Honey Bee Survival and Pathogen Prevalence: From the Perspective of Landscape and Exposure to Pesticides
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Landscape
2.2. Honey Bee Colonies
2.3. Varroa Mite Infestation
2.4. Viral and Nosema Infections
2.4.1. Sampling
2.4.2. RNA Extraction
2.4.3. RT-qPCR Steps
2.5. Detection of Pesticide Residues
2.5.1. Forager Bees
2.5.2. Honey and Wax
2.5.3. Winter and Dead Bees
2.5.4. Crop Flowers
2.6. Statistical Analysis
3. Results
3.1. Landscape Study
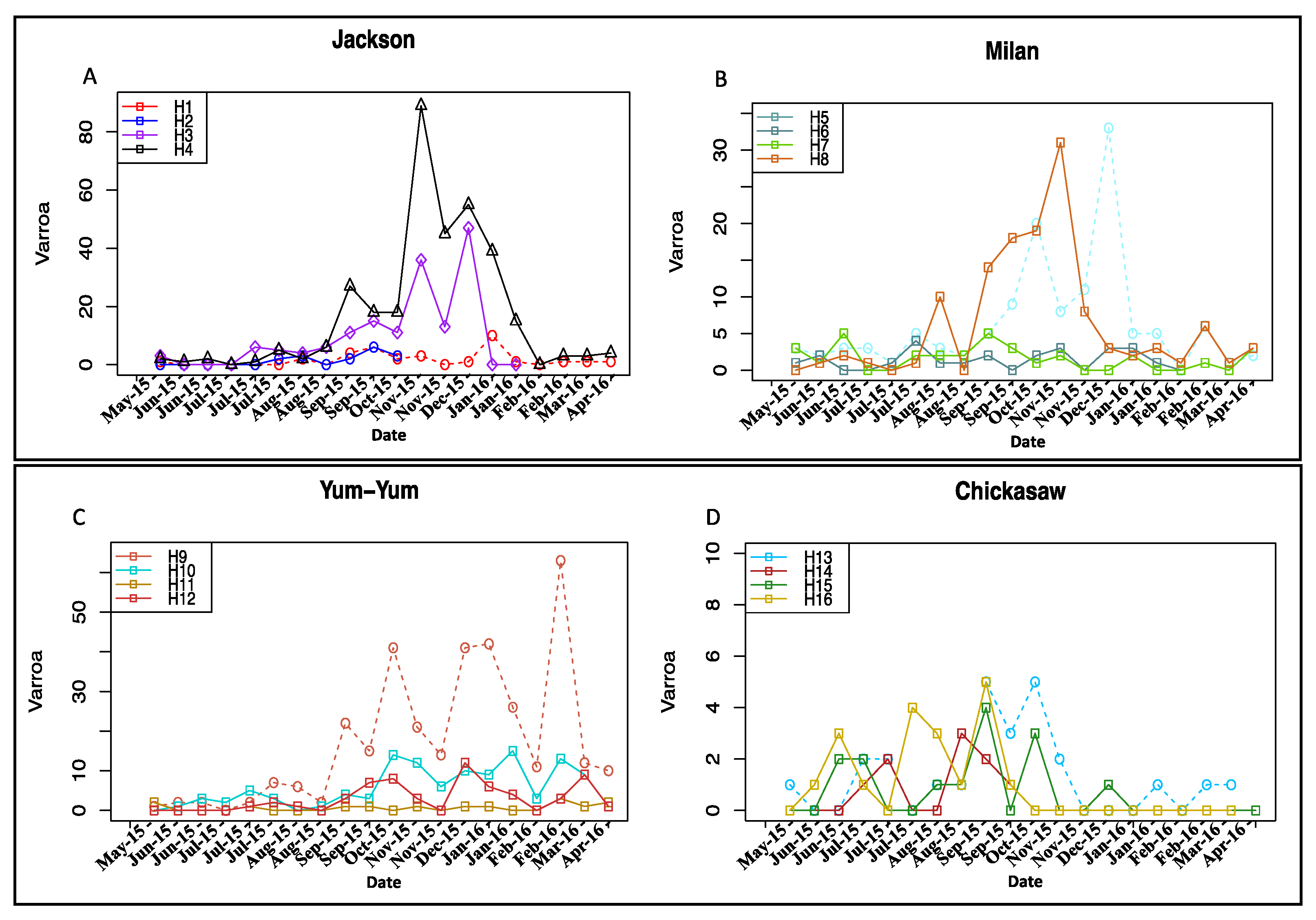
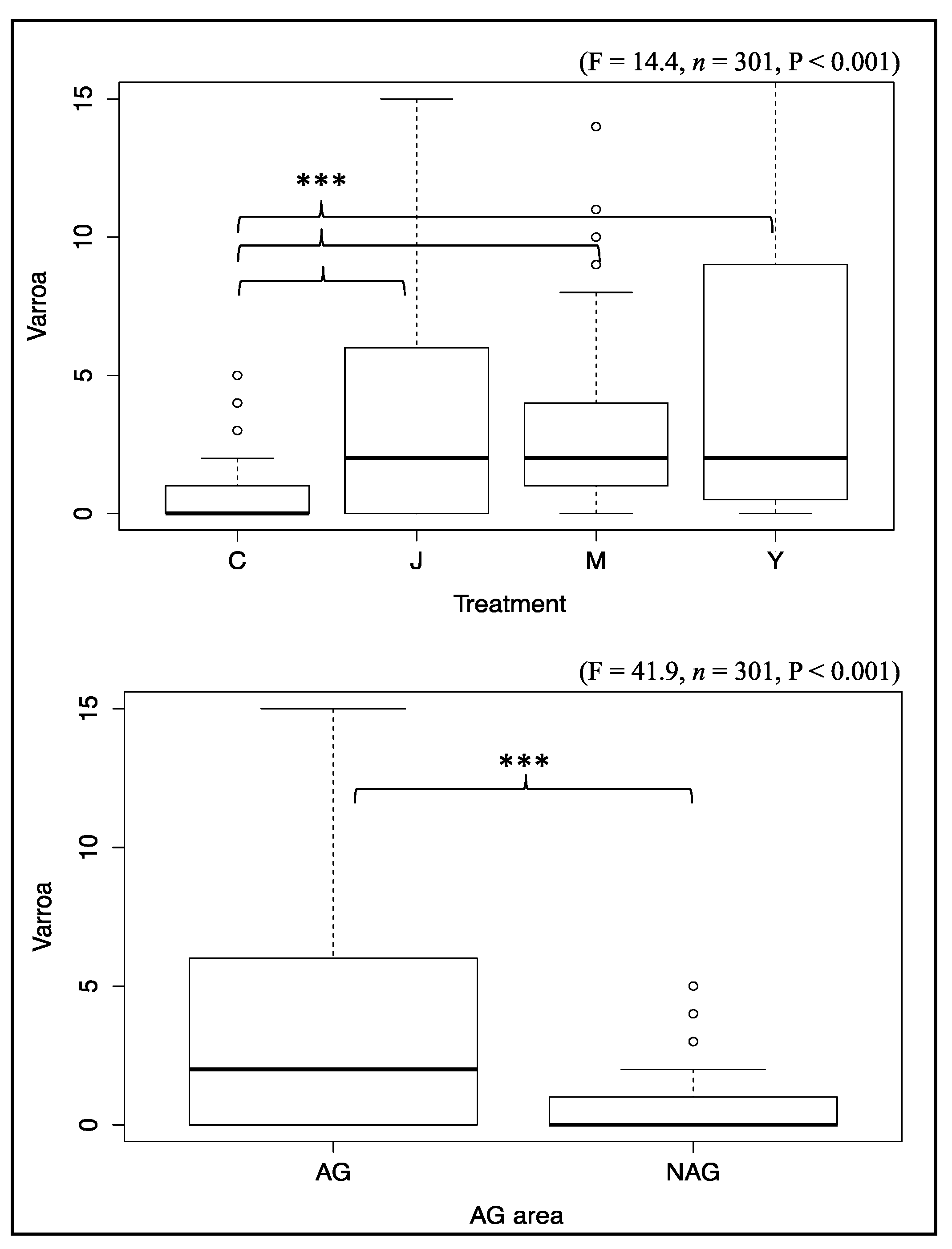
3.2. Varroa Infestation
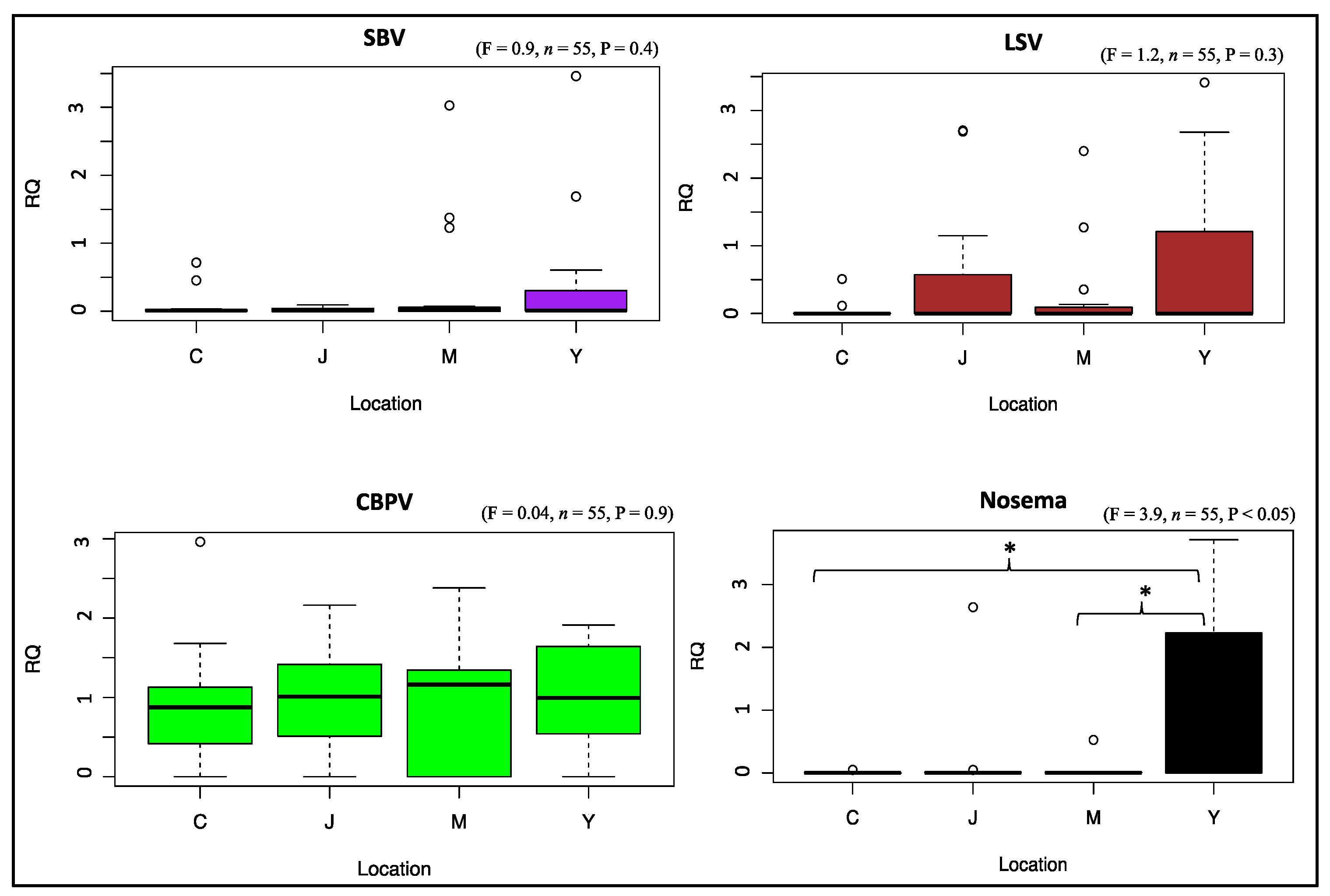
3.3. Viral and Nosema Infections
3.4. Overall Pathogen and Treatment Effects
3.5. Pesticide Residues
3.5.1. Forager Bees
3.5.2. Honey and Wax
3.5.3. Winter and Dead Bees
3.5.4. Crop Flowers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Engelsdorp, D.; Speybroeck, N.; Evans, J.D.; Nguyen, B.K.; Mullin, C.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y.; Tarpy, D.R.; et al. Weighing risk factors associated with bee colony collapse disorder by classification and regression tree analysis. J. Econ. Entomol. 2010, 103, 1517–1523. [Google Scholar] [CrossRef]
- Nazzi, F.; Brown, S.P.; Annoscia, D.; Del Piccolo, F.; Di Prisco, G.; Varricchio, P.; Della Vedova, G.; Cattonaro, F.; Caprio, E.; Pennacchio, F. Synergistic parasite-pathogen interactions mediated by host immunity can drive the collapse of honeybee colonies. PLoS Pathog. 2012, 8, e1002735. [Google Scholar] [CrossRef] [PubMed]
- Biesmeijer, J.C.; Roberts, S.P.; Reemer, M.; Ohlemuller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel declines in pollinators and insect-pollinated plants in britain and the netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Currie, R.W.; Pernal, S.F.; Guzman-Novoa, E. Honey bee colony losses in canada. J. Apic. Res. 2010, 49, 104–106. [Google Scholar] [CrossRef]
- Le Conte, Y.; Ellis, M.; Ritter, W. Varroa mites and honey bee health: Can varroa explain part of the colony losses? Apidologie 2010, 41, 353–363. [Google Scholar] [CrossRef]
- Guzman-Novoa, E.; Eccles, L.; Calvete, Y.; Mcgowan, J.; Kelly, P.G.; Correa-Benitez, A. Varroa destructor is the main culprit for the death and reduced populations of overwintered honey bee (Apis mellifera) colonies in ontario, canada. Apidologie 2010, 41, 443–450. [Google Scholar] [CrossRef]
- Fries, I. Nosema ceranae in european honey bees (Apis mellifera). J. Invertebr. Pathol. 2010, 103, S73–S79. [Google Scholar] [CrossRef] [PubMed]
- Van der Sluijs, J.P.; Simon-Delso, N.; Goulson, D.; Maxim, L.; Bonmatin, J.-M.; Belzunces, L.P. Neonicotinoids, bee disorders and the sustainability of pollinator services. Curr. Opin. Environ. Sustain. 2013, 5, 293–305. [Google Scholar] [CrossRef]
- Johnson, R.M.; Ellis, M.D.; Mullin, C.A.; Frazier, M. Pesticides and honey bee toxicity—USA. Apidologie 2010, 41, 312–331. [Google Scholar] [CrossRef]
- Dainat, B.; Evans, J.D.; Chen, Y.P.; Gauthier, L.; Neumann, P. Dead or alive: Deformed wing virus and Varroa destructor reduce the life span of winter honeybees. Appl. Environ. Microbiol. 2012, 78, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, P.; Aumeier, P.; Ziegelmann, B. Biology and control of varroa destructor. J. Invertebr. Pathol. 2010, 103, S96–S119. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.P.; Pettis, J.S.; Collins, A.; Feldlaufer, M.F. Prevalence and transmission of honeybee viruses. Appl. Environ. Microbiol. 2006, 72, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Calderone, N.W. Evaluation of mite-away-ii for fall control of varroa destructor (acari: Varroidae) in colonies of the honey bee apis mellifera (hymenoptera: Apidae) in the northeastern USA. Exp. Appl. Acarol. 2010, 50, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Maggi, M.D.; Ruffinengo, S.R.; Damiani, N.; Sardella, N.H.; Eguaras, M.J. First detection of varroa destructor resistance to coumaphos in argentina. Exp. Appl. Acarol. 2009, 47, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; Vanengelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in north american apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef] [PubMed]
- Ravoet, J.; Reybroeck, W.; de Graaf, D.C. Pesticides for apicultural and/or agricultural application found in belgian honey bee wax combs. Bull. Environ. Contam. Toxicol. 2015, 94, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Traynor, K.S.; Pettis, J.S.; Tarpy, D.R.; Mullin, C.A.; Frazier, J.L.; Frazier, M.; vanEngelsdorp, D. In-hive pesticide exposome: Assessing risks to migratory honey bees from in-hive pesticide contamination in the eastern united states. Sci. Rep. 2016, 6, 33207. [Google Scholar] [CrossRef] [PubMed]
- Straub, L.; Villamar-Bouza, L.; Bruckner, S.; Chantawannakul, P.; Gauthier, L.; Khongphinitbunjong, K.; Retschnig, G.; Troxler, A.; Vidondo, B.; Neumann, P.; et al. Neonicotinoid insecticides can serve as inadvertent insect contraceptives. Proc. R. Soc. Lond. B Biol. Sci. 2016, 283. [Google Scholar] [CrossRef] [PubMed]
- Decourtye, A.; Devillers, J.; Genecque, E.; Menach, K.L.; Budzinski, H.; Cluzeau, S.; Pham-Delègue, M.H. Comparative sublethal toxicity of nine pesticides on olfactory learning performances of the honeybee apis mellifera. Arch. Environ. Contam. Toxicol. 2005, 48, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Williamson, S.M.; Willis, S.J.; Wright, G.A. Exposure to neonicotinoids influences the motor function of adult worker honeybees. Ecotoxicology 2014, 23, 1409–1418. [Google Scholar] [CrossRef] [PubMed]
- Henry, M.; Béguin, M.; Requier, F.; Rollin, O.; Odoux, J.-F.; Aupinel, P.; Aptel, J.; Tchamitchian, S.; Decourtye, A. A common pesticide decreases foraging success and survival in honey bees. Science 2012, 336, 348–350. [Google Scholar] [CrossRef] [PubMed]
- Alburaki, M.; Gregorc, A.; Adamczyk, J.; Stewart, S. Insights on pollen diversity of honey bee apis mellifera l. Colonies located in various agricultural landscapes. Southeast. Nat. 2018, in press. [Google Scholar]
- Ruttner, F. Biogeography and Taxonomy of Honeybees; Springer: Berlin, Germany, 1988. [Google Scholar]
- Louveaux, J. The acclimatization of bees to a heather region. Bee World 1973, 54, 105–111. [Google Scholar] [CrossRef]
- Louveaux, J.; Albisetti, M.; Delangue, M.; Theurkauff, M. Les modalités de l’adaptation des abeilles (Apis mellifera L.) au milieu naturel. Ann. L’abeille 1966, 9, 323–350. [Google Scholar] [CrossRef]
- Alburaki, M.; Steckel, S.J.; Williams, M.T.; Skinner, J.A.; Tarpy, D.R.; Meikle, W.G.; Adamczyk, J.; Stewart, S.D. Agricultural landscape and pesticide effects on honey bee (hymenoptera: Apidae) biological traits. J. Econ. Entomol. 2017, 110, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Aufauvre, J.; Biron, D.G.; Vidau, C.; Fontbonne, R.; Roudel, M.; Diogon, M.; Vigues, B.; Belzunces, L.P.; Delbac, F.; Blot, N. Parasite-insecticide interactions: A case study of nosema ceranae and fipronil synergy on honeybee. Sci. Rep. 2012, 2, 326. [Google Scholar] [CrossRef] [PubMed]
- Cornman, R.S.; Tarpy, D.R.; Chen, Y.; Jeffreys, L.; Lopez, D.; Pettis, J.S.; vanEngelsdorp, D.; Evans, J.D. Pathogen webs in collapsing honey bee colonies. PLoS ONE 2012, 7, e43562. [Google Scholar] [CrossRef] [PubMed]
- Di Prisco, G.; Cavaliere, V.; Annoscia, D.; Varricchio, P.; Caprio, E.; Nazzi, F.; Gargiulo, G.; Pennacchio, F. Neonicotinoid clothianidin adversely affects insect immunity and promotes replication of a viral pathogen in honey bees. Proc. Natl. Acad. Sci. USA 2013, 110, 18466–18471. [Google Scholar] [CrossRef] [PubMed]
- Alaux, C.; Brunet, J.L.; Dussaubat, C.; Mondet, F.; Tchamitchan, S.; Cousin, M.; Brillard, J.; Baldy, A.; Belzunces, L.P.; Le Conte, Y. Interactions between nosema microspores and a neonicotinoid weaken honeybees (Apis mellifera). Environ. Microbiol. 2010, 12, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Alburaki, M.; Boutin, S.; Mercier, P.L.; Loublier, Y.; Chagnon, M.; Derome, N. Neonicotinoid-coated zea mays seeds indirectly affect honeybee performance and pathogen susceptibility in field trials. PLoS ONE 2015, 10, e0125790. [Google Scholar] [CrossRef] [PubMed]
- Alburaki, M.; Cheaib, B.; Quesnel, L.; Mercier, P.L.; Chagnon, M.; Derome, N. Performance of honeybee colonies located in neonicotinoid-treated and untreated cornfields in quebec. J. Appl. Entomol. 2016, 141, 112–121. [Google Scholar] [CrossRef]
- Traynor, K.S.; Rennich, K.; Forsgren, E.; Rose, R.; Pettis, J.; Kunkel, G.; Madella, S.; Evans, J.; Lopez, D.; vanEngelsdorp, D. Multiyear survey targeting disease incidence in us honey bees. Apidologie 2016, 47, 325–347. [Google Scholar] [CrossRef]
- Martin, S.J.; Highfield, A.C.; Brettell, L.; Villalobos, E.M.; Budge, G.E.; Powell, M.; Nikaido, S.; Schroeder, D.C. Global honey bee viral landscape altered by a parasitic mite. Science 2012, 336, 1304–1306. [Google Scholar] [CrossRef] [PubMed]
- Seeley, T.D. Honeybee Democracy; Princeton University Press: Princeton, NJ, USA, 2010; p. 273. [Google Scholar]
- ESRI. Arcgis Desktop: Release 10; Environmental Systems Research Institute: Redlands, CA, USA, 2011. [Google Scholar]
- Bourgeois, L.; Beaman, L.; Holloway, B.; Rinderer, T.E. External and internal detection of nosema ceranae on honey bees using real-time pcr. J. Invertebr. Pathol. 2012, 109, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P. A reagent for the single-step simultaneous isolation of rna, DNA and proteins from cell and tissue samples. Biotechniques 1993, 15, 532–534. [Google Scholar] [PubMed]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative rt-pcr data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]
- Glass, G.V.; Peckham, P.D.; Sanders, J.R. Consequences of failure to meet assumptions underlying the fixed effects analyses of variance and covariance. Rev. Educ. Res. 1972, 42, 237–288. [Google Scholar] [CrossRef]
- Harwell, M.R.; Rubinstein, E.N.; Hayes, W.S.; Olds, C.C. Summarizing monte carlo results in methodological research: The one- and two-factor fixed effects anova cases. J. Educ. Behav. Stat. 1992, 17, 315–339. [Google Scholar] [CrossRef]
- Lix, L.M.; Keselman, J.C.; Keselman, H.J. Consequences of assumption violations revisited: A quantitative review of alternatives to the one-way analysis of variance f test. Rev. Educ. Res. 1996, 66, 579–619. [Google Scholar]
- Sanchez-Bayo, F.; Goka, K. Pesticide residues and bees—A risk assessment. PLoS ONE 2014, 9, e94482. [Google Scholar] [CrossRef] [PubMed]
- Rolke, D.; Fuchs, S.; Grunewald, B.; Gao, Z.; Blenau, W. Large-scale monitoring of effects of clothianidin-dressed oilseed rape seeds on pollinating insects in northern germany: Effects on honey bees (Apis mellifera). Ecotoxicology 2016, 25, 1648–1665. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Zhang, P.; Wang, J.; Zheng, J. Determination of amitraz and its metabolites in whole blood using solid-phase extraction and liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2014, 951–952, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Haarmann, T.; Spivak, M.; Weaver, D.; Weaver, B.; Glenn, T. Effects of fluvalinate and coumaphos on queen honey bees (hymenoptera: Apidae) in two commercial queen rearing operations. J. Econ. Entomol. 2002, 95, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Rinderer, T.E.; De Guzman, L.I.; Lancaster, V.A.; Delatte, G.T.; Stelzer, J.A. Varroa in the mating yard: I. The effects of varroa jacobsoni and apistan® on drone honey bees. Am. Bee J. 1999, 139, 134–139. [Google Scholar]
- Bowen-Walker, P.; Martin, S.; Gunn, A. The transmission of deformed wing virus between honeybees (Apis mellifera L.) by the ectoparasitic mite varroa jacobsoni oud. J. Invertebr. Pathol. 1999, 73, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Bakonyi, T.; Farkas, R.; Szendroi, A.; Dobos-Kovacs, M.; Rusvai, M. Detection of acute bee paralysis virus by rt-pcr in honey bee and varroa destructor field samples: Rapid screening of representative hungarian apiaries. Apidologie 2002, 33, 63–74. [Google Scholar] [CrossRef]
- Gulmez, Y.; Bursali, A.; Tekin, S. First molecular detection and characterization of deformed wing virus (dwv) in honeybees (Apis mellifera L.) and mite (varroa destructor) in turkey. Afr. J. Biotechnol. 2009, 8, 3698–3702. [Google Scholar]
- Shen, M.; Yang, X.; Cox-Foster, D.; Cui, L. The role of varroa mites in infections of kashmir bee virus (kbv) and deformed wing virus (dwv) in honey bees. Virology 2005, 342, 141–149. [Google Scholar] [CrossRef] [PubMed]

| Apiary 1 | Apiary 2 | Apiary 3 | Apiary 4 | |
|---|---|---|---|---|
| Location | Jackson | Milan | Yum-Yum | Chickasaw |
| Hive Equipment | Varroa mite screen board | |||
| Varroa treatment | Once/October-2015/2 strips of Apivar per hive | |||
| N° of Sampling/Chemical analysis | ||||
| Forager | 5 times (May to September) 2015 | |||
| Worker (Viral) | 6 times (May to October) 2015 | |||
| Winter bees | One time (March 2016) | |||
| Dead bees | 29 July and 17 August 2015 | 26 June and 27 July 2015 | None | 27 July 2015 |
| Honey | 4 times (June to September) 2015 | |||
| Wax | 1 time (May 2015) | |||
| Model Number | Response/Explanatory Variable | Fixed Effects | Estimate Value | Std. Error | DF | T-Value | p-Value |
|---|---|---|---|---|---|---|---|
| (1) | Varroa~treatment | Jackson | 7.21 | 1.6 | 297 | 4.38 | <0.001 |
| Milan | 3.12 | 1.5 | 297 | 1.9 | 0.04 | ||
| Yum-Yum | 5.74 | 1.5 | 297 | 3.65 | <0.001 | ||
| (2) | Weight~treatment | Jackson | 0.4 | 1.2 | 315 | 0.3 | 0.7 |
| Milan | 4.65 | 1.2 | 315 | 3.9 | <0.001 | ||
| Yum-Yum | 5.74 | 1.2 | 315 | 4.81 | <0.001 | ||
| (3) | ABPV~treatment | Jackson | 0.2 | 71.8 | 92 | 0.003 | 0.9 |
| Milan | 173.9 | 62.2 | 92 | 2.79 | 0.006 | ||
| Yum-Yum | 0 | 58.9 | 92 | 0 | 0.9 | ||
| (4) | BQCV~treatment | Jackson | 110.7 | 57.8 | 92 | 1.9 | 0.057 |
| Milan | 1.7 | 50.1 | 92 | 0.03 | 0.9 | ||
| Yum-Yum | 1.8 | 47.5 | 92 | 0.03 | 0.9 | ||
| (5) | CBPV~treatment | Jackson | −83.4 | 45.9 | 92 | −1.8 | 0.07 |
| Milan | −70.9 | 39.7 | 92 | −1.7 | 0.07 | ||
| Yum-Yum | −82.1 | 37.6 | 92 | −2.1 | 0.03 | ||
| (6) | DWVa~treatment | Jackson | −2.7 | 283 | 92 | −0.01 | 0.9 |
| Milan | 44.1 | 246 | 92 | 0.1 | 0.8 | ||
| Yum-Yum | 492.2 | 232 | 92 | 2.1 | 0.03 | ||
| (7) | DWVb~treatment | Jackson | −102.3 | 54.4 | 92 | −1.8 | 0.06 |
| Milan | −113 | 47.1 | 92 | − 2.4 | 0.01 | ||
| Yum-Yum | −115 | 44.6 | 92 | − 2.5 | 0.01 | ||
| (8) | Varroa~treatment + weight | Jackson | 6.61 | 1.52 | 296 | 4.32 | <0.001 |
| Milan | 0.7 | 1.5 | 296 | 0.4 | 0.6 | ||
| Yum-Yum | 2.9 | 1.5 | 296 | 1.9 | 0.053 | ||
| Weight | 0.48 | 0.06 | 296 | 7.03 | <0.001 | ||
| (9) | Varroa~treatment + weight + all viruses | Jackson | 6.21 | 1.77 | 87 | 3.49 | <0.001 |
| Weight | 0.53 | 0.06 | 87 | 8.18 | <0.001 | ||
| (10) | Varroa~treatment + weight + all viruses + nosema | Jackson | 6.37 | 1.85 | 83 | 3.44 | <0.001 |
| Weight | 0.53 | 0.06 | 83 | 7.91 | <0.001 | ||
| (11) | Varroa~treatment + all viruses + nosema | Jackson | 7.05 | 1.64 | 84 | 2.90 | 0.004 |
| Sample | Pesticide | Apiary 1 (Jackson) PPB | Apiary 2 (Milan) PPB | Apiary 3 (Yum-Yum) PPB | Apiary 4 (Chickasaw) PPB | LD50 Oral (ng/bee) |
|---|---|---|---|---|---|---|
| Dead Bees | Imidacloprid | 3.3 | 190 | NA | 13 | |
| Imida. Olfen | 623 | NA | 28 | |||
| Clothianidin | 43 | 70 | NA | 4 | ||
| Thiamethoxam | 146 | NA | 5 | |||
| Carbaryl | 107 | 150 | ||||
| Methamidophos | 14.3 | 200 | ||||
| 1-Naphthol (carbaryl) | 230 | 10,500 | ||||
| Foragers | Imidacloprid | 3.1 | 13 | |||
| Azoxystrobin 1 | 7 | 25,000 | ||||
| Pendimethalin 2 | 111 | 665,000 | ||||
| Winter Bees | DMPF (amitraz) | 63.2 | 64.8 | 150 | 115 | 750,000 |
| Honey | Fluvalinate 3 | 5.4 | 5 | 45,000 | ||
| Wax | Imidacloprid | 3.7 | 13 | |||
| Fluvalinate 3 | 103 | 122 | 146 | 205 | 45,000 | |
| Dicofol 3 | 4.3 | 8 | 11 | 8 | 10,000 | |
| Carbendazim 1 | 5 | 5 | 5 | 5 | 50,000 | |
| Coumaphos | 6 | 5 | 5 | 6 | 4600 | |
| Fenpyroximate 3 | 5 | 5 | 5 | 8 | 1100 | |
| Metalaxyl 1 | 7 | 269,000 | ||||
| Atrazine 2 | 8 | 10 | 1000 | |||
| Crop flowers | ||||||
| Cotton | Imidacloprid | 25 | NA | 13 | ||
| Thiamethoxam | 24 | NA | 5 | |||
| Acephate | 309 | 57 | 4190 | NA | 230 | |
| Bifenthrin | 91 | NA | 200 | |||
| Cyhalothrin | 4 | NA | 22 | |||
| Methamidophos | 30.2 | 6504 | 1300 | NA | 200 | |
| Oxamyl 4 | 851 | 271 | NA | 380 | ||
| Soybean | Imidacloprid | 5.3 | 2.4 | NA | 13 | |
| Azoxystrobin 1 | 44 | NA | 25,000 | |||
| Fenpyroximate 3 | 5 | NA | 11,000 | |||
| Metolachlor 2 | 191 | NA | 1,260,000 | |||
| Pyridaben | 10 | NA | 550 | |||
| Sorghum | Acephate | 124 | NA | 230 | ||
| Cyhalothrin | 187 | 35 | NA | 22 | ||
| Methamidophos | 23 | NA | 200 | |||
| Azoxystrobin 1 | 15 | 17 | NA | 25,000 | ||
| Atrazine 2 | 8 | NA | 1000 | |||
| Chlorpyrifos | 3.2 | NA | 130 | |||
| Bifenthrin | 186 | NA | 240 | |||
| Oxamyl 4 | 5 | NA | 380 | |||
| Spinosad | 108 | NA | 57 | |||
| Pyraclostrobin 4 | 286 | NA | 73,000 | |||
| Corn | Metribuzin 2 | 9 | NA | 567,000 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alburaki, M.; Chen, D.; Skinner, J.A.; Meikle, W.G.; Tarpy, D.R.; Adamczyk, J.; Stewart, S.D. Honey Bee Survival and Pathogen Prevalence: From the Perspective of Landscape and Exposure to Pesticides. Insects 2018, 9, 65. https://doi.org/10.3390/insects9020065
Alburaki M, Chen D, Skinner JA, Meikle WG, Tarpy DR, Adamczyk J, Stewart SD. Honey Bee Survival and Pathogen Prevalence: From the Perspective of Landscape and Exposure to Pesticides. Insects. 2018; 9(2):65. https://doi.org/10.3390/insects9020065
Chicago/Turabian StyleAlburaki, Mohamed, Deniz Chen, John A. Skinner, William G. Meikle, David R. Tarpy, John Adamczyk, and Scott D. Stewart. 2018. "Honey Bee Survival and Pathogen Prevalence: From the Perspective of Landscape and Exposure to Pesticides" Insects 9, no. 2: 65. https://doi.org/10.3390/insects9020065
APA StyleAlburaki, M., Chen, D., Skinner, J. A., Meikle, W. G., Tarpy, D. R., Adamczyk, J., & Stewart, S. D. (2018). Honey Bee Survival and Pathogen Prevalence: From the Perspective of Landscape and Exposure to Pesticides. Insects, 9(2), 65. https://doi.org/10.3390/insects9020065








