Effects of a Combined High-Intensity Interval Training and Resistance Training Program in Patients Awaiting Bariatric Surgery: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Anthropometry, Body Composition and CRF Measurements
2.4. Cardiorespiratory Fitness Measurement
2.5. Muscle Strength Measurements
2.6. Exercise Training Program (ETP)
2.7. Statistical Analysis
3. Results
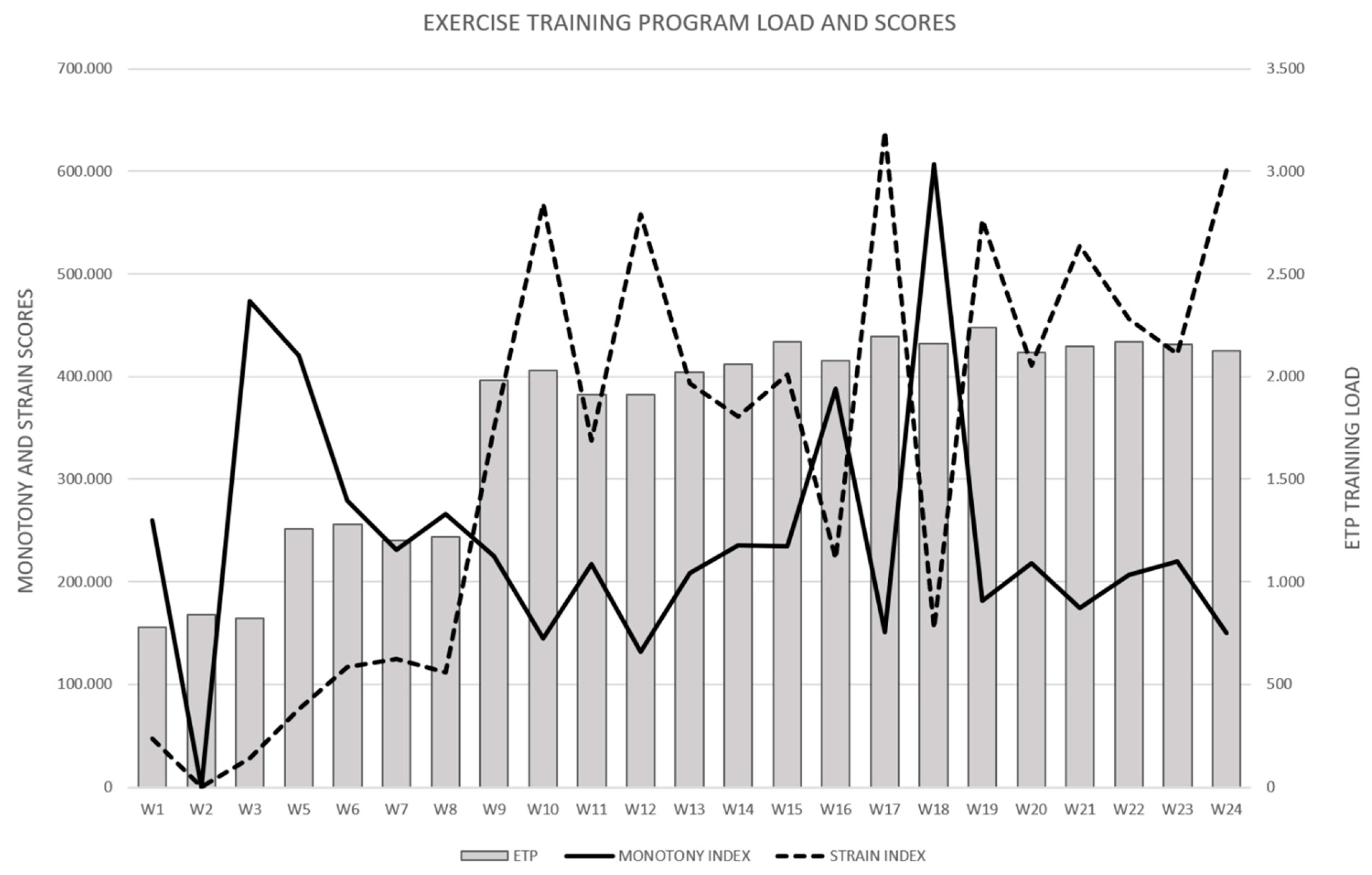
3.1. Description of the Dynamic Loads Performed by the Experimental Group
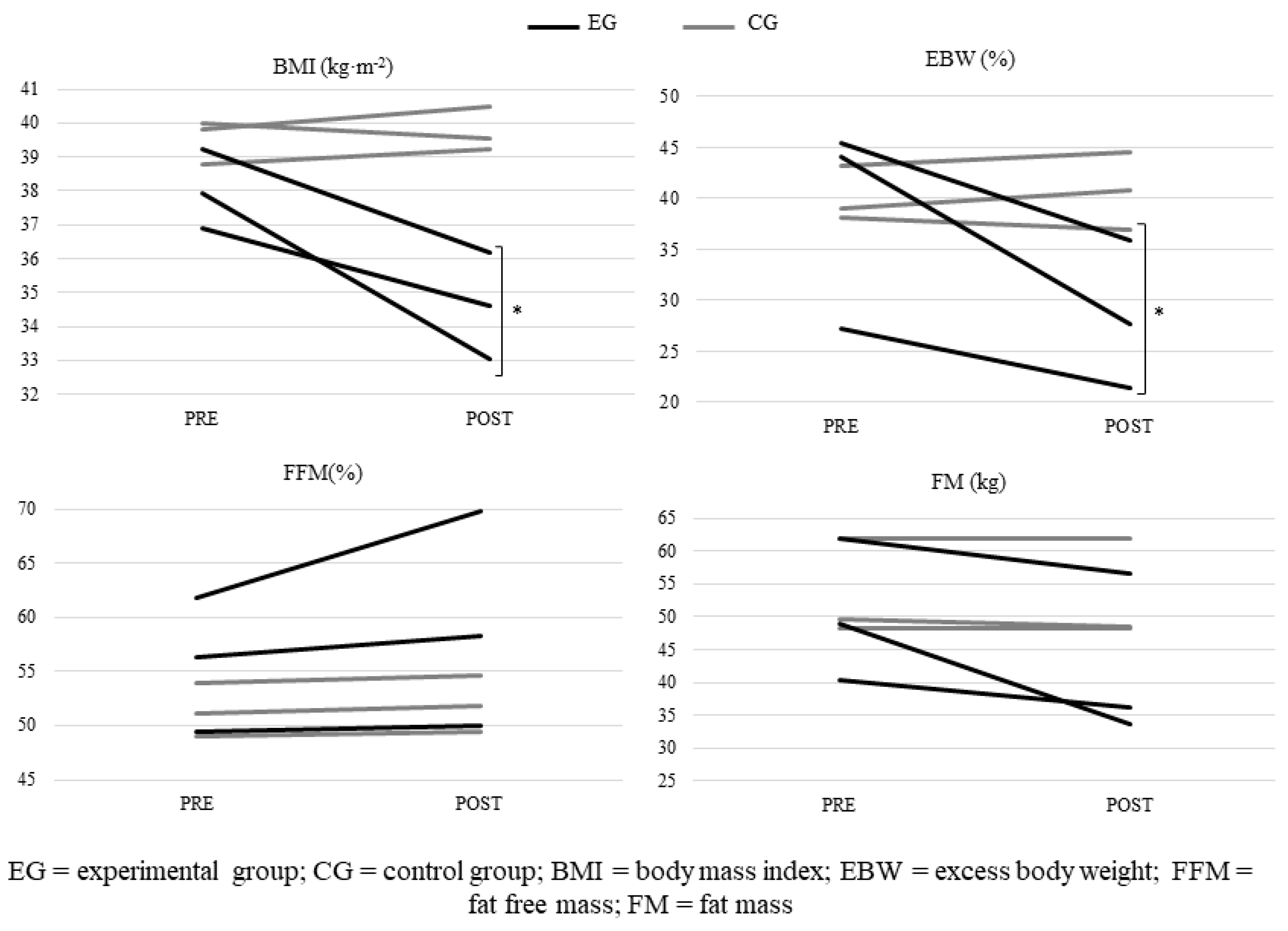
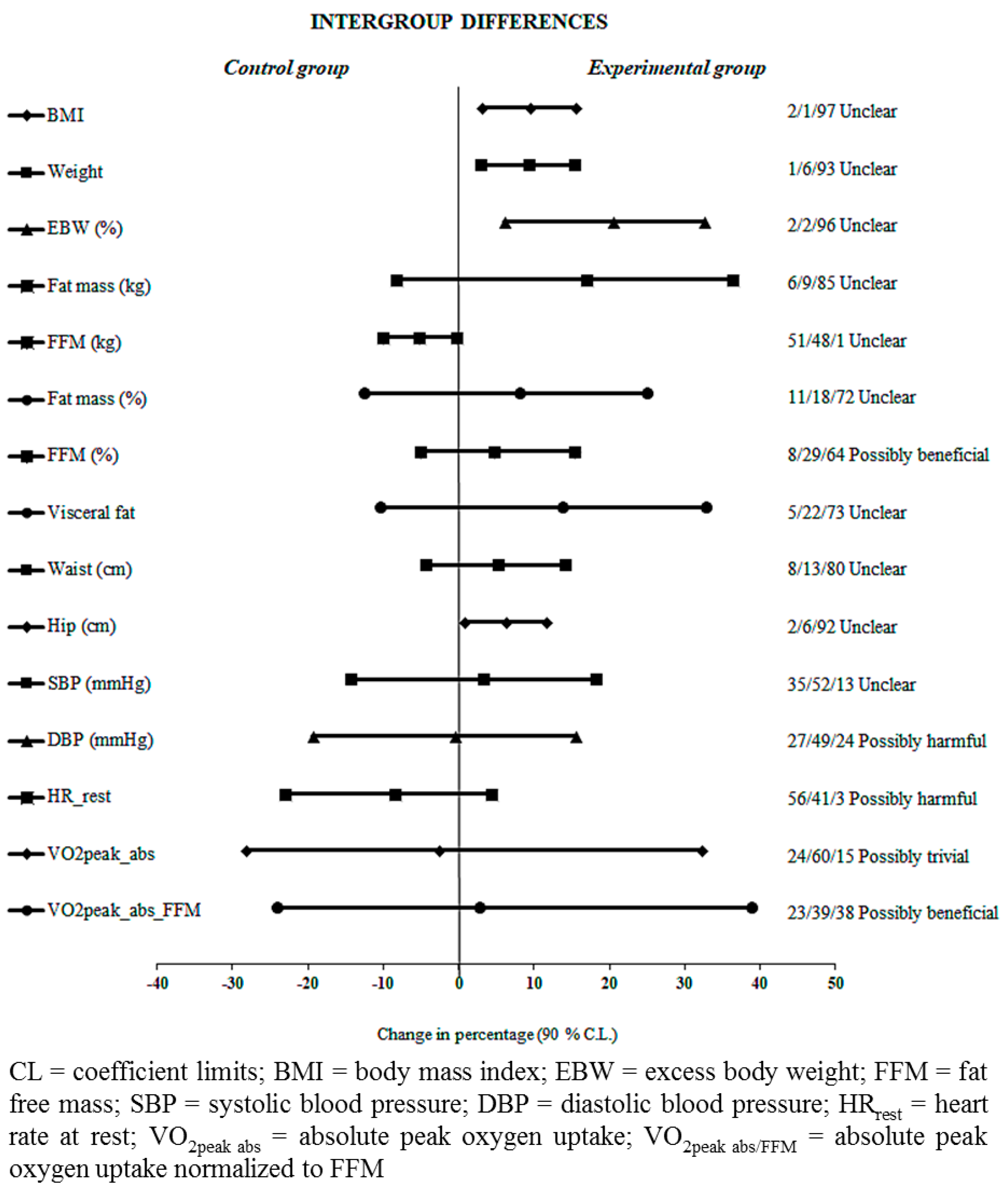
3.2. Anthropometry and Body Composition
3.3. Physical Fitness and Cardiometabolic Risk Factors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Banegas, J.R.; Lopez-Garcia, E.; Gutierrez-Fisac, J.L.; Guallar-Castillon, P.; Rodriguez-Artalejo, F. A simple estimate of mortality attributable to excess weight in the European Union. Eur. J. Clin. Nutr. 2003, 57, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.B.; Fontaine, K.R.; Manson, J.E.; Stevens, J.; VanItallie, T.B. Annual deaths attributable to obesity in the United States. JAMA 1999, 282, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- Floody, P.D.; Mayorga, D.J.; Navarrete, F.C.; Díaz, M.C.; Elgueta, H.O.; Poblete, A.O. Efectividad del tratamiento integral sobre las condiciones preoperatorias de mujeres obesas candidatas a cirugía bariátrica. Nutr. Hosp. 2015, 32, 2570–2575. [Google Scholar]
- Moya, M.; Hernández, A.; Sarabia, J.M.; Sánchez-Martos, M.A.; Hernández-Davó, J.L.; López-Grueso, R.; Aracil, A.; Pastor, D.; Fernández-Fernández, J. Bariatric surgery, weight loss and the role of physical activity: A systematic review. Eur. J. Hum. Mov. 2014, 32, 145–160. [Google Scholar]
- Clark, J.E. Diet, exercise or diet with exercise: Comparing the effectiveness of treatment options for weight-loss and changes in fitness for adults (18–65 years old) who are overfat, or obese; systematic review and meta-analysis. J. Diabetes Metab. Disord. 2015, 14, 1–28. [Google Scholar]
- Sjöström, L. Review of the key results from the Swedish Obese Subjects (SOS) trial–a prospective controlled intervention study of bariatric surgery. J. Intern. Med. 2013, 273, 219–234. [Google Scholar] [CrossRef]
- King, W.C.; Hsu, J.Y.; Belle, S.H.; Courcoulas, A.P.; Eid, G.M.; Flum, D.R.; Mitchell, J.E.; Pender, J.R.; Smith, M.D.; Steffen, K.J.; et al. Pre-to postoperative changes in physical activity: Report from the longitudinal assessment of bariatric surgery-2 (LABS-2). Surg. Obes. Relat. Dis. 2012, 8, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Sánchez Ortega, L.; Sánchez Juan, C.; Alfonso García, A. Valoración de un programa de ejercicio físico estructurado en pacientes con obesidad mórbida pendientes de cirugía bariátrica. Nutr. Hosp. 2014, 29, 64–72. [Google Scholar]
- Van Wissen, J.; Bakker, N.; Doodeman, H.J.; Jansma, E.P.; Bonjer, H.J.; Houdijk, A.P.J. Preoperative methods to reduce liver volume in bariatric surgery: A systematic review. Obes. Surg. 2016, 26, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Guida, B.; Cataldi, M.; Busetto, L.; Aiello, M.L.; Musella, M.; Capone, D.; Parolisi, S.; Policastro, V.; Ragozini, G.; Belfiore, A. Predictors of fat-free mass loss 1 year after laparoscopic sleeve gastrectomy. J. Endocrinol. Invest. 2018, 41, 1307–1315. [Google Scholar] [CrossRef] [PubMed]
- Castello, V.; Simões, R.P.; Bassi, D.; Catai, A.M.; Arena, R.; Borghi-Silva, A. Impact of aerobic exercise training on heart rate variability and functional capacity in obese women after gastric bypass surgery. Obes. Surg. 2011, 21, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Snell, P.G.; Rao, S.; Adams-Huet, B.; Quittner, C.; Livingston, E.H.; Garg, A. High-Volume Exercise Program in Obese Bariatric Surgery Patients: A Randomized, Controlled Trial. Obesity 2011, 19, 1826–1834. [Google Scholar] [CrossRef]
- Stegen, S.; Derave, W.; Calders, P.; Van Laethem, C.; Pattyn, P. Physical fitness in morbidly obese patients: Effect of gastric bypass surgery and exercise training. Obes. Surg. 2011, 21, 61–70. [Google Scholar] [CrossRef]
- Baillot, A.; Vallée, C.A.; Mampuya, W.M.; Dionne, I.J.; Comeau, E.; Méziat-Burdin, A.; Langlois, M.F. Effects of a Pre-surgery Supervised Exercise Training 1 Year After Bariatric Surgery: A Randomized Controlled Study. Obes. Surg. 2018, 28, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Bond, D.S.; Thomas, J.G.; King, W.C.; Vithiananthan, S.; Trautvetter, J.; Unick, J.L.; Ryder, B.A.; Pohl, D.; Roye, G.D.; Sax, H.C.; et al. Exercise improves quality of life in bariatric surgery candidates: Results from the Bari-Active trial. Obesity 2015, 23, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Floody, P.D.; Mayorga, D.J.; Navarrete, F.C.; Poblete, A.O.; Lepeley, N.T.; Hormazábal, M.A. Doce semanas de ejercicio físico intervalado con sobrecarga mejora las variables antropométricas de obesos mórbidos y obesos con comorbilidades postulantes a cirugía bariátrica. Nutr. Hosp. 2015, 32, 2007–2011. [Google Scholar]
- Baillot, A.; Mampuya, W.M.; Comeau, E.; Méziat-Burdin, A.; Langlois, M.F. Feasibility and impacts of supervised exercise training in subjects with obesity awaiting bariatric surgery: A pilot study. Obes. Surg. 2013, 23, 882–891. [Google Scholar] [CrossRef]
- Kalarchian, M.A.; Marcus, M.D.; Courcoulas, A.P.; Cheng, Y.; Levine, M.D. Preoperative lifestyle intervention in bariatric surgery: Initial results from a randomized, controlled trial. Obesity 2013, 21, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Adair, J.D.; Wollner, S.B.; DaCosta, M.E.; Blackburn, G.L. Progressive resistance training for patients with class III obesity. Obes. Weight Manag. 2010, 6, 115–118. [Google Scholar] [CrossRef]
- Keating, S.E.; Johnson, N.A.; Mielke, G.I.; Coombes, J.S. A systematic review and meta-analysis of interval training versus moderate-intensity continuous training on body adiposity. Obes. Rev. 2017, 18, 943–964. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.; Durrer, C.; Simtchouk, S.; Jung, M.E.; Bourne, J.E.; Voth, E.; Little, J.P. Short-term high-intensity interval and moderate-intensity continuous training reduce leukocyte TLR4 in inactive adults at elevated risk of type 2 diabetes. J. Appl. Physiol. 2015, 119, 508–516. [Google Scholar] [CrossRef]
- Lanzi, S.; Codecasa, F.; Cornacchia, M.; Maestrini, S.; Capodaglio, P.; Brunani, A.; Salvadori, A.; Malatesta, D. Short-term HIIT and Fatmax training increase aerobic and metabolic fitness in men with class II and III obesity. Obesity 2015, 23, 1987–1994. [Google Scholar] [CrossRef]
- Fisher, G.; Brown, A.W.; Bohan Brown, M.M.; Alcorn, A.; Noles, C.; Winwood, L.; Resuehr, H.; George, B.; Jeansonne, M.M.; Allison, D.B. High Intensity Interval-vs Moderate Intensity-Training for Improving Cardiometabolic Health in Overweight or Obese Males: A Randomized Controlled Trial. PLoS ONE 2015, 10, e0138853. [Google Scholar] [CrossRef]
- Little, J.P.; Jung, M.E.; Wright, A.E.; Wright, W.; Manders, R.J. Effects of high-intensity interval exercise versus continuous moderate-intensity exercise on postprandial glycemic control assessed by continuous glucose monitoring in obese adults. Appl. Physiol. Nutr. Metab. 2014, 39, 835–841. [Google Scholar] [CrossRef]
- Batacan, R.B.; Duncan, M.J.; Dalbo, V.J.; Tucker, P.S.; Fenning, A.S. Effects of high-intensity interval training on cardiometabolic health: A systematic review and meta-analysis of intervention studies. Br. J. Sports Med. 2017, 51, 494–503. [Google Scholar] [CrossRef]
- Keating, S.E.; Machan, E.A.; O’Connor, H.T.; Gerofi, J.A.; Sainsbury, A.; Caterson, I.D.; Johnson, N.A. Continuous exercise but not high intensity interval training improves fat distribution in overweight adults. J. Obes. 2014, 2014, 834865. [Google Scholar] [CrossRef] [PubMed]
- Jelleyman, C.; Yates, T.; O’Donovan, G.; Gray, L.J.; King, J.A.; Khunti, K.; Davies, M.J. The effects of high-intensity interval training on glucose regulation and insulin resistance: A meta-analysis. Obes. Rev. 2015, 16, 942–961. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Manuel Gómez, J.; Lilienthal Heitmann, B.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis—Part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef]
- Pickering, T.G.; Hall, J.E.; Appel, L.J.; Falkner, B.E.; Graves, J.; Hill, M.N.; Jones, D.W.; Kurtz, T.; Sheps, S.G.; Roccella, E.J. Recommendations for blood pressure measurement in humans and experimental animals: Part 1: Blood pressure measurement in humans: A statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension 2005, 45, 142–161. [Google Scholar] [PubMed]
- Marfell-Jones, M.; Stewart, A.; de Ridder, J.H. International Standards for Anthropometric Assessment; The International Society for the Advancement of Kinanthropometry: Wellington, New Zealand, 2012. [Google Scholar]
- Achten, J.; Gleeson, M.; Jeukendrup, A.E. Determination of the exercise intensity that elicits maximal fat oxidation. Med. Sci. Sports Exerc. 2002, 34, 92–97. [Google Scholar] [CrossRef]
- Symons, T.B.; Vandervoort, A.; Rice, C.L.; Overend, T.J.; Marsh, G.D. Effects of maximal isometric and isokinetic resistance training on strength and functional mobility in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 777–781. [Google Scholar] [CrossRef]
- Brzycki, M. Strength testing-predicting a one-rep max from reps-to-fatigue. J. Phys. Health Educ. Recreat. Dance 1993, 64, 88–90. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Foster, C.; Florhaug, J.A.; Franklin, J.; Gottschall, L.; Hrovatin, L.A.; Parker, S.; Doleshal, P.; Dodge, C. A new approach to monitoring exercise training. J. Strength Cond. Res. 2001, 15, 109–115. [Google Scholar] [PubMed]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Batterham, A.M.; Hopkins, W.G. Making meaningful inferences about magnitudes. Int. J. Sports Physiol. Perform. 2006, 1, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Herpertz, S.; Kessler, H.; Jongen, S. Psychosomatic and Psychosocial Questions Regarding Bariatric Surgery: What Do We Know, or What Do We Think We Know? Z. Psychosom. Med. Psychother. 2017, 63, 344–369. [Google Scholar] [CrossRef] [PubMed]
- Marcon, E.R.; Baglioni, S.; Bittencourt, L.; Lopes, C.L.N.; Neumann, C.R.; Trindade, M.R.M. What is the best treatment before bariatric surgery? Exercise, exercise and group therapy, or conventional waiting: A randomized controlled trial. Obes. Surg. 2017, 27, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Baillot, A.; Mampuya, W.M.; Dionne, I.J.; Comeau, E.; Méziat-Burdin, A.; Langlois, M.F. Impacts of supervised exercise training in addition to interdisciplinary lifestyle management in subjects awaiting bariatric surgery: A randomized controlled study. Obes. Surg. 2016, 26, 2602–2610. [Google Scholar] [CrossRef] [PubMed]
- Floody, P.D.; Navarrete, F.C.; Mayorga, D.J.; Jara, C.C.; Campillo, R.R.; Poblete, A.O.; Alarcón Hormazábal, M.; Thuillier Lepeley, N.; Mansilla, C.S. Efectos de un programa de tratamiento multidisciplinar en obesos mórbidos y obesos con comorbilidades candidatos a cirugía bariátrica. Nutr. Hosp. 2015, 31, 2011–2016. [Google Scholar]
- King, W.C.; Bond, D.S. The importance of pre and postoperative physical activity counseling in bariatric surgery. Exerc. Sport Sci. Rev. 2013, 41, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Gadducci, A.V.; de Cleva, R.; de Faria Santarém, G.C.; Silva, P.R.S.; Greve, J.M.D.; Santo, M.A. Muscle strength and body composition in severe obesity. Clinics 2017, 72, 272–275. [Google Scholar] [CrossRef]
| Variables | Total (n = 6) | EG (n = 3) | CG (n = 3) |
|---|---|---|---|
| Female (n) | 5 | 2 | 3 |
| Age (years) | 38.17 ± 12.06 | 39.67 ± 10.21 | 36.67 ± 15.88 |
| BMI (Kg·m−2) | 38.78 ± 1.18 | 38.02 ± 1.16 | 39.54 ± 0.64 |
| Weight (Kg) | 111.83 ± 14.10 | 114.43 ± 19.11 | 109.23 ± 10.57 |
| Fat mass (%) | 46.40 ± 4.89 | 44.17 ± 6.21 | 48.63 ± 2.51 |
| FFM (%) | 53.60 ± 4.89 | 55.83 ± 6.21 | 51.37 ± 2.51 |
| Visceral fat (%) | 14.00 ± 3.88 | 15 ± 5.51 | 13.00 ± 2.08 |
| VO2peak abs (L·min−1) | 2.20 ± 0.86 | 2.62 ± 1.11 | 1.78 ± 0.29 |
| VO2peak abs/FFM (mL·FFM−1·min−1) | 35.77 ± 7.14 | 39.92 ± 8.01 | 31.61 ± 3.40 |
| Systolic blood pressure (mmHg) | 131.89 ± 27.39 | 144.22 ± 37.39 | 119.56 ± 4.53 |
| Diastolic blood pressure (mmHg) | 77.94 ± 13.48 | 83.22 ± 18.68 | 72.67 ± 4.63 |
| Month | 1 | 2 | 3 | 4 | 5 | 6 | |
|---|---|---|---|---|---|---|---|
| MICT | Weekly frequency (sessions/week) | 2 | 1 | 2 | 2 | 2 | 2 |
| Volume (min) | 35 | 50 | 50 | 50 | 50 | 50 | |
| Intensity (% HRpeak) | 60–70 | 65–75 | 70–80 | 70–85 | 70–85 | 70–85 | |
| HIIT | Weekly frequency (sessions/week) | 2 | 2 | 2 | 2 | 2 | |
| Volume (min) | 20 | 20 | 20 | 20 | 20 | ||
| Intensity (% VO2peak) | 60–70 | 70–80 | 75–85 | 80–90 | 80–95 | ||
| Resistance training | Weekly frequency (sessions/week) | 2 | 2 | 2 | 2 | 2 | 2 |
| Volume (series × exer. × rep.) | 1 × 5 × 20 | 1 × 7 × 20 | 4 × 4 × 15 | 4 × 4 × 12 | 4 × 4 × 10 | 4 × 4 × 10 | |
| Intensity (% 1RM) | 55 | 60 | 65 | 70 | 75 | 75 | |
| Stretching training | Weekly frequency (sessions/week) | 2 | 3 | 4 | 4 | 4 | 4 |
| Volume (series × exer.) | 1 × 5 | 1 × 5 | 1 × 5 | 1 × 5 | 1 × 5 | 1 × 5 | |
| Duration (min) | 1 | 1 | 1 | 1 | 1 | 1 |
| EG (n = 3) | CG (n = 3) | |||||
|---|---|---|---|---|---|---|
| Variables | Pre | Post | ES (d) (90% CL) | Pre | Post | ES (d) (90% CL) |
| BMI (kg·m−2) | 38.02 ± 1.16 | 34.61 ± 1.56 * | 3.30 (1.05; 5.54) | 39.54 ± 0.64 | 39.75 ± 0.65 | −0.19 (−1.03; 0.66) |
| Weight (kg) | 114.43 ± 19.11 | 103.87 ± 14.80 | 0.57 (0.18; 0.57) | 109.23 ± 10.57 | 109.87 ± 11.43 | −0.03 (−0.18; 0.11) |
| EBW (kg) | 38.83 ± 10.14 | 28.27 ± 7.27 | 2.71 (0.79; 4.62) | 40.09 ± 2.70 | 40.72 ± 3.74 | −0.12 (−0.68; 0.43) |
| EBW (%) | 33.55 ± 3.89 | 27.00 ± 4.06 † | 4.42 (1.19; 7.65) | 36.77 ± 1.04 | 37.10 ± 1.02 | −0.18 (−1.00; 0.63) |
| Fat mass (kg) | 50.48 ± 10.87 | 42.16 ± 12.52 | 0.81 (−0.30; 1.91) | 53.25 ± 7.49 | 52.91 ± 7.91 | 0.03 (−0.08; 0.14) |
| Fat mass (%) | 44.17 ± 6.21 | 40.63 ± 9.94 | −1.08 (−1.15; 3.31) | 48.63 ± 2.51 | 48.03 ± 2.65 * | 0.14 (0.06; 0.21) |
| FFM (kg) | 63.95 ± 13.83 | 61.71 ± 14.39 | −0.32 (−0.70; 0.07) | 55.98 ± 3.80 | 56.96 ± 4.40 | 0.14 (−0.05; 0.33) |
| FFM (%) | 55.83 ± 6.21 | 59.37 ± 9.94 | 0.66 (−0.48; 1.80) | 51.37 ± 2.51 | 51.97 ± 2.65 * | 0.14 (0.08; 0.19) |
| Waist (cm) | 120.27 ± 4.07 | 114.26 ± 5.68 | 0.37 (−0.31; 1.05) | 115.62 ± 9.10 | 116.02 ± 8.18 | −0.03 (−0.21; 0.16) |
| Hip (cm) | 130.57 ± 10.76 | 122.27 ± 13.63 † | 0.69 (0.12; 1.26) | 134.04 ± 7.64 | 133.70 ± 8.72 | 0.03 (−0.14; 0.20) |
| WHR (cm) | 0.93 ± 0.09 | 0.94 ± 0.06 | −0.10 (−0.54; 0.35) | 0.86 ± 0.08 | 0.87 ± 0.08 | −0.04 (−0.10; 0.02) |
| Visceral fat (%) | 14.67 ± 5.51 | 12 ± 2.65 | 0.62 (−0.24; 1.47) | 12.67 ± 2.08 | 12.33 ± 1.53 | 0.08 (−0.16; 0.32) |
| EG (n = 3) | CG (n = 3) | |||||
|---|---|---|---|---|---|---|
| Variables | Pre | Post | ES (d) (90% CL) | Pre | Post | ES (d) (90% CL) |
| SBP (mmHg) | 144.22 ± 37.39 | 139.28 ± 28.76 | 0.41 (−2.02; 2.84) | 119.56 ± 4.53 | 120.61 ± 8.92 | −0.11 (−2.09; 1.86) |
| DBP (mmHg) | 83.22 ± 18.68 | 79.83 ± 18.37 | 0.37 (−0.53; 1.28) | 72.67 ± 4.63 | 69.39 ± 6.09 | 0.42 (−1.23; 2.06) |
| HRrest (bpm) | 56.33 ± 12.34 | 62.00 ± 11.53 | −0.26 (−0.54; 0.03) | 65.67 ± 13.58 | 66.67 ± 13.32 | −0.04 (−0.32; 0.23) |
| VO2peak abs (L·min−1) | 2.62 ± 1.11 | 2.69 ± 1.04 | 0.11 (−0.12; 0.35) | 1.78 ± 0.29 | 1.89 ± 0.37 | 0.21 (−0.85; 1.28) |
| VO2peak abs/FFM (mL*·FFM−1·min−1) | 39.92 ± 8.01 | 42.58 ± 6.24 | 0.38 (−0.23; 0.99) | 31.61 ± 3.40 | 33.07 ± 4.45 | 0.23 (−1.29; 1.76) |
| MDSD-Q (N·m) | 151.84 ± 36.10 | 155.16 ± 43.38 | 0.05 (−0.36; 0.45) | 144.12 ± 21.46 | 149.50 ± 18.35 | 0.14 (−0.16; 0.45) |
| MDSD-H (N·m) | 86.53 ± 24.87 | 84.54 ± 17.37 | −0.02 (−0.53; 0.48) | 66.06 ± 18.67 | 59.93 ± 14.97 | −0.17 (−0.40; 0.07) |
| MDSND-Q (N·m) | 149.88 ± 47.94 | 161.74 ± 54.86 | 0.35 (0.13; 0.58) | 141.14 ± 15.73 | 142.03 ± 19.00 | 0.02 (−0.44; 0.49) |
| MDSND-H (N·m) | 75.81 ± 14.93 | 85.66 ± 21.93 | 0.56 (−0.21; 1.32) | 57.03 ± 6.42 | 60.81 ± 6.69 | 0.32 (0.06; 0.57) |
| MISD-Q (N·m) | 141.90 ± 19.18 | 139.77 ± 21.19 | −0.07 (−0.30; 0.16) | 138.76 ± 19.35 | 130.11 ± 3.89 | −0.24 (−1.02; 0.54) |
| MISD-H (N·m) | 95.83 ± 23.39 | 90.53 ± 12.71 | −0.12 (−0.69; 0.45) | 72.16 ± 15.02 | 62.48 ± 14.31 | −0.37 (−1.20; 0.45) |
| MISND-Q (N·m) | 147.50 ± 3.47 | 151.73 ± 16.01 | 0.11 (−0.48; 0.69) | 133.61 ± 17.80 | 121.02 ± 8.40 | −0.40 (−0.90; 0.10) |
| MISND-H (N·m) | 90.30 ± 15.95 | 89.10 ± 15.30 | −0.02 (−0.32; 0.28) | 61.71 ± 19.85 | 61.18 ± 10.31 | −0.03 (−0.52; 0.46) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Picó-Sirvent, I.; Aracil-Marco, A.; Pastor, D.; Moya-Ramón, M. Effects of a Combined High-Intensity Interval Training and Resistance Training Program in Patients Awaiting Bariatric Surgery: A Pilot Study. Sports 2019, 7, 72. https://doi.org/10.3390/sports7030072
Picó-Sirvent I, Aracil-Marco A, Pastor D, Moya-Ramón M. Effects of a Combined High-Intensity Interval Training and Resistance Training Program in Patients Awaiting Bariatric Surgery: A Pilot Study. Sports. 2019; 7(3):72. https://doi.org/10.3390/sports7030072
Chicago/Turabian StylePicó-Sirvent, Inés, Adolfo Aracil-Marco, Diego Pastor, and Manuel Moya-Ramón. 2019. "Effects of a Combined High-Intensity Interval Training and Resistance Training Program in Patients Awaiting Bariatric Surgery: A Pilot Study" Sports 7, no. 3: 72. https://doi.org/10.3390/sports7030072
APA StylePicó-Sirvent, I., Aracil-Marco, A., Pastor, D., & Moya-Ramón, M. (2019). Effects of a Combined High-Intensity Interval Training and Resistance Training Program in Patients Awaiting Bariatric Surgery: A Pilot Study. Sports, 7(3), 72. https://doi.org/10.3390/sports7030072






