Exercise-Induced Muscle Damage and Recovery in Young and Middle-Aged Males with Different Resistance Training Experience
Abstract
1. Introduction
2. Materials and Methods
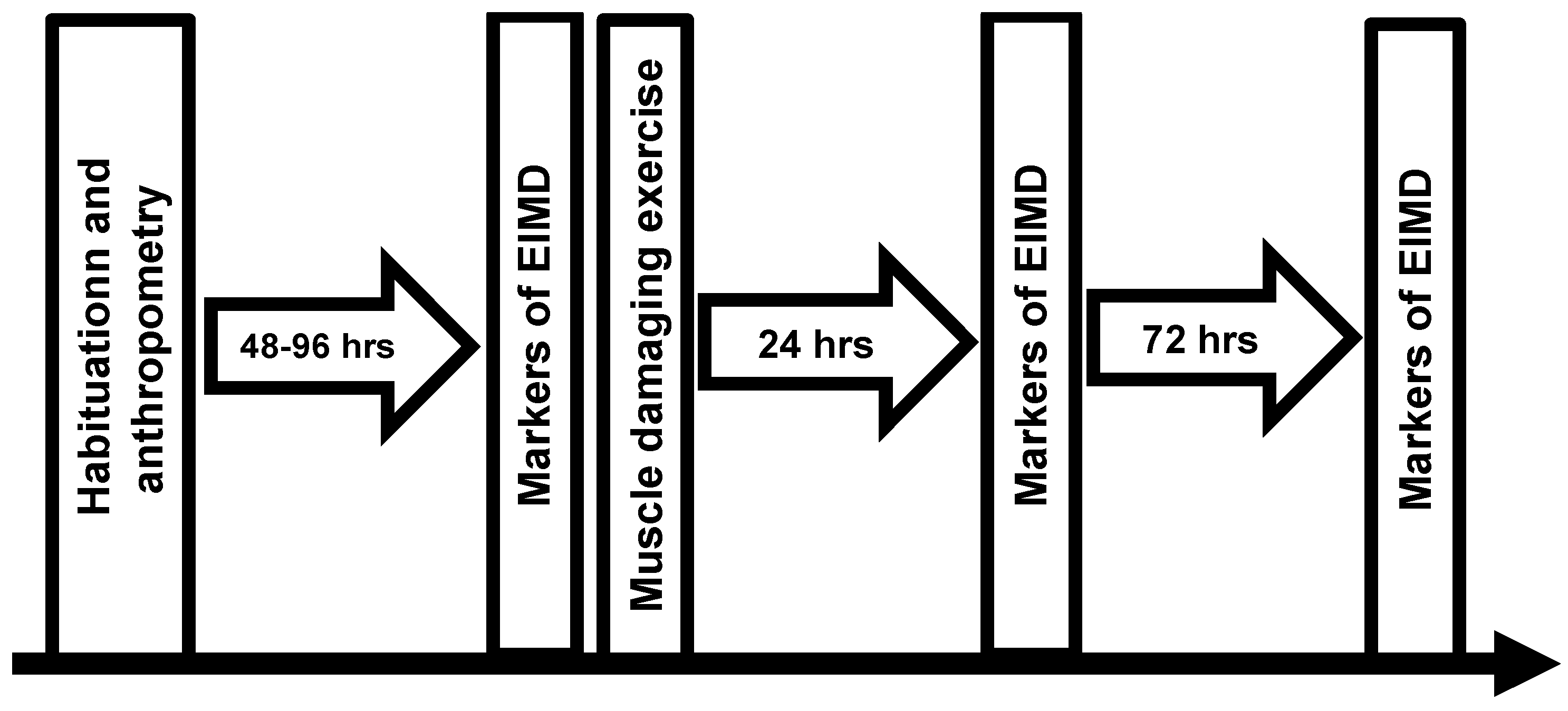
2.1. Design
2.2. Participants
2.3. Procedures
2.3.1. Anthropometric Measurements
2.3.2. Resistance Training History and Sports Participation
2.3.3. Maximal Strength Testing
2.3.4. Indirect Markers of Muscle Damage
2.3.5. Assessment of Maximal Voluntary Contraction and Voluntary Activation
2.3.6. Assessment of Peak Power During Squat
2.3.7. Muscle-Damaging Exercise Protocol
2.4. Statistical Analyses
3. Results
3.1. Biometric Measures and Training History
3.2. External Load Response during the Muscle-Damaging Protocol
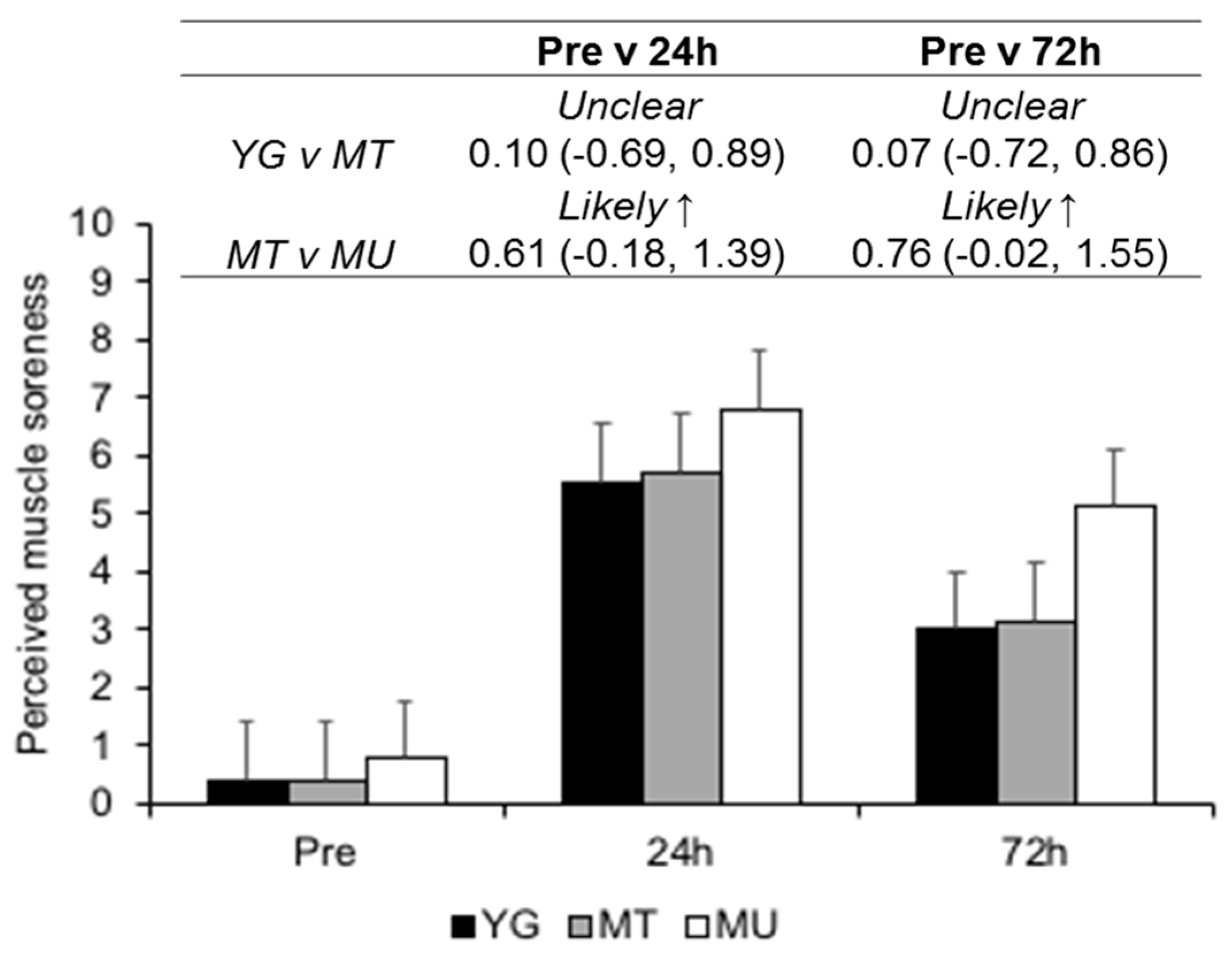
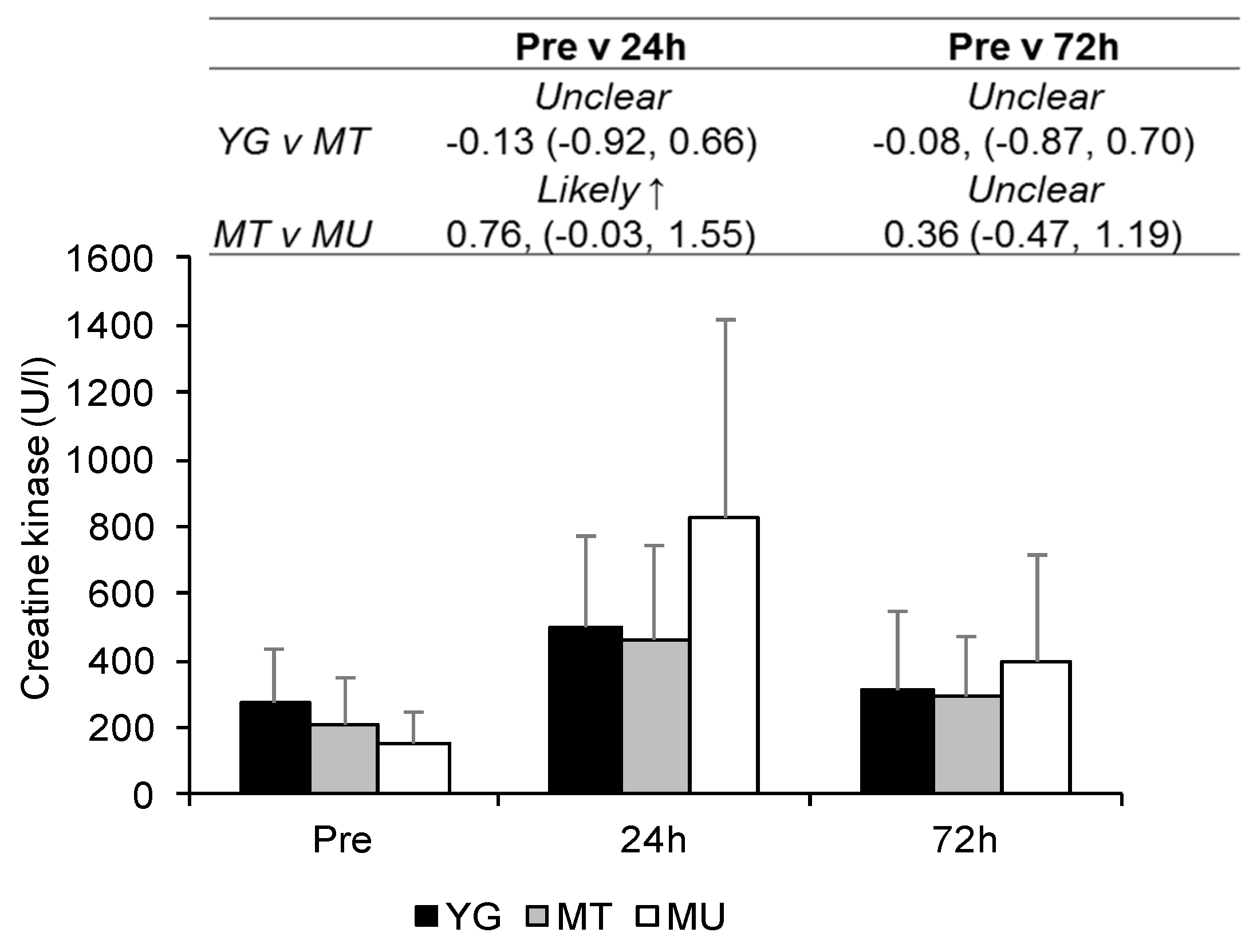
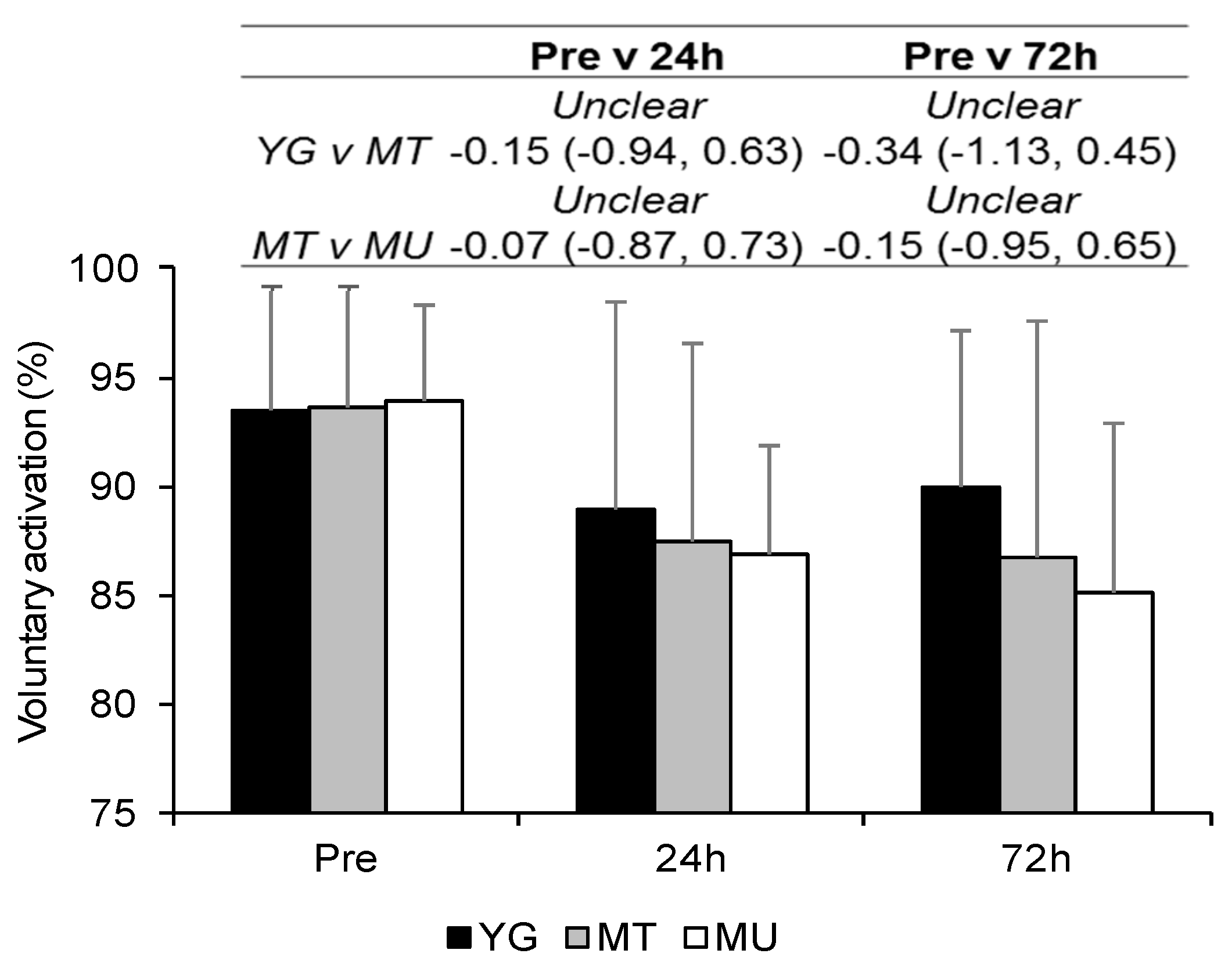
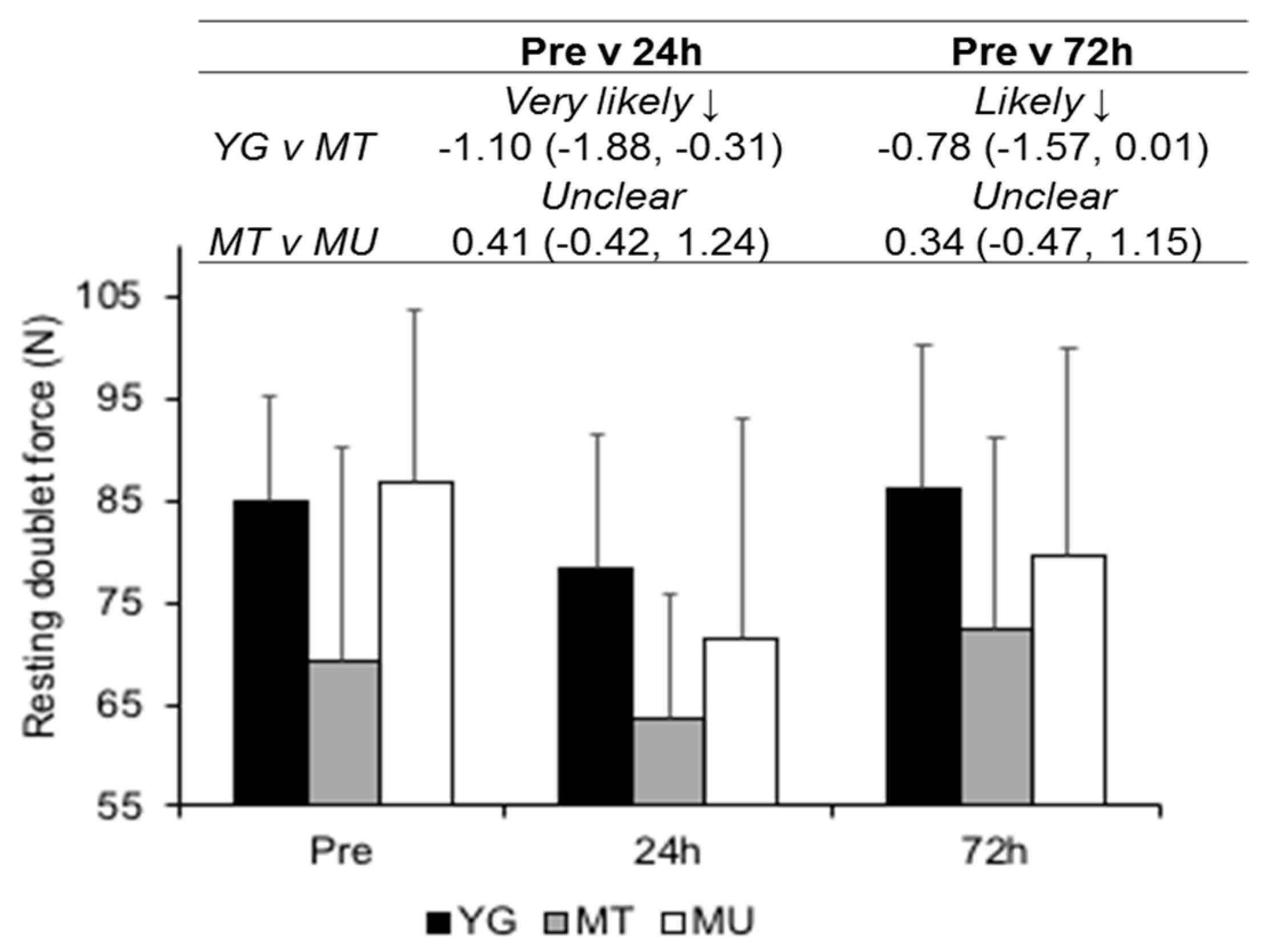
3.3. Indirect Markers of Muscle Damage
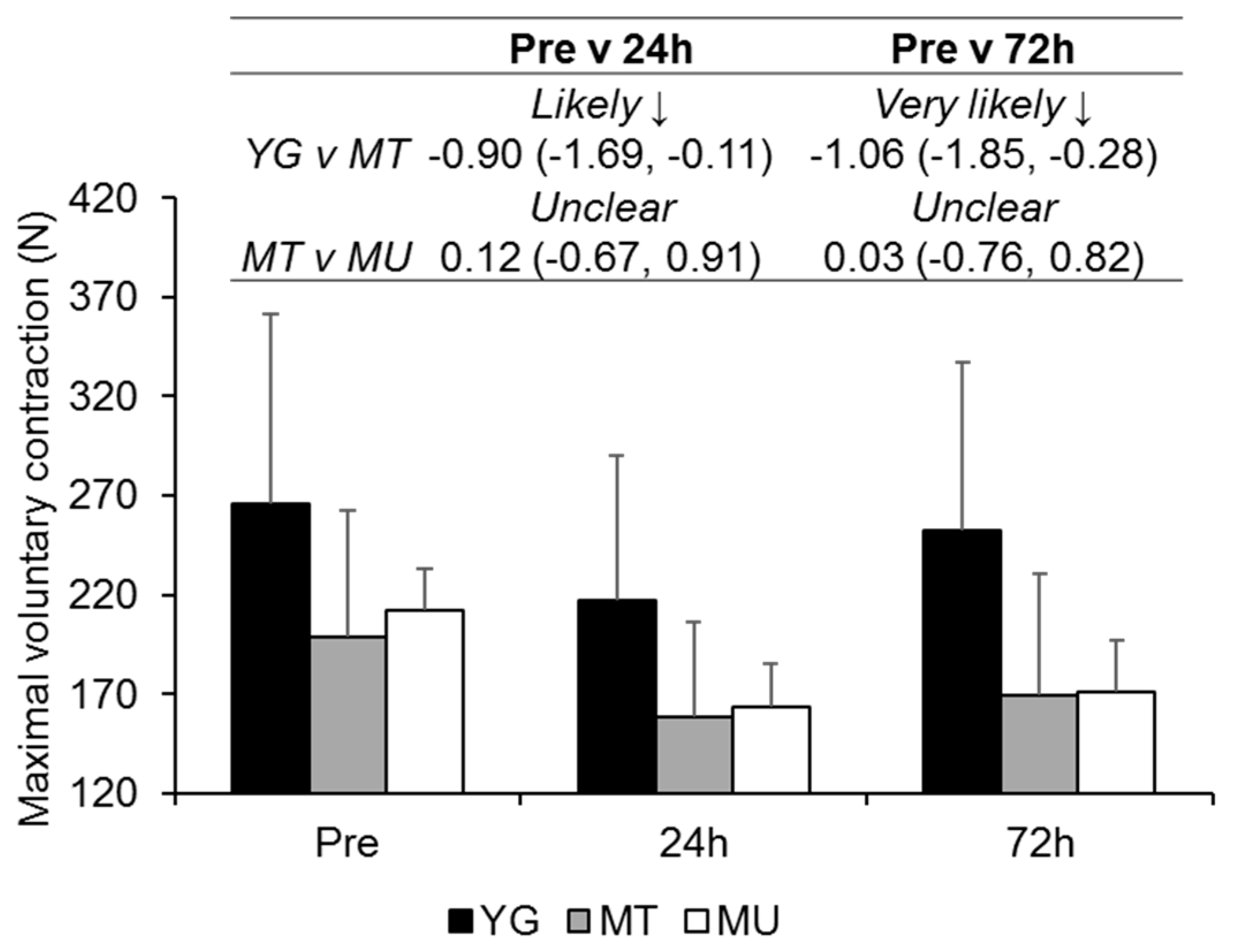
3.4. Peak Power during Squat Exercise
4. Discussion
4.1. Confirmation of EIMD
4.2. Changes in Indirect Markers of EIMD in Trained Young and Middle-Aged Males
4.3. Changes in Muscle Function in Trained Young and Middle-Aged Males
4.4. Differences in Recovery Between Trained and Untrained Middle-Aged Males
4.5. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Office for National Statistics. National Population Projections: 2014-Based Statistical Bulletin. Available online: https://www.ons.gov.uk/peoplepopulationandcommunity/populationandmigration/populationprojections/bulletins/nationalpopulationprojections/2015-10-29 (accessed on 28 May 2019).
- Pantoja, P.D.; Saez De Villarreal, E.; Brisswalter, J.; Peyré-Tartaruga, L.A.; Morin, J.B. Sprint acceleration mechanics in masters athletes. Med. Sci. Sports Exerc. 2016, 48, 2469–2474. [Google Scholar] [CrossRef] [PubMed]
- Frontera, W.R.; Suh, D.; Krivickas, L.S.; Hughes, V.A.; Goldstein, R.; Roubenoff, R. Skeletal muscle fiber quality in older men and women. Am. J. Physiol. Cell Physiol. 2000, 279, C611–C618. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Chilibeck, P.D. Differences in size, strength, and power of upper and lower body muscle groups in young and older men. J. Gerontol. Biol. Sci. 2005, 60, 148–156. [Google Scholar] [CrossRef]
- Fernandes, J.F.T.; Lamb, K.L.; Twist, C. A comparison of load-velocity and load-power relationships between well-trained young and middle-aged males during three popular resistance exercises. J. Strength Cond. Res. 2018, 32, 1440–1447. [Google Scholar] [CrossRef]
- Roth, S.M.; Martel, G.F.; Ivey, F.M.; Lemmer, J.T.; Tracy, B.L.; Hurlbut, D.E.; Metter, E.J.; Hurley, B.F.; Rogers, M.A. Ultrastructural muscle damage in young vs. older men after high-volume, heavy-resistance strength training. J. Appl. Physiol. 1999, 86, 1833–1840. [Google Scholar] [CrossRef]
- Hyldahl, R.D.; Hubal, M.J. Lengthening our perspective: Morphological, cellular, and molecular responses to eccentric exercise. Muscle Nerve 2014, 49, 155–170. [Google Scholar] [CrossRef]
- Damas, F.; Nosaka, K.; Libardi, C.A.; Chen, T.C.; Ugrinowitsch, C. Susceptibility to exercise-induced muscle damage: A cluster analysis with a large sample. Int. J. Sports Med. 2016, 37, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.; Willardson, J.M. Short recovery augments magnitude of muscle damage in high responders. Med. Sci. Sports Exerc. 2010, 42, 1370–1374. [Google Scholar] [CrossRef]
- Hyldahl, R.D.; Chen, T.C.; Nosaka, K. Mechanisms and mediators of the skeletal muscle repeated bout effect. Exerc. Sport Sci. Rev. 2017, 45, 24–33. [Google Scholar] [CrossRef]
- Nosaka, K.; Sakamoto, K.E.I.; Newton, M.; Sacco, P. How long does the protective effect on eccentric exercise-induced muscle damage last? Med. Sci. Sports Exerc. 2001, 33, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Lavender, A.P.; Nosaka, K. Responses of old men to repeated bouts of eccentric exercise of the elbow flexors in comparison with young men. Eur. J. Appl. Physiol. 2006, 97, 619–626. [Google Scholar] [CrossRef]
- Gorianovas, G.; Skurvydas, A.; Streckis, V.; Brazaitis, M.; Kamandulis, S.; McHugh, M.P. Repeated bout effect was more expressed in young adult males than in elderly males and boys. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Lavender, A.P.; Nosaka, K. Comparison between old and young men for changes in makers of muscle damage following voluntary eccentric exercise of the elbow flexors. Appl. Physiol. Nutr. Metab. 2006, 31, 218–225. [Google Scholar] [CrossRef]
- Lavender, A.P.; Nosaka, K. Fluctuations of isometric force after eccentric exercise of the elbow flexors of young, middle-aged, and old men. Eur. J. Appl. Physiol. 2007, 100, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Gordon, J., III; Hoffman, J.R.; Arroyo, E.; Varanoske, A.; Coker, N.; Gepner, Y.; Wells, A.; Stout, J.; Fukuda, D. Comparisons in the recovery response from resistance exercise between young and middle-aged men. J. Strength Cond. Res. 2017, 31, 3454–3462. [Google Scholar] [CrossRef] [PubMed]
- Lavender, A.P.; Nosaka, K. Changes in markers of muscle damage of middle-aged and young men following eccentric exercise of the elbow flexors. J. Sci. Med. Sport 2008, 11, 124–131. [Google Scholar] [CrossRef]
- Buford, T.W.; MacNeil, R.G.; Clough, L.G.; Dirain, M.; Sandesara, B.; Pahor, M.; Manini, T.M.; Leeuwenburgh, C. Active muscle regeneration following eccentric contraction-induced injury is similar between healthy young and older adults. J. Appl. Physiol. 2014, 116, 1481–1490. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.W.; Newton, M.; McGuigan, M.R.; Nosaka, K. Comparison between old and young men for responses to fast velocity maximal lengthening contractions of the elbow flexors. Eur. J. Appl. Physiol. 2008, 104, 531–539. [Google Scholar] [CrossRef]
- Batterham, A.; George, K. Reliability in evidence-based clinical practice: A primer for allied health professionals. Phys. Sport 2003, 4, 122–128. [Google Scholar] [CrossRef]
- Macdonald, G.Z.; Button, D.C.; Drinkwater, E.J.; Behm, D.G. Foam rolling as a recovery tool after an intense bout of physical activity. Med. Sci. Sports Exerc. 2014, 46, 131–142. [Google Scholar] [CrossRef]
- Batterham, A.M.; Atkinson, G. How big does my sample need to be? A primer on the murky world of sample size estimation. Phys. Sport 2005, 6, 153–163. [Google Scholar] [CrossRef]
- Fernandes, J.F.T.; Lamb, K.L.; Twist, C. The intra- and inter-day reproducibility of the FitroDyne as a measure of multi-jointed muscle function. Isokinet. Exerc. Sci. 2016, 24, 39–49. [Google Scholar] [CrossRef]
- Jackson, A.S.; Pollock, M.L. Generalized equations for predicting body density of men. Br. J. Nutr. 1978, 40, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Heyward, V.H.; Wagner, D.R. Applied Body Composition Assessment; Human Kinetics: Champaign, IL, USA, 2004. [Google Scholar]
- Wathen, D. Load Assingment. In Essenetials of Strength and Conditioning; Human Kinetics: Champaign, IL, USA, 1994; pp. 435–446. [Google Scholar]
- LeSuer, D.; McCormick, J.; Mayhew, J.; Wasserstein, R.; Arnold, M. The accuracy of prediction equations for estimating 1-RM performance in the bench press squat and deadlift. J. Strength Cond. Res. 1997, 11, 211–213. [Google Scholar]
- Morton, J.P.; Atkinson, G.; MacLaren, D.P.M.; Cable, N.T.; Gilbert, G.; Broome, C.; McArdle, A.; Drust, B. Reliability of maximal muscle force and voluntary activation as markers of exercise-induced muscle damage. Eur. J. Appl. Physiol. 2005, 94, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Burt, D.G.; Lamb, K.; Nicholas, C.; Twist, C. Effects of exercise-induced muscle damage on resting metabolic rate, sub-maximal running and post-exercise oxygen consumption. Eur. J. Sport Sci. 2014, 14, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–12. [Google Scholar] [CrossRef]
- Batterham, A.M.; Hopkins, W.G. Making meaningful inferences about magnitudes. Int. J. Sports Physiol. Perform. 2006, 1, 50–57. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Science; Lawrence Earlbaum Associates: Hilsdale, NJ, USA, 1988. [Google Scholar]
- Avela, J.; Kyröläinen, H.; Komi, P.V.; Rama, D. Reduced reflex sensitivity persists several days after long-lasting stretch-shortening cycle exercise. J. Appl. Physiol. 1999, 86, 1292–1300. [Google Scholar] [CrossRef]
- Manfredi, T.G.; Fielding, R.A.; O’Reilly, K.; Meredith, C.N.; Lee, Y.; Evans, W.J. Plasma creatine kinase actiivty and eimd in older men. Med. Sci. Sports Exerc. 1991, 23, 1028–1034. [Google Scholar] [CrossRef]
- Klass, M.; Baudry, S.; Duchateau, J. Voluntary activation during maximal contraction with advancing age: A brief review. Eur. J. Appl. Physiol. 2007, 100, 543–551. [Google Scholar] [CrossRef]
- Knight, C.A.; Kamen, G. Adaptations in muscular activation of the knee extensor muscles with strength training in young and older adults. J. Electromyogr. Kinesiol. 2001, 11, 405–412. [Google Scholar] [CrossRef]
- Cronin, J.B.; Hansen, K.T. Strength and power predictors of sports speed. J. Strength Cond. Res. 2005, 19, 349–357. [Google Scholar]
- Delaney, J.A.; Scott, T.J.; Ballard, D.A.; Duthie, G.M.; Hickmans, J.A.; Lockie, R.G.; Dascombe, B.J. Contributing factors to change-of-direction ability in professional rugby league players. J. Strength Cond. Res. 2015, 29, 2688–2696. [Google Scholar] [CrossRef] [PubMed]
- Toft, A.D.; Jensen, L.B.; Bruunsgaard, H.; Ibfelt, T.; Halkjaer-Kristensen, J.; Febbraio, M.; Pedersen, B.K. Cytokine response to eccentric exercise in young and elderly humans. Am. J. Physiol. Cell Physiol. 2002, 283, C289–C295. [Google Scholar] [CrossRef] [PubMed]
- Verdijk, L.B.; Gleeson, B.G.; Jonkers, R.A.M.; Meijer, K.; Savelberg, H.H.C.M.; Dendale, P.; Van Loon, L.J.C. Skeletal muscle hypertrophy following resistance training is accompanied by a fiber type-specific increase in satellite cell content in elderly men. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2009, 64, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Friden, J.; Lieber, R.L. Eccentric exercise-induced injuries to contractile and cytoskeletal muscle fibre components. Acta Physiol. Scand. 2001, 171, 321–326. [Google Scholar] [CrossRef]
| Measure | Group | Comparison | |||
|---|---|---|---|---|---|
| YG (n = 9) | MT (n = 9) | MU (n = 9) | YG v MT | MT v MU | |
| Age (years) | 22.3 ± 1.7 | 39.9 ± 6.2 | 44.4 ± 6.3 | Most likely ↑ 3.70 (2.87, 4.53) | Likely ↑ 0.71 (−0.10, 1.52) |
| Mass (kg) | 82.0 ± 9.0 | 79.1 ± 10.3 | 83.4 ± 9.56 | Unclear 0.29 (−1.10, 0.52) | Unclear 0.42 (−0.39, 1.23) |
| Fat-free mass (kg) | 71.4 ± 7.9 | 63.9 ± 6.5 | 68.6 ± 7.1 | Very likely ↓ −1.02 (−1.83, −0.22) | Likely ↑ 0.68 (−0.13, 1.49) |
| Fat-mass (kg) | 10.5 ± 4.5 | 15.2 ± 5.7 | 14.8 ± 7.0 | Likely ↑ 0.89 (0.09, 1.70) | Unclear −0.07 (−0.88, 0.74) |
| Body fat (%) | 12.8 ± 4.7 | 18.8 ± 5.8 | 17.4 ± 6.7 | Very likely ↑ 1.13 (0.32, 1.94) | Unclear −0.23 (−1.04, 0.58) |
| Sum of skinfolds (mm) | 82.3 ± 24.6 | 102.4 ± 31.9 | 91.7 ± 32.7 | Likely ↑ 0.69 (−0.12, 1.50) | Unclear −0.32 (−1.13, 0.48) |
| Squat 1RM (kg) | 130.8 ± 26.8 | 109.3 ± 22.5 | 98.4 ± 14.25 | Unclear −0.85 (−1.65, −0.04) | Unclear −0.56 (−1.37, 0.25) |
| Resistance Training Characteristics | YG (n = 9) | MT (n = 9) | |
|---|---|---|---|
| Years of resistance training (mean ± SD) | 4.6 ± 1.3 | 18.0 ± 5.6 | |
| Weekly frequency * | 1 to 2 | 2 (22.2) | 6 (66.7) |
| 3 to 4 | 4 (44.4) | 2 (22.2) | |
| 5+ | 3 (33.3) | 1 (11.1) | |
| Session duration * | 0 to 30 min | 0 (0.0) | 1 (11.1) |
| 31 to 60 min | 3 (33.3) | 7 (77.8) | |
| 61 to 90 min | 5 (55.6) | 1 (11.1) | |
| 90+ min | 1 (11.1) | 0 (0.0) | |
| Reason for resistance training * | Strength | 6 (66.7) | 4 (44.4) |
| Hypertrophy | 1 (11.1) | 0 (0.0) | |
| Fat loss | 1 (11.1) | 4 (44.4) | |
| Health | 1 (11.1) | 1 (11.1) | |
| Sports Participation Characteristics | YG (n = 9) | MT (n = 9) | MU (n = 9) | |
|---|---|---|---|---|
| Years of sports participation (mean ± SD) | 11.2 ± 4.8 | 22.0 ± 7.8 | 30.3 ± 7.8 | |
| Weekly frequency | 1 to 2 | 4 (44.4) | 2 (22.2) | 0 (0.0) |
| 3 to 4 | 4 (44.4) | 4 (44.4) | 6 (66.7) | |
| 5+ | 1 (11.1) | 3 (33.3) | 3 (33.3) | |
| Session duration | 0 to 30 min | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| 31 to 60 min | 3 (33.3) | 4 (44.4) | 7 (77.8) | |
| 61 to 90 min | 3 (33.3) | 3 (33.3) | 1 (11.1) | |
| 90+ min | 3 (33.3) | 2 (22.2) | 1 (11.1) | |
| Type of sport | Team | 5 (55.6) | 3 (33.3) | 3 (33.3) |
| Endurance | 3 (33.3) | 5 (55.6) | 4 (44.4) | |
| Racket | 0 (0.0) | 1 (11.1) | 2 (22.2) | |
| Other | 1 (11.1) | 0 (0.0) | 0 (0.0) | |
| Intensity | Group | Pre | 24 h | 72 h | Comparison (90% CI) | |
|---|---|---|---|---|---|---|
| Pre v 24 h | Pre v 72 h | |||||
| 20% 1RM (W) | YG | 507.9 ± 134.6 | 473.8 ± 119.9 | 476.6 ± 119.7 | YG v MT | |
| Very likely ↓ | Very likely ↓ | |||||
| MT | 387.4 ± 87.9 | 360.3 ± 76.1 | 366.3 ± 76.4 | −1.07(−1.85, −0.28) | −1.04 (−1.82, −0.25) | |
| MT v MU | ||||||
| MU | 320.7 ± 47.9 | 291.7 ± 40.1 | 289.7 ± 40.2 | Very likely ↓ | Very likely ↓ | |
| −1.06 (−1.84, −0.27) | −1.17 (−1.96, −0.39) | |||||
| 80% 1RM (W) | YG | 1295.3 ± 369.1 | 1207.5 ± 328.2 | 1275.9 ± 338.3 | YG v MT | |
| Very likely ↓ | Very likely ↓ | |||||
| MT | 977.1 ± 211.1 | 869.8 ± 195.0 | 964.9 ± 212.1 | −1.07 (−1.96, −0.39) | −1.04 (−1.83, −0.25) | |
| MT v MU | ||||||
| MU | 886.0 ± 163.2 | 746.7 ± 153.3 | 735.1 ± 134.8 | Likely ↓ | Very likely ↓ | |
| −0.67 (−1.45, 0.12) | −1.22 (−2.01, −0.43) | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, J.F.T.; Lamb, K.L.; Twist, C. Exercise-Induced Muscle Damage and Recovery in Young and Middle-Aged Males with Different Resistance Training Experience. Sports 2019, 7, 132. https://doi.org/10.3390/sports7060132
Fernandes JFT, Lamb KL, Twist C. Exercise-Induced Muscle Damage and Recovery in Young and Middle-Aged Males with Different Resistance Training Experience. Sports. 2019; 7(6):132. https://doi.org/10.3390/sports7060132
Chicago/Turabian StyleFernandes, John F. T., Kevin L. Lamb, and Craig Twist. 2019. "Exercise-Induced Muscle Damage and Recovery in Young and Middle-Aged Males with Different Resistance Training Experience" Sports 7, no. 6: 132. https://doi.org/10.3390/sports7060132
APA StyleFernandes, J. F. T., Lamb, K. L., & Twist, C. (2019). Exercise-Induced Muscle Damage and Recovery in Young and Middle-Aged Males with Different Resistance Training Experience. Sports, 7(6), 132. https://doi.org/10.3390/sports7060132







