3.1. General Body Plan
Insect vectors of Chagas disease are opportunistic feeders that display a body plan designed for accommodating a large blood meal. As seen in the insects of this study (
Figure 2), these insects have a characteristically large abdomen compared to the head and thorax. While they may vary in color (for color prints see [
9,
10]), the color patches are located in the same regions reflecting the similarity in the underlying tissue and muscle attachments. For the most part, the dorsal cuticle of the adult lacks pigmentation, especially towards the medial regions, and the internal organs can be visualized through the cuticle when the lighting has been adjusted to reduce glare. The ability to see through the cuticle of an intact insect was used previously to show that the heart rate of
R. prolixus is inhibited by tactile stimulation of the sensory hairs along the ventral posterior side of the animal [
8]. What cannot be seen from the dorsal aspect are the lateral abdominal folds in adults of
R. prolixus and
R. brethesi. These folds are adult features for
Rhodnius and provide the surface area needed to allow the abdomen to stretch.
Another morphological feature often overlooked in the abdominal cuticle of these insects is the first pair of abdominal spiracles. Unlike the rest of the abdominal spiracles, these are located on the dorsal side of the insect in a narrow flap of cuticle that is immediately in front of the first full-sized abdominal segment. These spiracles are not found along the lateral edge of the abdomen, but are positioned approximately one quarter of the distance to the midline (see
Figure 3). The location of this first pair of spiracles identifies the first full-sized abdominal segment on the dorsal cuticle of these insects to be a fusion of abdominal Segments 1 and 2. Consistent numbering of the abdominal segments is important to ensure accuracy when comparing the work done on these insects by different researchers. For this study, and for comparisons to previous work, the abdominal segments are numbered Segment 1–2, Segment 3, Segment 4,
etc. By applying this terminology, the most posterior full sized abdominal segment that houses the genitalia (not visible in the dorsal views in
Figure 2) is abdominal Segment 7.
Figure 2.
Dorsal view of the adult males of five species of Chagas disease vectors in which a cardio-inhibitor has been detected in crude extracts of the testes. Wings have been removed to show the similarity in the appearance of their dorsal abdominal cuticles. In these insects, the dorsal cuticle is mainly transparent and internal organs are visible when the lighting is adjusted to reduce glare. In these pictures, the dorsal vessel is clearly visible in Neotriatoma bruneri and Rhodnius prolixus as a dark line extending along the midline of the abdomen. It appears as a faint white line in R. bresethi. Scale bar: 5 mm.
Figure 2.
Dorsal view of the adult males of five species of Chagas disease vectors in which a cardio-inhibitor has been detected in crude extracts of the testes. Wings have been removed to show the similarity in the appearance of their dorsal abdominal cuticles. In these insects, the dorsal cuticle is mainly transparent and internal organs are visible when the lighting is adjusted to reduce glare. In these pictures, the dorsal vessel is clearly visible in Neotriatoma bruneri and Rhodnius prolixus as a dark line extending along the midline of the abdomen. It appears as a faint white line in R. bresethi. Scale bar: 5 mm.
Figure 3.
Dorsal view of the metathorax and the first full-sized abdominal segment (Segment 1–2) of an adult male Rhodnius prolixus covered with Rhodnius saline to increase the transparency of the cuticle. Wings have been removed leaving only their bases attached (w). Black arrowheads point to the first set of abdominal spiracles that are located on the dorsal side of the animal. The spiracles are not visible, but their corresponding tracheal trunks can be viewed through the transparent cuticle. The aorta of the dorsal vessel (ao) can also be seen through the cuticle. Scale bar: 1 mm.
Figure 3.
Dorsal view of the metathorax and the first full-sized abdominal segment (Segment 1–2) of an adult male Rhodnius prolixus covered with Rhodnius saline to increase the transparency of the cuticle. Wings have been removed leaving only their bases attached (w). Black arrowheads point to the first set of abdominal spiracles that are located on the dorsal side of the animal. The spiracles are not visible, but their corresponding tracheal trunks can be viewed through the transparent cuticle. The aorta of the dorsal vessel (ao) can also be seen through the cuticle. Scale bar: 1 mm.
In addition to the abdomen being large, the structural components of both the nervous system and the circulatory system are arranged to permit these systems to continue to function when the abdomen becomes fully extended to accommodate the blood meal. The ventral nerve cord of many insects contains a ganglion in each of the thoracic and abdominal segments, but in Reduviidae, segmental ganglia in the abdomen are absent [
11]. Rather than being located in the abdomen where they could experience considerable pressure from the expanded stomach, the abdominal segments are supplied by segmental nerves arising from the posterior ganglion located in the mesothorax. These abdominal nerves display some slack allowing them to be extended to a limited extent, and being located on the ventral side of the abdomen, these nerves are less subject to the stretch of the stomach. In adults, most stretch associated with feeding occurs in the dorsal cuticle.
Circulation is also maintained after engorging a blood meal because the alary muscles and ostia of the heart are located on the dorsal vessel to the rear of the abdomen at the level of the last two abdominal segments [
12]. This region is not congested by the expanded stomach, and hemolymph is free to enter the heart which can propel a pulse of hemolymph into the aorta. The aorta stretches considerably along its length when the stomach expands, and it becomes flattened against the inner surface of the dorsal cuticle. Nevertheless, it maintains the ability to produce peristalsis which pushes the pulse of hemolymph up into the head, as can be visualized through the transparent cuticle. Although these insects appear unaffected if circulation is stopped, previous studies have shown that continued circulation to the head is important for long-term processes, such as egg production [
13]. By localizing the heart to a region not congested by the engorged stomach, circulation can continue unimpeded.
3.2. Heartbeats
To carry out the heart bioassay, the dorsal vessel was exposed as shown in
Figure 4. The heartbeat has been previously described in detail for
R. prolixus [
12], and the same beating pattern was observed in the other species examined here. In brief, a single beat in a spontaneously active heart begins with (1) alary muscle contractions which expand the heart chamber at the posterior end of the dorsal vessel allowing hemolymph to enter the heart, (2) constriction of the heart chamber by the heart muscles to push hemolymph into the aorta, and (3) peristaltic contraction of the aorta to push hemolymph anteriorly. Even though each action associated with the dorsal vessel can occur independently, all three actions are needed to ensure maximal flow of hemolymph. In a regularly beating heart, the heart rate can range from 1 bpm to close to 40 bpm. Rates above 40 bpm reduce circulation because the heart is not in diastole long enough for maximal filling.
Figure 4.
A live preparation covered with Rhodnius saline showing the posterior region of the dorsal vessel of an adult female R. prolixus exposed by removing the dorsal cuticle. When the alary muscles (two labeled as ‘a’) contract, the hemolymph enters the dorsal vessel through ostia located in the walls of the heart (h). The heart chamber contracts to propel hemolymph into the aorta (ao). The aorta initiates a peristaltic wave (at arrowhead), which constricts the lumen of the aorta pushing hemolymph up into the head. Only the dorsal alary muscles on the one side of abdominal Segments 6 and 7 are labeled. Scale bar measures 1 mm.
Figure 4.
A live preparation covered with Rhodnius saline showing the posterior region of the dorsal vessel of an adult female R. prolixus exposed by removing the dorsal cuticle. When the alary muscles (two labeled as ‘a’) contract, the hemolymph enters the dorsal vessel through ostia located in the walls of the heart (h). The heart chamber contracts to propel hemolymph into the aorta (ao). The aorta initiates a peristaltic wave (at arrowhead), which constricts the lumen of the aorta pushing hemolymph up into the head. Only the dorsal alary muscles on the one side of abdominal Segments 6 and 7 are labeled. Scale bar measures 1 mm.
For determining the effect of a test solution on the heartbeat, a single constriction of the heart chamber that pushes hemolymph into the aorta was counted as a single beat. In some cases, the heart may appear to be actively beating as the alary muscles contract to expand the heart, and then relax to allow the heart to return to rest by elastic recoil. Unlike a contraction of the heart, this action does not constrict the heart chamber and no hemolymph is pumped forward into the aorta. In a regularly beating heart, a single contraction and relaxation of the alary muscles is usually followed by contraction of the heart. In irregularly beating hearts, the alary muscles may contract two or more times before a heartbeat occurs, and if counted as a beat, the heart rate would be overestimated.
3.3. Male Reproductive System
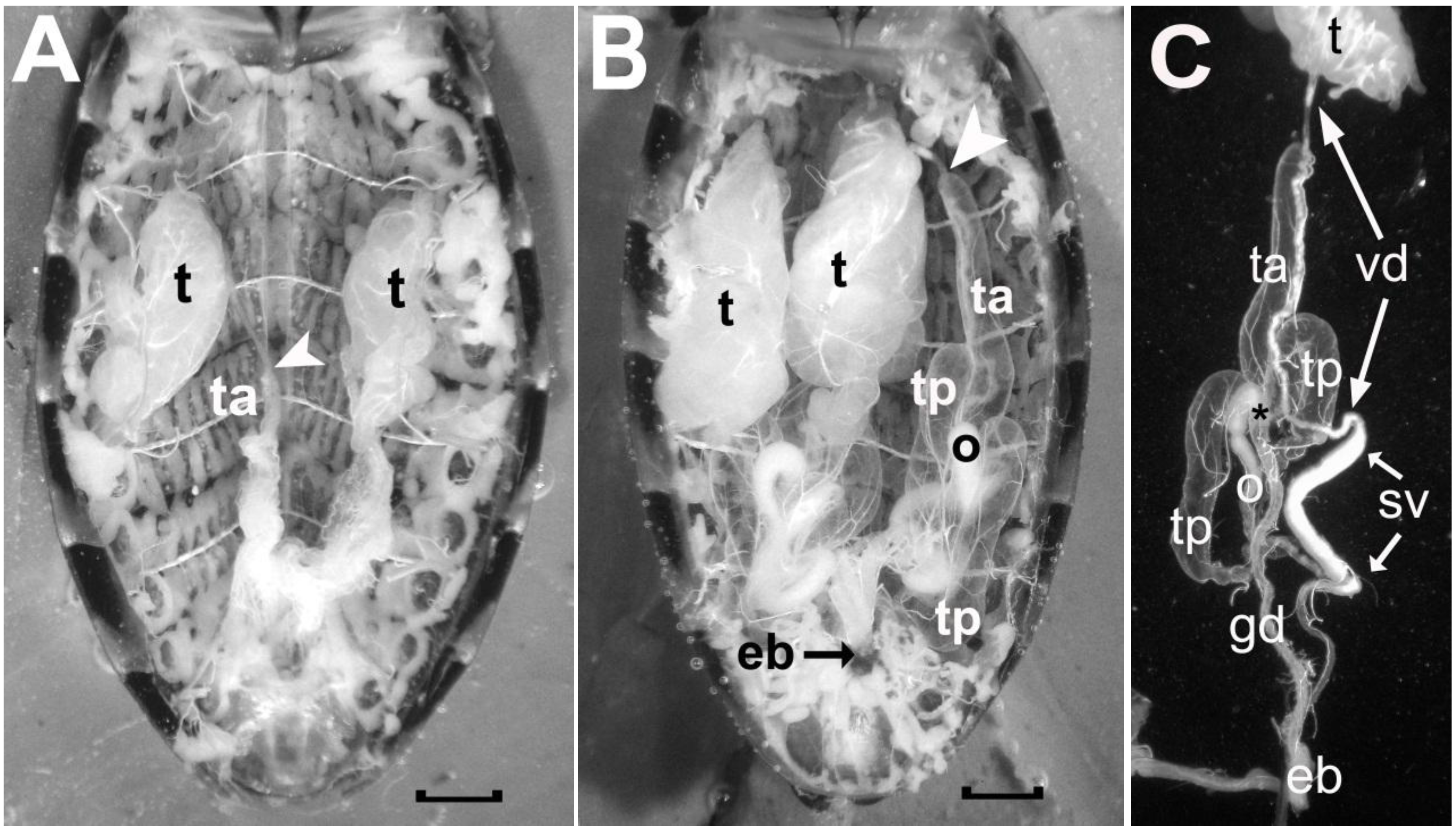
The male reproductive system in Reduviidae is bilaterally symmetrical and each side consists of a testis and four accessory glands (see
Figure 5). The testis is located laterally in the mid region of the abdomen and is supplied by trachea connected to tracheal trunks attached to Spiracles 3, 4 and 5 on the ipsilateral side. In recently emerged adult insects, the testes are well developed and extend between abdominal Segments 3 and 4 (
Figure 5A). As they mature, they increase in girth and length and fill a large portion of the abdominal cavity from Segment 1–2 to Segment 5 (
Figure 5B). The spermatozoa travel from the testis (t) down the vas deferens (vd) to be stored in the seminal vesicle (sv) before being delivered to the vagina by way of the ejaculatory bulb (eb) (refer to
Figure 5C).
Figure 5.
Photographs comparing the male reproductive system of a recently emerged adult male R. prolixus prior to ingesting a blood meal (A) and that of a two-month old adult male that has ingested more than one blood meal and has been mating (B). In C, the different components on one side of the reproductive tract of the older male are illustrated after these were teased apart. Symbols in A: t, testis; ta, empty anterior transparent gland; white arrowhead, location where the vas deferens from the testis attaches to the anterior tip of the anterior transparent gland. Symbols in B as in A with the addition of: ta, a filled anterior accessory transparent gland; tp, the two posterior accessory glands; o, opaque gland; eb, ejaculatory bulb. Symbols in C as in A and B with the addition of: gd, common duct of the accessory glands; vd, vas deferens; sv, seminal vesicle; asterisk, calyx of the glandular duct. Scale bars measure 1 mm.
Figure 5.
Photographs comparing the male reproductive system of a recently emerged adult male R. prolixus prior to ingesting a blood meal (A) and that of a two-month old adult male that has ingested more than one blood meal and has been mating (B). In C, the different components on one side of the reproductive tract of the older male are illustrated after these were teased apart. Symbols in A: t, testis; ta, empty anterior transparent gland; white arrowhead, location where the vas deferens from the testis attaches to the anterior tip of the anterior transparent gland. Symbols in B as in A with the addition of: ta, a filled anterior accessory transparent gland; tp, the two posterior accessory glands; o, opaque gland; eb, ejaculatory bulb. Symbols in C as in A and B with the addition of: gd, common duct of the accessory glands; vd, vas deferens; sv, seminal vesicle; asterisk, calyx of the glandular duct. Scale bars measure 1 mm.
The accessory glands produce two morphologically distinct sets of secretions that the male delivers to the female after he has placed the semen into the vagina. During copulation, these two secretions are delivered one after the other [
14]. The first secretion delivered after the semen is opaque in appearance, and is produced and stored in the opaque gland (‘o’ in
Figure 5B and
Figure 5C). The second secretion is transparent in appearance, and it constitutes the bulk of the spermatophore. This transparent material is made and stored in three transparent accessory glands. One transparent gland extends its blind end anteriorly (‘ta’ in
Figure 5B and
Figure 5C), and two extend posteriorly (‘tp’ in
Figure 5B and
Figure 5C). The bases of these glands converge at the calyx of the gland duct (asterisk in
Figure 5C), and this common gland duct carries their secretions to the ejaculatory bulb. Unlike the testes, which are fully formed by the time of adult emergence, the accessory glands in recently emerged insects appear as small flattened sacs (see
Figure 5A). Following a feed, they increase their girth and stretch longitudinally as they become filled with excretory material. In the older male (
Figure 5B), the anterior transparent gland has stretched alongside its corresponding testis, and its anterior tip remains structurally associated with the vas deferens as indicated by comparing the white arrowhead in the younger insect to that in the older male.
Our experience has shown that recently emerged unfed males do not mate, and to ensure that copulation occurs, adult males need to be fed a few days prior to mating. This observation, coupled with the appearance of the accessory glands in the unfed male, suggests that a lack of accessory gland material, and not the absence of spermatozoa, delays the mating behavior. When enough accessory gland material is available for mating, it is possible that a signal from the accessory glands to the nervous system provides the trigger for mating to begin. And it is possible that such a trigger could be carried by a hormone (an endocrine signal) or by the nervous system, because previous studies have shown that both mechanisms are available to the male. First, Sevala and Davey [
15] found that the transparent accessory glands produce a polypeptide that is secreted into the body cavity. This substance could serve as a hormone. Second, Chiang and Davey [
16] discovered a set of pressure receptors in the ventral body wall. These receptors are ideally situated to respond to pressure imposed by the enlarging accessory glands on either side of the insect.
Fully formed testes first appear in larval Stage 5 (see
Figure 6). They do not undergo metamorphosis following feeding, but increase in size until the adult insect emerges. From the L5 stage to the older adults, the testes appear as a slightly flattened clump of tubules held together by a thin overlying membrane, which is associated with a fine mesh of tracheae. This membrane does not fully enclose the tubules of the testes, and their blind ends extend from the clump (see
Figure 6B). These tubules are the testicular follicles, and in Reduviidae, each testis has seven follicles, which converge at their base to attach to the calyx of the vas deferens. Freitas
et al. [
17,
18,
19] have described the testicular follicles of several species of Reduviidae, and in all cases, two of the seven follicles are broader and several times longer than the other five follicles. We also observe this arrangement in
R. prolixus, an arrangement, which is maintained from the L5 to the older actively mating male (see
Figure 6).
In the male involved in mating, the bulk of the testis consists of the two larger follicles, which contain material not visible in the smaller follicles. This morphological difference suggests different processes occurring in these two groups of follicles. The two larger testicular follicles may be involved in spermatogenesis and the production of spermatozoa, whereas the smaller testicular follicles could be involved in producing other secretions necessary for reproduction. Experiments are being planned to determine if the cardio-inhibitor from the testes is present in all the follicles or is localized in one of the two groups.
Figure 6.
The testicular follicles and testis in R. prolixus taken from an L5 (A), a recently emerged adult (B), and a two-month old reproductively active adult (C). The five smaller follicles and two larger follicles (asterisks) are easily identified in the L5 (A). Even in an intact testis in B, the tips of the smaller follicles (arrows) can be distinguished from the two larger follicles. Testis growth with age is primarily due to growth of the two larger follicles. The larger follicles are approximately one and a half times longer than the smaller follicles in the young testis, but up to five to six times longer than the small follicles of the older testis (C). The vas deferens and the anterior transparent gland are not present in the L5, since these are adult structures that emerge in the process of metamorphosis. In C, the arrowhead denotes the attachment of the vas deferens to the transparent accessory gland. Scale bars: 0.2 mm.
Figure 6.
The testicular follicles and testis in R. prolixus taken from an L5 (A), a recently emerged adult (B), and a two-month old reproductively active adult (C). The five smaller follicles and two larger follicles (asterisks) are easily identified in the L5 (A). Even in an intact testis in B, the tips of the smaller follicles (arrows) can be distinguished from the two larger follicles. Testis growth with age is primarily due to growth of the two larger follicles. The larger follicles are approximately one and a half times longer than the smaller follicles in the young testis, but up to five to six times longer than the small follicles of the older testis (C). The vas deferens and the anterior transparent gland are not present in the L5, since these are adult structures that emerge in the process of metamorphosis. In C, the arrowhead denotes the attachment of the vas deferens to the transparent accessory gland. Scale bars: 0.2 mm.
3.5. Cardio-Inhibitor in the Spermatophore
For rhodtestolin to have an effect on the female, it needs to be delivered during copulation as the male creates the spermatophore in the vagina. As noted above, the spermatophore is produced by the male first injecting semen into the vagina followed by a small amount of material from the opaque gland and then a larger amount of material from the transparent gland. The spermatophore is not immediately ejected after copulation, but remains in the vagina for several hours. This timing allows the spermatozoa to migrate to the spermatheca, a pair of blind-ended tubes, one attached to each side of the common oviduct [
20]. Once a spermatophore casting is ejected from the vagina, the female can copulate again.
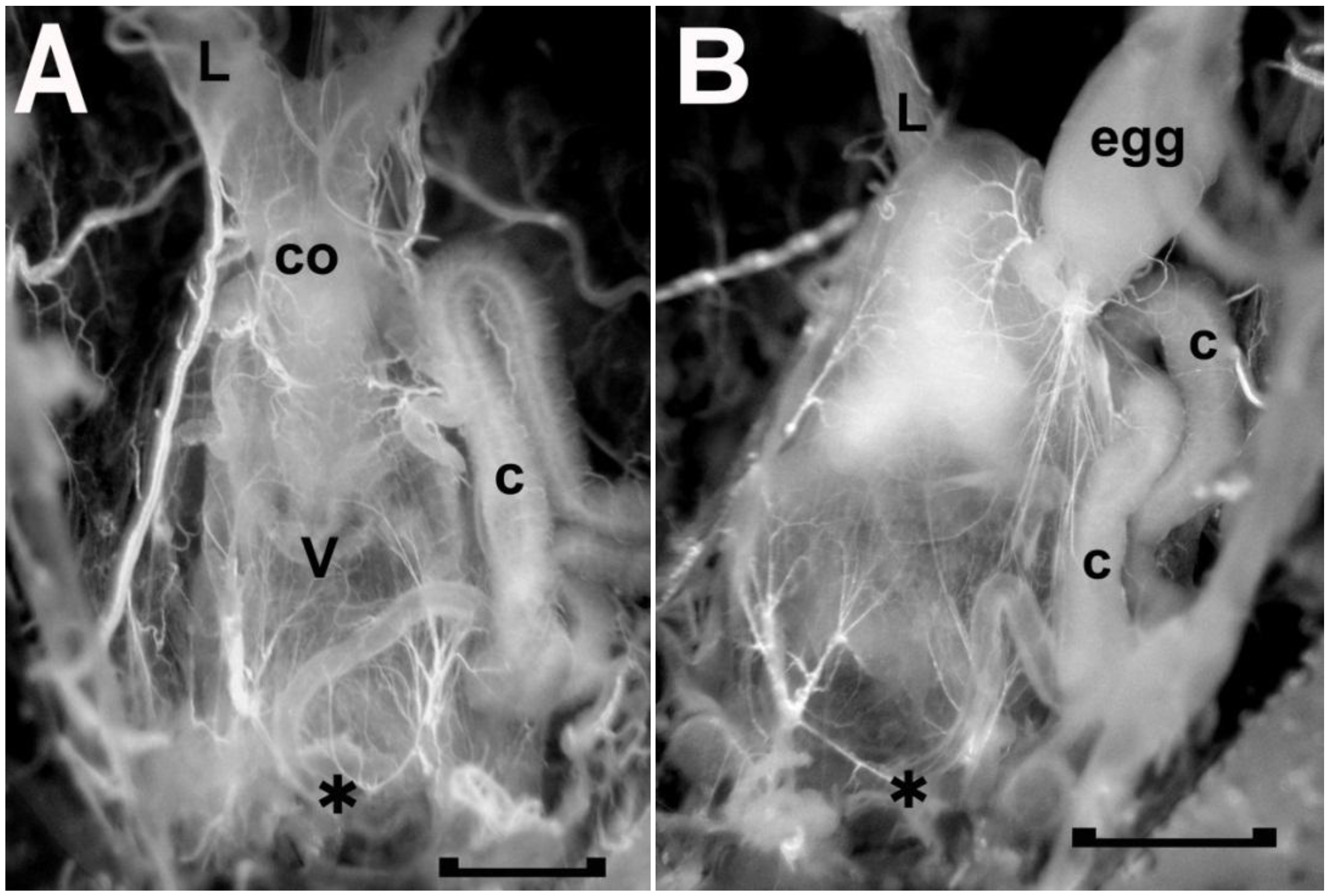
To remove spermatophores from recently copulating females, copulation was visually monitored. It lasted 52 ± 14 min (n = 26), and once the pairs separated, the female reproductive system was exposed and the vagina with the spermatophore removed. A spermataphore in the vagina can be seen through the walls of the vagina (
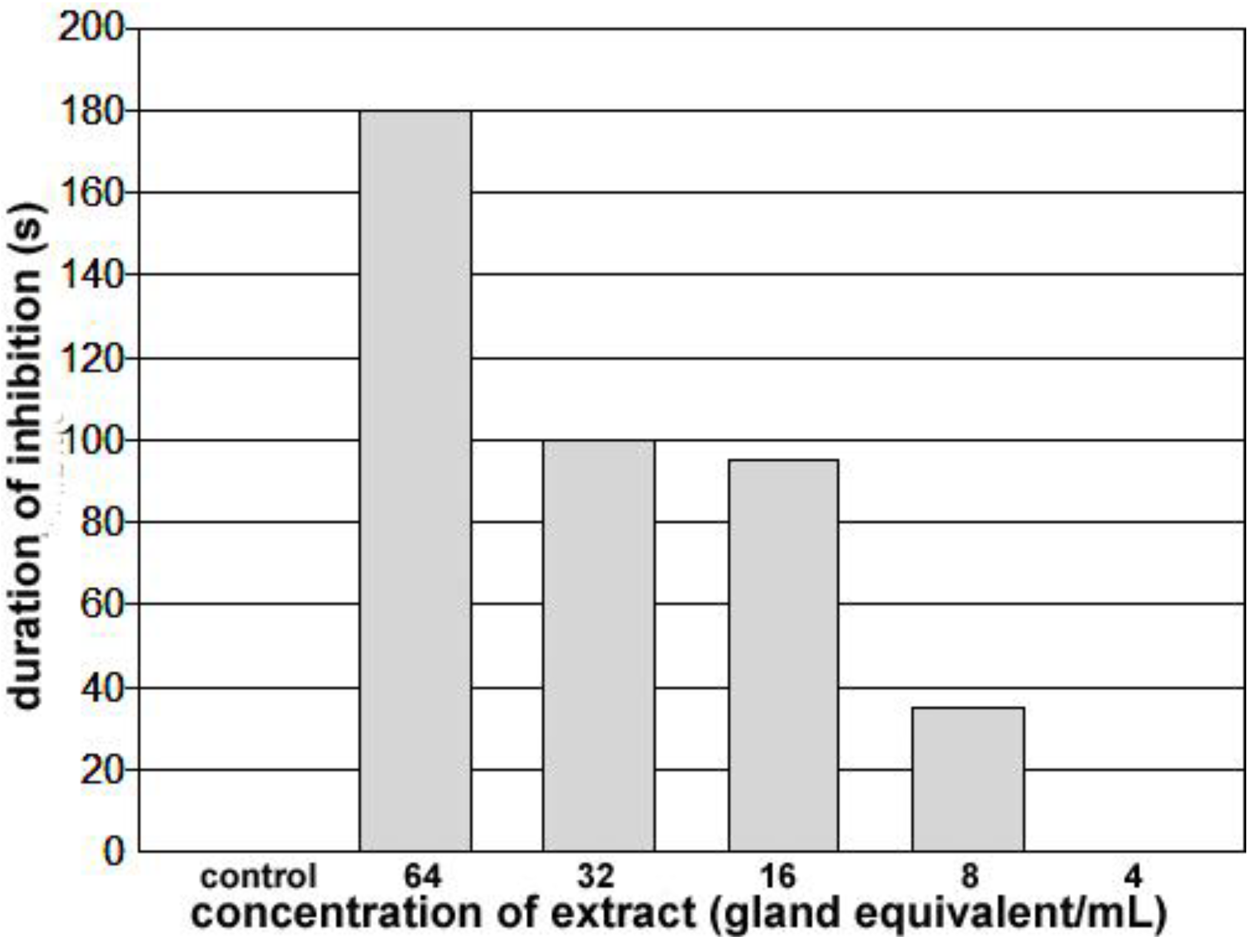
Figure 7B). Being delivered first, the semen and opaque material are present at the anterior end of the vagina next to the common oviduct, and transparent material, delivered last, fills the bulk of this chamber. An extract was made from the spermatophores collected, and concentrations adjusted for testing. The bioassay was carried out on the same bug, and although the concentration effect needs to be verified with further tests, these results do establish the presence of a cardio-inhibitor in the spermatophore. The highest concentration tested (64 spermatophore equivalents/mL) inhibited the heart beyond the 3 min cut-off period, and inhibition was not observed below 8 spermatophore equivalents/mL (see
Figure 8). As with the initial paper describing rhodtestolin [
3], accessory gland extracts were also tested for the same species, but these extracts did not inhibit the heartbeat.
Assuming that the cardio-inhibitor plays an important role during sexual reproduction, a physiologically significant amount must be delivered in a single spermatophore. Although more tests need to be completed to verify the concentration effects described above, it is possible that only one spermatophore equivalent should be needed to inhibit the heartbeat if rhodtestolin’s role in sexual physiology is directly related to heartbeat inhibition. Our concentration effects therefore suggest that rhodtestolin may play a role that functions at lower concentrations. One such role would be to prevent the vaginal muscles from contracting since their contractions would expel the spermatophore from the vagina. The functional anatomy of vaginal muscles has been described previously [
21], showing that they will contract in response to stretch. Since the spermatophore, which stretches the vagina, remains in position for several hours, any stretch reflex associated with the vaginal muscles must be delayed. The role of rhodtestolin on the contractions of the vaginal muscles is currently being investigated.
Figure 7.
The freshly dissected female reproductive tract of two adult Rhodnius prolixus, one without (A) and one with (B) a spermatophore in the vagina (v). As seen in A, the anterior end of the vagina is attached to the common oviduct (co), which, in turn, leads to the two lateral oviducts (L, left lateral oviduct). The presence of a spermatophore, as seen in B, obscures the common oviduct as the spermatophore expands the vagina anteriorly. The spermatophore mainly consists of transparent material with opaque material at the anterior end next to the opening to the common oviduct. C, secretory portion of cement gland; asterisk, vulva and location where the duct of the cement gland secretes the cement onto the passing egg; egg, mature egg in base of right oviduct. Scale bar, 0.5 mm.
Figure 7.
The freshly dissected female reproductive tract of two adult Rhodnius prolixus, one without (A) and one with (B) a spermatophore in the vagina (v). As seen in A, the anterior end of the vagina is attached to the common oviduct (co), which, in turn, leads to the two lateral oviducts (L, left lateral oviduct). The presence of a spermatophore, as seen in B, obscures the common oviduct as the spermatophore expands the vagina anteriorly. The spermatophore mainly consists of transparent material with opaque material at the anterior end next to the opening to the common oviduct. C, secretory portion of cement gland; asterisk, vulva and location where the duct of the cement gland secretes the cement onto the passing egg; egg, mature egg in base of right oviduct. Scale bar, 0.5 mm.
Figure 8.
Duration of inhibition of the heartbeat for spermatophore extracts applied to a heart beating spontaneously at approximately 5 bpm for at least 10 minutes. Application of a 25–50 µL of control saline increased heart rate for approximately 30 s and application of less than 8 gland equivalents/mL did not slow the heartbeat. Application of concentrations of 8 or higher increased the duration of inhibition progressively. At 64 glands equivalents/mL, the heart remained quiet for greater than 3 minutes.
Figure 8.
Duration of inhibition of the heartbeat for spermatophore extracts applied to a heart beating spontaneously at approximately 5 bpm for at least 10 minutes. Application of a 25–50 µL of control saline increased heart rate for approximately 30 s and application of less than 8 gland equivalents/mL did not slow the heartbeat. Application of concentrations of 8 or higher increased the duration of inhibition progressively. At 64 glands equivalents/mL, the heart remained quiet for greater than 3 minutes.