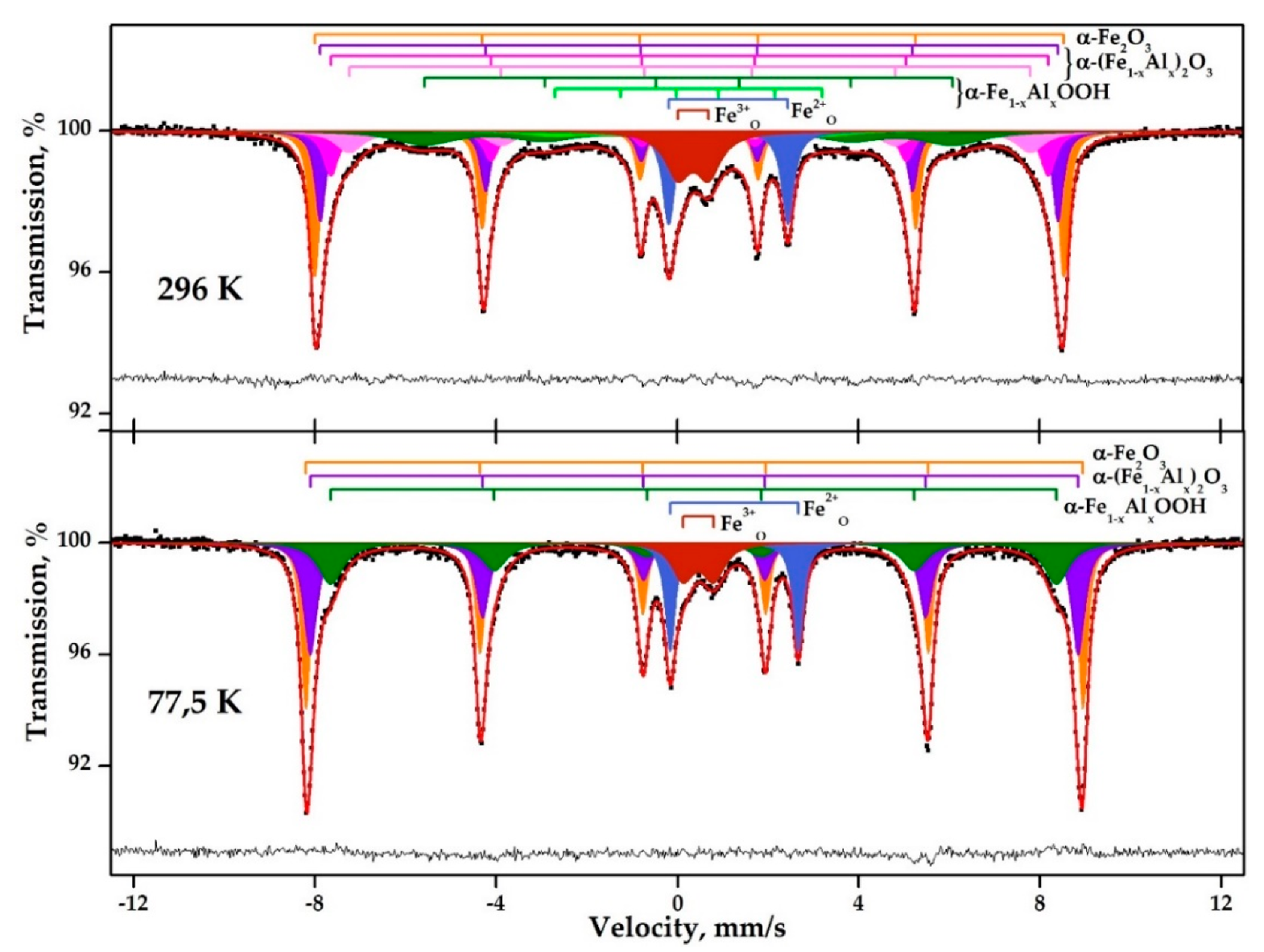
3.1. Composition of the Neutralized Red Mud
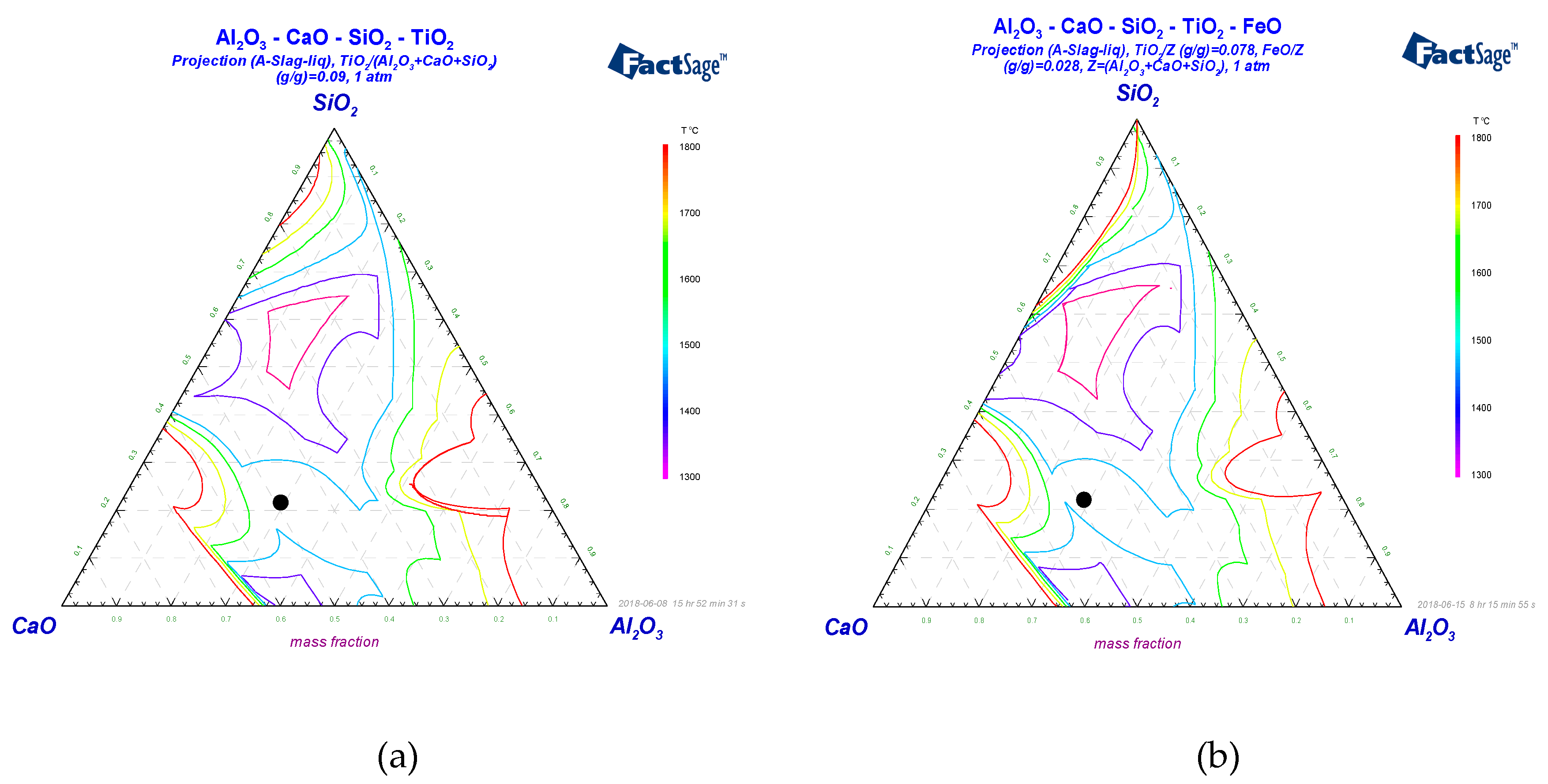
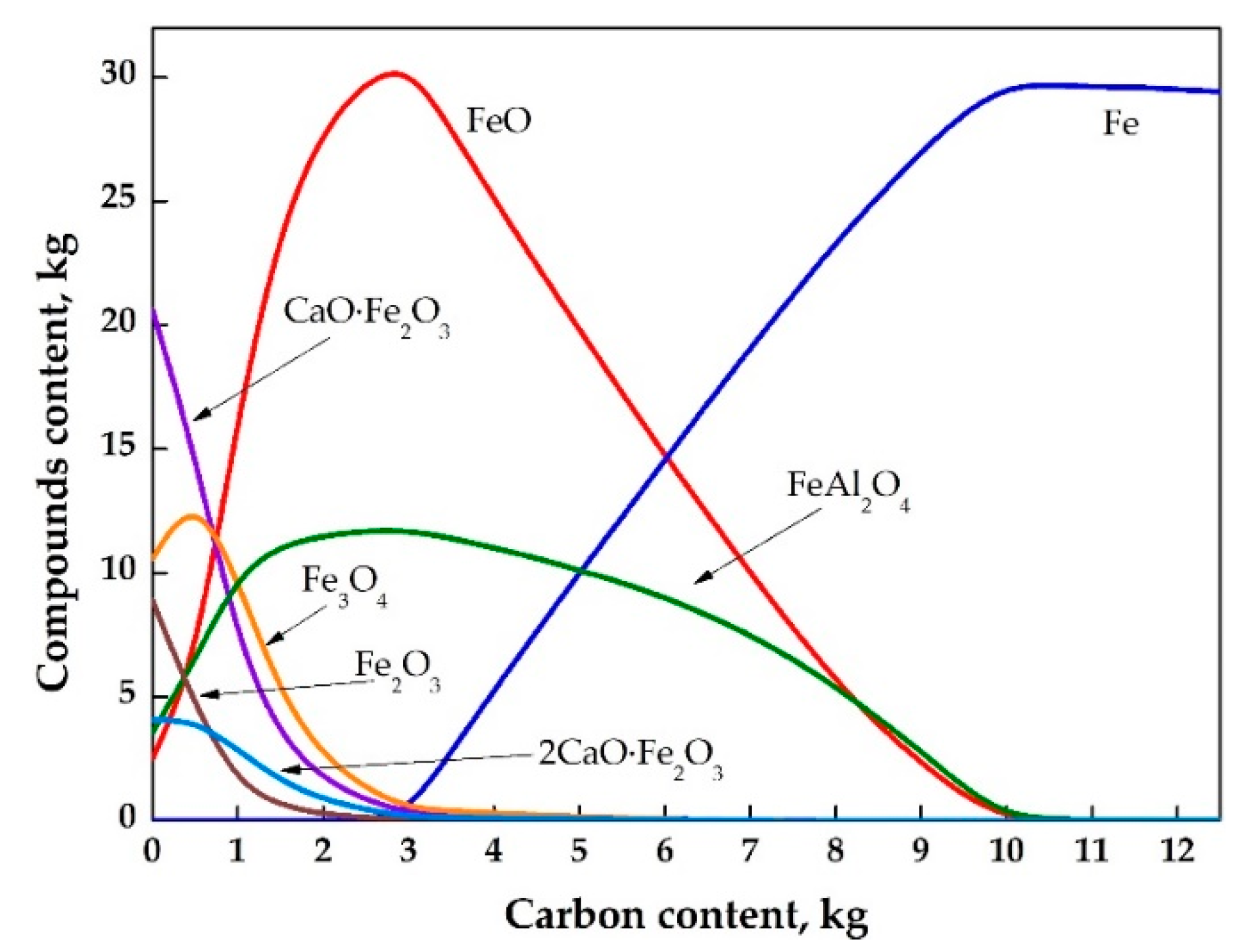
3.2. Thermodynamic Calculations
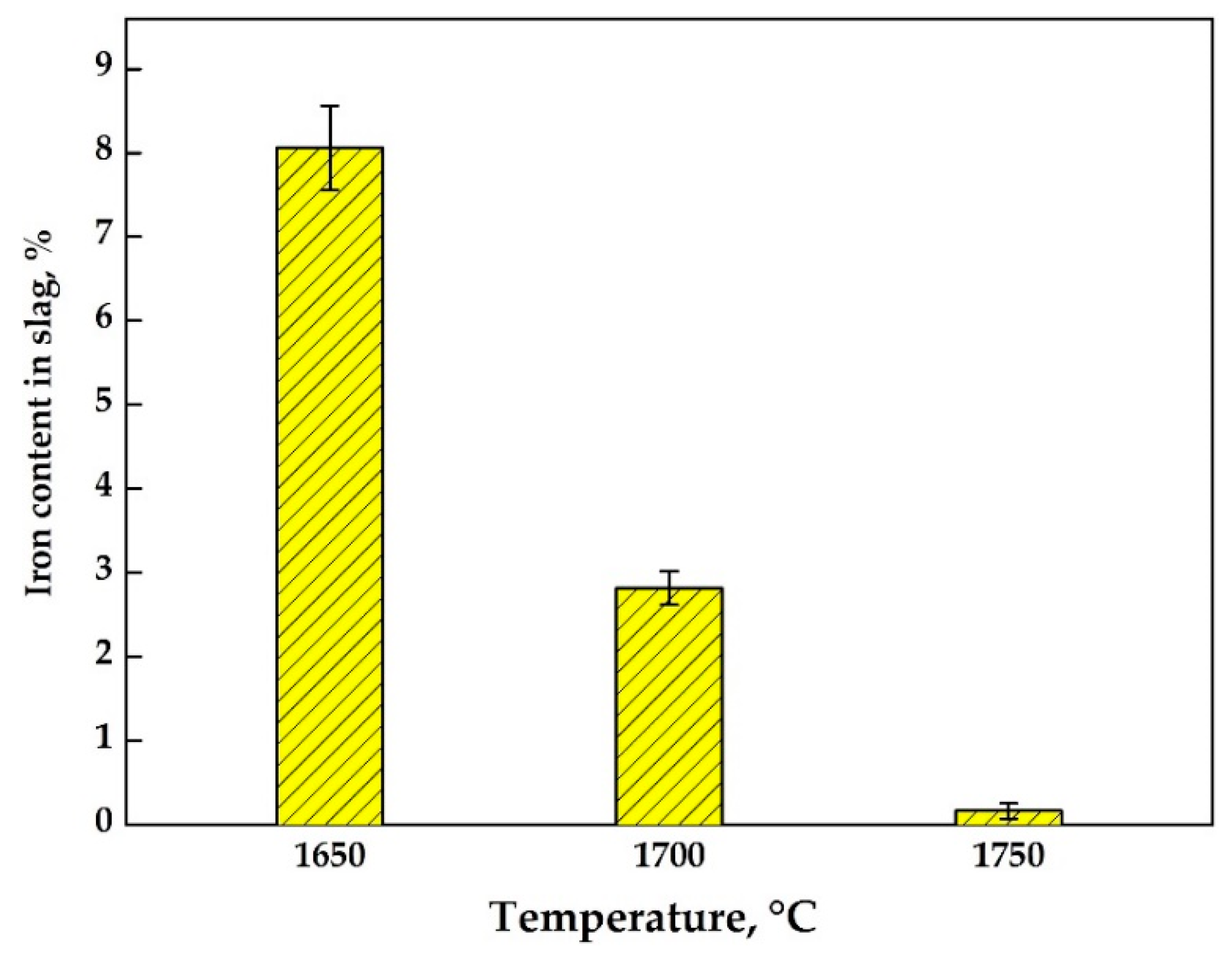
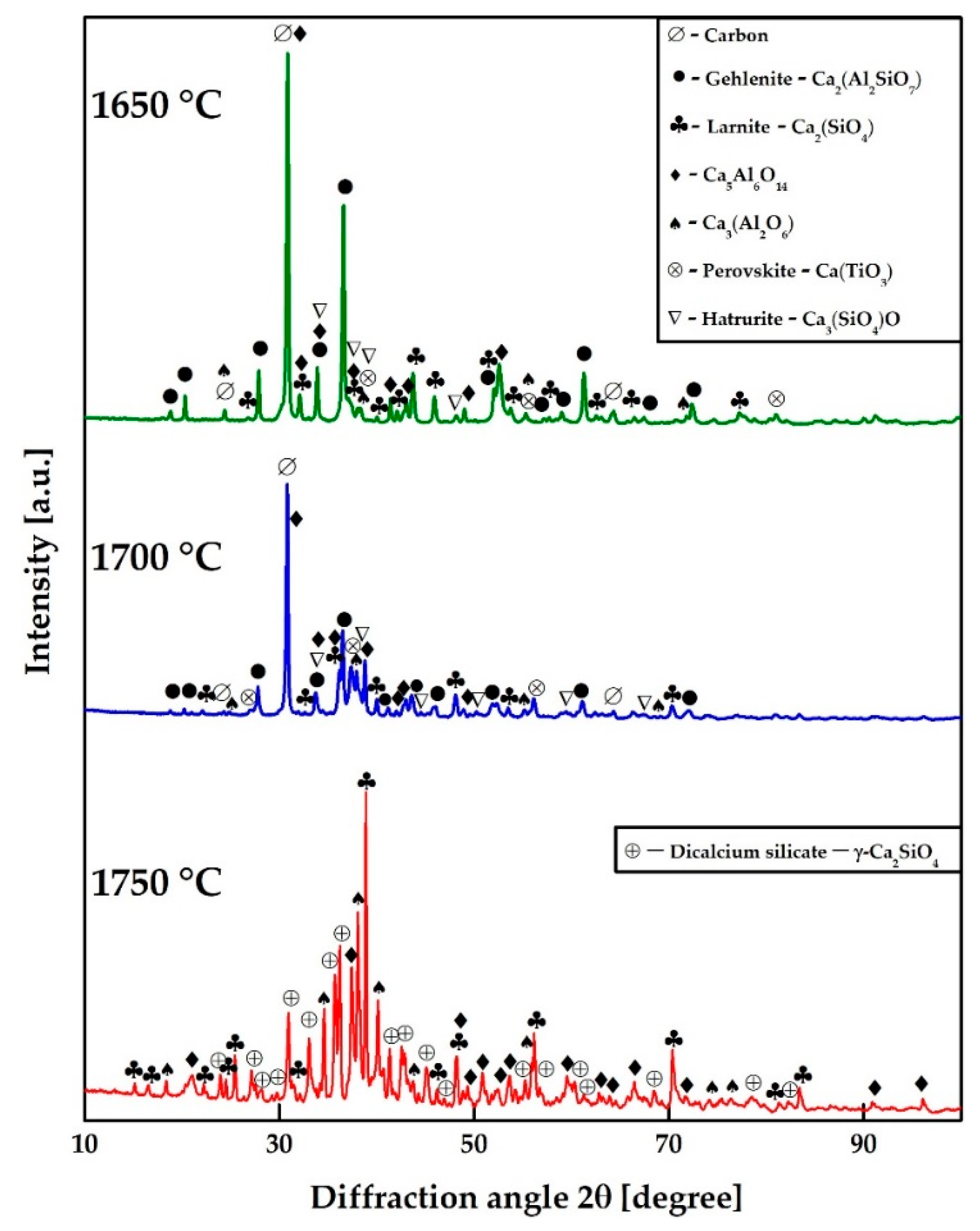
3.3. Reductive Smelting of the Neutralized Red Mud
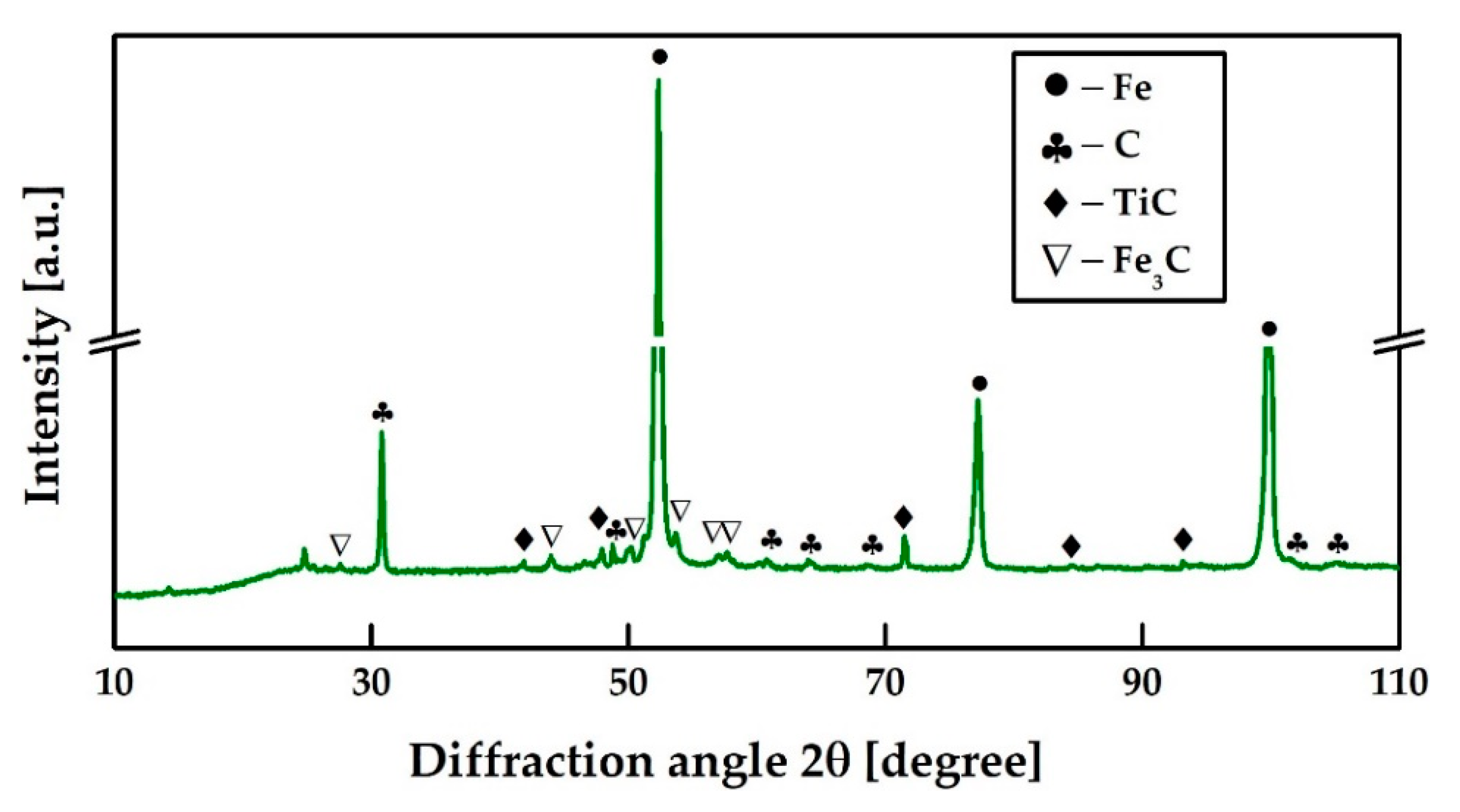
3.4. Composition of Pig Iron
3.5. Pig Iron Application
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Evans, K. The History, Challenges, and New Developments in the Management and Use of Bauxite Residue. J. Sustain. Metall. 2016, 2, 316–331. [Google Scholar] [CrossRef]
- Zhang, R.; Zheng, S.; Ma, S.; Zhang, Y. Recovery of alumina and alkali in Bayer red mud by the formation of andradite-grossular hydrogarnet in hydrothermal process. J. Hazard. Mater. 2011, 189, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Dmitriev, A. The Comprehensive Utilisation of Red Mud Utilisation in Blast Furnace. In Recovery and Utilization of Metallurgical Solid Waste; Zhang, Y., Ed.; IntechOpen: London, UK, 2019; ISBN 978-1-78985-102-1. [Google Scholar]
- Tabereaux, A.T. Hungarian Red Mud Disaster: Addressing Environmental Liabilities of Alumina Residue Storage & Disposal. Light Met. Age 2010, 68, 22–24. [Google Scholar]
- Khairul, M.A.; Zanganeh, J.; Moghtaderi, B. The composition, recycling and utilisation of Bayer red mud. Resour. Conserv. Recycl. 2019, 141, 483–498. [Google Scholar] [CrossRef]
- Vind, J.; Alexandri, A.; Vassiliadou, V.; Panias, D.; Vind, J.; Alexandri, A.; Vassiliadou, V.; Panias, D. Distribution of Selected Trace Elements in the Bayer Process. Metals 2018, 8, 327. [Google Scholar] [CrossRef]
- Vind, J.; Malfliet, A.; Blanpain, B.; Tsakiridis, P.; Tkaczyk, A.; Vassiliadou, V.; Panias, D.; Vind, J.; Malfliet, A.; Blanpain, B.; et al. Rare Earth Element Phases in Bauxite Residue. Minerals 2018, 8, 77. [Google Scholar] [CrossRef]
- Akcil, A.; Akhmadiyeva, N.; Abdulvaliyev, R.; Meshram, P. Overview on Extraction and Separation of Rare Earth Elements from Red Mud: Focus on Scandium. Miner. Process. Extr. Metall. Rev. 2018, 39, 145–151. [Google Scholar] [CrossRef]
- Gräfe, M.; Power, G.; Klauber, C. Bauxite residue issues: III. Alkalinity and associated chemistry. Hydrometallurgy 2011, 108, 60–79. [Google Scholar] [CrossRef]
- Kaußen, F.; Friedrich, B. Reductive Smelting of Red Mud for Iron Recovery. Chem. Ing. Tech. 2015, 87, 1535–1542. [Google Scholar] [CrossRef]
- Borra, C.R.; Blanpain, B.; Pontikes, Y.; Binnemans, K.; Van Gerven, T. Smelting of Bauxite Residue (Red Mud) in View of Iron and Selective Rare Earths Recovery. J. Sustain. Metall. 2016, 2, 28–37. [Google Scholar] [CrossRef]
- Kaußen, F.M.; Friedrich, B. Phase characterization and thermochemical simulation of (landfilled) bauxite residue (“red mud”) in different alkaline processes optimized for aluminum recovery. Hydrometallurgy 2018, 176, 49–61. [Google Scholar] [CrossRef]
- Anisonyan, K.G.; Kopyev, D.Y.; Goncharov, K.V.; Sadykhov, G.B. An investigation of a single-stage red mud reducing roasting process with the cast iron and aluminate slag production. Non-Ferr. Met. 2018, 44, 18–23. [Google Scholar] [CrossRef]
- Ning, G.; Zhang, B.; Liu, C.; Li, S.; Ye, Y.; Jiang, M.; Ning, G.; Zhang, B.; Liu, C.; Li, S.; et al. Large-Scale Consumption and Zero-Waste Recycling Method of Red Mud in Steel Making Process. Minerals 2018, 8, 102. [Google Scholar] [CrossRef]
- Luo, M.; Qi, X.; Zhang, Y.; Ren, Y.; Tong, J.; Chen, Z.; Hou, Y.; Yeerkebai, N.; Wang, H.; Feng, S.; et al. Study on dealkalization and settling performance of red mud. Environ. Sci. Pollut. Res. 2017, 24, 1794–1802. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, T.; Lyu, G.; Guo, F.; Zhang, W.; Zhang, Y. Recovery of alkali and alumina from bauxite residue (red mud) and complete reuse of the treated residue. J. Clean. Prod. 2018, 188, 456–465. [Google Scholar] [CrossRef]
- Geerdes, M.; Chaigneau, R.; Kurunov, I. Modern Blast Furnace Ironmaking: An Introduction, 3rd ed.; IOS Press: Amsterdam, The Netherlands, 2015; ISBN 9781614994992. [Google Scholar]
- Deng, B.; Li, G.; Luo, J.; Ye, Q.; Liu, M.; Peng, Z.; Jiang, T. Enrichment of Sc2O3 and TiO2 from bauxite ore residues. J. Hazard. Mater. 2017, 331, 71–80. [Google Scholar] [CrossRef]
- Liu, Z.; Li, H. Metallurgical process for valuable elements recovery from red mud—A review. Hydrometallurgy 2015, 155, 29–43. [Google Scholar] [CrossRef]
- Yuan, M.; Qiao, X.; Yu, J. Phase equilibria of AlCl3 + FeCl3 + H2O, AlCl3 + CaCl2 + H2O, and FeCl3 + CaCl2 + H2O at 298.15 K. J. Chem. Eng. Data 2016, 61, 1749–1755. [Google Scholar] [CrossRef]
- Wang, J.; Petit, C.; Zhang, X.; Cui, S. Phase Equilibrium Study of the AlCl3-CaCl2-H2O System for the Production of Aluminum Chloride Hexahydrate from Ca-Rich Flue Ash. J. Chem. Eng. Data 2016, 61, 359–369. [Google Scholar] [CrossRef]
- Cui, L.; Cheng, F.; Zhou, J. Preparation of high purity AlCl3·6H2O crystals from coal mining waste based on iron (III) removal using undiluted ionic liquids. Sep. Purif. Technol. 2016, 167, 45–54. [Google Scholar] [CrossRef]
- Guo, Y.; Yang, X.; Cui, H.; Cheng, F.; Yang, F. Crystallization behavior of AlCl3·6H2O in hydrochloric system. Huagong Xuebao/CIESC J. 2014, 65, 3960–3967. [Google Scholar] [CrossRef]
- Bonomi, C.; Alexandri, A.; Vind, J.; Panagiotopoulou, A.; Tsakiridis, P.; Panias, D.; Bonomi, C.; Alexandri, A.; Vind, J.; Panagiotopoulou, A.; et al. Scandium and Titanium Recovery from Bauxite Residue by Direct Leaching with a Brønsted Acidic Ionic Liquid. Metals 2018, 8, 834. [Google Scholar] [CrossRef]
- Yagmurlu, B.; Dittrich, C.; Friedrich, B. Effect of aqueous media on the recovery of scandium by selective precipitation. Metals 2018, 8, 314. [Google Scholar] [CrossRef]
- Onghena, B.; Borra, C.R.; Van Gerven, T.; Binnemans, K. Recovery of scandium from sulfation-roasted leachates of bauxite residue by solvent extraction with the ionic liquid betainium bis (trifluoromethylsulfonyl) imide. Sep. Purif. Technol. 2017, 176, 208–219. [Google Scholar] [CrossRef]
- Zhu, X.; Li, W.; Tang, S.; Zeng, M.; Bai, P.; Chen, L. Selective recovery of vanadium and scandium by ion exchange with D201 and solvent extraction using P507 from hydrochloric acid leaching solution of red mud. Chemosphere 2017, 175, 365–372. [Google Scholar] [CrossRef]
- Alkan, G.; Schier, C.; Gronen, L.; Stopic, S.; Friedrich, B. A Mineralogical Assessment on Residues after Acidic Leaching of Bauxite Residue (Red Mud) for Titanium Recovery. Metals 2017, 7, 458. [Google Scholar] [CrossRef]
- Zhu, X.; Li, W.; Guan, X. Kinetics of titanium leaching with citric acid in sulfuric acid from red mud. Trans. Nonferr. Met. Soc. China 2015, 25, 3139–3145. [Google Scholar] [CrossRef]
- Shiryaeva, E.V.; Podgorodetskiy, G.S.; Malyscheva, T.Y.; Gorbunov, V.B.; Zavodyaniy, A.V.; Schapovalov, A.N. Influence of low alkaline red mud on the properties and microstructure of the agglomerates from the charge materials JSC “Ural steel. ” Izv. Vyss. Uchebnykh Zaved. Chernaya Metall. 2014, 57, 14–19. [Google Scholar] [CrossRef][Green Version]
- Liu, G.; Liu, Y.; Zhang, T. Approaches to improve alumina extraction based on the phase transformation mechanism of recovering alkali and extracting alumina by the calcification-carbonization method. Hydrometallurgy 2019, 189. [Google Scholar] [CrossRef]
- Zhu, X.-F.; Zhang, T.-A.; Wang, Y.-X.; Lü, G.-Z.; Zhang, W.-G. Recovery of alkali and alumina from Bayer red mud by the calcification–carbonation method. Int. J. Miner. Metall. Mater. 2016, 23, 257–268. [Google Scholar] [CrossRef]
- Bale, C.; Chartrand, P.; Degterov, S.A.; Eriksson, G.; Hack, K.; Ben Mahfoud, R.; Melancon, J.; Pelton, A.D.; Petersen, S. FactSage thermochemical software and databases. Calphad 2002, 26, 189–228. [Google Scholar] [CrossRef]
- Roine, A.; Kotiranta, T. Development of sustainable processes with new HSC Chemistry 6.0 software. In Proceedings of the European Metallurgical Conference, EMC 2007, Dusseldorf, Germany, 11–14 June 2007; Volume 4, pp. 1877–1888. [Google Scholar]
- Zinoveev, D.; Grudinsky, P.; Korneev, V.; Dyubanov, V.; Zheleznyi, M. Recycling red mud of JSC ural aluminum plant with the recovery of iron and construction materials. Key Eng. Mater. 2017, 743, 331–337. [Google Scholar] [CrossRef]
- Pankratov, D.A. Mössbauer study of oxo derivatives of iron in the Fe2O3-Na2O2 system. Inorg. Mater. 2014, 50, 82–89. [Google Scholar] [CrossRef]
- Raspopov, N.A.; Korneev, V.P.; Averin, V.V.; Lainer, Y.A.; Zinoveev, D.V.; Dyubanov, V.G. Reduction of iron oxides during the pyrometallurgical processing of red mud. Russ. Metall. 2013, 2013, 33–37. [Google Scholar] [CrossRef]
- Karimi, E.; Teixeira, I.F.; Ribeiro, L.P.; Gomez, A.; Lago, R.M.; Penner, G.; Kycia, S.W.; Schlaf, M. Ketonization and deoxygenation of alkanoic acids and conversion of levulinic acid to hydrocarbons using a Red Mud bauxite mining waste as the catalyst. Catal. Today 2012, 190, 73–88. [Google Scholar] [CrossRef]
- Seitz, G.; Penin, N.; Decoux, L.; Wattiaux, A.; Duttine, M.; Gaudon, M. Near the Ferric Pseudobrookite Composition (Fe2TiO5). Inorg. Chem. 2016, 55, 2499–2507. [Google Scholar] [CrossRef]
- Muranaka, S.; Shinjo, T.; Bando, Y.; Takada, T. Mossbauer Study of Fe2TiO5 and FeTi2O5. J. Phys. Soc. Jpn. 1971, 30, 890. [Google Scholar] [CrossRef]
- Onodera, H.; Fujita, A.; Yamamoto, H.; Sagawa, M.; Hirosawa, S. Mössbauer study of the intermetallic compound Nd2Fe14B. I. interpretation of complex spectrum. J. Magn. Magn. Mater. 1987, 68, 6–14. [Google Scholar] [CrossRef]
- Rostovshchikova, T.N.; Smirnov, V.V.; Tsodikov, M.V.; Bukhtenko, O.V.; Maksimov, Y.V.; Kiseleva, O.I.; Pankratov, D.A. Catalytic conversions of chloroolefins over iron oxide nanoparticles: 1. Isomerization of dichlorobutenes in the presence of iron oxide nanopaticles immobilized on silicas with different structures. Russ. Chem. Bull. 2005, 54, 1418–1424. [Google Scholar] [CrossRef]
- Oh, S.J.; Cook, D.C.; Townsend, H.E. Characterization of Iron Oxides Commonly Formed as Corrosion Products on Steel. Hyperfine Interact. 1998, 112, 59–66. [Google Scholar] [CrossRef]
- Pankratov, D.A.; Yurev, A.I. Mössbauer diagnostics of the isomorphic substitution of iron for aluminum in triclinic iron vanadate. Bull. Russ. Acad. Sci. Phys. 2013, 77, 759–764. [Google Scholar] [CrossRef]
- Jónás, K.; Solymár, K.; Zöldi, J. Some applications of mössbauer spectroscopy for the quantitative analysis of minerals and mineral mixtures. J. Mol. Struct. 1980, 60, 449–452. [Google Scholar] [CrossRef]
- Morin, F.J. Magnetic susceptibility of α-Fe2O3 and α-Fe2O3 with added titanium. Phys. Rev. 1950, 78, 819–820. [Google Scholar] [CrossRef]
- Van San, E.; De Grave, E.; Vandenberghe, R.E.; Desseyn, H.O.; Datas, L.; Barrón, V.; Rousset, A. Study of Al-substituted hematites, prepared from thermal treatment of lepidocrocite. Phys. Chem. Miner. 2001, 28, 488–497. [Google Scholar] [CrossRef]
- Azof, F.I.; Kolbeinsen, L.; Safarian, J. Characteristics of Calcium-Aluminate Slags and Pig Iron Produced from Smelting-Reduction of Low-Grade Bauxites. Metall. Mater. Trans. B Process Metall. Mater. Process. Sci. 2018, 49, 2400–2420. [Google Scholar] [CrossRef]
- Sajitha, E.P.; Prasad, V.; Subramanyam, S.V.; Mishra, A.K.; Sarkar, S.; Bansal, C. Size-dependent magnetic properties of iron carbide nanoparticles embedded in a carbon matrix. J. Phys. Condens. Matter 2007, 19. [Google Scholar] [CrossRef]
- Yurkov, G.Y.; Shashkeev, K.A.; Kondrashov, S.V.; Popkov, O.V.; Shcherbakova, G.I.; Zhigalov, D.V.; Pankratov, D.A.; Ovchenkov, E.A.; Koksharov, Y.A. Synthesis and magnetic properties of cobalt ferrite nanoparticles in polycarbosilane ceramic matrix. J. Alloys Compd. 2016, 686, 421–430. [Google Scholar] [CrossRef]
- Fruchart, R.; Roger, A.; Senateur, J.P. Crystallographic and magnetic properties of solid solutions of the phosphides M2P, M = Cr, Mn, Fe, Co, and Ni. J. Appl. Phys. 1969, 40, 1250–1257. [Google Scholar] [CrossRef]
- Bailey, R.E.; Duncan, J.F. Moessbauer And Nuclear Magnetic Resonance Studies of Several Iron Phosphides. Inorg. Chem. 1967, 6, 1444–1447. [Google Scholar] [CrossRef]
- Presniakov, I.A.; Sobolev, A.V.; Chernyavskii, I.O.; Pankratov, D.A.; Morozov, I.V. Anisotropic magnetic hyperfine interactions in phosphide FeP. Bull. Russ. Acad. Sci. Phys. 2015, 79, 984–989. [Google Scholar] [CrossRef]
- Alleg, S.; Brahimi, A.; Azzaza, S.; Souilah, S.; Zergoug, M.; Suňol, J.J.; Greneche, J.M. X-ray diffraction, Mössbauer spectrometry and thermal studies of the mechanically alloyed (Fe1−xMnx)2P powders. Adv. Powder Technol. 2018, 29, 257–265. [Google Scholar] [CrossRef]
- Fionov, A.S.; Yurkov, G.Y.; Kolesov, V.V.; Pankratov, D.A.; Ovchenkov, E.A.; Koksharov, Y.A. Composite material based on iron containing nanoparticles for applications in the problems of electromagnetic compatibility. J. Commun. Technol. Electron. 2012, 57, 543–552. [Google Scholar] [CrossRef]
- Sauer, W.E.; Reynik, R.J. Electronic and Magnetic Structure of Dilute Iron-Base Alloys. J. Appl. Phys. 1971, 42, 1604–1605. [Google Scholar] [CrossRef]
- Lubyanoi, D.A.; Drobyshev, A.N.; Samsonov, Y.A.; Kaminskaya, I.A. Higher Efficiency of Manufacturing of Steel Casting Equipment from Pig Iron. Steel Transl. 1994, 6, 40–41. [Google Scholar]
- Andreev, V.V.; Lubyanoi, D.A.; Samsonov, Y.N.; Kaminskaya, I.A.; Lubyanaya, S.V. Development of Extra-Furnace Treatment Technology for Blast-Furnace Iron in Order to Manufacture Replacement Metallurgical Equipment with Improved Operating Life. Metallurgist 2014, 58, 492–495. [Google Scholar] [CrossRef]
- Berns, H.; Wewers, B. Development of an abrasion resistant steel composite with in situ TiC particles. Wear 2001, 251, 1386–1395. [Google Scholar] [CrossRef]
- Lubyanoi, D.A.; Sofroshenkov, A.F.; Sinyavskii, I.A.; Makarov, E.S.; Gorkavenko, V.V. Technology of neutralizing the harmful effect of phosphorus in iron of heat-resistant castings. Steel Transl. 1999, 29, 86–90. [Google Scholar]
- Lubyanoj, D.A.; Sinyavskij, I.A.; Selyanin, I.F.; Sofroshenkov, A.F.; Morozova, O.V. Influence of conditions of ladle treatment on structure, casting and mechanical properties of phosphorus-containing cast iron produced in blast furnaces. Izv. Ferr. Metall. 2001, 6, 53–55. [Google Scholar]
- Lubyanoi, D.A.; Gorkavenko, V.V.; Makarov, É.S.; Kaminskaya, I.A.; Frolov, A.G.; Yakovenko, N.A. Phosphorous cast iron for heat-resistant castings. Met. Sci. Heat Treat. 2002, 44, 452–453. [Google Scholar] [CrossRef]





| Fe2O3 | SiO2 | Al2O3 | TiO2 | CaO | MgO | MnO | Na2O | P2O5 | S | LOI * |
|---|---|---|---|---|---|---|---|---|---|---|
| 36.9 | 8.71 | 11.8 | 3.54 | 23.8 | 1.01 | 0.95 | 0.27 | 0.42 | 0.14 | 12.46 |
| Temperature, K | # | Compound or Site | δ | Δ = 2ε | Γexp (mm/s) | a+ | a− | Heff (kOe) | S# (at%) |
|---|---|---|---|---|---|---|---|---|---|
| 296 | 1 | α-Fe2O3 | 0.37 | −0.21 | 0.26 | - | - | 513.2 | 25 |
| 2 | α-(Fe1−xAlx)2O3 | 0.37 | −0.22 | 0.27 | - | - | 505.4 | 16 | |
| 3 | 0.37 | −0.20 | 0.39 | - | - | 491.3 | 11 | ||
| 4 | 0.37 | −0.18 | 0.72 | - | - | 467 | 10 | ||
| 5 | α-Fe1−xAlxOOH | 0.35 | −0.20 | 1.55 | - | - | 362 | 14 | |
| 6 | - | - | 183 | 4 | |||||
| 7 | Fe2+O | 1.13 | 2.64 | 0.32 | - | - | - | 10 | |
| 8 | Fe3+O | 0.35 | 0.68 | 0.67 | - | - | - | 10 | |
| 77.5 | 1 | α-Fe2O3 | 0.48 | −0.21 | 0.26 | −0.016 | −0.007 | 530.8 | 32 |
| 2 | α-(Fe1−xAlx)2O3 | 0.48 | −0.21 | 0.37 | - | - | 524.5 | 30 | |
| 3 | α-Fe1−xAlxOOH | 0.47 | −0.23 | 0.67 | - | - | 496.7 | 20 | |
| 4 | Fe2+O | 1.25 | 2.83 | 0.30 | - | - | - | 11 | |
| 5 | Fe3+O | 0.46 | 0.69 | 0.58 | - | - | - | 8 |
| Temperature | T.Fe * | CaO | Al2O3 | SiO2 | TiO2 | MgO | MnO | P2O5 | S |
|---|---|---|---|---|---|---|---|---|---|
| 1650 °C | 8.06 | 43.34 | 18.18 | 22.23 | 3.75 | 1.53 | 0.31 | 0.084 | 0.31 |
| 1700 °C | 2.82 | 50.41 | 21.46 | 14.46 | 6.64 | 1.71 | 0.13 | 0.032 | 0.33 |
| 1750 °C | 0.16 | 61.79 | 24.37 | 11.47 | 0.2 | 0.57 | 0.02 | 0.03 | 0.29 |
| T, °C | ΔG, kJ·mol−1 |
|---|---|
| 1500 | 22.756 |
| 1550 | 6.914 |
| 1600 | −8.887 |
| 1650 | −24.650 |
| 1700 | −40.376 |
| 1750 | −56.068 |
| 1800 | −71.725 |
| Temperature | Si | Mn | Ti | V | P | S |
|---|---|---|---|---|---|---|
| 1700 °C | 0.35 | 0.45 | 0.45 | 0.05 | 0.32 | <0.005 |
| 1750 °C | 1.17 | 1.35 | 1.12 | 0.49 | 0.96 | <0.005 |
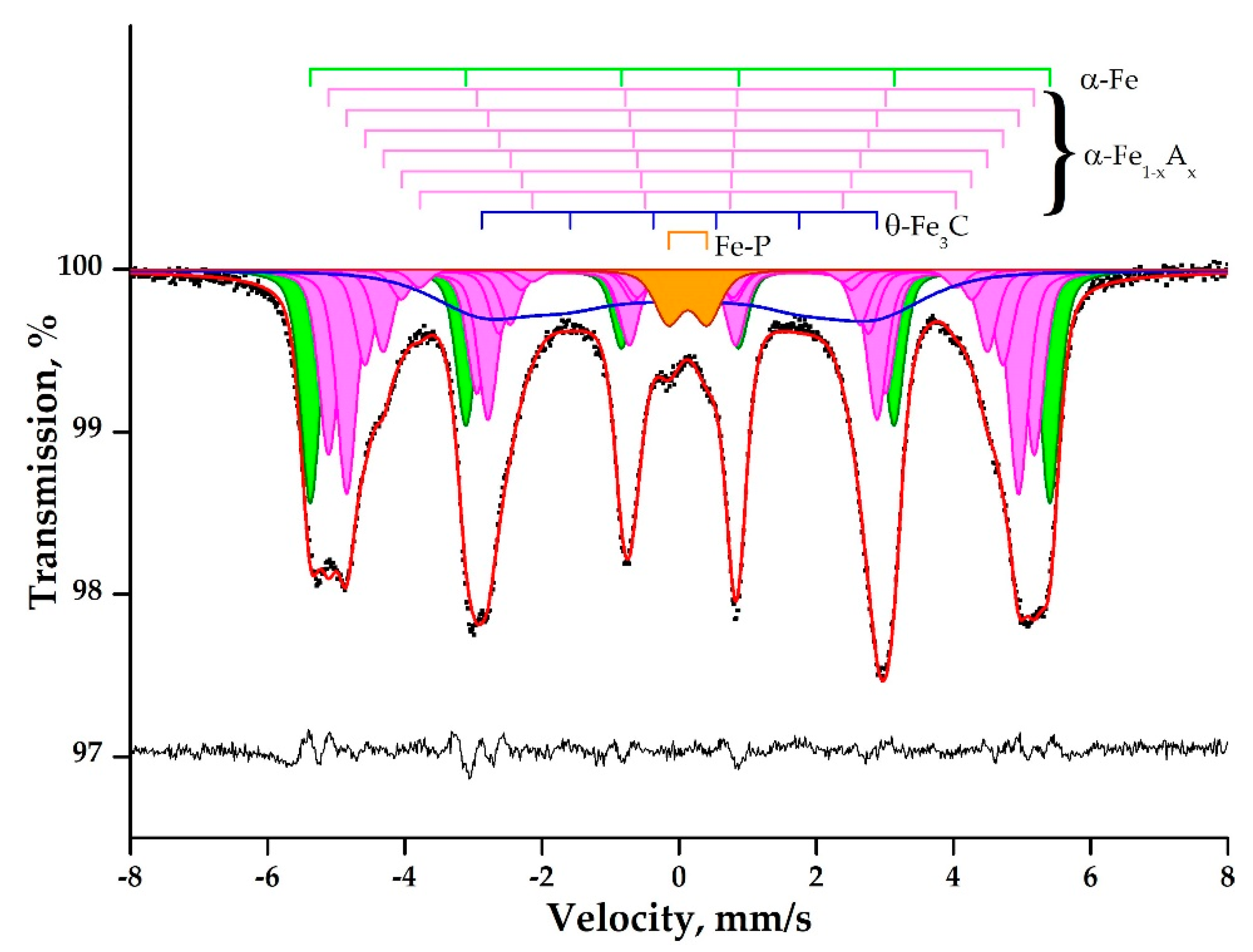
| # | Compound or Site | δ | dδ | Δ = 2ε | Γexp | Heff (kOe) | dH | S# |
|---|---|---|---|---|---|---|---|---|
| (mm/s) | (at %) | |||||||
| 1 | α-Fe | 0.013 | 0.0188 | 0.001 | 0.343 | 334.4 | 15.36 | 20.7 |
| 2 | α-Fe1−xAx | 0.032 | 319.0 | 16.5 | ||||
| 3 | 0.051 | 303.6 | 19.9 | |||||
| 4 | 0.070 | 288.3 | 8.4 | |||||
| 5 | 0.089 | 272.9 | 7.3 | |||||
| 6 | 0.108 | 257.6 | 2.6 | |||||
| 7 | 0.126 | 242.2 | 1.7 | |||||
| 8 | θ-Fe3C | 0.04 | - | −0.04 | 1.63 | 178.6 | - | 19.9 |
| 9 | Fe-P | 0.13 | - | 0.55 | 0.45 | - | - | 3.0 |
| Phase | Impression Diagonal, µm | Average Diagonal, µm | Vickers Hardness |
|---|---|---|---|
| TiC | 4.46; 4.13; 4.13; 4.39; 4.01; 4.4 | 4.25 | 5130 |
| Fe3P | 9.9; 9.9; 10.24; 9.8; | 9.96 | 934 |
| Perlite | 16.06; 16.27; 17.47 | 16.60 | 336 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valeev, D.; Zinoveev, D.; Kondratiev, A.; Lubyanoi, D.; Pankratov, D. Reductive Smelting of Neutralized Red Mud for Iron Recovery and Produced Pig Iron for Heat-Resistant Castings. Metals 2020, 10, 32. https://doi.org/10.3390/met10010032
Valeev D, Zinoveev D, Kondratiev A, Lubyanoi D, Pankratov D. Reductive Smelting of Neutralized Red Mud for Iron Recovery and Produced Pig Iron for Heat-Resistant Castings. Metals. 2020; 10(1):32. https://doi.org/10.3390/met10010032
Chicago/Turabian StyleValeev, Dmitry, Dmitry Zinoveev, Alex Kondratiev, Dmitry Lubyanoi, and Denis Pankratov. 2020. "Reductive Smelting of Neutralized Red Mud for Iron Recovery and Produced Pig Iron for Heat-Resistant Castings" Metals 10, no. 1: 32. https://doi.org/10.3390/met10010032
APA StyleValeev, D., Zinoveev, D., Kondratiev, A., Lubyanoi, D., & Pankratov, D. (2020). Reductive Smelting of Neutralized Red Mud for Iron Recovery and Produced Pig Iron for Heat-Resistant Castings. Metals, 10(1), 32. https://doi.org/10.3390/met10010032








