Regional Bioelectrical Phase Angle Is More Informative than Whole-Body Phase Angle for Monitoring Neuromuscular Performance: A Pilot Study in Elite Young Soccer Players
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
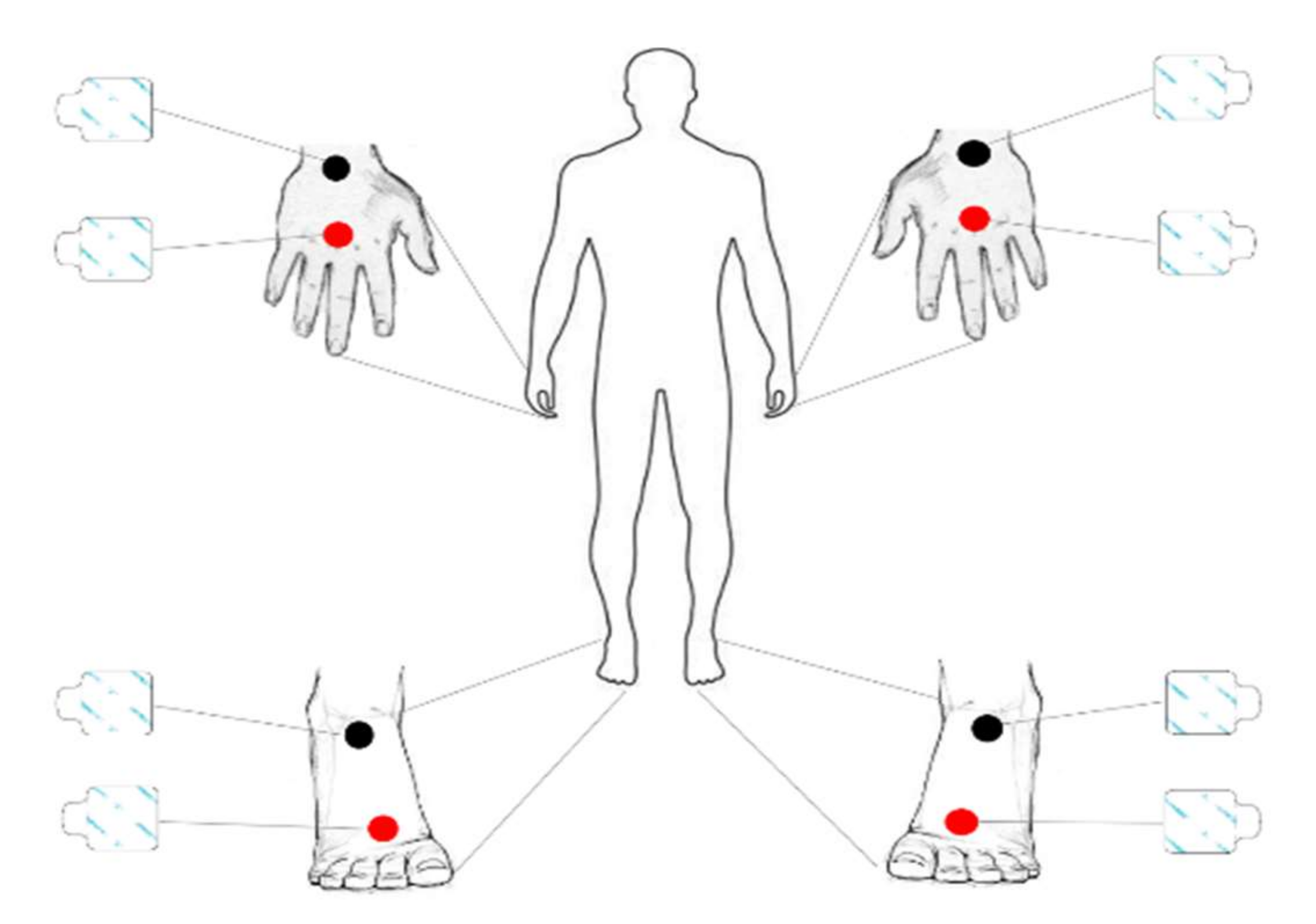
2.3. Bioimpedance and Body Composition Analysis
2.4. Urine
2.5. Lower-Limb Neuromuscular Performance
2.6. Statistical Analysis
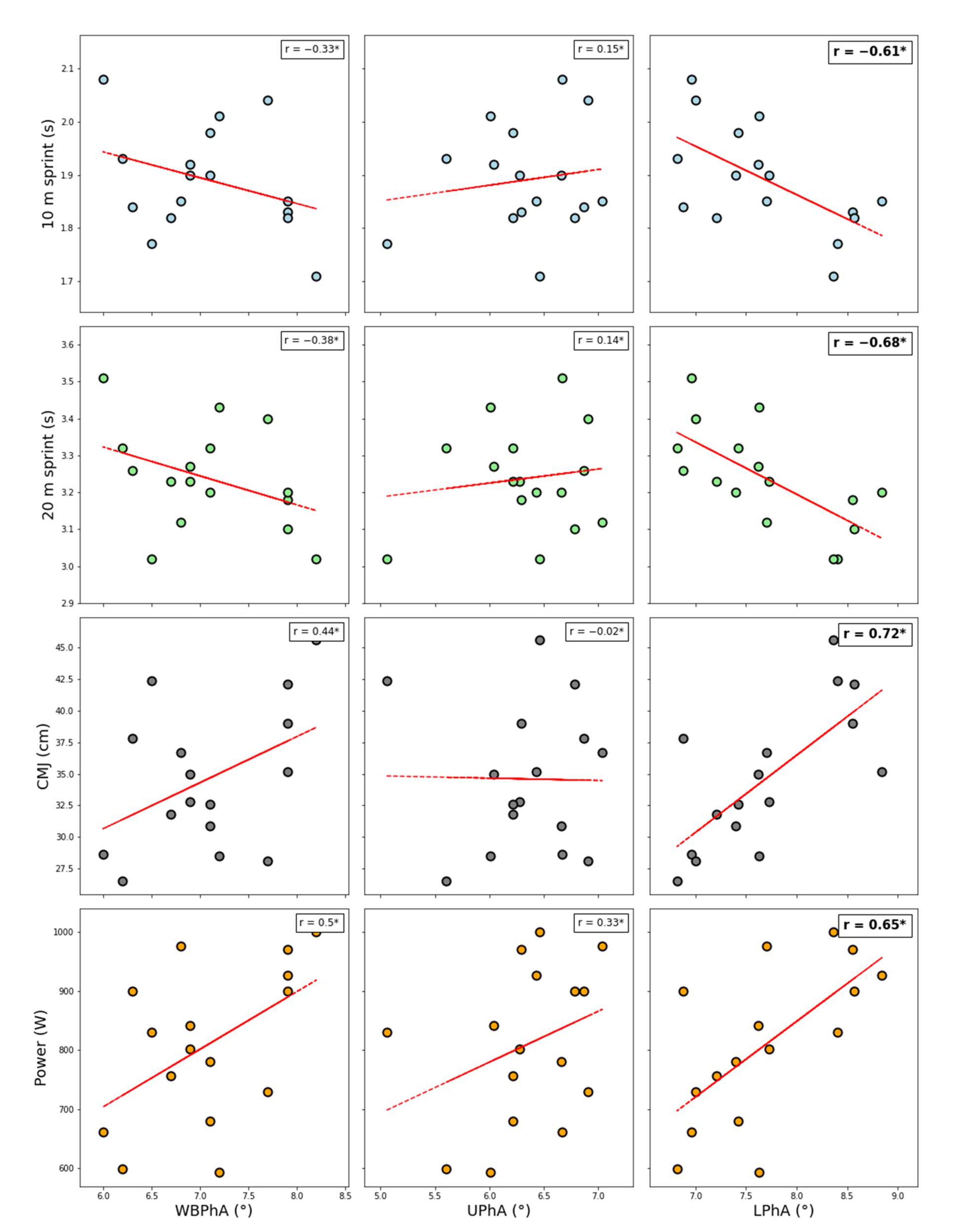
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lukaski, H.; Raymond-Pope, C.J. New Frontiers of Body Composition in Sport. Int. J. Sports Med. 2021, 42, 588–601. [Google Scholar] [CrossRef] [PubMed]
- Campa, F.; Gobbo, L.A.; Stagi, S.; Cyrino, L.T.; Toselli, S.; Marini, E.; Coratella, G. Bioelectrical impedance analysis versus reference methods in the assessment of body composition in athletes. Eur. J. Appl. Physiol. 2022, 122, 561–589. [Google Scholar] [CrossRef] [PubMed]
- Campa, F.; Silva, A.M.; Matias, C.N.; Monteiro, C.P.; Paoli, A.; Nunes, J.P.; Talluri, J.; Lukaski, H.; Toselli, S. Body Water Content and Morphological Characteristics Modify Bioimpedance Vector Patterns in Volleyball, Soccer, and Rugby Players. Int. J. Environ. Res. Public Health 2020, 17, 6604. [Google Scholar] [CrossRef] [PubMed]
- Milanese, C.; Cavedon, V.; Corradini, G.; De Vita, F.; Zancanaro, C. Seasonal DXA-measured body composition changes in professional male soccer players. J. Sports Sci. 2015, 33, 1219–1228. [Google Scholar] [CrossRef]
- Bongiovanni, T.; Rossi, A.; Iaia, F.M.; DI Baldassarre, A.; Pasta, G.; Manetti, P.; Alberti, G.; Trecroci, A. Relationship of regional and whole body morphology to vertical jump in elite soccer players: A data-driven approach. J. Sports Med. Phys. Fitness 2021, 34080815. [Google Scholar] [CrossRef]
- Harley, J.A.; Hind, K.; O’hara, J.P. Three-compartment body composition changes in elite rugby league players during a super league season, measured by dual-energy X-ray absorptiometry. J. Strength Cond. Res. 2011, 25, 1024–1029. [Google Scholar] [CrossRef]
- Nana, A.; Slater, G.J.; Stewart, A.D.; Burke, L.M. Methodology review: Using dual-energy X-ray absorptiometry (DXA) for the assessment of body composition in athletes and active people. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 198–215. [Google Scholar] [CrossRef]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Manuel Gómez, J.; Lilienthal Heitmann, B.; Kent-Smith, L.; Melchior, J.-C.; Pirlich, M.; et al. Bioelectrical impedance analysis-part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef]
- Campa, F.; Bongiovanni, T.; Trecroci, A.; Rossi, A.; Greco, G.; Pasta, G.; Coratella, G. Effects of the COVID-19 Lockdown on Body Composition and Bioelectrical Phase Angle in Serie A Soccer Players: A Comparison of Two Consecutive Seasons. Biology (Basel) 2021, 10, 1175. [Google Scholar] [CrossRef]
- Marini, E.; Campa, F.; Buffa, R.; Stagi, S.; Matias, C.N.; Toselli, S.; Sardinha, L.B.; Silva, A.M. Phase angle and bioelectrical impedance vector analysis in the evaluation of body composition in athletes. Clin. Nutr. 2020, 39, 447–454. [Google Scholar] [CrossRef]
- Gonzalez, M.C.; Barbosa-Silva, T.G.; Bielemann, R.M.; Gallagher, D.; Heymsfield, S.B. Phase angle and its determinants in healthy subjects: Influence of body composition. Am. J. Clin. Nutr. 2016, 103, 712–716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campa, F.; Toselli, S.; Mazzilli, M.; Gobbo, L.A.; Coratella, G. Assessment of Body Composition in Athletes: A Narrative Review of Available Methods with Special Reference to Quantitative and Qualitative Bioimpedance Analysis. Nutrients 2021, 13, 1620. [Google Scholar] [CrossRef] [PubMed]
- Martins, P.C.; Teixeira, A.S.; Guglielmo, L.G.A.; Francisco, J.S.; Silva, D.A.S.; Nakamura, F.Y.; de Lima, L.R.A. Phase Angle Is Related to 10 m and 30 m Sprint Time and Repeated-Sprint Ability in Young Male Soccer Players. Int. J. Environ. Res. Public Health 2021, 18, 4405. [Google Scholar] [CrossRef] [PubMed]
- Bongiovanni, T.; Rossi, A.; Iaia, F.M.; Alberti, G.; Pasta, G.; Trecroci, A. Association of phase angle and appendicular upper and lower body lean soft tissue with physical performance in young elite soccer players: A pilot study. J. Sports Med. Phys. Fitness 2021, 34546027. [Google Scholar] [CrossRef]
- Nabuco, H.C.G.; Silva, A.M.; Sardinha, L.B.; Rodrigues, F.B.; Tomeleri, C.M.; Ravagnani, F.C.P.; Cyrino, E.S.; Ravagnani, C.F.C. Phase Angle is Moderately Associated with Short-term Maximal Intensity Efforts in Soccer Players. Int. J. Sports Med. 2019, 40, 739–743. [Google Scholar] [CrossRef]
- Hetherington-Rauth, M.; Leu, C.G.; Júdice, P.B.; Correia, I.R.; Magalhães, J.P.; Sardinha, L.B. Whole body and regional phase angle as indicators of muscular performance in athletes. Eur. J. Sport Sci. 2021, 21, 1684–1692. [Google Scholar] [CrossRef]
- Obayashi, H.; Ikuta, Y.; Fujishita, H.; Fukuhara, K.; Sakamitsu, T.; Ushio, K.; Kimura, H.; Adachi, N. The relevance of whole or segmental body bioelectrical impedance phase angle and physical performance in adolescent athletes. Physiol. Meas. 2021, 42, 035011. [Google Scholar] [CrossRef]
- Pollastri, L.; Lanfranconi, F.; Tredici, G.; Burtscher, M.; Gatterer, H. Body Water Status and Short-term Maximal Power Output during a Multistage Road Bicycle Race (Giro d’Italia 2014). Int. J. Sports Med. 2016, 37, 329–333. [Google Scholar] [CrossRef]
- Moore, M.L.; Benavides, M.L.; Dellinger, J.R.; Adamson, B.T.; Tinsley, G.M. Segmental body composition evaluation by bioelectrical impedance analysis and dual-energy X-ray absorptiometry: Quantifying agreement between methods. Clin. Nutr. 2020, 39, 2802–2810. [Google Scholar] [CrossRef]
- Organ, L.W.; Bradham, G.B.; Gore, D.T.; Lozier, S.L. Segmental bioelectrical impedance analysis: Theory and application of a new technique. J. Appl. Physiol. 1994, 77, 98–112. [Google Scholar] [CrossRef]
- Bongiovanni, T.; Trecroci, A.; Rossi, A.; Iaia, F.M.; Pasta, G.; Campa, F. Association between Change in Regional Phase Angle and Jump Performance: A Pilot Study in Serie A Soccer Players. Eur. J. Investig. Health Psychol. Educ. 2021, 11, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Di Vincenzo, O.; Marra, M.; Sammarco, R.; Speranza, E.; Cioffi, I.; Scalfi, L. Body composition, segmental bioimpedance phase angle and muscular strength in professional volleyball players compared to a control group. J. Sports Med. Phys. Fitness 2020, 60, 870–874. [Google Scholar] [CrossRef] [PubMed]
- Marra, M.; Da Prat, B.; Montagnese, C.; Caldara, A.; Sammarco, R.; Pasanisi, F.; Corsetti, R. Segmental bioimpedance analysis in professional cyclists during a three week stage race. Physiol. Meas. 2016, 37, 1035–1040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bongiovanni, T.; Trecroci, A.; Cavaggioni, L.; Rossi, A.; Perri, E.; Pasta, G.; Iaia, F.M.; Alberti, G. Importance of anthropometric features to predict physical performance in elite youth soccer: A machine learning approach. Res. Sports Med. 2020, 29, 213–224. [Google Scholar] [CrossRef]
- Going, S.; Nichols, J.; Loftin, M.; Stewart, D.; Lohman, T.; Tuuri, G.; Ring, K.; Pickrel, J.; Blew, R.; Stevens, J. Validation of bioelectrical impedance analysis (BIA) for estimation of body composition in Black, White and Hispanic adolescent girls. Int. J. Body Compos. Res. 2006, 4, 161–167. [Google Scholar]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [Green Version]
- Bongiovanni, T.; Mascherini, G.; Genovesi, F.; Pasta, G.; Iaia, F.M.; Trecroci, A.; Ventimiglia, M.; Alberti, G.; Campa, F. Bioimpedance Vector References Need to Be Period-Specific for Assessing Body Composition and Cellular Health in Elite Soccer Players: A Brief Report. J. Funct. Morphol. Kinesiol. 2020, 5, 73. [Google Scholar] [CrossRef]
- Matias, C.N.; Campa, F.; Santos, D.A.; Lukaski, H.; Sardinha, L.B.; Silva, A.M. Fat-free Mass Bioelectrical Impedance Analysis Predictive Equation for Athletes using a 4-Compartment Model. Int. J. Sports Med. 2021, 42, 27–32. [Google Scholar] [CrossRef]
- Sardinha, L.B.; Correia, I.R.; Magalhães, J.P.; Júdice, P.B.; Silva, A.M.; Hetherington-Rauth, M. Development and validation of BIA prediction equations of upper and lower limb lean soft tissue in athletes. Eur. J. Clin. Nutr. 2020, 74, 1646–1652. [Google Scholar] [CrossRef]
- Nuccio, R.P.; Barnes, K.A.; Carter, J.M.; Baker, L.B. Fluid Balance in Team Sport Athletes and the Effect of Hypohydration on Cognitive, Technical, and Physical Performance. Sports Med. 2017, 47, 1951–1982. [Google Scholar] [CrossRef] [Green Version]
- Holland, J.J.; Skinner, T.L.; Irwin, C.G.; Leveritt, M.D.; Goulet, E.D.B. The Influence of Drinking Fluid on Endurance Cycling Performance: A Meta-Analysis. Sports Med. 2017, 47, 2269–2284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.Y.; Khong, T.K.; Ramliy, N.; Yusof, A. Relationship between objective and subjective hydration measures on sprint performance among soccer players during actual matches in hot and humid environment. J. Sports Med. Phys. Fitness 2021, 34931790. [Google Scholar] [CrossRef] [PubMed]
- Pintus, R.; Bongiovanni, T.; Corbu, S.; Francavilla, V.C.; DessÌ, A.; Noto, A.; Corsello, G.; Finco, G.; Fanos, V.; Cesare Marincola, F. Sportomics in professional soccer players: Metabolomics results during preseason. J. Sports Med. Phys. Fitness 2021, 61, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Perrier, E.T.; Armstrong, L.E.; Daudon, M.; Kavouras, S.; Lafontan, M.; Lang, F.; Péronnet, F.; Stookey, J.D.; Tack, I.; Klein, A. From state to process: Defining hydration. Obes. Facts 2014, 7 (Suppl. 2), 6–12. [Google Scholar] [CrossRef]
- Manz, F.; Wentz, A. 24-h hydration status: Parameters, epidemiology and recommendations. Eur. J. Clin. Nutr. 2003, 57 (Suppl. 2), S10–S18. [Google Scholar] [CrossRef] [Green Version]
- American College of Sports Medicine; Sawka, M.N.; Burke, L.M.; Eichner, E.R.; Maughan, R.J.; Montain, S.J.; Stachenfeld, N.S. American College of Sports Medicine position stand. Exercise and fluid replacement. Med. Sci. Sports Exerc. 2007, 39, 377–390. [Google Scholar] [CrossRef] [Green Version]
- Armstrong, L.E.; Maresh, C.M.; Castellani, J.W.; Bergeron, M.F.; Kenefick, R.W.; LaGasse, K.E.; Riebe, D. Urinary indices of hydration status. Int. J. Sport Nutr. 1994, 4, 265–279. [Google Scholar] [CrossRef]
- Trecroci, A.; Duca, M.; Formenti, D.; Alberti, G.; Iaia, F.M.; Longo, S. Short-Term Compound Training on Physical Performance in Young Soccer Players. Sports (Basel) 2020, 8, 108. [Google Scholar] [CrossRef]
- Lewis, F.L.; Xie, L.; Popa, D. Optimal and Robust Estimation: With an Introduction to Stochastic Control Theory; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Mohr, M.; Krustrup, P.; Bangsbo, J. Match performance of high-standard soccer players with special reference to development of fatigue. J. Sports Sci. 2003, 21, 519–528. [Google Scholar] [CrossRef] [Green Version]
- Rossi, A.; Perri, E.; Trecroci, A.; Savino, M.; Alberti, G.; Iaia, M.F. Characterization of In-season Elite Football Trainings by GPS Features: The Identity Card of a Short-Term Football Training Cycle. In Proceedings of the 2016 IEEE 16th International Conference on Data Mining Workshops (ICDMW), Barcelona, Spain, 12–15 December 2016; pp. 160–166. [Google Scholar]
- D’Hondt, J.; Chapelle, L.; Van Droogenbroeck, L.; Aerenhouts, D.; Clarys, P.; D’Hondt, E. Bioelectrical impedance analysis as a means of quantifying upper and lower limb asymmetry in youth elite tennis players: An explorative study. Eur. J. Sport Sci. 2021, 1–12. [Google Scholar] [CrossRef]

| Characteristics | Feature | Mean | SD | 95% CI |
|---|---|---|---|---|
| Anthropometry | age (years) | 14.3 | 1.2 | [11.95, 16.65] |
| Body weight (kg) | 63.2 | 6.8 | [49.87, 76.53] | |
| Height (cm) | 176.0 | 5.7 | [164.83, 187.17] | |
| BMI | 20.4 | 1.4 | [17.66, 23.14] | |
| BIA | Resistance | 518.26 | 45.96 | [428.18, 608.34] |
| Reactance | 64.26 | 6.74 | [51.05, 77.47] | |
| WBPhA (°) | 7.08 | 0.68 | [5.75, 8.41] | |
| Upper-hemisome resistance (Ohm) | 527.79 | 43.69 | [442.16, 613.42] | |
| Upper-hemisome reactance (Ohm) | 58.62 | 5.73 | [47.39, 69.85] | |
| UPhA (°) | 6.34 | 0.51 | [5.34, 7.34] | |
| Lower-hemisome resistance (Ohm) | 456.72 | 45.35 | [367.83, 545.61] | |
| Lower-hemisome reactance (Ohm) | 61.12 | 6.35 | [48.67, 73.57] | |
| LPhA (°) | 7.69 | 0.67 | [6.38, 9] | |
| FFM (Kg) | 56.11 | 5.50 | [45.33, 66.89] | |
| FM (Kg) | 7.13 | 1.85 | [3.5, 10.76] | |
| FFM (%) | 88.83 | 2.16 | [84.6, 93.06] | |
| FM (%) | 11.17 | 2.16 | [6.94, 15.4] | |
| ALST (Kg) | 5.09 | 0.77 | [3.58, 6.6] | |
| LLST(Kg) | 16.25 | 1.81 | [12.7, 19.8] | |
| Urine | UOsm (mOsm/KgH2O) | 856.88 | 226.24 | [413.45, 1300.31] |
| USG (g/cm3) | 1.019 | 0.007 | [1.01, 1.03] | |
| Performance variables | 10 m sprint (s) | 1.89 | 0.10 | [1.69, 2.09] |
| 20 m sprint (s) | 3.24 | 0.14 | [2.97, 3.51] | |
| CMJ (cm) | 34.6 | 5.7 | [23.43, 45.77] | |
| Power (W) | 808.9 | 132.0 | [550.18, 1,067.62] |
| Variables | Parameters | Coef [95% CI] | SD | t | p-Value | r2 |
|---|---|---|---|---|---|---|
| 10 m sprint | const | 2.80 [2.34, 3.26] | 0.21 | 13.27 | <0.001 | 0.62 |
| LPhA | −0.09 [−0.15, −0.04] | 0.03 | −3.64 | 0.003 | ||
| UOsm | −2.27 × 10−4 [−8.35 × 10−4, −5.67 × 10−5] | 7.57 × 10−5 | −2.91 | 0.012 | ||
| 20 m sprint | const | 4.52 [4.05, 4.99] | 0.22 | 21.02 | <0.001 | 0.81 |
| LPhA | −0.21 [−0.30, −0.13] | 0.04 | −5.75 | <0.001 | ||
| UOsm | 4.11 × 10−4 [1.13 × 10−4, 1.51 × 10−5] | 8.16 × 10−5 | −4.65 | 0.001 | ||
| WBPhA | 0.10 [0.02, 0.18] | 0.04 | 2.58 | 0.024 | ||
| CMJ height | const | −21.97 [−45.72, 1.78] | 10.99 | −2.00 | 0.05 | 0.68 |
| LPhA | 6.23 [3.33, 9.13] | 1.34 | 4.64 | <0.001 | ||
| UOsm | 0.01 [0.002, 0.02] | 0.00 | 2.56 | 0.024 | ||
| CMJ power | const | −311.88 [−585.99, −37.77] | 125.81 | −2.48 | 0.029 | 0.93 |
| ALST | 146.12 [110.97, 118.27] | 16.13 | 9.06 | <0.001 | ||
| LPhA | 113.78 [66.97, 160.60] | 21.49 | 5.30 | <0.001 | ||
| WBPhA | −70.44 [−121.45, −19.44] | 23.41 | −3.01 | 0.011 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bongiovanni, T.; Rossi, A.; Trecroci, A.; Martera, G.; Iaia, F.M.; Alberti, G.; Pasta, G.; Lacome, M. Regional Bioelectrical Phase Angle Is More Informative than Whole-Body Phase Angle for Monitoring Neuromuscular Performance: A Pilot Study in Elite Young Soccer Players. Sports 2022, 10, 66. https://doi.org/10.3390/sports10050066
Bongiovanni T, Rossi A, Trecroci A, Martera G, Iaia FM, Alberti G, Pasta G, Lacome M. Regional Bioelectrical Phase Angle Is More Informative than Whole-Body Phase Angle for Monitoring Neuromuscular Performance: A Pilot Study in Elite Young Soccer Players. Sports. 2022; 10(5):66. https://doi.org/10.3390/sports10050066
Chicago/Turabian StyleBongiovanni, Tindaro, Alessio Rossi, Athos Trecroci, Giulia Martera, F. Marcello Iaia, Giampietro Alberti, Giulio Pasta, and Mathieu Lacome. 2022. "Regional Bioelectrical Phase Angle Is More Informative than Whole-Body Phase Angle for Monitoring Neuromuscular Performance: A Pilot Study in Elite Young Soccer Players" Sports 10, no. 5: 66. https://doi.org/10.3390/sports10050066
APA StyleBongiovanni, T., Rossi, A., Trecroci, A., Martera, G., Iaia, F. M., Alberti, G., Pasta, G., & Lacome, M. (2022). Regional Bioelectrical Phase Angle Is More Informative than Whole-Body Phase Angle for Monitoring Neuromuscular Performance: A Pilot Study in Elite Young Soccer Players. Sports, 10(5), 66. https://doi.org/10.3390/sports10050066








