Abstract
Fourteen polymorphic microsatellite loci were isolated in this transcriptome-based data analysis for Cotesia plutellae, which is an important larval parasitoid of the worldwide pest Plutella xylostella. A subsequent test was performed for a wild C. plutellae population (N = 32) from Fuzhou, Fujian, southeastern China, to verify the effectiveness of the 14 microsatellite loci in future studies on C. plutellae genetic diversity. The observed number of alleles ranged from two to six. The expected and observed heterozygosity ranged from 0.123 to 0.316 and from 0.141 to 0.281, respectively. The polymorphism information content (PIC) value ranged from 0.272 to 0.622. Potentially due to the substructure of the sampled population, three of the 14 microsatellite loci deviated from Hardy—Weinberg equilibrium (HWE). Further, loci C6, C22, and C31 could be amplified in Cocobius fulvus and Encarsia japonica, suggesting the transferability of these three polymorphic loci to other species of Hymenoptera.
1. Introduction
The diamondback moth (Plutella xylostella) is one of the most devastating pests for the global Brassica industry, and the estimated annual losses and management costs are approximately US $4–5 billion [1]. Traditional agrochemical-based management generates considerable drawbacks, such as pollution, habitat destruction, and loss of biodiversity. P. xylostella is attacked by natural enemies in all developmental stages; biological factors (e.g., parasitoids, arthropod predators, and pathogenic microorganisms, etc.) thus play a significant role in the integrated management of this species. There are more than 60 reported parasitoid species of P. xylostella, of which 12 have been widely applied for pest management in the field [2]. Cotesia plutellae is a larval parasitoid of P. xylostella, and its global distribution covers 38 countries on all continents except Antarctica [1]. As one of the most important parasitoids of P. xylostella, successful introduction of C. plutellae has been documented more than 20 times [1]. Parasitization of P. xylostella larvae by C. plutellae may increase their demand for food [3], extend the duration of the 4th-instar, and pupation (or the larva-to pupa metamorphosis) may be inhibited [4,5]. Before cocooning, C. plutellae moves out of the body of its host, eventually leading to its death.
Knowledge of the genetic variation and structure of the parasitoid populations favors parasitoid conservation and better pest control. Polymerase Chain Reaction (PCR) and molecular marker-based techniques are increasingly becoming recognized as valuable tools in ecological studies, and have been widely used in evaluating the genetic diversity of natural enemy populations [6]. Various advantages, such as high polymorphism, genome-wide distribution, co-dominant inheritance, and reproducibility, have made microsatellites one of the most widely applied types of marker in the field of molecular ecology [7]—especially in detecting genetic variation at the population level, evaluating population genetic diversity, and identifying genetic maps and kinship [8]. To date, however, no microsatellite-based studies have been performed on C. plutellae.
A comprehensive understanding of the biological characteristics, ecological characteristics, genetic variation, and interrelationships in different geographical populations of pests are prerequisite for the development of an effective pest control strategy [9]. Using molecular markers, we can understand the potential threat areas and effectively control the spread of pests [10]. So, the analysis and development of SSR markers with polymorphism has great application value and significance.
2. Material and Methods
2.1. Primer Design
A total of 19,309 microsatellite loci—among which 17,070 had complete motifs—were detected by MISA (MicroSAtellite identification) [11] based on the C. plutellae transcriptome dataset [12], downloaded from (National Center of Biotechnology Information) (NCBI) (http://www.ncbi.nlm.nih.gov/sra/DRX001445). Primer 3.0 [13] was used for primer design for the complete microsatellite loci, of which 85 trimers were randomly selected for validation with PCR.
2.2. Proved the Effectiveness of Primers
Primer effectiveness was verified on a lab-reared C. plutellae colony (Fuzhou, May 2014), and the subsequent identification of polymorphic loci and population genetic analysis was performed using individuals from wild C. plutellae populations (Fuzhou, April 2014). The C. plutellae were collected from nine 20 × 30 m plots with an average distance of 1.47 km (range, 0.65–3.68 km) between plots. Considering the arrhenotokous parthenogenesis of Hymenopteran species (i.e., diploid females develop from fertilized eggs and haploid males develop from unfertilized eggs) [14,15], only female C. plutellae individuals were used for population genetic analysis (N = 32).
Genomic DNA was individually isolated with the DNeasy Blood and Tissue Kit (QIAGEN, Hilden, Germany) following the manufacturer's instructions. We initially performed PCR to validate the effectiveness of the primer pairs. Polymorphism identification was based on the primer pairs and the expected product sizes. The forward primers of the validated primer pairs were linked with the universal primer M-13 (TGT AAA ACG ACG GCC AGT) at their 5’ ends, and M-13 labeled with FAM, HEX, or TAMRA fluorescent dye was also used in the genotyping system.
The total volume of each PCR mixture was 25 µL, containing 12.5 µL Mix (Promega), 0.2 µL forward primer, 0.8 µL reverse primer, and 0.8 µL M-13. The temperature conditions were at 94 °C for 10 min, and then 30 cycles at 94 °C for 30 s, Tm (the optimal annealing temperatures, see Table 1) for 45 s, 72 °C for 45 s, followed by eight cycles at 94 °C for 30 s, 53 °C for 45 s, 72 °C for 45 s, and a final extension at 72 °C for 10 min.

Table 1.
Characterization of the 14 polymorphic microsatellite loci developed for C. plutellae.
2.3. Data Analysis
After testing by agarose gel electrophoresis (AGE), sizes of the amplification products were detected using an ABI 3730 sequencer (Applied Biosystems, Foster City, CA, USA). GeneMapper 4.1 (Applied Biosystems, Foster City, CA, USA) was used to assign alleles based on the sizes of PCR amplifications.
Fourteen polymorphic loci were isolated in this study. Number of alleles, expected heterozygosity (HO), observed heterozygosity (HE), and polymorphism information content (PIC) were calculated using genepop V4 (Montpellier, France) [16]. Hardy—Weinberg equilibrium (HWE) and linkage disequilibrium (LD) were also tested using genepop V4.
3. Results and Discussion
As one of the important indicators of population genetic variation, the number of alleles ranged from two to six for the 14 identified microsatellite loci in the tested population (Table 1). Comparable results can be found in recent studies. For example, the number of alleles per locus ranges from 2 to 19 for 30 female Blastophaga javana (Hymenoptera: Agaoninae) [17], the number of alleles ranges from 3 to 11 for Trypoxylon (Trypargilum) albitarse (Hymenoptera: Crabronidae) [18]; and the allelic number per locus varies from three to seven (N = 30) for Pachycrepoideus vindemmiae (Rondani) (Hymenoptera: Pteromalidae) [19]. A locus was considered to show low polymorphism with an associated PIC value of <0.25; and to show high polymorphism with an associated PIC value of >0.5. The polymorphism level of the 14 identified microsatellite loci in the present study were medium-to-high, with PIC values ranging from 0.272 to 0.622. The HE and HO values of the 14 microsatellite loci ranged from 0.123 to 0.316 and 0.141 to 0.281, respectively. Compared with the values of HE, the relatively lower HO values suggest a lack of heterozygosity in the examined population (Table 1).
4. Conclusions
Loci C6, C32, and C53 deviated significantly from HWE, and significant LD was observed between loci C6 and C32. Samples for this study were collected from different fields patchily distributed within the sampling site. Considering the weak migration capacity of C. plutellae, the structures of sub-populations from different fields might be responsible for such a deviation from LD and HWE.
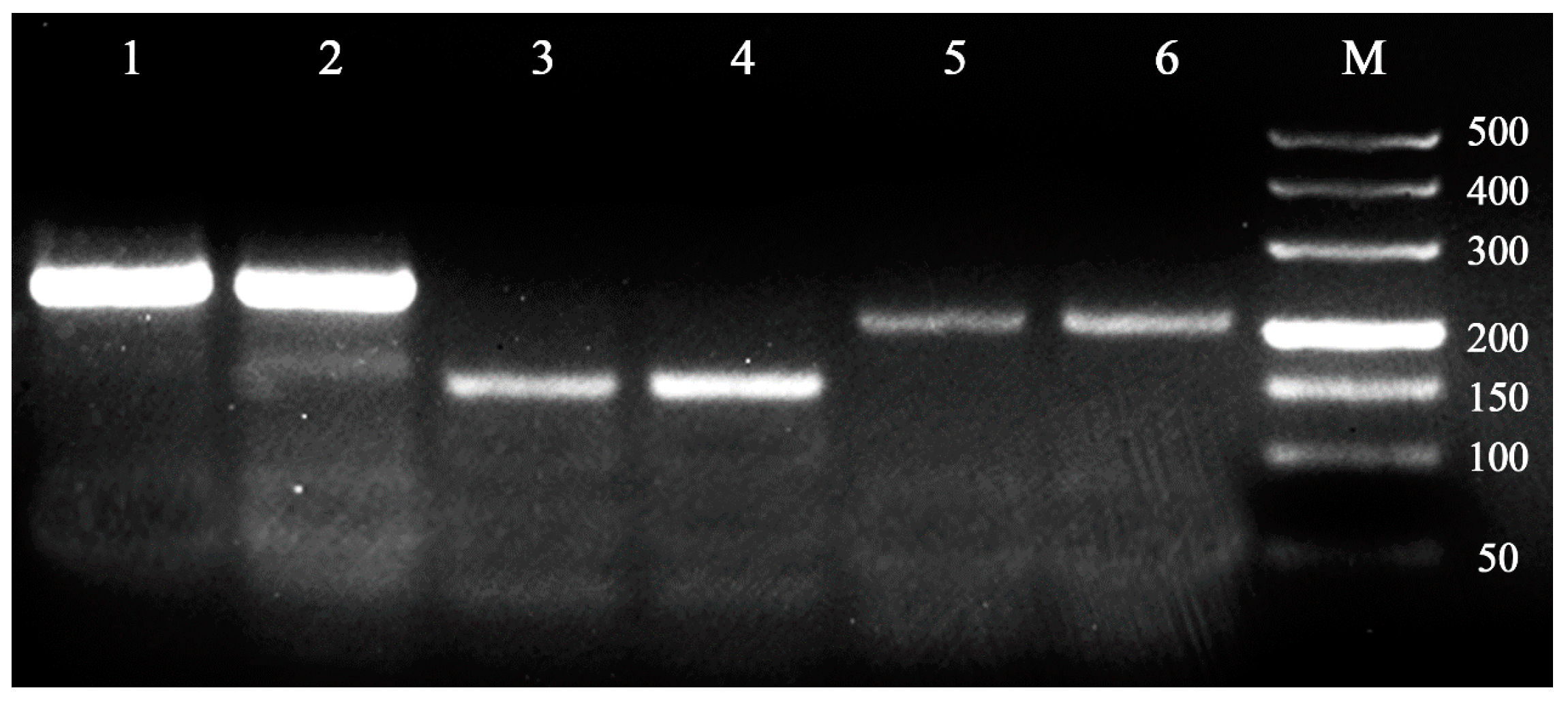
The transferability of these primers (i.e., with detectable amplification products) was also verified in Cocobius fulvus and Encarsia japonica, and three of the fourteen microsatellite loci showed amplified products with the expected sizes (Figure 1). Therefore, the microsatellite loci characterized in this study will favor studies of genetic diversity and population structure of not only C. plutellae, but also other Hymenopteran parasitoids.
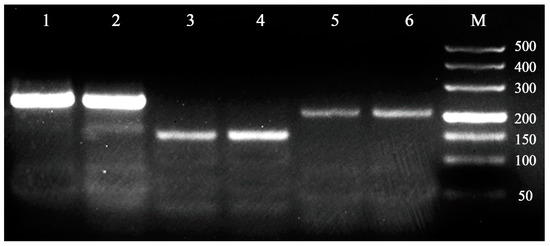
Figure 1.
Gel electrophoretogram showing the effectiveness of three microsatellite loci (C6, C22, C31) in C. fulvus and E. japonica, with 1, 3, 5 for C. fulvus, and 2, 4, 6 for E. japonica. M: DL500 bp DNA marker.
Acknowledgments
The work was supported by National Natural Science Foundation of China (No. 31320103922 and No. 31230061).
Author Contributions
Minsheng You managed the project. Tiansheng Liu and Fushi Ke designed experiments and analysis. Wenbin Chen prepared insect samples. Weiyi He and Shijun You revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Furlong, M.J.; Wright, D.J.; Dosdall, M. Diamondback moth ecology and management: problems, progress, and prospects. Annu. Rev. Entomol. 2013, 58, 517–541. [Google Scholar] [CrossRef] [PubMed]
- Delvare, G.; Kirk, A.A.; Bordet, D. The taxonomic status and role of Hymenoptera in biological control of DBM, Plutella xylostella (L.) (Lepidoptera: Plutellidae) Improving biocontrol of Plutella xylostella. In Proceedings of the International Symposium on CIRAD, Montpellier, France, 22–24 October 2004; pp. 17–49. [Google Scholar]
- Shi, Z.H.; Liu, S.S.; Li, Y.X. Cotesia plutellae parasitizing Plutella xylostella: Host-age dependent parasitism and its effect on host development and food consumption. Biocontrol 2002, 47, 499–511. [Google Scholar] [CrossRef]
- Ali, M.R.; Seo, J.; Lee, D.; Kim, Y. Teratocyte-secreting proteins of an endoparasitoid wasp, Cotesia plutellae, prevent host metamorphosis by altering endocrine signals. Comp. Biochem. Phys. 2013, 166, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Hepat, R.; Lee, D.; Kim, Y. Protein tyrosine phosphatase encoded in Cotesia plutellae bracovirus suppresses a larva-to-pupa metamorphosis of the diamondback moth, Plutella xylostella. Comp. Biochem. Phys. 2013, 166, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Gariepy, T.D.; Kuhlmann, U.; Gillott, C.; Erlandson, M. Parasitoids, predators and PCR: The use of diagnostic molecular markers in biological control of Arthropods. J. Appl. Entomol. 2007, 131, 225–240. [Google Scholar] [CrossRef]
- Selkoe, K.A.; Toonen, R.J. Microsatellites for ecologists: A practical guide to using and evaluating microsatellite markers. Ecol. Lett. 2006, 9, 615–629. [Google Scholar] [CrossRef] [PubMed]
- Vaiman, D.; Pailhoux, E.; Payen, E.; Saidi-Mehtar, N.; Cotinot, C. Evolutionary conservation of a microsatellite in the Wilms Tumour (WT) gene: Mapping in sheep and cattle. Cytogenet. Genome. Res. 1995, 70, 112–115. [Google Scholar] [CrossRef]
- Roderick, G.K.; Navajas, M. Genes in new environments: Genetics and evolution in biological control. Nat. Rev. Genet. 2003, 4, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Cullingham, C.I.; Roe, A.D.; Sperling, F.A.H.; Coltman, D.W. Phylogeographic insights into an irruptive pest outbreak. Ecol. Evol. 2012, 2, 908–919. [Google Scholar] [CrossRef] [PubMed]
- Thiel, T.; Michalek, W.; Varshney, R.K.; Graner, A. Exploiting EST databases for the development and characterization of gene-derived SSR-markers in barley (Hordeum Vulgare L.). Theor. Appl. Genet. 2003, 106, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, O.; Brillada, C.; Yazawa, S.; Maffei, M.E.; Arimura, G.I. Transcriptome pyrosequencing of the parasitoid wasp Cotesia plutellae: genes involved in the antennal odorant-sensory system. PLoS ONE 2012, 7, e50664. [Google Scholar] [CrossRef] [PubMed]
- Rozen, S.; Skaletsky, H. Primer 3. Available online: http://primer3.sourceforge.net/ (accessed on 6 September 2015).
- Cook, J.M. Sex determination in the Hymenoptera: A review of models and evidence. Heredity 1993, 71, 421–435. [Google Scholar] [CrossRef]
- Heimpel, G.E.; De Boer, J.G. Sex determination in the Hymenoptera. Annu. Rev. Entomol. 2008, 53, 209–230. [Google Scholar] [CrossRef] [PubMed]
- Rousset, F. Genepop’007: A complete re-implementation of the genepop software for Windows and Linux. Mol. Ecol. Resour. 2008, 8, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Tian, E.W.; Yu, H.; Zhang, D.Y.; Nason, J.D. Development of microsatellite loci for Blastophaga javana (Agaonidae), the pollinating wasp of Ficus hirta (Moraceae). Am. J. Bot. 2011, 98, E41–E43. [Google Scholar] [CrossRef] [PubMed]
- Almeida, J.C.; Bergamaschi, A.B.; Sanches, A.; Hatanaka, T.; Del Lama, M.A. Isolation and characterization of microsatellite loci for the mud-dauber wasp Trypoxylon (Trypargilum) albitarse (Hymenoptera: Crabronidae). Eur. J. Entomol. 2013, 110, 541–543. [Google Scholar] [CrossRef]
- Chen, W.; Fang, L.; Liu, J.; He, Z.; Hu, H. Isolation and characterization of polymorphic microsatellite loci for Pachycrepoideus vindemmiae (Rondani) (Hymenoptera: Pteromalidae). Genet. Mol. Res. 2015, 14, 1798–1801. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).