Effect of New Zealand Blackcurrant Extract on Isometric Contraction-Induced Fatigue and Recovery: Potential Muscle-Fiber Specific Effects
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Experimental Design
2.2. Mechanical Testing and Surface Electrical Stimulation of the Quadriceps Femoris Muscles
2.3. New Zealand Blackcurrant Extract Supplementation
2.4. Physical Activity and Dietary Analysis
2.5. Statistical Analysis
3. Results
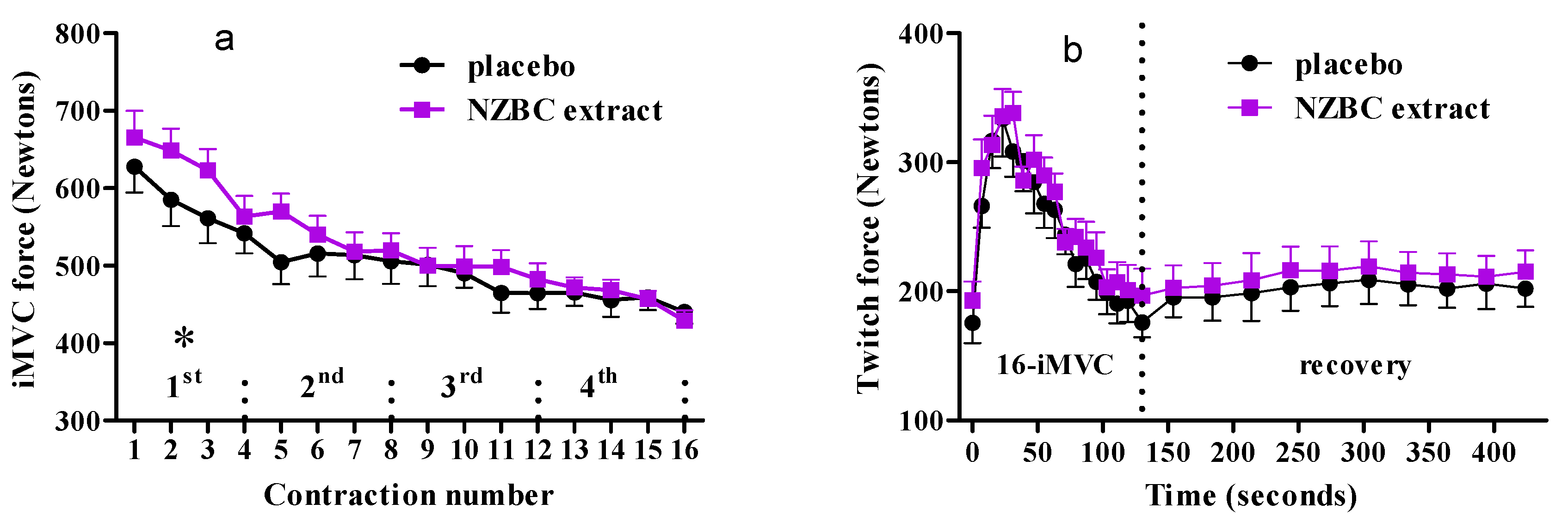
3.1. Performance of Maximal Voluntary Isometric Contractions during the Fatiguing Exercise Protocol
3.2. Twitch Forces during Fatiguing Exercise Protocol and Recovery
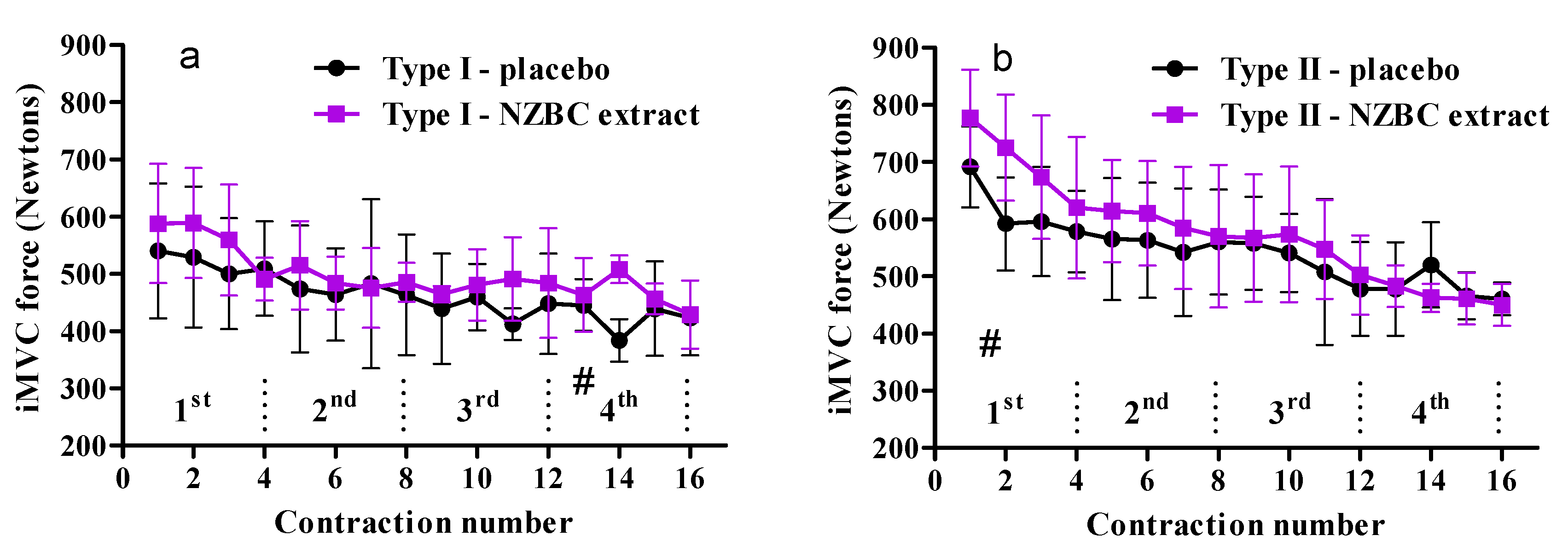
3.3. Fiber-Type Specific Responses on Performance of Maximal Voluntary Isometric Contractions during Fatiguing Exercise Protocol
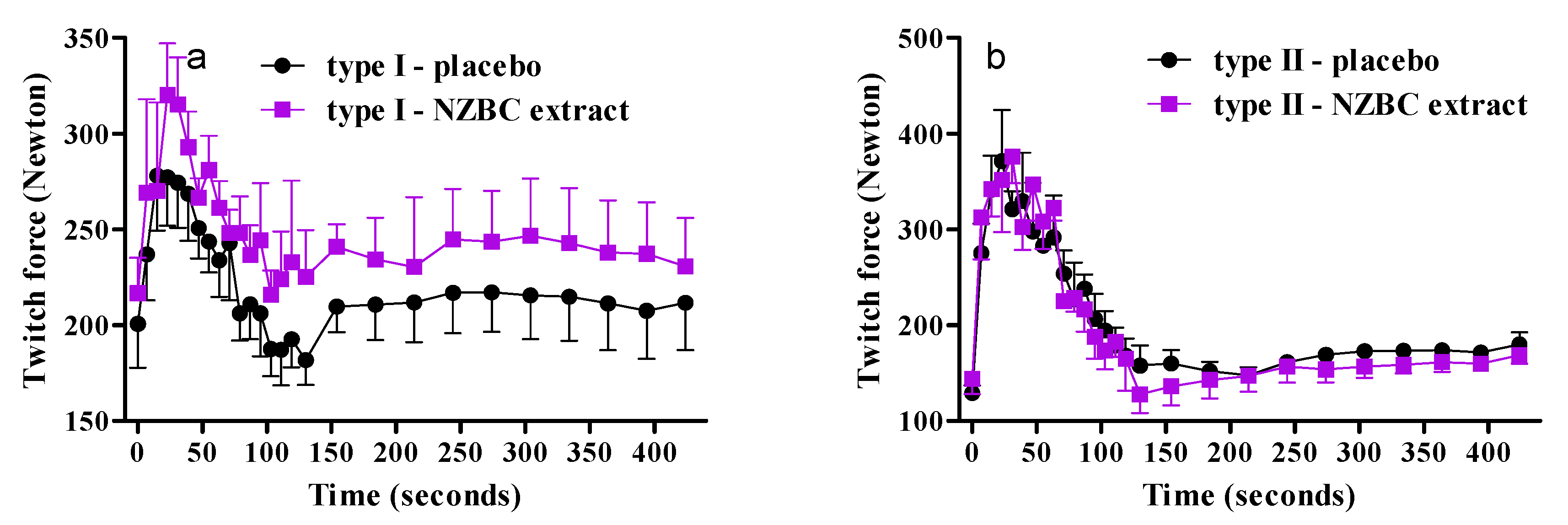
3.4. Fiber-Type Specific Responses on Twitch Forces during Fatiguing Exercise Protocol and Recovery
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bailey, S.J.; Winyard, P.; Vanhatalo, A.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Tarr, J.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation reduces the O2cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J. Appl. Physiol. 2009, 107, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Bowtell, J.; Kelly, V. Fruit-Derived Polyphenol Supplementation for Athlete Recovery and Performance. Sports Med. 2019, 49, 3–23. [Google Scholar] [CrossRef] [PubMed]
- Cortez, R.E.; De Mejia, E.G. Blackcurrants (Ribes nigrum): A Review on Chemistry, Processing, and Health Benefits. J. Food Sci. 2019, 84, 2387–2401. [Google Scholar] [CrossRef]
- Cook, M.D.; Myers, S.D.; Blacker, S.D.; Willems, M.E.T. New Zealand blackcurrant extract improves cycling performance and fat oxidation in cyclists. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 115, 2357–2365. [Google Scholar] [CrossRef]
- Perkins, I.C.; Vine, S.A.; Blacker, S.D.; Willems, M.E.T. New Zealand Blackcurrant Extract Improves High-Intensity Intermittent Running. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 487–493. [Google Scholar] [CrossRef]
- Murphy, C.A.; Cook, M.D.; Willems, M.E.T. Effect of New Zealand Blackcurrant Extract on Repeated Cycling Time Trial Performance. Sports 2017, 5, 25. [Google Scholar] [CrossRef] [PubMed]
- Godwin, C.; Cook, M.D.; Willems, M.E.T. Effect of New Zealand Blackcurrant Extract on Performance during the Running Based Anaerobic Sprint Test in Trained Youth and Recreationally Active Male Football Players. Sports 2017, 5, 69. [Google Scholar] [CrossRef]
- Willems, M.E.T.; Cousins, L.; Williams, D.; Blacker, S.D. Beneficial Effects of New Zealand Blackcurrant Extract on Maximal Sprint Speed during the Loughborough Intermittent Shuttle Test. Sports 2016, 4, 42. [Google Scholar] [CrossRef]
- Potter, J.A.; Hodgson, C.I.; Broadhurst, M.; Howell, L.; Gilbert, J.; Willems, M.E.T.; Perkins, I.C. Effects of New Zealand blackcurrant extract on sport climbing performance. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 120, 67–75. [Google Scholar] [CrossRef]
- Cook, M.D.; Myers, S.D.; Gault, M.L.; Willems, M.E.T. Blackcurrant Alters Physiological Responses and Femoral Artery Diameter during Sustained Isometric Contraction. Nutrients 2017, 9, 556. [Google Scholar] [CrossRef]
- Fryer, S.; Paterson, C.; Perkins, I.C.; Gloster, C.; Willems, M.E.; Potter, J.A. New Zealand Blackcurrant Extract Enhances Muscle Oxygenation During Forearm Exercise in Intermediate-Level Rock Climbers. Int. J. Sport Nutr. Exerc. Metab. 2020, 30, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Lomiwes, D.; Ha, B.; Ngametua, N.; Burr, N.S.; Cooney, J.M.; Trower, T.M.; Sawyer, G.; Hedderley, D.; Hurst, R.D.; Hurst, S.M. Timed consumption of a New Zealand blackcurrant juice support positive affective responses during a self-motivated moderate walking exercise in healthy sedentary adults. J. Int. Soc. Sports Nutr. 2019, 16, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hamada, T.; Sale, D.G.; MacDougall, J.D.; Tarnopolsky, M.A. Interaction of fibre type, potentiation and fatigue in human knee extensor muscles. Acta Physiol. Scand. 2003, 178, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Bell, P.G.; Stevenson, E.J.; Davison, G.W.; Howatson, G. The Effects of Montmorency Tart Cherry Concentrate Supplementation on Recovery Following Prolonged, Intermittent Exercise. Nutrients 2016, 8, 441. [Google Scholar] [CrossRef]
- Hargreaves, M. Fatigue mechanisms determining exercise performance: Integrative physiology is systems biology. J. Appl. Physiol. 2008, 104, 1541–1542. [Google Scholar] [CrossRef]
- Colliander, E.B.; Dudley, G.A.; Tesch, P.A. Skeletal muscle fiber type composition and performance during repeated bouts of maximal, concentric contractions. Graefe’s Arch. Clin. Exp. Ophthalmol. 1988, 58, 81–86. [Google Scholar] [CrossRef]
- Lievens, E.; Klass, M.; Bex, T.; Derave, W. Muscle fiber typology substantially influences time to recover from high-intensity exercise. J. Appl. Physiol. 2020, 128, 648–659. [Google Scholar] [CrossRef]
- Breese, B.C.; McNarry, M.A.; Marwood, S.; Blackwell, J.R.; Bailey, S.J.; Jones, A.M. Beetroot juice supplementation speeds O2 uptake kinetics and improves exercise tolerance during severe-intensity exercise initiated from an elevated metabolic rate. Am. J. Physiol. Integr. Comp. Physiol. 2013, 305, R1441–R1450. [Google Scholar] [CrossRef]
- Hamada, T.; Sale, D.G.; MacDougall, J.D.; Tarnopolsky, M.A. Postactivation potentiation, fiber type, and twitch contraction time in human knee extensor muscles. J. Appl. Physiol. 2000, 88, 2131–2137. [Google Scholar] [CrossRef]
- Cook, M.D.; Myers, S.D.; Gault, M.L.; Edwards, V.C.; Willems, M.E.T. Dose effects of New Zealand blackcurrant on substrate oxidation and physiological responses during prolonged cycling. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 117, 1207–1216. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; Du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef] [PubMed]
- Keller, M.L.; Pruse, J.; Yoon, T.; Schlinder-Delap, B.; Harkins, A.; Hunter, S.K. Supraspinal Fatigue is Similar in Men and Women for a Low-Force Fatiguing Contraction. Med. Sci. Sports Exerc. 2011, 43, 1873–1883. [Google Scholar] [CrossRef] [PubMed]
- Søgaard, K.; Gandevia, S.C.; Todd, G.; Petersen, N.T.; Taylor, J.L. The effect of sustained low-intensity contractions on supraspinal fatigue in human elbow flexor muscles. J. Physiol. 2006, 573, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Curran-Everett, D.; Benos, D.J. Guidelines for reporting statistics in journals published by the American Physiological Society. Am. J. Physiol. Integr. Comp. Physiol. 2004, 287, R247–R249. [Google Scholar] [CrossRef] [PubMed]
- Cairns, S.P.; Inman, L.A.G.; MacManus, C.P.; Van De Port, I.G.L.; Ruell, P.A.; Thom, J.; Thompson, M.W. Central activation, metabolites, and calcium handling during fatigue with repeated maximal isometric contractions in human muscle. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 117, 1557–1571. [Google Scholar] [CrossRef]
- Henriksson, J.; Katz, A.; Sahlin, K. Redox state changes in human skeletal muscle after isometric contraction. J. Physiol. 1986, 380, 441–451. [Google Scholar] [CrossRef]
- Westerblad, H.; Allen, D.G.; Bruton, J.D.; Andrade, F.H.; Lännergren, J. Mechanisms underlying the reduction of isometric force in skeletal muscle fatigue. Acta Physiol. Scand. 1998, 162, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Trump, M.E.; Heigenhauser, G.J.; Putman, C.T.; Spriet, L.L. Importance of muscle phosphocreatine during intermittent maximal cycling. J. Appl. Physiol. 1996, 80, 1574–1580. [Google Scholar] [CrossRef]
- Matsumoto, H.; Takenami, E.; Iwasaki-Kurashige, K.; Osada, T.; Katsumura, T.; Hamaoka, T. Effects of blackcurrant anthocyanin intake on peripheral muscle circulation during typing work in humans. Graefe’s Arch. Clin. Exp. Ophthalmol. 2004, 94, 36–45. [Google Scholar] [CrossRef]
- Kushmerick, M.J.; Moerland, T.S.; Wiseman, R.W. Mammalian skeletal muscle fibers distinguished by contents of phosphocreatine, ATP, and Pi. Proc. Natl. Acad. Sci. USA 1992, 89, 7521–7525. [Google Scholar] [CrossRef]
- Greenhaff, P.L.; Nevill, M.E.; Soderlund, K.; Bodin, K.; Boobis, L.H.; Williams, C.; Hultman, E. The metabolic responses of human type I and II muscle fibres during maximal treadmill sprinting. J. Physiol. 1994, 478, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Egginton, S.; Amigo-Benavent, M.; Mannion, A.; Ross, H. Is human skeletal muscle capillary supply modelled according to fibre size or fibre type? Exp. Physiol. 1997, 82, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Bernûs, G.; De Suso, J.M.G.; Alonso, J.; Martin, P.A.; Prat, J.A.; Arús, C. 31P-MRS of quadriceps reveals quantitative differences between sprinters and long-distance runners. Med. Sci. Sports Exerc. 1993, 11, 479–484. [Google Scholar] [CrossRef]
- Haider, G.; Folland, J.P. Nitrate Supplementation Enhances the Contractile Properties of Human Skeletal Muscle. Med. Sci. Sports Exerc. 2014, 46, 2234–2243. [Google Scholar] [CrossRef] [PubMed]
- Vandenboom, R. Modulation of Skeletal Muscle Contraction by Myosin Phosphorylation. Compr. Physiol. 2016, 7, 171–212. [Google Scholar] [CrossRef]
- Zimmermann, H.B.; MacIntosh, B.R.; Pupo, J.D. Does postactivation potentiation (PAP) increase voluntary performance? Appl. Physiol. Nutr. Metab. 2020, 45, 349–356. [Google Scholar] [CrossRef]
- Abt, G.; Boreham, C.; Davison, G.; Jackson, R.; Nevill, A.; Wallace, E.; Williams, M. Power, precision, and sample size estimation in sport and exercise science research. J. Sports Sci. 2020, 38, 1933–1935. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Willems, M.E.T.; Bradley, M.; Blacker, S.D.; Perkins, I.C. Effect of New Zealand Blackcurrant Extract on Isometric Contraction-Induced Fatigue and Recovery: Potential Muscle-Fiber Specific Effects. Sports 2020, 8, 135. https://doi.org/10.3390/sports8100135
Willems MET, Bradley M, Blacker SD, Perkins IC. Effect of New Zealand Blackcurrant Extract on Isometric Contraction-Induced Fatigue and Recovery: Potential Muscle-Fiber Specific Effects. Sports. 2020; 8(10):135. https://doi.org/10.3390/sports8100135
Chicago/Turabian StyleWillems, Mark E. T., Megan Bradley, Sam D. Blacker, and Ian C. Perkins. 2020. "Effect of New Zealand Blackcurrant Extract on Isometric Contraction-Induced Fatigue and Recovery: Potential Muscle-Fiber Specific Effects" Sports 8, no. 10: 135. https://doi.org/10.3390/sports8100135
APA StyleWillems, M. E. T., Bradley, M., Blacker, S. D., & Perkins, I. C. (2020). Effect of New Zealand Blackcurrant Extract on Isometric Contraction-Induced Fatigue and Recovery: Potential Muscle-Fiber Specific Effects. Sports, 8(10), 135. https://doi.org/10.3390/sports8100135





