From Ni–P Metastable Alloy Nanoparticles to Bulk Submicrometer Grain-Sized MMCs with Tunable Mechanical and Magnetic Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis
2.2. Characterization Techniques
3. Results
3.1. X-Ray Diffraction and TEM Characterizations
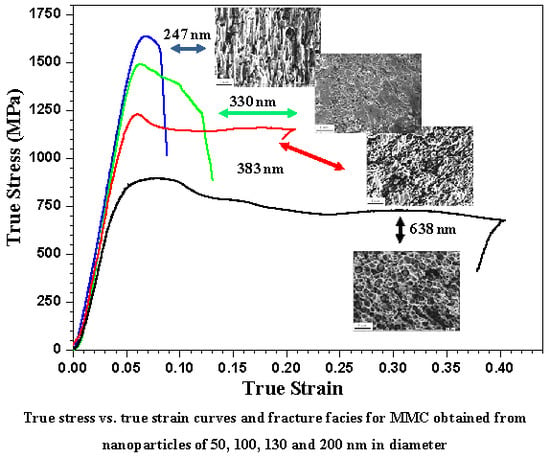
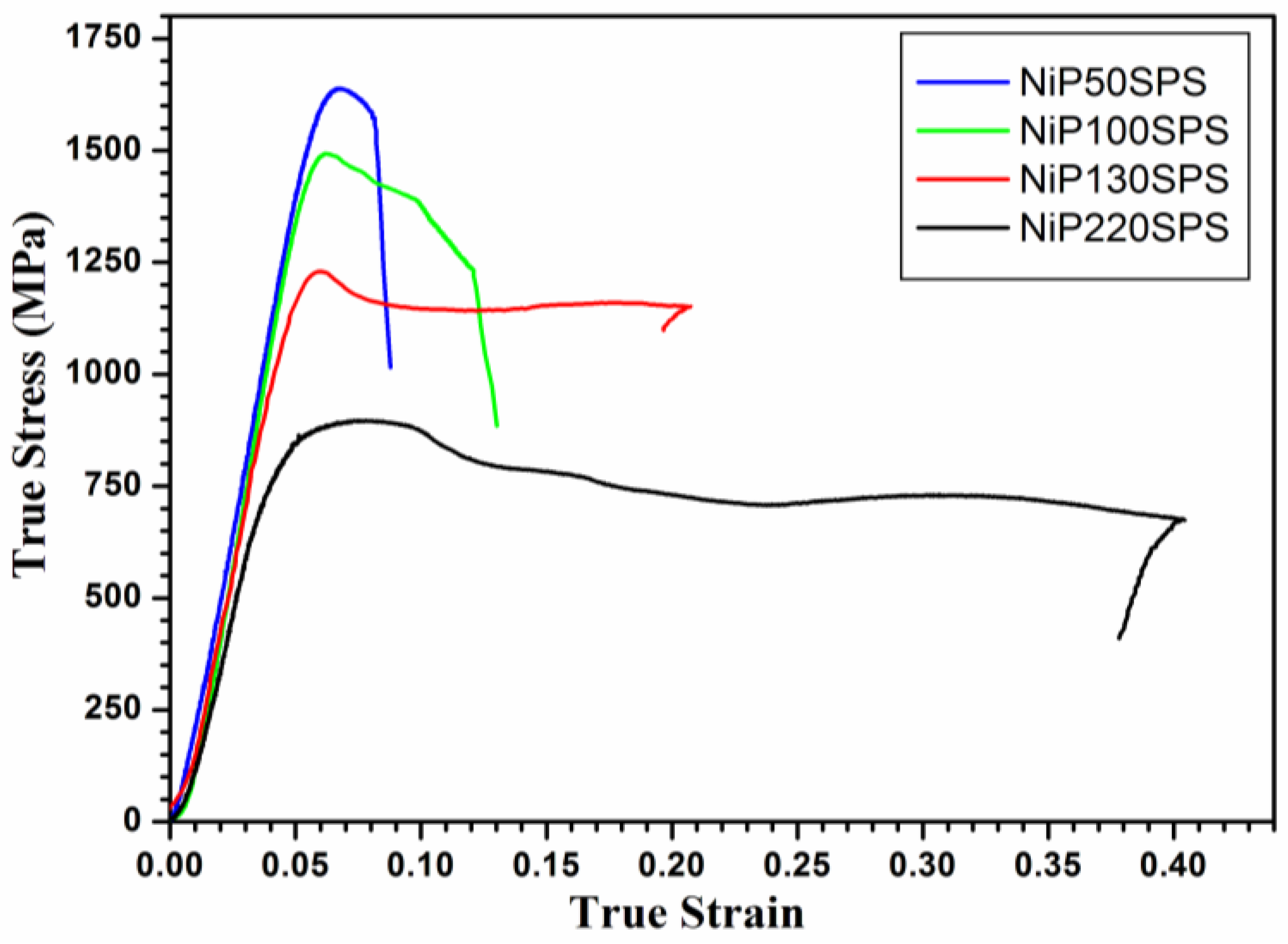
3.2. Compressive Test
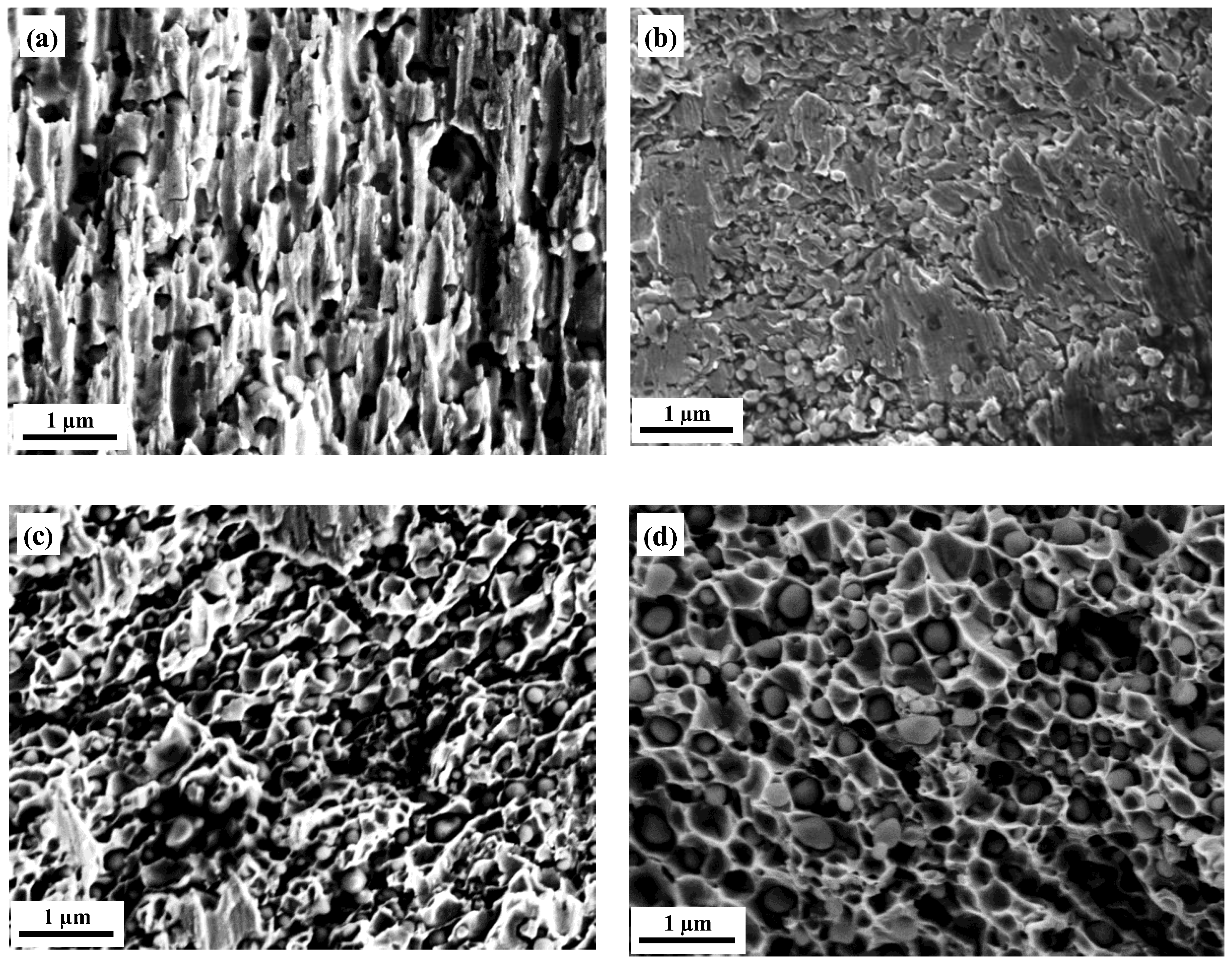
3.3. Fracture Behaviors
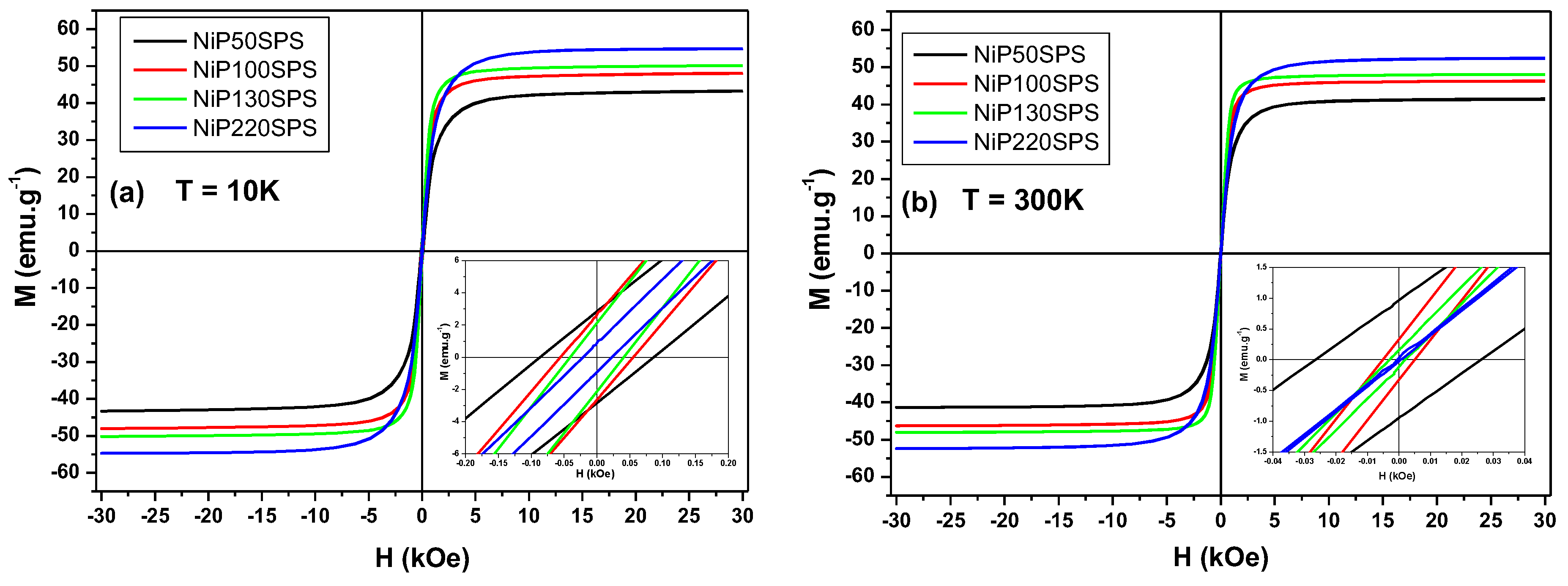
3.4. Magnetic Properties
4. Discussion
- -
- -
- The difference in microstructure of these materials induced by the elaboration process. In the present study, the R-SPS process led to MMCs based on nickel with Ni3P as a reinforcement starting from nanoparticles of Ni–P metastable alloys. The formation of Ni3P occurred in situ by diffusion of P from the inside of the nanoparticles to their boundaries. This mechanism leads to strong chemical bonding between matrix and reinforcement components [38].
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Anselmi-Tamburini, U.; Garay, J.E.; Munir, Z.A. Fast low-temperature consolidation of bulk nanometric ceramic materials. Scripta Mater. 2006, 54, 823–828. [Google Scholar] [CrossRef]
- Cabouro, G.; Le Gallet, S.; Chevalier, S.; Gaffet, E.; Grin, Y.; Bernard, F. Dense Mosi2 produced by reactive flash sintering: Control of Mo/Si agglomerates prepared by high-energy ball milling. Powder Technol. 2011, 208, 526–531. [Google Scholar] [CrossRef]
- Orrù, R.; Cao, G. Comparison of Reactive and Non-Reactive Spark Plasma Sintering Routes for the Fabrication of Monolithic and Composite Ultra High Temperature Ceramics (UHTC) Materials. Materials 2013, 6, 1566. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, J. Rapid reactive synthesis and sintering of textured Ca3Co4O9 ceramics by spark plasma sintering. J. Mater. Process. Tech. 2008, 208, 70–74. [Google Scholar] [CrossRef]
- Turki, F.; Abderrazak, H.; Schoenstein, F.; Abdellaoui, M.; Jouini, N. SPS parameters influence on Ti3SiC2 formation from Si/TiC: Mechanical properties of the bulk materials. J. Alloys Compd. 2017, 708, 123–133. [Google Scholar] [CrossRef]
- Patissier, A.; Paul-Boncour, V. Fast synthesis of LaFe13−xSix magnetocaloric compounds by reactive Spark Plasma Sintering. J. Alloys Compd. 2015, 645, 143–150. [Google Scholar] [CrossRef]
- Bousnina, M.A.; Schoenstein, F.; Smiri, L.S.; Jouini, N. Facile synthesis of metastable Ni–P nanostructured materials by a novel bottom-up strategy. Solid State Sci. 2015, 40, 13–19. [Google Scholar] [CrossRef]
- Bousnina, M.A.; Turki, F.; Schoenstein, F.; Têtard, F.; Rabu, P.; Smiri, L.S.; Jouini, N. Bulk nanostructured NiP alloys: Elaboration from metastable NiP nanoparticles by spark plasma sintering; mechanical and magnetic properties. J. Alloys Compd. 2016, 686, 252–266. [Google Scholar] [CrossRef]
- Dakhlaoui-Omrani, A.; Bousnina, M.A.; Smiri, L.S.; Taibi, M.; Leone, P.; Schoenstein, F.; Jouini, N. Elaboration of nickel nanoparticles by modified polyol process and their spark plasma sintering, characterization and magnetic properties of the nanoparticles and the dense nano-structured material. Mater. Chem. Phys. 2010, 123, 821–828. [Google Scholar] [CrossRef]
- Roisnel, T.; Rodríguez-Carvajal, J. WinPLOTR: A Windows tool for powder diffraction patterns analysis. Mater. Sci. Forum 2001, 378, 118–123, www.cdifx.univ-rennes1.fr/winplotr/winplotr.htm. [Google Scholar] [CrossRef]
- Rietveld, H. A profile refinement method for nuclear and magnetic structures. J. Appl. Cryst. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Schmetterer, C.; Vizdal, J.; Ipser, H. A new investigation of the system Ni–P. Intermetallics 2009, 17, 826–834. [Google Scholar] [CrossRef]
- Bui, Q.H.; Dirras, G.; Ramtani, S.; Gubicza, J. On the strengthening behavior of ultrafine-grained nickel processed from nanopowders. Mater. Sci. Eng. A 2010, 527, 3227–3235. [Google Scholar] [CrossRef]
- Gubicza, J.; Bui, H.Q.; Fellah, F.; Dirras, G.F. Microstructure and mechanical behavior of ultrafine-grained Ni processed by different powder metallurgy methods. J. Mater. Res. 2009, 24, 217–226. [Google Scholar] [CrossRef]
- Prasad, M.J.N.V.; Chokshi, A.H. Microstructural stability and superplasticity in an electrodeposited nanocrystalline Ni–P alloy. Acta Mater. 2011, 59, 4055–4067. [Google Scholar] [CrossRef]
- Weil, L.; Marfoure, S. Variation thermique du champ coercitif du nickel aggloméré. J. Phys. Radium 1947, 8, 358–361. [Google Scholar] [CrossRef]
- Ni, X.; Zhao, Q.; Zheng, H.; Li, B.; Song, J.; Zhang, D.; Zhang, X. A Novel Chemical Reduction Route towards the Synthesis of Crystalline Nickel Nanoflowers from a Mixed Source. Eur. J. Inorg. Chem. 2005, 223, 4788–4793. [Google Scholar] [CrossRef]
- You, Y.; Gu, C.; Wang, X.; Tu, J. Electrochemical Synthesis and Characterization of Ni–P Alloy Coatings from Eutectic–Based Ionic Liquid. J. Electrochem. Soc. 2012, 159, D642–D648. [Google Scholar] [CrossRef]
- Gu, C.; Lian, J.; Li, G.; Niu, L.; Jiang, Z. High corrosion-resistant Ni–P/Ni/Ni–P multilayer coatings on steel. Surf. Coat. Tech. 2005, 197, 61–67. [Google Scholar] [CrossRef]
- Changgeng, X.; Xinmin, H.; Zonggang, D.; Yanwen, W. Properties of electroless Ni-P and Ni-P-SiC. Plat. Surf. Finish. 1989, 76, 90–93. [Google Scholar]
- Keong, K.G.; Sha, W.; Malinov, S. Hardness evolution of electroless nickel–phosphorus deposits with thermal processing. Surf. Coat. Technol. 2003, 168, 263–274. [Google Scholar] [CrossRef]
- Guo, Z.; Keong, K.G.; Sha, W. Crystallisation and phase transformation behaviour of electroless nickel phosphorus platings during continuous heating. J. Alloys Compd. 2003, 358, 112–119. [Google Scholar] [CrossRef]
- Deng, J.; Chen, H.; Bao, X.; Muhler, M. The effect of cyclic oxidation-reduction pretreatments on an amorphous Ni80P20 catalyst: An XPS/UPS/ISS study. Appl. Surf. Sci. 1994, 81, 341–346. [Google Scholar] [CrossRef]
- Wang, Y.; Brogan, K.; Tung, S.C. Wear and scuffing characteristics of composite polymer and nickel/ceramic composite coated piston skirts against aluminum and cast iron cylinder bores. Wear 2001, 250, 706–717. [Google Scholar] [CrossRef]
- Krishnan, K.H.; John, S.; Srinivasan, K.N.; Praveen, J.; Ganesan, M.; Kavimani, P.M. An overall aspect of electroless Ni-P depositions-A review article. Metall. Mater. Trans. A 2006, 37, 1917–1926. [Google Scholar] [CrossRef]
- Bousnina, M.A.; Dakhlaoui-Omrani, A.; Schoenstein, F.; Madec, P.; Haddadi, H.; Smiri, L.S.; Jouini, N. Spark plasma sintering and hot isostatic pressing of nickel nanopowders elaborated by a modified polyol process and their microstructure, magnetic and mechanical characterization. J. Alloys Compd. 2010, 504, S323–S327. [Google Scholar] [CrossRef]
- Panagopoulos, C.N.; Papachristos, V.D.; Sigalas, C. Tensile behaviour of as deposited and heat-treated electroless Ni-P deposits. J. Mater. Sci. 1999, 34, 2587–2600. [Google Scholar] [CrossRef]
- Deng, J.-F.; Li, H.; Wang, W. Progress in design of new amorphous alloy catalysts. Catal. Today 1999, 51, 113–125. [Google Scholar] [CrossRef]
- Gubicza, J.; Nauyoks, S.; Balogh, L.; Labar, J.; Zerda, T.W.; Ungár, T. Influence of sintering temperature and pressure on crystallite size and lattice defect structure in nanocrystalline SiC. J. Mater. Res. 2007, 22, 1314–1321. [Google Scholar] [CrossRef]
- Kvackaj, T.; Kovacova, A.; Kocisko, R.; Bidulska, J.; Lityńska–Dobrzyńska, L.; Jenei, P.; Gubicza, J. Microstructure evolution and mechanical performance of copper processed by equal channel angular rolling. Mater. Charact. 2017, 134, 246–252. [Google Scholar] [CrossRef]
- Leslie-Pelecky, D.L.; Rieke, R.D. Magnetic Properties of Nanostructured Materials. Chem. Mater. 1996, 8, 1770–1783. [Google Scholar] [CrossRef]
- Hall, E.O. The Deformation and Ageing of Mild Steel: III Discussion of Results. Proc. Phys. Soc. B 1951, 64, 747–753. [Google Scholar] [CrossRef]
- Petch, N.J. The Cleavage Strength of Polycrystals. J. Iron Steel Inst. 1953, 174, 25–28. [Google Scholar]
- Carlton, C.E.; Ferreira, P.J. What is behind the inverse Hall–Petch effect in nanocrystalline materials? Acta Mater. 2007, 55, 3749–3756. [Google Scholar] [CrossRef]
- Krasilnikov, N.; Lojkowski, W.; Pakiela, Z.; Valiev, R. Tensile strength and ductility of ultra-fine-grained nickel processed by severe plastic deformation. Mater. Sci. Eng. A 2005, 397, 330–337. [Google Scholar] [CrossRef]
- Saboori, A.; Dadkhah, M.; Fino, P.; Pavese, M. An Overview of Metal Matrix Nanocomposites Reinforced with Graphene Nanoplatelets: Mechanical, Electrical and Thermophysical Properties. Metals 2018, 8, 423. [Google Scholar] [CrossRef]
- Ferguson, J.B.; Sheykh-Jaberi, F.; Kim, C.-S.; Rohatgi, P.K.; Cho, K. On the strength and strain to failure in particle-reinforced magnesium metal-matrix nanocomposites (Mg MMNCs). Mater. Sci. Eng. A 2012, 558, 193–204. [Google Scholar] [CrossRef]
- Chu, K.; Liu, Z.; Jia, C.; Chen, H.; Liang, X.; Gao, W.; Tian, W.; Guo, H. Thermal conductivity of SPS consolidated Cu/diamond composites with Cr-coated diamond particles. J. Alloys Compd. 2010, 490, 453–458. [Google Scholar] [CrossRef]
- Zheng, M.; Wu, X.C.; Zou, B.S.; Wang, Y.J. Magnetic properties of nanosized MnFe2O4 particles. J. Magn. Magn. Mater. 1998, 183, 152–156. [Google Scholar] [CrossRef]
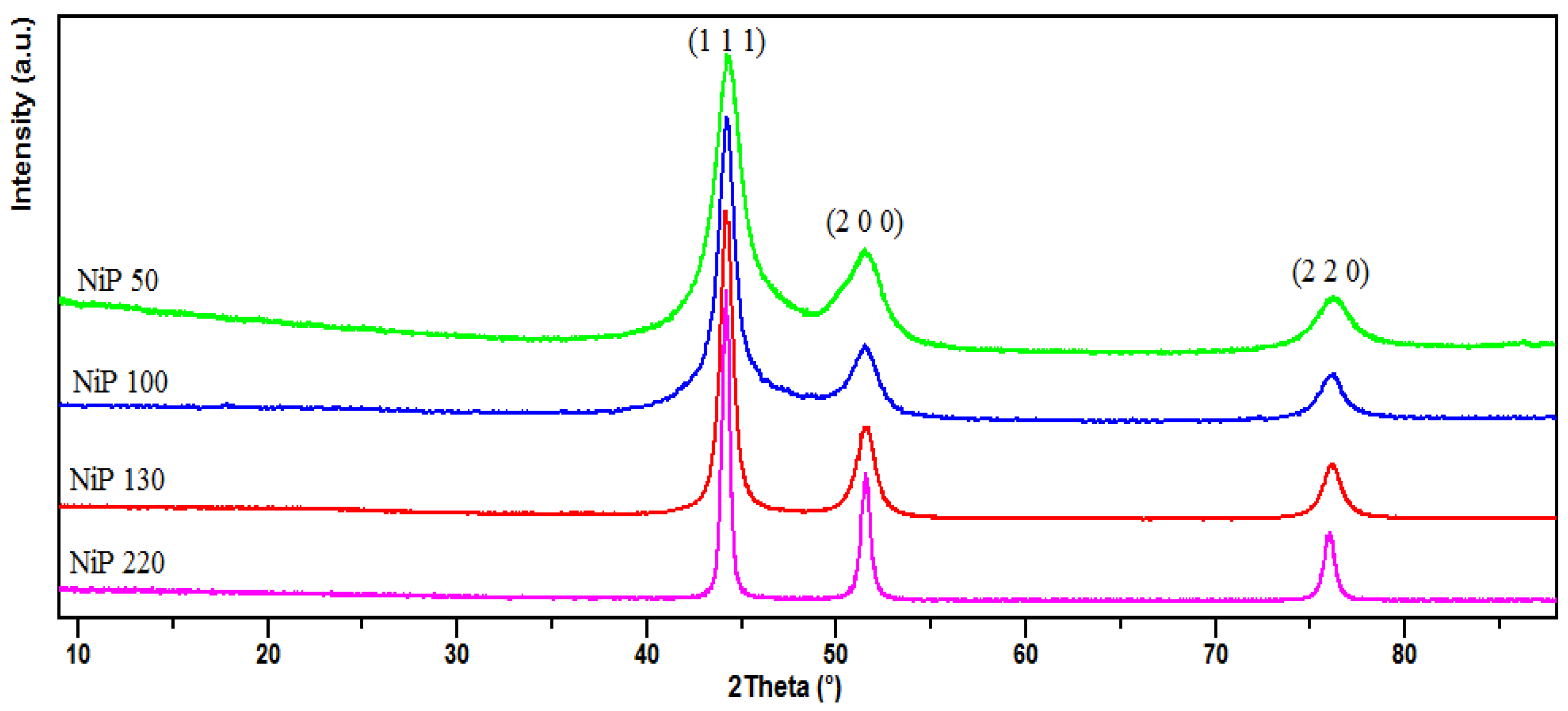
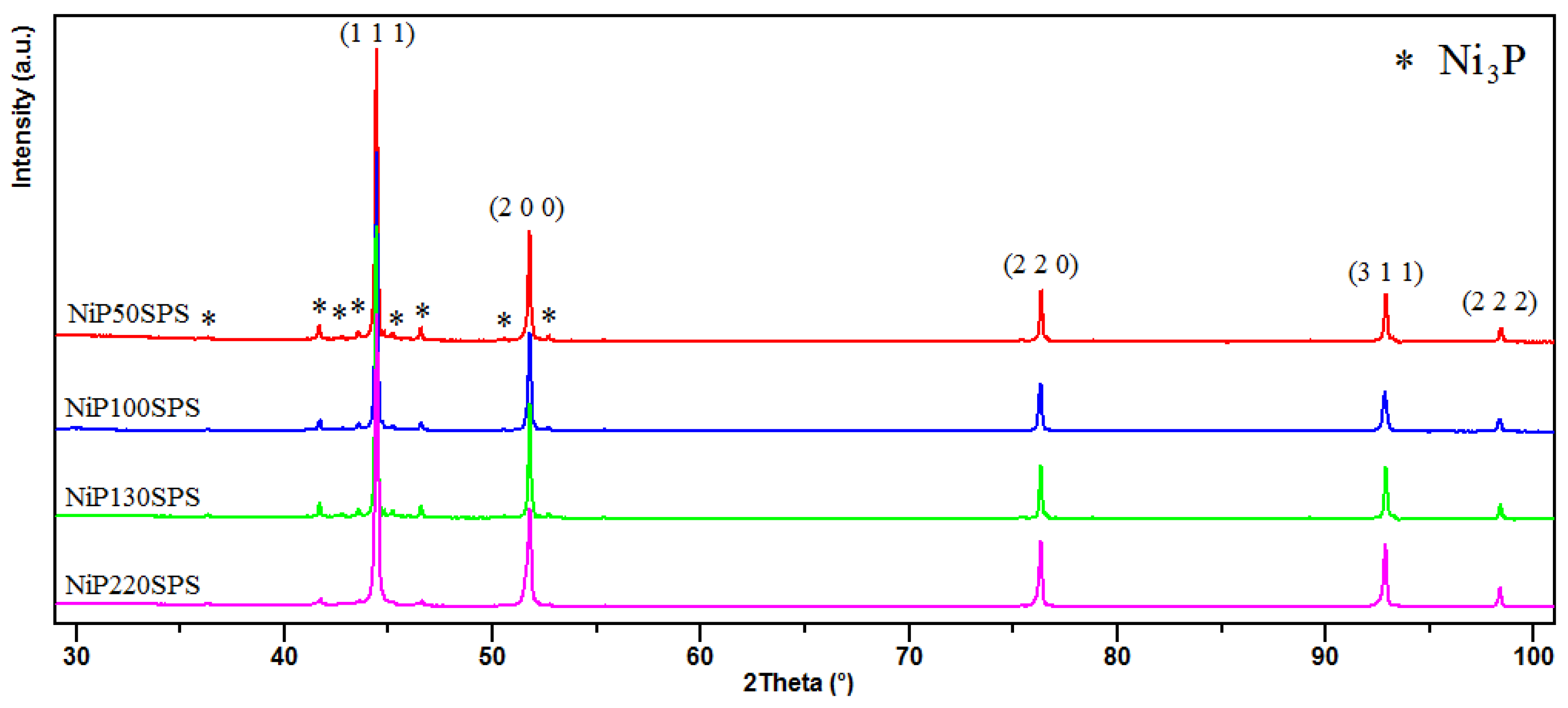
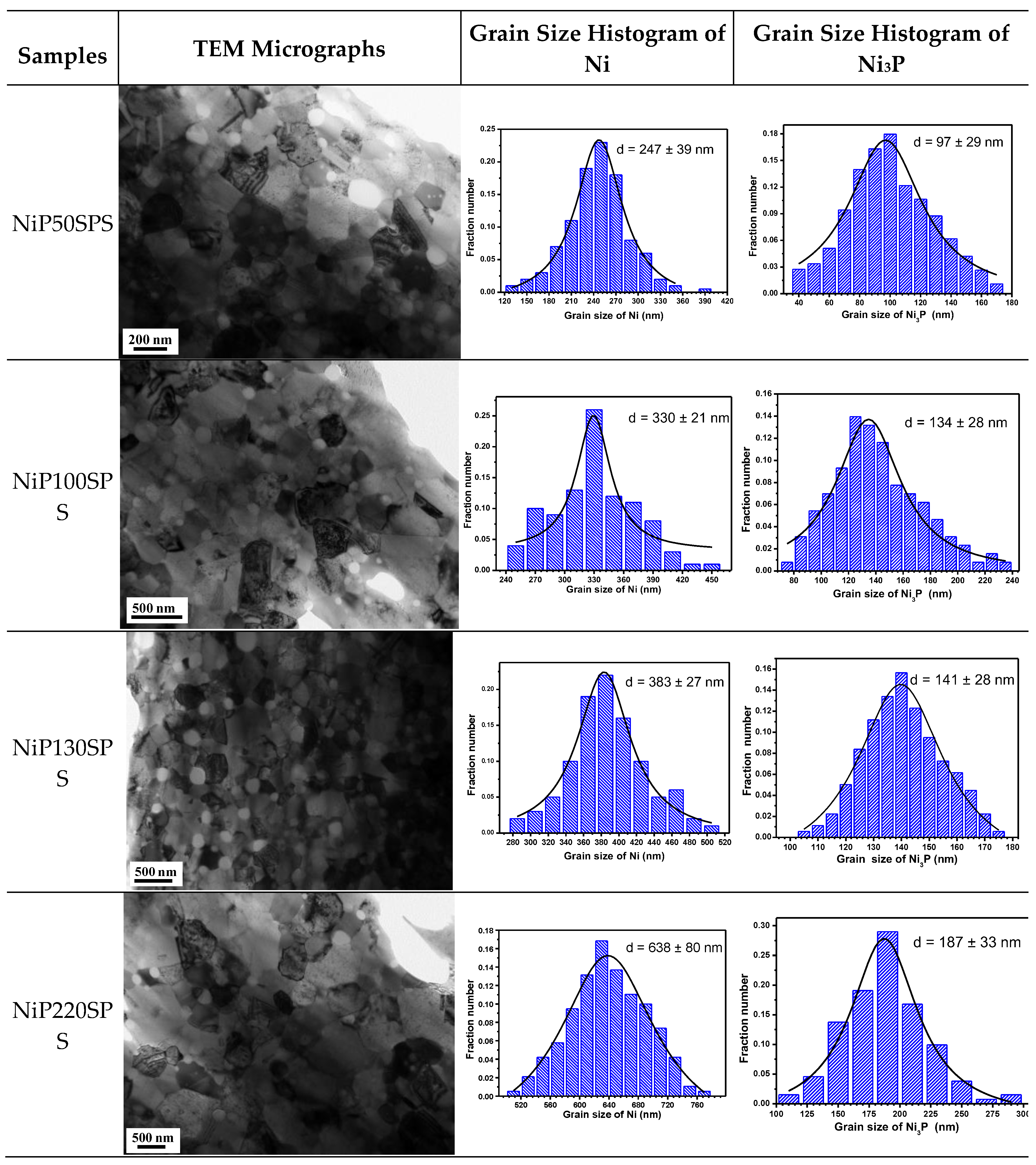
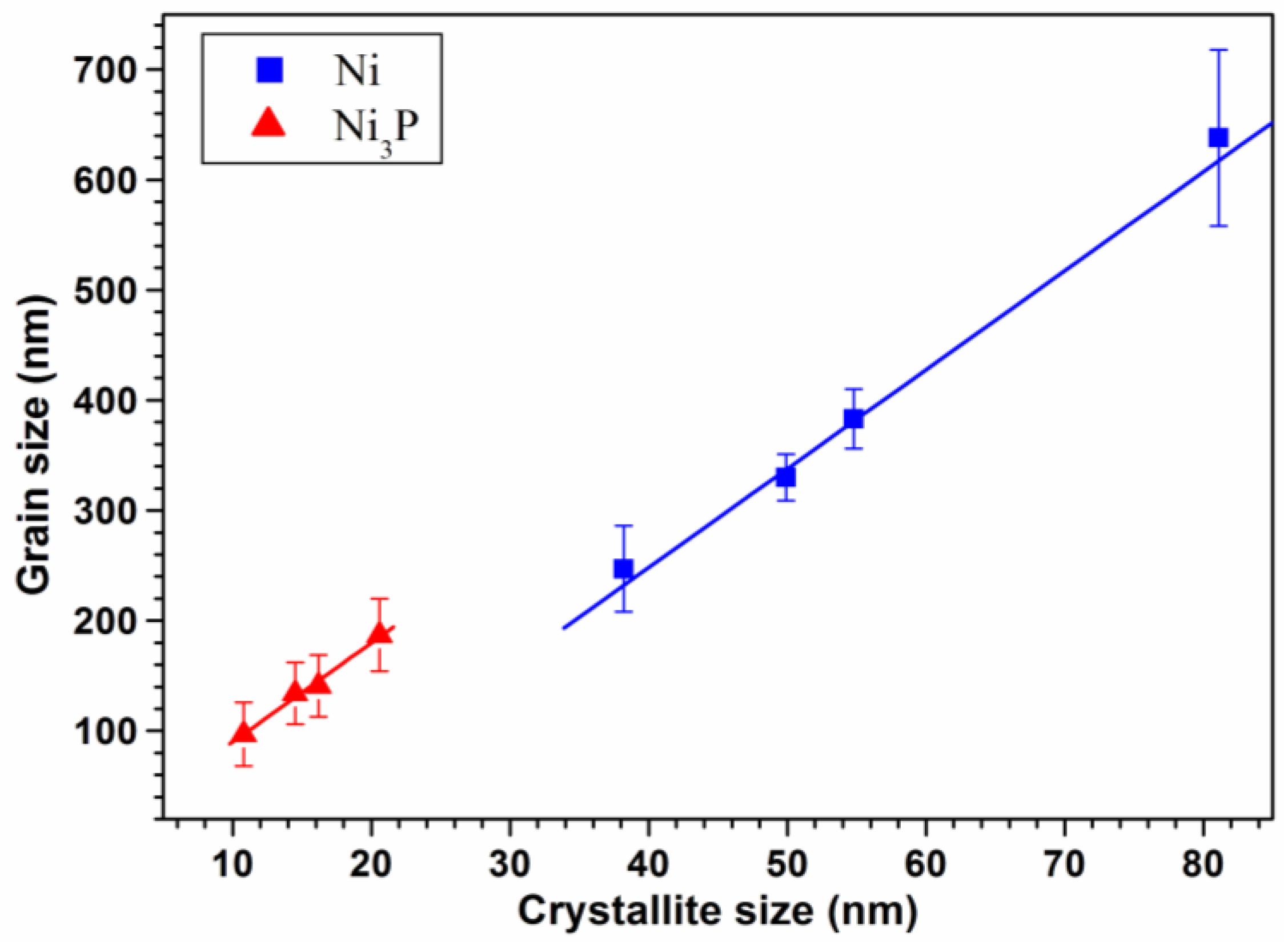
| Samples | Particle Size of Ni-P Powder (nm) | Crystallite Size of Ni (nm) | Crystallite Size of Ni3P (nm) | Grain Size of Ni (nm) | Grain Size of Ni3P (nm) | % Mass P (ICP) | % Mass Ni3P (Rietveld) |
|---|---|---|---|---|---|---|---|
| NiP50SPS | 50 ± 9 | 38.2 | 10.8 | 247 ± 39 | 97 ± 29 | 4.16 | 9.84 |
| NiP100SPS | 100 ± 18 | 49.9 | 14.5 | 330 ± 21 | 134 ± 28 | 4.01 | 9.40 |
| NiP130SPS | 130 ± 19 | 54.8 | 16.2 | 383 ± 27 | 141 ± 28 | 3.72 | 7.19 |
| NiP220SPS | 220 ± 12 | 81.1 | 20.6 | 638 ± 80 | 187 ± 33 | 2.22 | 4.97 |
| Samples | Grain Size of Ni (nm) | Grain Size of Ni3P (nm) | Relative Densities (%) | Hv |
|---|---|---|---|---|
| NiP50SPS | 247 ± 39 | 97 ± 29 | 94.5 | 522 ± 4 |
| NiP100SPS | 330 ± 21 | 134 ± 28 | 95.6 | 593 ± 5 |
| NiP130SPS | 383 ± 27 | 141 ± 28 | 96.2 | 615 ± 5 |
| NiP220SPS | 638 ± 80 | 187 ± 33 | 97.8 | 355 ± 6 |
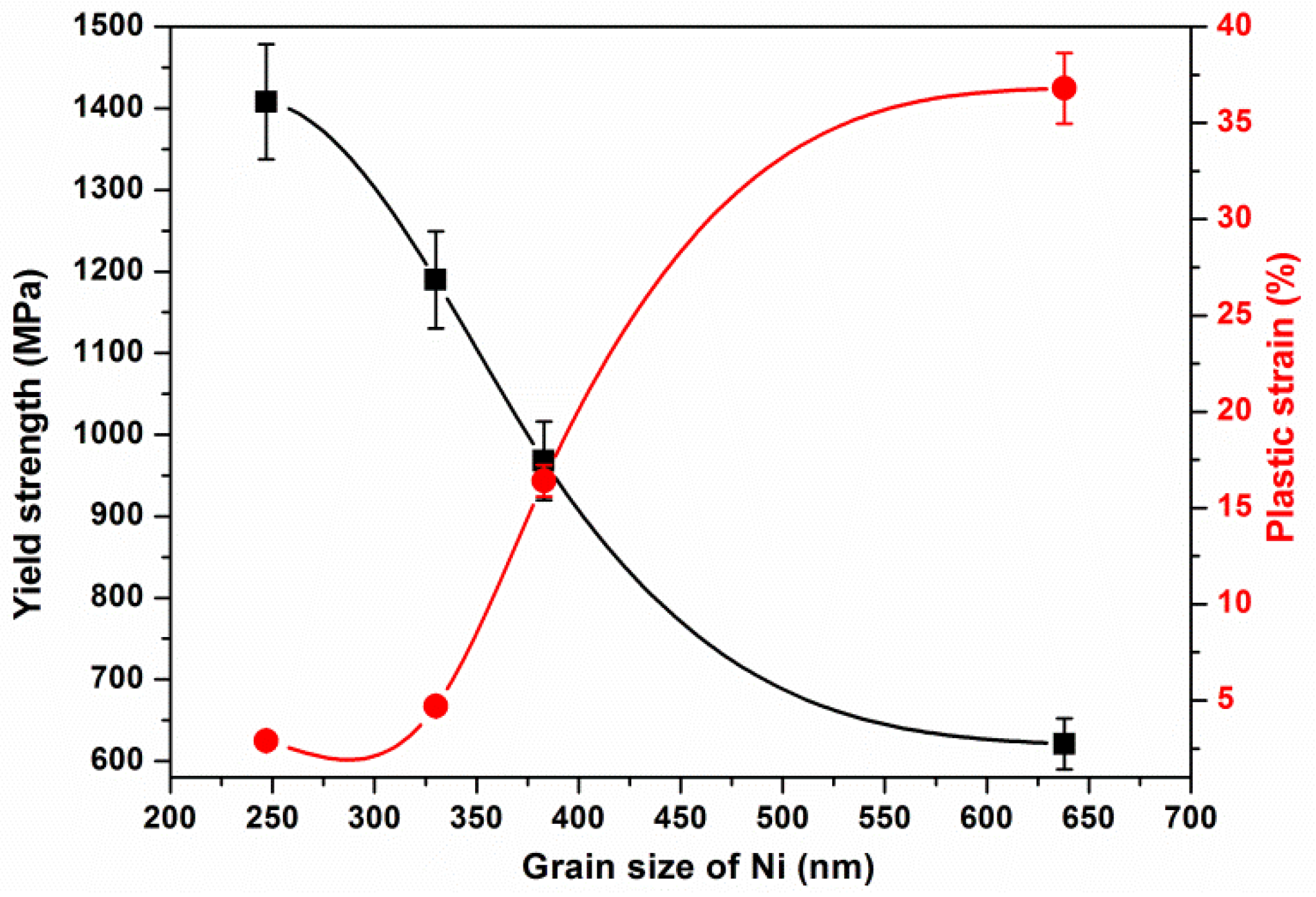
| Samples | Grain Size of Ni (nm) | Yield Strength (MPa) | Maximum Stress (MPa) | Plastic Strain (%) |
|---|---|---|---|---|
| NiP50SPS | 247 ± 39 | 1408 | 1638 | 2.9 |
| NiP100SPS | 330 ± 21 | 1190 | 1493 | 4.7 |
| NiP130SPS | 383 ± 27 | 968 | 1229 | 16.4 |
| NiP220SPS | 638 ± 80 | 621 | 894 | 36.8 |
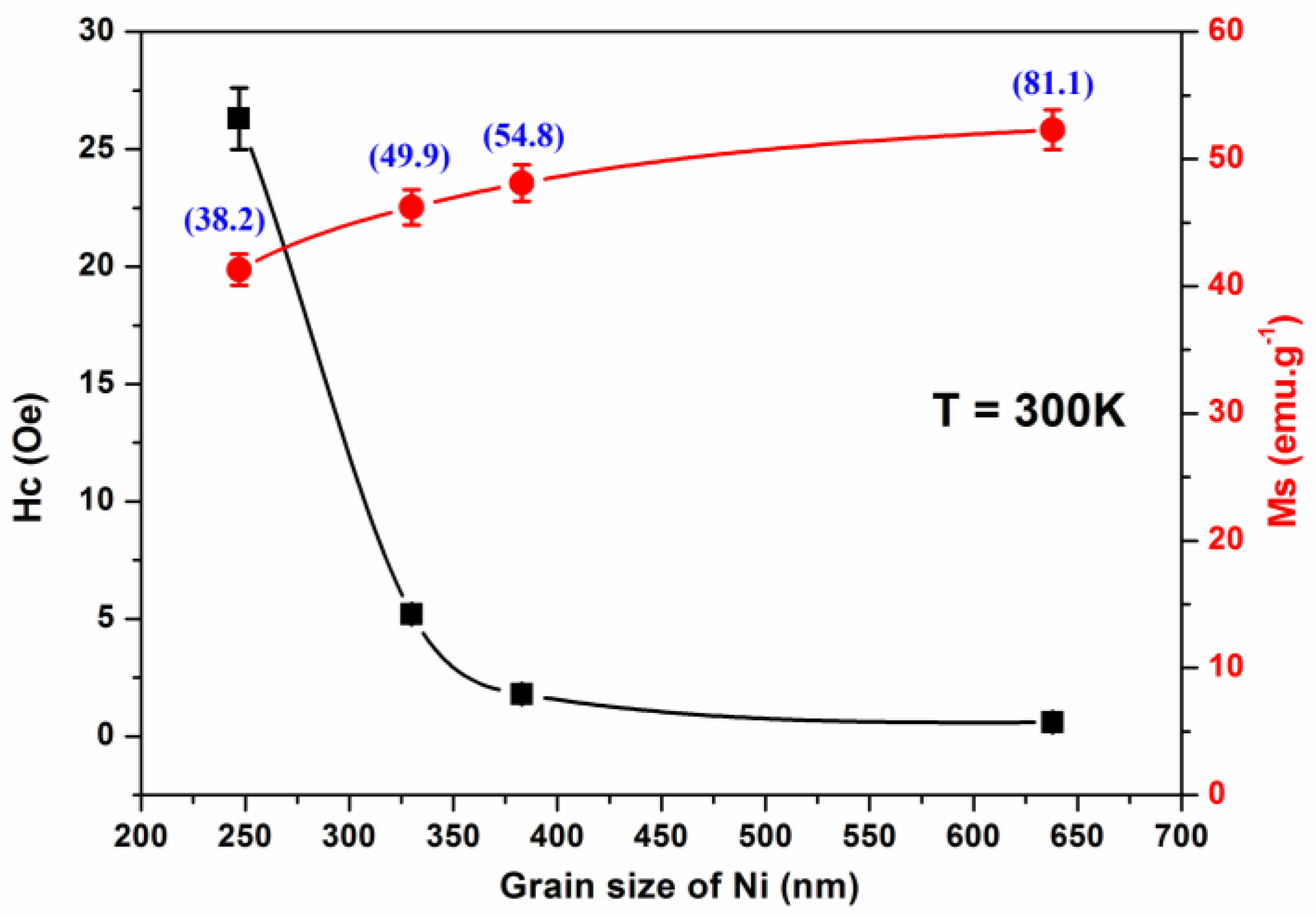
| Samples | Grain Size of Nickel (nm) | Crystallite Size of Nickel (nm) | Hc (Oe) | Mr (emu g−1) | Ms (emu g−1) | |||
|---|---|---|---|---|---|---|---|---|
| 10 K | 300 K | 10 K | 300 K | 10 K | 300 K | |||
| NiP50SPS | 247 ± 39 | 38.2 | 86.7 | 26.3 | 2.9 | 0.96 | 43.3 | 41.3 |
| NiP100SPS | 330 ± 21 | 49.9 | 55.4 | 5.2 | 2.6 | 0.33 | 48.1 | 46.2 |
| NiP130SPS | 383 ± 27 | 54.8 | 40.6 | 1.8 | 2.1 | 0.15 | 50.2 | 48.1 |
| NiP220SPS | 638 ± 80 | 81.8 | 22.8 | 0.6 | 0.9 | 0.03 | 54.7 | 52.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bousnina, M.A.; Schoenstein, F.; Mercone, S.; Jouini, N. From Ni–P Metastable Alloy Nanoparticles to Bulk Submicrometer Grain-Sized MMCs with Tunable Mechanical and Magnetic Properties. Metals 2020, 10, 112. https://doi.org/10.3390/met10010112
Bousnina MA, Schoenstein F, Mercone S, Jouini N. From Ni–P Metastable Alloy Nanoparticles to Bulk Submicrometer Grain-Sized MMCs with Tunable Mechanical and Magnetic Properties. Metals. 2020; 10(1):112. https://doi.org/10.3390/met10010112
Chicago/Turabian StyleBousnina, Mohamed Ali, Frédéric Schoenstein, Silvana Mercone, and Noureddine Jouini. 2020. "From Ni–P Metastable Alloy Nanoparticles to Bulk Submicrometer Grain-Sized MMCs with Tunable Mechanical and Magnetic Properties" Metals 10, no. 1: 112. https://doi.org/10.3390/met10010112
APA StyleBousnina, M. A., Schoenstein, F., Mercone, S., & Jouini, N. (2020). From Ni–P Metastable Alloy Nanoparticles to Bulk Submicrometer Grain-Sized MMCs with Tunable Mechanical and Magnetic Properties. Metals, 10(1), 112. https://doi.org/10.3390/met10010112