Golf as a Physical Activity to Potentially Reduce the Risk of Falls in Older Adults with Parkinson’s Disease
Abstract
1. Introduction
1.1. Parkinson’s Disease
1.2. Neurodegenerative Diseases and the Positive Impact of Exercise
1.3. Maintaining Balance in the Presence of Parkinson’s Disease
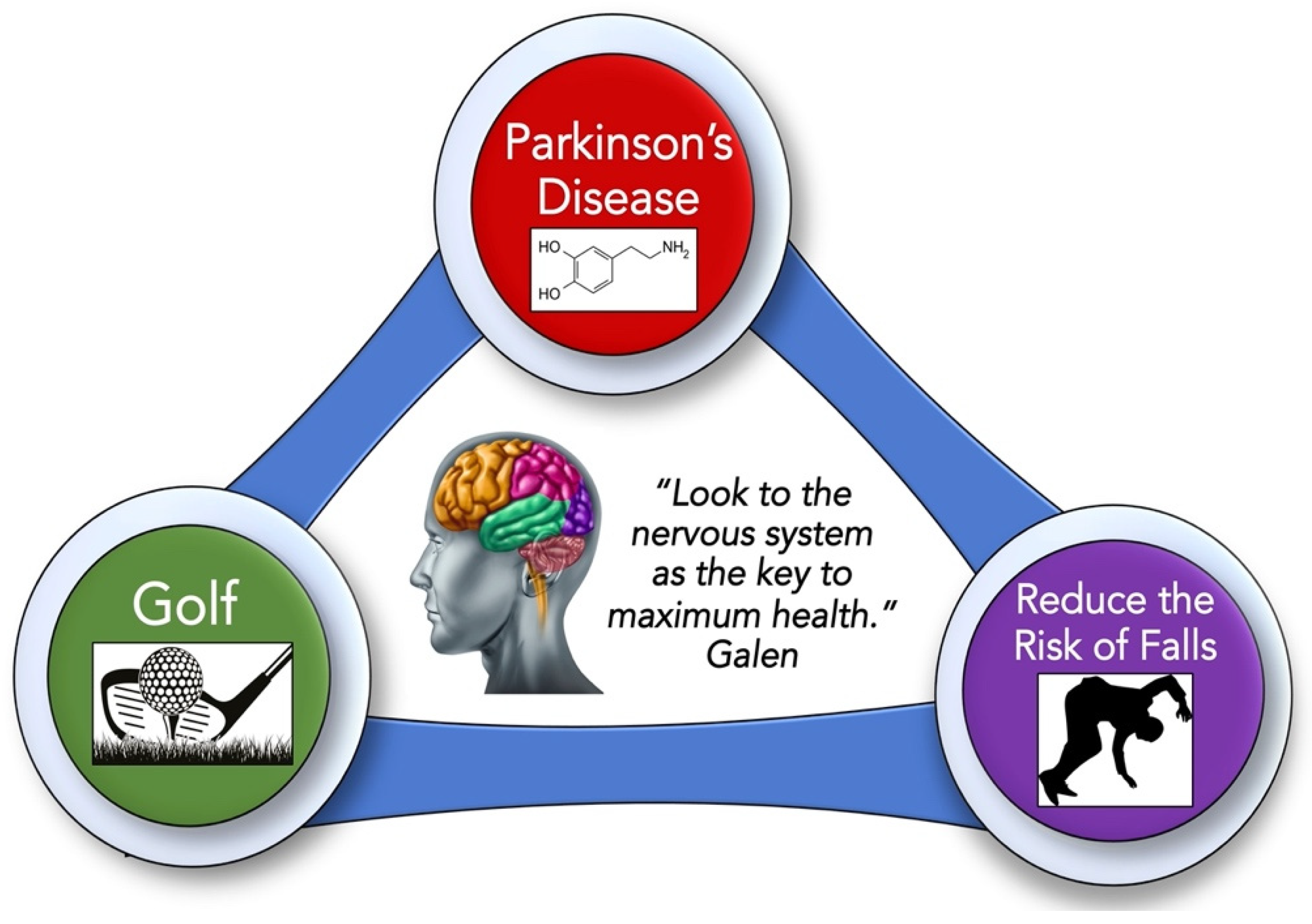
1.4. Can the Game of Golf Reduce Falls in Older Adults with Parkinson’s Disease?
2. Increased Risk of Falling in Older Adults
2.1. Causes of Falls
2.2. Role of Exercise to Lower the Risk of Falls
3. Parkinson’s Disease and the Increased Risk of Falls
3.1. PwP Have an Increased Probability of Falling
3.2. Exercise Has Proven Beneficial in Reducing Falls in Parkinson’s Disease
4. Golf as an Exercise to Reduce Falls in Older Adults
4.1. Overview of Golf Compared to PD-Specific Exercises
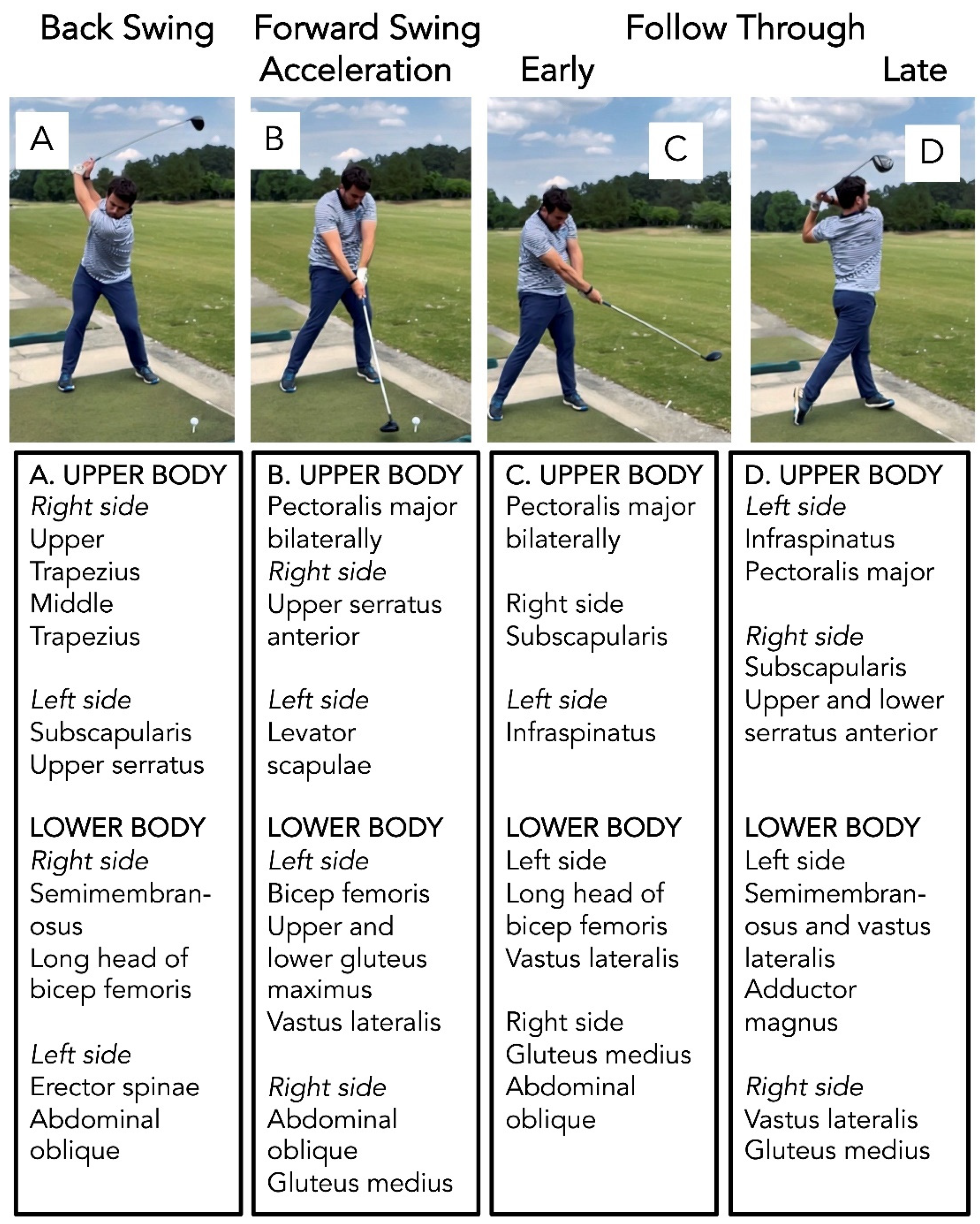
4.2. The Golf Swing
4.3. Playing Golf Promotes Wellness and Reduces Falls in Older Adults
5. Golf Could Improve Functional Ability in Older Adults with Parkinson’s Disease
5.1. Comparing Regular Physical Activity to Playing Golf
5.2. Postural Instability: Anticipatory Movements and Bradykinesia
5.3. Postural Instability: Axial Rigidity and Coordination
5.4. Motivation and External Engagement
5.5. Hitting Golf Balls and Playing Golf
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kalia, L.; Lang, A. Parkinson’s Disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef]
- Poewe, W.; Seppi, K.; Tanner, C.M.; Halliday, G.M.; Brundin, P.; Volkmann, J.; Schrag, A.-E.; Lang, A.E. Parkinson Disease. Nat. Rev. Dis. Primers 2017, 3, 17013. [Google Scholar] [CrossRef]
- Ahlskog, J.E. The New Parkinson’s Disease Treatment Book: Partnering with Your Doctor to Get the Most from Your Medications; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Santens, P.; Boon, P.; Van Roost, D.; Caemaert, J. The pathophysiology of motor symptoms in Parkinson’s disease. Acta Neurol. Belg. 2003, 103, 103. [Google Scholar]
- Fox, S.H.; Katzenschlager, R.; Lim, S.Y.; Barton, B.; de Bie, R.M.; Seppi, K.; Coelho, M.; Sampaio, C.; Movement Disorder Society Evidence-Based Medicine Committee. International Parkinson and Movement Disorder Society Evidence-Based Medicine Review: Update on Treatments for the Motor Symptoms of Parkinson’s Disease. Mov. Disord. 2018, 33, 1248–1266. [Google Scholar] [CrossRef]
- Berganzo, K.; Tijero, B.; Gonzalez-Eizaguirre, A.; Somme, J.; Lezcano, E.; Gabilondo, I.; Fernandez, M.; Zarranz, J.; Gómez-Esteban, J. Motor and Non-Motor Symptoms of Parkinson’s Disease and Their Impact on Quality of Life and on Different Clinical Subgroups. Neurología 2016, 31, 585–591. [Google Scholar] [CrossRef]
- Church, F. Treatment Options for Motor and Non-Motor Symptoms of Parkinson’s Disease. Biomolecules 2021, 11, 612. [Google Scholar] [CrossRef]
- Vuletić, V. Non-Motor Symptoms in Parkinson’s Disease. Mind Brain 2020, 109–118. [Google Scholar] [CrossRef]
- Schapira, A.H.; Chaudhuri, K.R.; Jenner, P. Non-Motor Features of Parkinson Disease. Nat. Rev. Neurosci. 2017, 18, 435. [Google Scholar] [CrossRef]
- Chaudhuri, K.R.; Schapira, A.H. Non-Motor Symptoms of Parkinson’s Disease: Dopaminergic Pathophysiology and Treatment. Lancet Neurol. 2009, 8, 464–474. [Google Scholar] [CrossRef]
- Chaudhuri, K.R.; Healy, D.G.; Schapira, A.H. Non-Motor Symptoms of Parkinson’s Disease: Diagnosis and Management. Lancet Neurol. 2006, 5, 235–245. [Google Scholar] [CrossRef]
- Maiti, P.; Manna, J.; Dunbar, G.L. Current Understanding of the Molecular Mechanisms in Parkinson’s Disease: Targets for Potential Treatments. Transl. Neurodegener. 2017, 6, 28. [Google Scholar] [CrossRef]
- Parkinson’s Foundation Statistics; Parkinson’s Foundation: Miami, FL, USA, 2019.
- Rekatsina, M.; Paladini, A.; Piroli, A.; Zis, P.; Pergolizzi, J.V.; Varrassi, G. Pathophysiology and Therapeutic Perspectives of Oxidative Stress and Neurodegenerative Diseases: A Narrative Review. Adv. Ther. 2020, 37, 113–139. [Google Scholar] [CrossRef]
- Lázaro, D.F.; Bellucci, A.; Brundin, P.; Outeiro, T.F. Editorial: Protein Misfolding and Spreading Pathology in Neurodegenerative Diseases. Front. Mol. Neurosci. 2020, 12, 312. [Google Scholar] [CrossRef]
- Hung, C.-W.; Chen, Y.-C.; Hsieh, W.-L.; Chiou, S.-H.; Kao, C.-L. Ageing and neurodegenerative diseases. Ageing Res. Rev. 2010, 9, S36–S46. [Google Scholar] [CrossRef]
- Quan, H.; Koltai, E.; Suzuki, K.; Aguiar, A.S., Jr.; Pinho, R.; Boldogh, I.; Berkes, I.; Radak, Z. Exercise, Redox System and Neurodegenerative Diseases. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165778. [Google Scholar] [CrossRef]
- Paillard, T.; Rolland, Y.; de Souto Barreto, P. Protective Effects of Physical Exercise in Alzheimer’s Disease and Parkinson’s Disease: A Narrative Review. J. Clin. Neurol. 2015, 11, 212. [Google Scholar] [CrossRef]
- Marques-Aleixo, I.; Beleza, J.; Sampaio, A.; Stevanović, J.; Coxito, P.; Gonçalves, I.; Ascensão, A.; Magalhães, J. Preventive and Therapeutic Potential of Physical Exercise in Neurodegenerative Diseases. Antioxid. Redox Signal. 2021, 34, 674–693. [Google Scholar] [CrossRef]
- Campos, C.; Rocha, N.B.F.; Lattari, E.; Paes, F.; Nardi, A.E.; Machado, S. Exercise-induced neuroprotective effects on neurodegenerative diseases: The key role of trophic factors. Expert Rev. Neurother. 2016, 16, 723–734. [Google Scholar] [CrossRef]
- Liu, Y.; Yan, T.; Chu, J.M.-T.; Chen, Y.; Dunnett, S.; Ho, Y.-S.; Wong, G.T.-C.; Chang, R.C.-C. The beneficial effects of physical exercise in the brain and related pathophysiological mechanisms in neurodegenerative diseases. Lab. Investig. 2019, 99, 943–957. [Google Scholar] [CrossRef]
- Schenkman, M.; Shipp, K.M.; Chandler, J.; Studenski, S.A.; Kuchibhatla, M. Relationships Between Mobility of Axial Structures and Physical Performance. Phys. Ther. 1996, 76, 276–285. [Google Scholar] [CrossRef]
- Duchesne, C.; Lungu, O.V.; Nadeau, A.; Robillard, M.; Bore, A.; Bobeuf, F.; Lafontaine, A.; Gheysen, F.; Bherer, L.; Doyon, J. Enhancing both motor and cognitive functioning in Parkinson’s disease: Aerobic exercise as a rehabilitative intervention. Brain Cogn. 2015, 99, 68–77. [Google Scholar] [CrossRef]
- Hirsch, M.A.; Farley, B.G. Exercise and Neuroplasticity in Persons Living with Parkinson’s Disease. Eur. J. Phys. Rehabil. Med. 2009, 45, 215–229. [Google Scholar]
- Alamgir, H.; Muazzam, S.; Nasrullah, M. Unintentional falls mortality among elderly in the United States: Time for action. Injury 2012, 43, 2065–2071. [Google Scholar] [CrossRef]
- Rubenstein, L.Z. Falls in older people: Epidemiology, risk factors and strategies for prevention. Age Ageing 2006, 35 (Suppl. 2), ii37–ii41. [Google Scholar] [CrossRef]
- Dibble, L.E.; Lange, M. Predicting Falls in Individuals with Parkinson Disease: A Reconsideration of Clinical Balance Measures. J. Neurol. Phys. Ther. 2006, 30, 60–67. [Google Scholar] [CrossRef]
- Dennison, A.C.; Noorigian, J.V.; Robinson, K.M.; Fisman, D.N.; Cianci, H.J.; Moberg, P.; Bunting-Perry, L.; Martine, R.; Duda, J.; Stern, M.B. Falling in Parkinson Disease: Identifying and Prioritizing Risk Factors in Recurrent Fallers. Am. J. Phys. Med. Rehabil. 2007, 86, 621–632. [Google Scholar] [CrossRef]
- Siegel, R.E. Galen on the Affected Parts. Translation from the Greek Text with Explanatory Notes. Med. Hist. 1977, 21, 212. [Google Scholar]
- Tinetti, M.E.; Speechley, M.; Ginter, S.F. Risk Factors for Falls among Elderly Persons Living in the Community. N. Engl. J. Med. 1988, 319, 1701–1707. [Google Scholar] [CrossRef]
- Ambrose, A.F.; Paul, G.; Hausdorff, J.M. Risk factors for falls among older adults: A review of the literature. Maturitas 2013, 75, 51–61. [Google Scholar] [CrossRef] [PubMed]
- James, S.L.; Lucchesi, L.R.; Bisignano, C.; Castle, C.D.; Dingels, Z.V.; Fox, J.T.; Hamilton, E.B.; Henry, N.J.; Krohn, K.J.; Liu, Z.; et al. The global burden of falls: Global, regional and national estimates of morbidity and mortality from the Global Burden of Disease Study. Inj. Prev. 2020, 26, i3–i11. [Google Scholar] [CrossRef]
- Gregg, E.W.; Pereira, M.A.; Caspersen, C.J. Physical Activity, Falls, and Fractures Among Older Adults: A Review of the Epidemiologic Evidence. J. Am. Geriatr. Soc. 2000, 48, 883–893. [Google Scholar] [CrossRef]
- Tow, B.P.B.; Chua, B.S.Y.; Fook-Chong, S.; Howe, T.S. Concurrent fractures of the hip and wrist: A matched analysis of elderly patients. Injury 2009, 40, 385–387. [Google Scholar] [CrossRef]
- Ensrud, K.E. Epidemiology of Fracture Risk with Advancing Age. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2013, 68, 1236–1242. [Google Scholar] [CrossRef] [PubMed]
- Franko, J.; Kish, K.J.; O’Connell, B.G.; Subramanian, S.; Yuschak, J.V. Advanced Age and Preinjury Warfarin Anticoagulation Increase the Risk of Mortality After Head Trauma. J. Trauma Inj. Infect. Crit. Care 2006, 61, 107–110. [Google Scholar] [CrossRef]
- Moreland, J.D.; Richardson, J.A.; Goldsmith, C.H.; Clase, C.M. Muscle Weakness and Falls in Older Adults: A Systematic Review and Meta-Analysis. J. Am. Geriatr. Soc. 2004, 52, 1121–1129. [Google Scholar] [CrossRef]
- Borges, E.G.D.S.; Vale, R.G.D.S.; Cader, S.A.; Leal, S.; Miguel, F.; Pernambuco, C.S.; Dantas, E.H. Postural balance and falls in elderly nursing home residents enrolled in a ballroom dancing program. Arch. Gerontol. Geriatr. 2014, 59, 312–316. [Google Scholar] [CrossRef] [PubMed]
- Dunsky, A.; Zeev, A.; Netz, Y. Balance Performance Is Task Specific in Older Adults. BioMed Res. Int. 2017, 2017, 6987017. [Google Scholar] [CrossRef] [PubMed]
- Moncada, L.V.V.; Mire, L.G. Preventing Falls in Older Persons. Am. Fam. Phys. 2017, 96, 240–247. [Google Scholar]
- Lee, P.G.; Jackson, E.A.; Richardson, C.R. Exercise Prescriptions in Older Adults. Am. Fam. Phys. 2017, 95, 425–432. [Google Scholar]
- Sherrington, C.; Tiedemann, A.; Fairhall, N.; Close, J.C.; Lord, S.R. Exercise to prevent falls in older adults: An updated meta-analysis and best practice recommendations. NSW Public Health Bull. 2011, 22, 78–83. [Google Scholar] [CrossRef]
- Lesinski, M.; Hortobágyi, T.; Muehlbauer, T.; Gollhofer, A.; Granacher, U. Effects of Balance Training on Balance Performance in Healthy Older Adults: A Systematic Review and Meta-Analysis. Sports Med. 2015, 45, 1721–1738. [Google Scholar] [CrossRef] [PubMed]
- Gschwind, Y.J.; Kressig, R.W.; Lacroix, A.; Muehlbauer, T.; Pfenninger, B.; Granacher, U. A best practice fall prevention exercise program to improve balance, strength/power, and psychosocial health in older adults: Study protocol for a randomized controlled trial. BMC Geriatr. 2013, 13, 105. [Google Scholar] [CrossRef] [PubMed]
- Bloem, B.R.; Grimbergen, Y.A.; Cramer, M.; Willemsen, M.; Zwinderman, A.H. Prospective Assessment of Falls in Parkinson’s Disease. J. Neurol. 2001, 248, 950–958. [Google Scholar] [CrossRef]
- Stolze, H.; Klebe, S.; Zechlin, C.; Baecker, C.; Friege, L.; Deuschl, G. Falls in Frequent Neurologicaldiseases. J. Neurol. 2004, 251, 79–84. [Google Scholar] [CrossRef]
- Gray, P.; Hildebrand, K. Fall Risk Factors in Parkinson’s Disease. J. Neurosci. Nurs. 2000, 32, 222–228. [Google Scholar] [CrossRef]
- Wood, B.H.; Bilclough, J.A.; Bowron, A.; Walker, R.W. Incidence and Prediction of Falls in Parkinson’s Disease: A Prospective Multidisciplinary Study. J. Neurol. Neurosurg. Psychiatry 2002, 72, 721–725. [Google Scholar] [CrossRef]
- Almeida, L.R.S.; Paul, S.S.; Valenca, G.T. Recurrent Falls in People with Parkinson’s Disease. Diagn. Manag. Parkinson’s Dis. 2020, 1, 157–183. [Google Scholar]
- Ashburn, A.; Stack, E.; Pickering, R.M.; Ward, C.D. A Community-Dwelling Sample of People with Parkinson’s Disease: Characteristics of Fallers and Non-Fallers. Age Ageing 2001, 30, 47–52. [Google Scholar] [CrossRef]
- Almeida, L.R.; Sherrington, C.; Allen, N.E.; Paul, S.S.; Valenca, G.T.; Oliveira-Filho, J.; Canning, C.G. Disability is an Independent Predictor of Falls and Recurrent Falls in People with Parkinson’s Disease without a History of Falls: A One-Year Prospective Study. J. Park. Dis. 2015, 5, 855–864. [Google Scholar] [CrossRef]
- Almeida, L.R.; Valenca, G.T.; Negreiros, N.N.; Pinto, E.B.; Oliveira-Filho, J. Predictors of Recurrent Falls in People with Parkinson’s Disease and Proposal for a Predictive Tool. J. Park. Dis. 2017, 7, 313–324. [Google Scholar] [CrossRef]
- Koller, W.C.; Glatt, S.; Vetere-Overfield, B.; Hassanein, R. Falls and Parkinson’s Disease. Clin. Neuropharmacol. 1989, 12, 98–105. [Google Scholar] [PubMed]
- Okuma, Y. Freezing of Gait and Falls in Parkinson’s Disease. J. Parkinson’s Dis. 2014, 4, 255–260. [Google Scholar] [CrossRef]
- Bloem, B.R.; Hausdorff, J.M.; Visser, J.E.; Giladi, N. Falls and Freezing of Gait in Parkinson’s Disease: A Review of Two Interconnected, Episodic Phenomena. Mov. Disord. 2004, 19, 871–884. [Google Scholar] [CrossRef] [PubMed]
- Cole, M.H.; Rippey, J.; Naughton, G.A.; Silburn, P.A. Use of a Short-Form Balance Confidence Scale to Predict Future Recurrent Falls in People with Parkinson Disease. Arch. Phys. Med. Rehabil. 2016, 97, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Camicioli, R.; Majumdar, S.R. Relationship between Mild Cognitive Impairment and Falls in Older People with and without Parkinson’s Disease: 1-Year Prospective Cohort Study. Gait Posture 2010, 32, 87–91. [Google Scholar] [CrossRef] [PubMed]
- McKay, J.L.; Hackney, M.E.; Factor, S.A.; Ting, L.H. Lower Limb Rigidity Is Associated with Frequent Falls in Parkinson’s Disease. Mov. Disord. Clin. Pract. 2019, 6, 446–451. [Google Scholar] [CrossRef]
- Adkin, A.L.; Frank, J.S.; Jog, M.S. Fear of Falling and Postural Control in Parkinson’s Disease. Mov. Disord. 2003, 18, 496–502. [Google Scholar] [CrossRef]
- Mak, M.K.; Wong, A.; Pang, M.Y. Impaired Executive Function Can Predict Recurrent Falls in Parkinson’s Disease. Arch. Phys. Med. Rehabil. 2014, 95, 2390–2395. [Google Scholar] [CrossRef]
- Öztürk, E.A.; Koçer, B.G. Can Recurrent Falls Be Predicted in Parkinson’s Disease? Ankara Üniversitesi Tıp Fakültesi Mecmuası. 2017, 70, 1–6. [Google Scholar] [CrossRef]
- Gazibara, T.; Tepavcevic, D.K.; Svetel, M.; Tomic, A.; Stankovic, I.; Kostic, V.S.; Pekmezovic, T. Recurrent falls in Parkinson’s disease after one year of follow-up: A nested case-control study. Arch. Gerontol. Geriatr. 2016, 65, 17–24. [Google Scholar] [CrossRef]
- Allen, N.E.; Sherrington, C.; Paul, S.S.; Canning, C.G. Balance and Falls in Parkinson’s Disease: A Meta-Analysis of the Effect of Exercise and Motor Training. Mov. Disord. 2011, 26, 1605–1615. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Wong-Yu, I.S.; Mak, M.K. Effects of Exercise on Falls, Balance, and Gait Ability in Parkinson’s Disease: A Meta-Analysis. Neurorehabilit. Neural Repair 2016, 30, 512–527. [Google Scholar] [CrossRef] [PubMed]
- Mirelman, A.; Rochester, L.; Maidan, I.; Del Din, S.; Alcock, L.; Nieuwhof, F.; Rikkert, M.O.; Bloem, B.R.; Pelosin, E.; Avanzino, L.; et al. Addition of a non-immersive virtual reality component to treadmill training to reduce fall risk in older adults (V-TIME): A randomised controlled trial. Lancet 2016, 388, 1170–1182. [Google Scholar] [CrossRef]
- Sparrow, D.; DeAngelis, T.R.; Hendron, K.; Thomas, C.A.; Saint-Hilaire, M.; Ellis, T. Highly Challenging Balance Program Reduces Fall Rate in Parkinson Disease. J. Neurol. Phys. Ther. 2016, 40, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Ashburn, A.; Fazakarley, L.; Ballinger, C.; Pickering, R.; McLellan, L.D.; Fitton, C. A randomised controlled trial of a home based exercise programme to reduce the risk of falling among people with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2006, 78, 678–684. [Google Scholar] [CrossRef]
- Goodwin, V.A.; Richards, S.H.; Henley, W.; Ewings, P.; Taylor, A.H.; Campbell, J.L. An Exercise Intervention to Prevent Falls in People with Parkinson’s Disease: A Pragmatic Randomised Controlled Trial. J. Neurol. Neurosurg. Psychiatry 2011, 82, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Song, R.; Grabowska, W.; Park, M.; Osypiuk, K.; Vergara-Diaz, G.; Bonato, P.; Hausdorff, J.; Fox, M.; Sudarsky, L.; Macklin, E. The Impact of Tai Chi and Qigong Mind-Body Exercises on Motor and Non-Motor Function and Quality of Life in Parkinson’s Disease: A Systematic Review and Meta-Analysis. Parkinsonism Relat. Disord. 2017, 41, 3–13. [Google Scholar] [CrossRef]
- Fuzhong, L.; Peter, H.; Kathleen, F.; Elizabeth, E.; Ronald, S.; Johnny, G.; Gianni, M.; Sara, S. Tai Chi and Postural Stability in Patients with Parkinson’s Disease. N. Engl. J. Med. 2012, 366, 511–519. [Google Scholar]
- Shen, X.; Mak, M.K. Technology-Assisted Balance and Gait Training Reduces Falls in Patients with Parkinson’s Disease: A Randomized Controlled Trial with 12-Month Follow-Up. Neurorehabilit. Neural Repair 2015, 29, 103–111. [Google Scholar] [CrossRef]
- Morris, M.E.; Menz, H.B.; McGinley, J.L.; Watts, J.J.; Huxham, F.E.; Murphy, A.T.; Danoudis, M.E.; Iansek, R. A Randomized Controlled Trial to Reduce Falls in People with Parkinson’s Disease. Neurorehabilit. Neural Repair 2015, 29, 777–785. [Google Scholar] [CrossRef]
- Canning, C.G.; Sherrington, C.; Lord, S.R.; Close, J.C.; Heritier, S.; Heller, G.Z.; Howard, K.; Allen, N.E.; Latt, M.D.; Murray, S.M.; et al. Exercise for falls prevention in Parkinson disease: A randomized controlled trial. Neurology 2015, 84, 304–312. [Google Scholar] [CrossRef] [PubMed]
- van der Marck, M.A.; Klok, M.P.C.; Okun, M.S.; Giladi, N.; Munneke, M.; Bloem, B.R.; Force, N.F.T. Consensus-Based Clinical Practice Recommendations for the Examination and Management of Falls in Patients with Parkinson’s Disease. Parkinsonism Relat. Disord. 2014, 20, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A.; Canning, C.G.; Hausdorff, J.M.; Lord, S.; Rochester, L. Falls in Parkinson’s Disease: A Complex and Evolving Picture. Mov. Disord. 2017, 32, 1524–1536. [Google Scholar] [CrossRef]
- Amara, A.W.; Memon, A.A. Effects of Exercise on Non-motor Symptoms in Parkinson’s Disease. Clin. Ther. 2018, 40, 8–15. [Google Scholar] [CrossRef]
- Crowley, E.K.; Nolan, Y.M.; Sullivan, A.M. Exercise as Therapy for Parkinson’s? Aging 2018, 10, 1536. [Google Scholar] [CrossRef] [PubMed]
- Crowley, E.; Nolan, Y.; Sullivan, A. Exercise as a therapeutic intervention for motor and non-motor symptoms in Parkinson’s disease: Evidence from rodent models. Prog. Neurobiol. 2019, 172, 2–22. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, A.O.; Filho, A.S.S.; Murillo-Rodriguez, E.; Rocha, N.B.; Carta, M.G.; Machado, S. Physical Exercise for Parkinson’s Disease: Clinical and Experimental Evidence. Clin. Pr. Epidemiol. Ment. Health 2018, 14, 89–98. [Google Scholar] [CrossRef]
- Ebersbach, G.; Grust, U.; Ebersbach, A.; Wegner, B.; Gandor, F.; Kühn, A.A. Amplitude-Oriented Exercise in Parkinson’s Disease: A Randomized Study Comparing Lsvt-Big and a Short Training Protocol. J. Neural Transm. 2015, 122, 253–256. [Google Scholar] [CrossRef]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-Induced Neuroplasticity: A Mechanistic Model and Prospects for Promoting Plasticity. Neuroscientist 2018, 25, 65–85. [Google Scholar] [CrossRef]
- Ellis, T.; Rochester, L. Mobilizing Parkinson’s Disease: The Future of Exercise. J. Park. Dis. 2018, 8, S95–S100. [Google Scholar] [CrossRef] [PubMed]
- Petzinger, G.M.; Fisher, B.E.; Van Leeuwen, J.E.; Vukovic, M.; Akopian, G.; Meshul, C.K.; Holschneider, D.P.; Nacca, A.; Walsh, J.P.; Jakowec, M.W. Enhancing Neuroplasticity in the Basal Ganglia: The Role of Exercise in Parkinson’s Disease. Mov. Disord. 2010, 25, S141–S145. [Google Scholar] [CrossRef]
- Schenkman, M.; Moore, C.G.; Kohrt, W.M.; Hall, D.A.; Delitto, A.; Comella, C.L.; Josbeno, D.A.; Christiansen, C.L.; Berman, B.D.; Kluger, B.M. Effect of High-Intensity Treadmill Exercise on Motor Symptoms in Patients with De Novo Parkinson Disease: A Phase 2 Randomized Clinical Trial. JAMA Neurol. 2018, 75, 219–226. [Google Scholar] [CrossRef]
- Silveira, C.R.; Roy, E.A.; Intzandt, B.N.; Almeida, Q.J. Aerobic exercise is more effective than goal-based exercise for the treatment of cognition in Parkinson’s disease. Brain Cogn. 2018, 122, 1–8. [Google Scholar] [CrossRef]
- Hall, M.-F.E.; Church, F.C. Integrative Medicine and Health Therapy for Parkinson Disease. Top. Geriatr. Rehabil. 2020, 36, 176–186. [Google Scholar] [CrossRef]
- David, F.J.; Rafferty, M.R.; Robichaud, J.A.; Prodoehl, J.; Kohrt, W.M.; Vaillancourt, D.E.; Corcos, D.M. Progressive Resistance Exercise and Parkinson’s Disease: A Review of Potential Mechanisms. Parkinson’s Dis. 2012, 2012, 124527. [Google Scholar] [CrossRef]
- Lamotte, G.; Rafferty, M.R.; Prodoehl, J.; Kohrt, W.M.; Comella, C.L.; Simuni, T.; Corcos, D.M. Effects of Endurance Exercise Training on the Motor and Non-Motor Features of Parkinson’s Disease: A Review. J. Parkinson’s Dis. 2015, 5, 21–41. [Google Scholar] [CrossRef]
- Ahlskog, J.E. Does vigorous exercise have a neuroprotective effect in Parkinson disease? Neurology 2011, 77, 288–294. [Google Scholar] [CrossRef] [PubMed]
- van der Kolk, N.M.; de Vries, N.M.; Kessels, R.P.; Joosten, H.; Zwinderman, A.H.; Post, B.; Bloem, B.R. Effectiveness of Home-Based and Remotely Supervised Aerobic Exercise in Parkinson’s Disease: A Double-Blind, Randomised Controlled Trial. Lancet Neurol. 2019, 18, 998–1008. [Google Scholar] [CrossRef]
- Ridgel, A.L.; Vitek, J.L.; Alberts, J.L. Forced, Not Voluntary, Exercise Improves Motor Function in Parkinson’s Disease Patients. Neurorehabilit. Neural Repair 2009, 23, 600–608. [Google Scholar] [CrossRef]
- Ferrazzoli, D.; Ortelli, P.; Cucca, A.; Bakdounes, L.; Canesi, M.; Volpe, D. Motor-Cognitive Approach and Aerobic Training: A Synergism for Rehabilitative Intervention in Parkinson’s Disease. Neurodegener. Dis. Manag. 2020, 10, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Sell, T.C.; Abt, J.P.; Lephart, S.M. Physical Activity-Related Benefits of Walking During Golf. In Science and Golf V: Proceedings of the World Scientific Congress of Golf (V); Crews, D., Lutz., R., Eds.; World Scientific Congress of Golf Trust: Mesa, AZ, USA, 2008; ISBN 9780978873417. [Google Scholar]
- Cann, A.P.; Vandervoort, A.A.; Lindsay, D.M. Optimizing the Benefits versus Risks of Golf Participation by Older People. J. Geriatr. Phys. Ther. 2005, 28, 85–92. [Google Scholar] [CrossRef]
- Luscombe, J.; Murray, A.D.; Jenkins, E.; Archibald, D. A rapid review to identify physical activity accrued while playing golf. BMJ Open 2017, 7, 018993. [Google Scholar]
- Farahmand, B.; Broman, G.; De Faire, U.; Vågerö, D.; Ahlbom, A. Golf: A Game of Life and Death–Reduced Mortality in Swedish Golf Players. Scand. J. Med. Sci. Sports 2009, 19, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Stenner, B.J.; Mosewich, A.D.; Buckley, J.D. An exploratory investigation into the reasons why older people play golf. Qual. Res. Sport Exerc. Health 2016, 8, 257–272. [Google Scholar] [CrossRef]
- Murray, A.D.; Daines, L.; Archibald, D.; Hawkes, R.A.; Schiphorst, C.; Kelly, P.; Grant, L.; Mutrie, N. The relationships between golf and health: A scoping review. Br. J. Sports Med. 2017, 51, 12–19. [Google Scholar] [CrossRef]
- Vaile, P.A. The Soul of Golf. N. Am. Rev. 1910, 192, 359–368. [Google Scholar]
- Mun, F.; Suh, S.W.; Park, H.-J.; Choi, A. Kinematic relationship between rotation of lumbar spine and hip joints during golf swing in professional golfers. Biomed. Eng. Online 2015, 14, 41. [Google Scholar] [CrossRef] [PubMed]
- Adlington, G.S. Proper Swing Technique and Biomechanics of Golf. Clin. Sports Med. 1996, 15, 9–26. [Google Scholar] [CrossRef]
- Myers, J.; Lephart, S.; Tsai, Y.-S.; Sell, T.; Smoliga, J.; Jolly, J. The role of upper torso and pelvis rotation in driving performance during the golf swing. J. Sports Sci. 2008, 26, 181–188. [Google Scholar] [CrossRef]
- Nesbit, S.M.; Serrano, M. Work and power analysis of the golf swing. J. Sports Sci. Med. 2005, 4, 520–533. [Google Scholar]
- Nesbit, S.M.; McGinnis, R. Kinematic Analyses of the Golf Swing Hub Path and its Role in Golfer/Club Kinetic Transfers. J. Sports Sci. Med. 2009, 8, 235–246. [Google Scholar]
- Meister, D.W.; Ladd, A.; Butler, E.E.; Zhao, B.; Rogers, A.P.; Ray, C.J.; Rose, J. Rotational Biomechanics of the Elite Golf Swing: Benchmarks for Amateurs. J. Appl. Biomech. 2011, 27, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Gluck, G.S.; Bendo, J.A.; Spivak, J. The lumbar spine and low back pain in golf: A literature review of swing biomechanics and injury prevention. Spine J. 2008, 8, 778–788. [Google Scholar] [CrossRef]
- McHardy, A.; Pollard, H.; Garbutt, P.J. Muscle activity during the golf swing * Commentary. Br. J. Sports Med. 2005, 39, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Hume, A.P.P.A.; Keogh, J.; Reid, D. The Role of Biomechanics in Maximising Distance and Accuracy of Golf Shots. Sports Med. 2005, 35, 429–449. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.; Kang, T.G.; Mun, J.H. Biomechanical Evaluation of Dynamic Balance Control Ability During Golf Swing. J. Med. Biol. Eng. 2016, 36, 430–439. [Google Scholar] [CrossRef]
- Mitchell, K.; Banks, S.; Morgan, D.; Sugaya, H. Shoulder Motions During the Golf Swing in Male Amateur Golfers. J. Orthop. Sports Phys. Ther. 2003, 33, 196–203. [Google Scholar] [CrossRef]
- Ball, K.; Best, R. Centre of pressure patterns in the golf swing: Individual-based analysis. Sports Biomech. 2012, 11, 175–189. [Google Scholar] [CrossRef]
- Neumann, D.A. Axial Skeleton: Muscle and Joint Interactions. In Kinesiology of the Musculoskeletal System, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 379–422. ISBN 9780323287531. [Google Scholar]
- Tsang, W.W.; Hui-Chan, C.W. Static and Dynamic Balance Control in Older Golfers. J. Aging Phys. Act. 2010, 18, 1–13. [Google Scholar] [CrossRef]
- Gao, K.L.; Hui-Chan, C.W.Y.; Tsang, W.W.N. Golfers have better balance control and confidence than healthy controls. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 111, 2805–2812. [Google Scholar] [CrossRef] [PubMed]
- Tsang, W.W.; Hui-Chan, C.W. Effects of Exercise on Joint Sense and Balance in Elderly Men: Tai Chi versus Golf. Med. Sci. Sports Exerc. 2004, 36, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Du Bois, A.M.; Marcione, N.A.; Castle, S.C.; Salem, G.J. Golf as Therapeutic Exercise for Older Adults. In Proceedings of the World Scientific Congress of Golf, Abbotsford, BC, Canada, 11–13 July 2008. [Google Scholar]
- Bustelo, S.M.; Simon, B.; Warner, M.; Jácome, A.; Wootton, J.; Welch, D.; Samuel, D. Between-side symmetry of quadriceps thickness using ultrasound imaging in female golfers and non-golfers aged over 80 years. Osteoarthr. Cartil. 2016, 24, S65. [Google Scholar] [CrossRef][Green Version]
- Du Bois, A.; Marcione, N.; Powers, C.; Flanagan, S.; Schroeder, T.; Castle, S.; Moore, J.; Salem, G.J. The Effects of a Comprehensive Golf Training Program on Measures of Physical Performance and Dynamic Balance in Older Military Veterans. Int. J. Golf Sci. 2021, 9. in press. [Google Scholar]
- Johnson, R.; Plummer, L.; Chan, J.; Willis, A.M. Feasibility and Tolerability Randomized Clinical Trial of Golf Versus Tai Chi for People with Moderate Parkinson’s Disease (1962). Neurology 2021, 96, 15 Supplement. [Google Scholar]
- Sacheli, M.A.; Murray, D.K.; Vafai, N.; Cherkasova, M.V.; Dinelle, K.; Shahinfard, E.; Neilson, N.; McKenzie, J.; Schulzer, M.; Appel-Cresswell, S. Habitual Exercisers Versus Sedentary Subjects with Parkinson’s Disease: Multimodal Pet and Fmri Study. Mov. Disord. 2018, 33, 1945–1950. [Google Scholar] [CrossRef] [PubMed]
- Rafferty, M.R.; Schmidt, P.N.; Luo, S.T.; Li, K.; Marras, C.; Davis, T.L.; Guttman, M.; Cubillos, F.; Simuni, T. Regular Exercise, Quality of Life, and Mobility in Parkinson’s Disease: A Longitudinal Analysis of National Parkinson Foundation Quality Improvement Initiative Data. J. Parkinson’s Dis. 2017, 7, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Lauzé, M.; Daneault, J.-F.; Duval, C. The Effects of Physical Activity in Parkinson’s Disease: A Review. J. Park. Dis. 2016, 6, 685–698. [Google Scholar] [CrossRef]
- Palakurthi, B.; Burugupally, S.P. Postural Instability in Parkinson’s Disease: A Review. Brain Sci. 2019, 9, 239. [Google Scholar] [CrossRef]
- Kim, S.D.; Allen, N.E.; Canning, C.G.; Fung, V.S.C. Postural Instability in Patients with Parkinson’s Disease. CNS Drugs 2013, 27, 97–112. [Google Scholar] [CrossRef]
- Levy, G.; Louis, E.D.; Côté, L.; Pérez, M.; Mejia-Santana, H.; Andrews, H.; Harris, J.; Waters, C.; Ford, B.; Frucht, S.; et al. Contribution of Aging to the Severity of Different Motor Signs in Parkinson Disease. Arch. Neurol. 2005, 62, 467–472. [Google Scholar] [CrossRef]
- King, L.A.; Horak, F.B. Lateral Stepping for Postural Correction in Parkinson’s Disease. Arch. Phys. Med. Rehabil. 2008, 89, 492–499. [Google Scholar] [CrossRef]
- Lin, C.-C.; Creath, R.A.; Rogers, M.W. Variability of Anticipatory Postural Adjustments During Gait Initiation in Individuals with Parkinson Disease. J. Neurol. Phys. Ther. 2016, 40, 40–46. [Google Scholar] [CrossRef]
- Dijkstra, B.W.; Gilat, M.; Lizama, L.E.C.; Mancini, M.; Bergmans, B.; Verschueren, S.M.; Nieuwboer, A. Impaired Weight-Shift Amplitude in People with Parkinson’s Disease with Freezing of Gait. J. Park. Dis. 2021, 1–14. [Google Scholar] [CrossRef]
- Horak, F.; Nutt, J.; Nashner, L. Postural inflexibility in parkinsonian subjects. J. Neurol. Sci. 1992, 111, 46–58. [Google Scholar] [CrossRef]
- Carpenter, M.G.; Allum, J.H.J.; Honegger, F.; Adkin, A.L.; Bloem, B.R. Postural abnormalities to multidirectional stance perturbations in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1245–1254. [Google Scholar] [CrossRef] [PubMed]
- Bertram, C.P.; Lemay, M.; Stelmach, G.E. The effect of Parkinson’s disease on the control of multi-segmental coordination. Brain Cogn. 2005, 57, 16–20. [Google Scholar] [CrossRef]
- Wang, J.; Bohan, M.; Leis, B.C.; Stelmach, G.E. Altered coordination patterns in parkinsonian patients during trunk-assisted prehension. Parkinsonism Relat. Disord. 2006, 12, 211–222. [Google Scholar] [CrossRef]
- Nieuwboer, A.; Rochester, L.; Müncks, L.; Swinnen, S.P. Motor Learning in Parkinson’s Disease: Limitations and Potential for Rehabilitation. Parkinsonism Relat. Disord. 2009, 15, S53–S58. [Google Scholar] [CrossRef]
- Georgiou, N.; Bradshaw, J.L.; Iansek, R.; Phillips, J.G.; Mattingley, J.B.; Bradshaw, J.A. Reduction in External Cues and Movement Sequencing in Parkinson’s Disease. J. Neurol. Neurosurg. Psychiatry 1994, 57, 368–370. [Google Scholar] [CrossRef]
- Rocha, P.A.; Porfírio, G.M.; Ferraz, H.B.; Trevisani, V.F. Effects of External Cues on Gait Parameters of Parkinson’s Disease Patients: A Systematic Review. Clin. Neurol. Neurosurg. 2014, 124, 127–134. [Google Scholar] [CrossRef]
- Chong, T.T.-J.; Bonnelle, V.; Manohar, S.; Veromann, K.-R.; Muhammed, K.; Tofaris, G.K.; Hu, M.; Husain, M. Dopamine Enhances Willingness to Exert Effort for Reward in Parkinson’s Disease. Cortex 2015, 69, 40–46. [Google Scholar] [CrossRef] [PubMed]
| Risk Factor | Reference(s) Cited |
|---|---|
| Exercise/Sports-Related: | |
| Balance-mobility | [47,48,50,51,52] |
| Decreased arm swing | [48] |
| Gait disturbances | [28,47,50] |
| Inability to get up from a sitting position | [28,53] |
| Orthostasis | [28,47] |
| PD-specific clinical symptoms: | |
| Bradykinesia | [53] |
| Dyskinesia | [51] |
| Freezing | [51,52,54,55,56,57] |
| Rigidity | [50,58] |
| General Neurological/PD-Related: | |
| Advanced age | [53,59] |
| Cognition | [57,60,61] |
| Depression and anxiety | [48,50,62] |
| Disease severity | [51,52,56] |
| Be safe, be careful, and realize that most PwP typically have gait and balance issues |
| To ensure safety, the benefit of a new exercise program will only work if you have (i) talked with your neurologist; (ii) worked out a plan with your physical therapist or personal trainer that includes stretching exercises for pre- and post-golf and discussion on the optimal dose and frequency as you prepare for a new exercise; (iii) if you know how to play golf and you are starting anew, welcome back; and (iv) if you are a beginner golfer, welcome, and have fun. |
| Plan #1: Practice the golf swing at home |
| This plan requires only a couple of golf clubs, a golf mat to swing on and likely plastic golf balls (and possibly a net to capture balls). If weather permits, practice outside. The goal is to swing the golf club 100 times/day for 3–5 days per week. |
| Plan #2: Hitting golf balls at a golf driving range |
| The next level is going to a golf driving range, where you are hitting real golf balls and likely have a few more golf clubs. Similar goal, try to hit 100 golf balls/day for 3–5 days per week. |
| Plan #3: Play a round of golf at a local golf course/club |
| The third level is to play a round of golf, playing either 9 holes or 18 holes at a local golf course. If you are able to walk, you enhance the exercise routine. Try to play once (or twice) per week. Golf has many rules, yet it is a very honorable and social sport. You can enjoy it playing alone or with friends. Two downsides to golf include the time it takes to play 9 or 18 holes of golf, and it is not without significant equipment and clothes expenditures. However, plans #1 and #2 described above are much more reasonable in terms of time and cost. |
| Plan #4: Virtual Golf |
| “Park Place Golf Club” is designed specifically for Parkinson’s disease. This is a virtual 18-hole golf and fitness training program that provides instructional golf videos paired with functional fitness workouts where you can track your progress. Park Place Virtual Golf was created by Taunya Foerster [from https://parkplacegolfclub.com (accessed on 16 April 2021)]. |
| Learning how to play golf |
| “Golf for Beginners: So You Want To Play Golf” [from https://www.golfdigest.com/gallery/golf-beginners-tips/amp (accessed on 16 April 2021)]; “Beginner Basics” [from https://schools.golfdigest.com/series/will-robins-beginner-basics-rsuzob7 (accessed on 16 April 2021)]; “The best way to learn golf now” [from https://www.golfdigest. com/story/how-to-play-golf-now/amp (accessed on 16 April 2021)]; “A Beginner’s Golf Guide: What every new golfer should know when picking up the game” [from https://www.golfdigest.com/story/the-complete-beginner-s-guide-to-golf (accessed on 16 April 2021)]; “Golf 101: Dos and don’ts for beginners” [from https://golf.com/instruction/golf-101-dos-and-donts-for-beginners/ (accessed on 16 April 2021)]; “How to Play Golf | The Beginner’s Guide” [from https://golfworkoutprogram.com/how-to-play-golf/ (accessed on 16 April 2021)]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bliss, R.R.; Church, F.C. Golf as a Physical Activity to Potentially Reduce the Risk of Falls in Older Adults with Parkinson’s Disease. Sports 2021, 9, 72. https://doi.org/10.3390/sports9060072
Bliss RR, Church FC. Golf as a Physical Activity to Potentially Reduce the Risk of Falls in Older Adults with Parkinson’s Disease. Sports. 2021; 9(6):72. https://doi.org/10.3390/sports9060072
Chicago/Turabian StyleBliss, Rebecca R., and Frank C. Church. 2021. "Golf as a Physical Activity to Potentially Reduce the Risk of Falls in Older Adults with Parkinson’s Disease" Sports 9, no. 6: 72. https://doi.org/10.3390/sports9060072
APA StyleBliss, R. R., & Church, F. C. (2021). Golf as a Physical Activity to Potentially Reduce the Risk of Falls in Older Adults with Parkinson’s Disease. Sports, 9(6), 72. https://doi.org/10.3390/sports9060072







