Peak Power Output Is Similarly Recovered After Three- and Five-Days’ Rest Following Sprint Interval Training in Young and Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Session 1: Baseline Peak Power Output
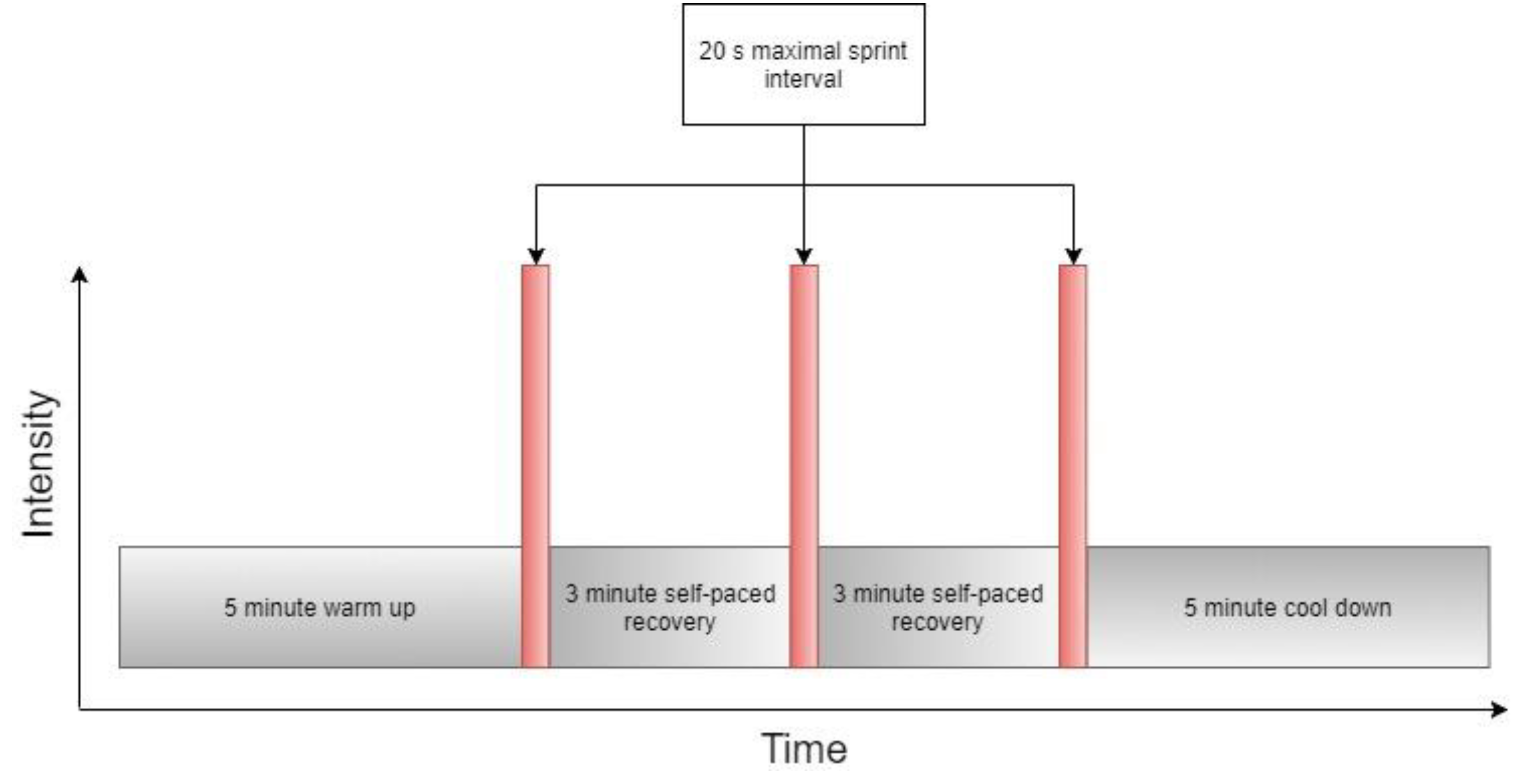
2.3. Session 2 and 4: Sprint Interval Training and Peak Power Assessment
2.4. Statistical Analysis
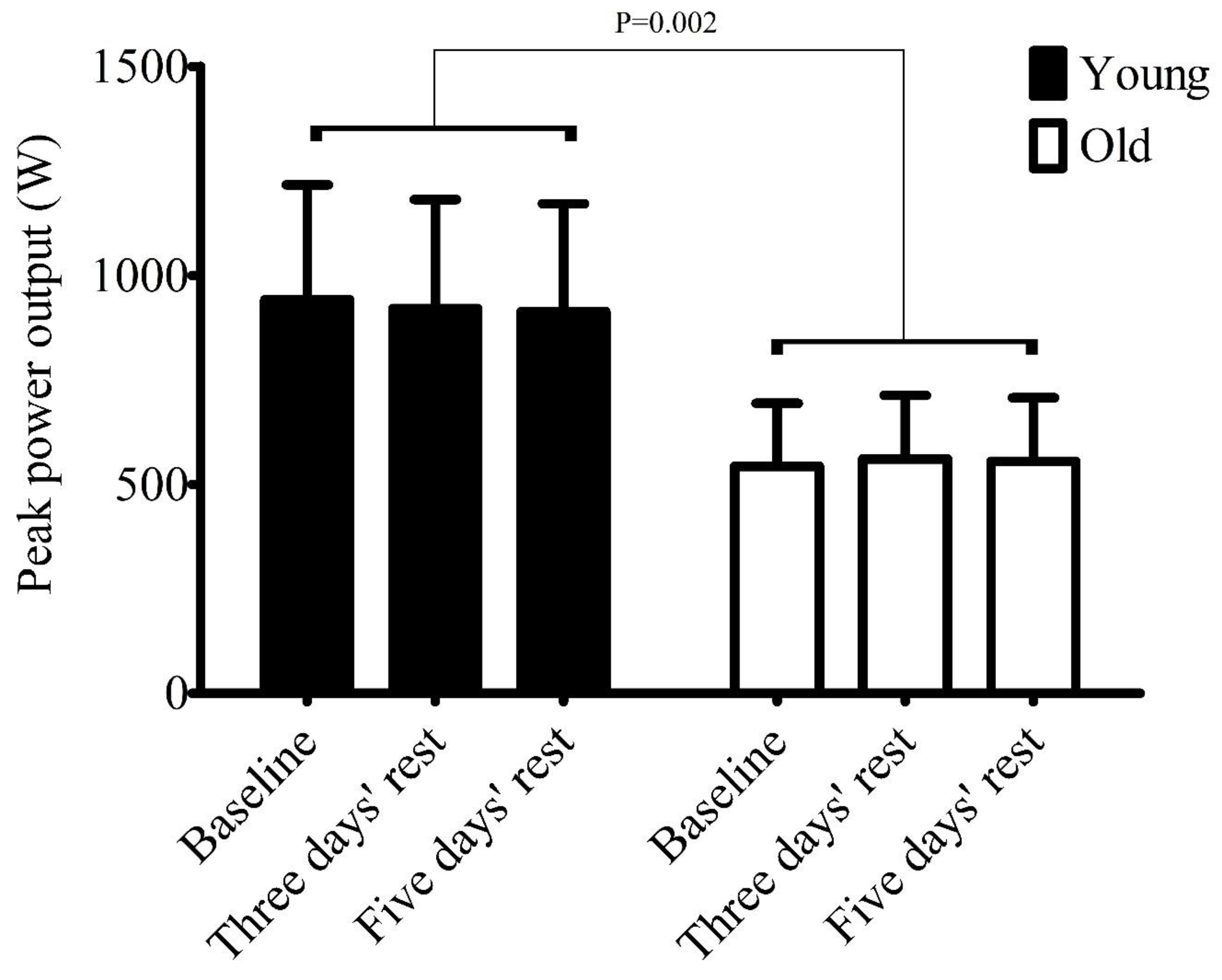
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- MacInnis, M.J.; Gibala, M.J. Physiological adaptations to interval training and the role of exercise intensity. J. Physiol. 2017, 595, 2915–2930. [Google Scholar] [CrossRef] [PubMed]
- Gist, N.H.; Fedewa, M.V.; Dishman, R.K.; Cureton, K.J. Sprint interval training effects on aerobic capacity: A systematic review and meta-analysis. Sports Med. 2014, 44, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Batacan, R.B.; Duncan, M.J.; Dalbo, V.J.; Tucker, P.S.; Fenning, A.S. Effects of high-intensity interval training on cardiometabolic health: a systematic review and meta-analysis of intervention studies. Br. J. Sports Med. 2017, 51, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Gibala, M.J.; Little, J.P.; Macdonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 590, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Burgomaster, K.A.; Howarth, K.R.; Phillips, S.M.; Rakobowchuk, M.; MacDonald, M.J.; McGee, S.L.; Gibala, M.J. Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans: Metabolic adaptations to sprint or endurance training in humans. J. Physiol. 2008, 586, 151–160. [Google Scholar] [CrossRef]
- Mandsager, K.; Harb, S.; Cremer, P.; Phelan, D.; Nissen, S.E.; Jaber, W. Association of cardiorespiratory fitness with long-term mortality among adults undergoing exercise treadmill testing. JAMA Netw. Open 2018, 1, e183605. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.D.; Close, G.L.; MacLaren, D.P.M.; Gregson, W.; Drust, B.; Morton, J.P. High-intensity interval running is perceived to be more enjoyable than moderate-intensity continuous exercise: Implications for exercise adherence. J. Sports Sci. 2011, 29, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Thum, J.S.; Parsons, G.; Whittle, T.; Astorino, T.A. High-intensity interval training elicits higher enjoyment than moderate intensity continuous exercise. PLoS ONE 2017, 12, e0166299. [Google Scholar] [CrossRef]
- Gillen, J.B.; Gibala, M.J. Is high-intensity interval training a time-efficient exercise strategy to improve health and fitness? Appl. Physiol. Nutr. Metab. 2014, 39, 409–412. [Google Scholar] [CrossRef]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle: Part I: cardiopulmonary emphasis. Sports Med. 2013, 43, 313–338. [Google Scholar] [CrossRef]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle. Part II: anaerobic energy, neuromuscular load and practical applications. Sports Med. 2013, 43, 927–954. [Google Scholar] [CrossRef]
- Robinson, M.M.; Dasari, S.; Konopka, A.R.; Johnson, M.L.; Manjunatha, S.; Esponda, R.R.; Carter, R.E.; Lanza, I.R.; Nair, K.S. Enhanced protein translation underlies improved metabolic and physical adaptations to different exercise training modes in young and old humans. Cell Metab. 2017, 25, 581–592. [Google Scholar] [CrossRef]
- Søgaard, D.; Lund, M.T.; Scheuer, C.M.; Dehlbaek, M.S.; Dideriksen, S.G.; Abildskov, C.V.; Christensen, K.K.; Dohlmann, T.L.; Larsen, S.; Vigelsø, A.H.; et al. High-intensity interval training improves insulin sensitivity in older individuals. Acta Physiol. 2018, 222, e13009. [Google Scholar] [CrossRef]
- Støren, Ø.; Helgerud, J.; Sæbø, M.; Støa, E.M.; Bratland-Sanda, S.; Unhjem, R.J.; Hoff, J.; Wang, E. The effect of age on the VO2max response to high-intensity interval training. Med. Sci. Sports Exerc. 2017, 49, 78–85. [Google Scholar] [CrossRef]
- Grace, F.; Herbert, P.; Elliott, A.D.; Richards, J.; Beaumont, A.; Sculthorpe, N.F. High intensity interval training (HIIT) improves resting blood pressure, metabolic (MET) capacity and heart rate reserve without compromising cardiac function in sedentary aging men. Exp. Gerontol. 2018, 109, 75–81. [Google Scholar] [CrossRef]
- Hayes, L.D.; Herbert, P.; Sculthorpe, N.F.; Grace, F.M. Exercise training improves free testosterone in lifelong sedentary aging men. Endocr. Connect. 2017, 6, 306–310. [Google Scholar] [CrossRef]
- Herbert, P.; Hayes, L.D.; Sculthorpe, N.; Grace, F.M. High-intensity interval training (HIIT) increases insulin-like growth factor-I (IGF-I) in sedentary aging men but not masters’ athletes: An observational study. The Aging Male 2016, 20, 1–6. [Google Scholar] [CrossRef][Green Version]
- Sculthorpe, N.F.; Herbert, P.; Grace, F. One session of high-intensity interval training (HIIT) every 5 days, improves muscle power but not static balance in lifelong sedentary ageing men: A randomized controlled trial. Medicine (Baltimore) 2017, 96, e6040. [Google Scholar] [CrossRef]
- Herbert, P.; Hayes, L.D.; Sculthorpe, N.F.; Grace, F.M. HIIT produces increases in muscle power and free testosterone in male masters athletes. Endocr. Connect. 2017, 6, 430–436. [Google Scholar] [CrossRef]
- Byrne, C.; Faure, C.; Keene, D.J.; Lamb, S.E. Ageing, Muscle power and physical function: A systematic review and implications for pragmatic training interventions. Sports Med. 2016, 46, 1311–1332. [Google Scholar] [CrossRef]
- Herbert, P.; Grace, F.M.; Sculthorpe, N.F. Exercising caution: Prolonged recovery from a single session of high-intensity interval training in older men. J. Am. Geriatr. Soc. 2015, 63, 817–818. [Google Scholar] [CrossRef]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef]
- Joseph, A.-M.; Adhihetty, P.J.; Leeuwenburgh, C. Beneficial effects of exercise on age-related mitochondrial dysfunction and oxidative stress in skeletal muscle. J. Physiol. 2016, 594, 5105–5123. [Google Scholar] [CrossRef]
- Viña, J.; Borras, C.; Abdelaziz, K.M.; Garcia-Valles, R.; Gomez-Cabrera, M.C. The free radical theory of aging revisited: the cell signaling disruption theory of aging. Antioxid. Redox Signal. 2013, 19, 779–787. [Google Scholar] [CrossRef]
- Gomes, M.J.; Martinez, P.F.; Pagan, L.U.; Damatto, R.L.; Mariano Cezar, M.D.; Ruiz Lima, A.R.; Okoshi, K.; Okoshi, M.P. Skeletal muscle aging: Influence of oxidative stress and physical exercise. Oncotarget 2017, 8, 20428–20440. [Google Scholar] [CrossRef]
- Clark, B.C.; Manini, T.M. Functional consequences of sarcopenia and dynapenia in the elderly. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 271–276. [Google Scholar] [CrossRef]
- Manini, T.M.; Clark, B.C. Dynapenia and Aging: An Update. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67A, 28–40. [Google Scholar] [CrossRef]
- Farzad, B.; Gharakhanlou, R.; Agha-Alinejad, H.; Curby, D.G.; Bayati, M.; Bahraminejad, M.; Mäestu, J. Physiological and performance changes from the addition of a sprint interval program to wrestling training. J. Strength. Cond. Res. 2011, 25, 2392–2399. [Google Scholar] [CrossRef]
- Kim, J.; Lee, N.; Trilk, J.; Kim, E.-J.; Kim, S.-Y.; Lee, M.; Cho, H.-C. Effects of sprint interval training on elite judoists. Int. J. Sports Med. 2011, 32, 929–934. [Google Scholar]
- MacDougall, J.D.; Hicks, A.L.; MacDonald, J.R.; McKelvie, R.S.; Green, H.J.; Smith, K.M. Muscle performance and enzymatic adaptations to sprint interval training. J. Appl. Physiol. 1998, 84, 2138–2142. [Google Scholar] [CrossRef]
- Weston, M.; Taylor, K.L.; Batterham, A.M.; Hopkins, W.G. Effects of low-volume high-intensity interval training (HIT) on fitness in adults: a meta-analysis of controlled and non-controlled trials. Sports Med. 2014, 44, 1005–1017. [Google Scholar] [CrossRef]
- Whyte, L.J.; Gill, J.M.R.; Cathcart, A.J. Effect of 2 weeks of sprint interval training on health-related outcomes in sedentary overweight/obese men. Metab. Clin. Exp. 2010, 59, 1421–1428. [Google Scholar] [CrossRef]
- Hazell, T.J.; Macpherson, R.E.K.; Gravelle, B.M.R.; Lemon, P.W.R. 10 or 30-s sprint interval training bouts enhance both aerobic and anaerobic performance. Eur. J. Appl. Physiol. 2010, 110, 153–160. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; van Essen, M.; Wilkin, G.P.; Burgomaster, K.A.; Safdar, A.; Raha, S.; Tarnopolsky, M.A. Short-term sprint interval versus traditional endurance training: similar initial adaptations in human skeletal muscle and exercise performance. J. Physiol. 2006, 575, 901–911. [Google Scholar] [CrossRef]
- Cocks, M.; Shaw, C.S.; Shepherd, S.O.; Fisher, J.P.; Ranasinghe, A.M.; Barker, T.A.; Tipton, K.D.; Wagenmakers, A.J.M. Sprint interval and endurance training are equally effective in increasing muscle microvascular density and eNOS content in sedentary males. J. Physiol. 2013, 591, 641–656. [Google Scholar] [CrossRef]
- Vollaard, N.B.J.; Metcalfe, R.S. Research into the health benefits of sprint interval training should focus on protocols with fewer and shorter sprints. Sports Med. 2017, 47, 2443–2451. [Google Scholar] [CrossRef]
- Vollaard, N.B.J.; Metcalfe, R.S.; Williams, S. Effect of number of sprints in an sit session on change in VO2max: A Meta-analysis. Med. Sci. Sports Exerc. 2017, 49, 1147–1156. [Google Scholar] [CrossRef]
- Riebe, D.; Franklin, B.A.; Thompson, P.D.; Garber, C.E.; Whitfield, G.P.; Magal, M.; Pescatello, L.S. Updating ACSM’s recommendations for exercise preparticipation health screening. Med. Sci. Sports Exerc. 2015, 47, 2473–2479. [Google Scholar] [CrossRef]
- Herbert, P.; Sculthorpe, N.; Baker, J.S.; Grace, F.M. Validation of a six second cycle test for the determination of peak power output. Res. Sports Med. 2015, 23, 115–125. [Google Scholar] [CrossRef]
- Chodzko-Zajko, W.J.; Proctor, D.N.; Singh, M.A.F.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. Exercise and physical activity for older adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef]
- He, F.; Li, J.; Liu, Z.; Chuang, C.-C.; Yang, W.; Zuo, L. Redox mechanism of reactive oxygen species in exercise. Front. Physiol. 2016, 7. [Google Scholar] [CrossRef]
- Austin, S.; Klimcakova, E.; St-Pierre, J. Impact of PGC-1α on the topology and rate of superoxide production by the mitochondrial electron transport chain. Free Radical Biol Med. 2011, 51, 2243–2248. [Google Scholar] [CrossRef]
- Powers, S.K.; Nelson, W.B.; Hudson, M.B. Exercise-induced oxidative stress in humans: Cause and consequences. Free Radical Biol. Med. 2011, 51, 942–950. [Google Scholar] [CrossRef]
- Steinbacher, P.; Eckl, P.; Steinbacher, P.; Eckl, P. Impact of oxidative stress on exercising skeletal muscle. Biomolecules 2015, 5, 356–377. [Google Scholar] [CrossRef] [PubMed]
- Farinatti, P.T.V.; Geraldes, A.A.R.; Bottaro, M.F.; Lima, M.V.I.C.; Albuquerque, R.B.; Fleck, S.J. Effects of different resistance training frequencies on the muscle strength and functional performance of active women older than 60 years. J. Strength Cond. Res. 2013, 27, 2225–2234. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, R.; Kruel, L.F.M.; Cadore, E.L.; Alberton, C.L.; Izquierdo, M.; Conceição, M.; Pinto, R.S.; Radaelli, R.; Wilhelm, E.; Bottaro, M.; et al. Efficiency of twice weekly concurrent training in trained elderly men. Exp. Gerontol. 2013, 48, 1236–1242. [Google Scholar] [CrossRef] [PubMed]
- Holviala, J.; Häkkinen, A.; Alen, M.; Sallinen, J.; Kraemer, W.; Häkkinen, K. Effects of prolonged and maintenance strength training on force production, walking, and balance in aging women and men: Functional capacity after strength training. Scand. J. Med. Sci. Sports 2014, 24, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Wang, R.; Chen, P.; Huang, S.C.; Donnelly, J.E.; Mehlferber, J.P. Dose-response relationship of cardiorespiratory fitness adaptation to controlled endurance training in sedentary older adults. Eur. J. Prev. Cardiol. 2016, 23, 518–529. [Google Scholar] [CrossRef]
- Hawley, J.A.; Hargreaves, M.; Joyner, M.J.; Zierath, J.R. Integrative biology of exercise. Cell 2014, 159, 738–749. [Google Scholar] [CrossRef]
- Adamson, S.; Kavaliauskas, M.; Yamagishi, T.; Phillips, S.; Lorimer, R.; Babraj, J. Extremely short duration sprint interval training improves vascular health in older adults. Sport Sci. Health 2018, 15, 123–131. [Google Scholar] [CrossRef]
- Fiorenza, M.; Gunnarsson, T.P.; Hostrup, M.; Iaia, F.M.; Schena, F.; Pilegaard, H.; Bangsbo, J. Metabolic stress-dependent regulation of the mitochondrial biogenic molecular response to high-intensity exercise in human skeletal muscle. J. Physiol. 2018, 596, 2823–2840. [Google Scholar] [CrossRef] [PubMed]
- Guadalupe-Grau, A.; Aznar-Laín, S.; Mañas, A.; Castellanos, J.; Alcázar, J.; Ara, I.; Mata, E.; Daimiel, R.; García-García, F.J. Short- and long-term effects of concurrent strength and hiit training in octogenarians with COPD. J. Aging Phys. Act. 2017, 25, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, B.B.; Westheim, A.; Risberg, M.A. Long-term effects of a group-based high-intensity aerobic interval-training program in patients with chronic heart failure. Am. J. Cardiol. 2008, 102, 1220–1224. [Google Scholar] [CrossRef] [PubMed]
- Bijker, K.E.; de Groot, G.; Hollander, A.P. Differences in leg muscle activity during running and cycling in humans. Eur. J. Appl. Physiol. 2002, 87, 556–561. [Google Scholar]
| Peak Power (W) | Mean Power (W) | |||||
|---|---|---|---|---|---|---|
| Group | Sprint 1 | Sprint 2 | Sprint 3 | Sprint 1 | Sprint 2 | Sprint 3 |
| Older | 541 ± 135 | 528 ± 139 | 498 ± 146 | 402 ± 93 | 384 ± 93 | 362 ± 88 |
| Younger | 897 ± 246 | 828 ± 219 | 788 ± 215 | 579 ± 139 | 513 ± 148 | 473 ± 156 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yasar, Z.; Dewhurst, S.; Hayes, L.D. Peak Power Output Is Similarly Recovered After Three- and Five-Days’ Rest Following Sprint Interval Training in Young and Older Adults. Sports 2019, 7, 94. https://doi.org/10.3390/sports7040094
Yasar Z, Dewhurst S, Hayes LD. Peak Power Output Is Similarly Recovered After Three- and Five-Days’ Rest Following Sprint Interval Training in Young and Older Adults. Sports. 2019; 7(4):94. https://doi.org/10.3390/sports7040094
Chicago/Turabian StyleYasar, Zerbu, Susan Dewhurst, and Lawrence D. Hayes. 2019. "Peak Power Output Is Similarly Recovered After Three- and Five-Days’ Rest Following Sprint Interval Training in Young and Older Adults" Sports 7, no. 4: 94. https://doi.org/10.3390/sports7040094
APA StyleYasar, Z., Dewhurst, S., & Hayes, L. D. (2019). Peak Power Output Is Similarly Recovered After Three- and Five-Days’ Rest Following Sprint Interval Training in Young and Older Adults. Sports, 7(4), 94. https://doi.org/10.3390/sports7040094





