Grooming Behavior as a Mechanism of Insect Disease Defense
Abstract
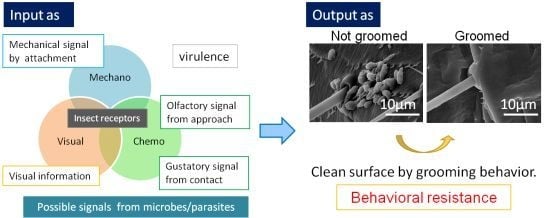
:1. Introduction
2. Function of Grooming
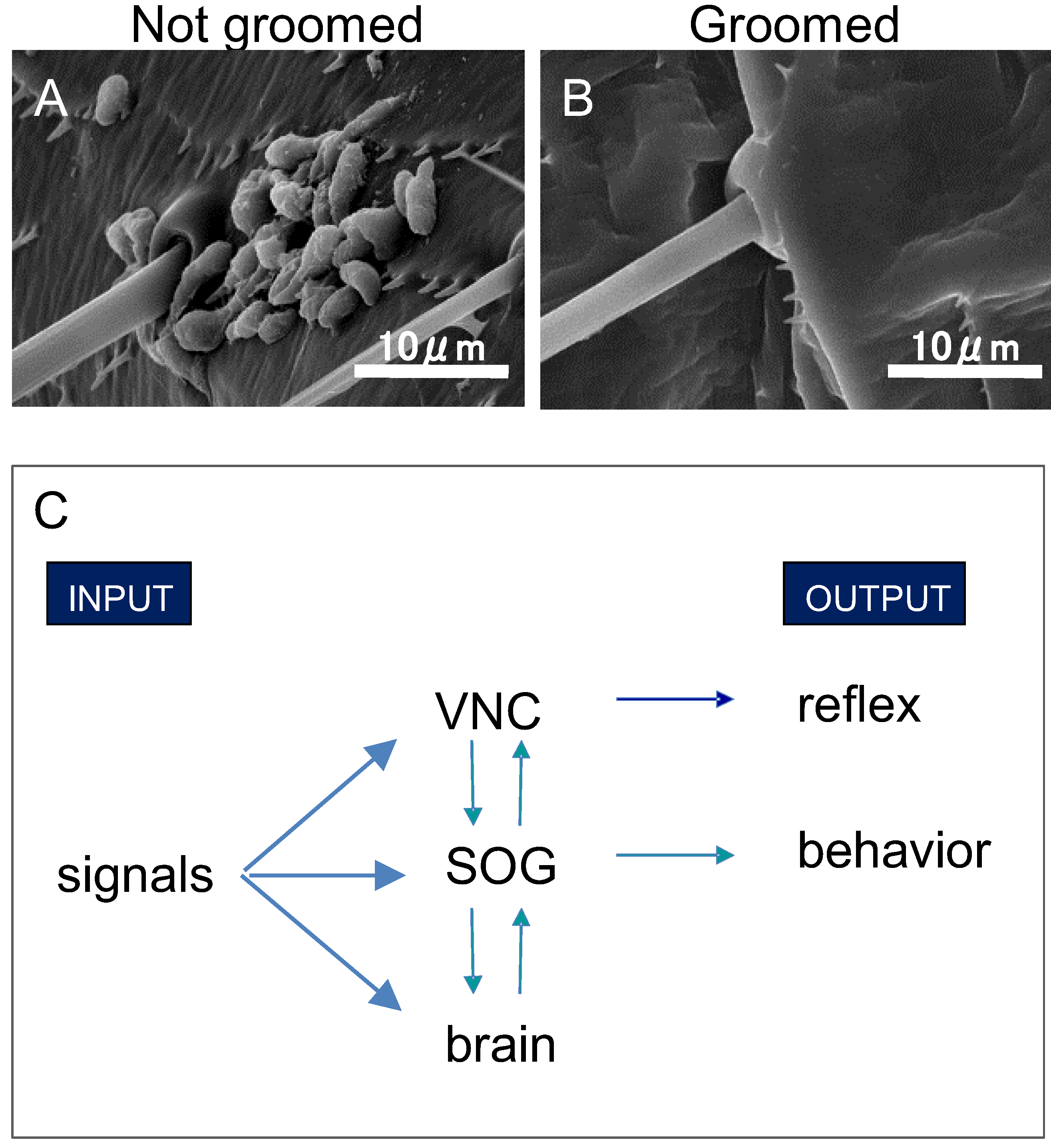
3. Neurobiology of Grooming
4. Chemosensory Signatures
5. Detection of Pathogens

6. Disease Prevention
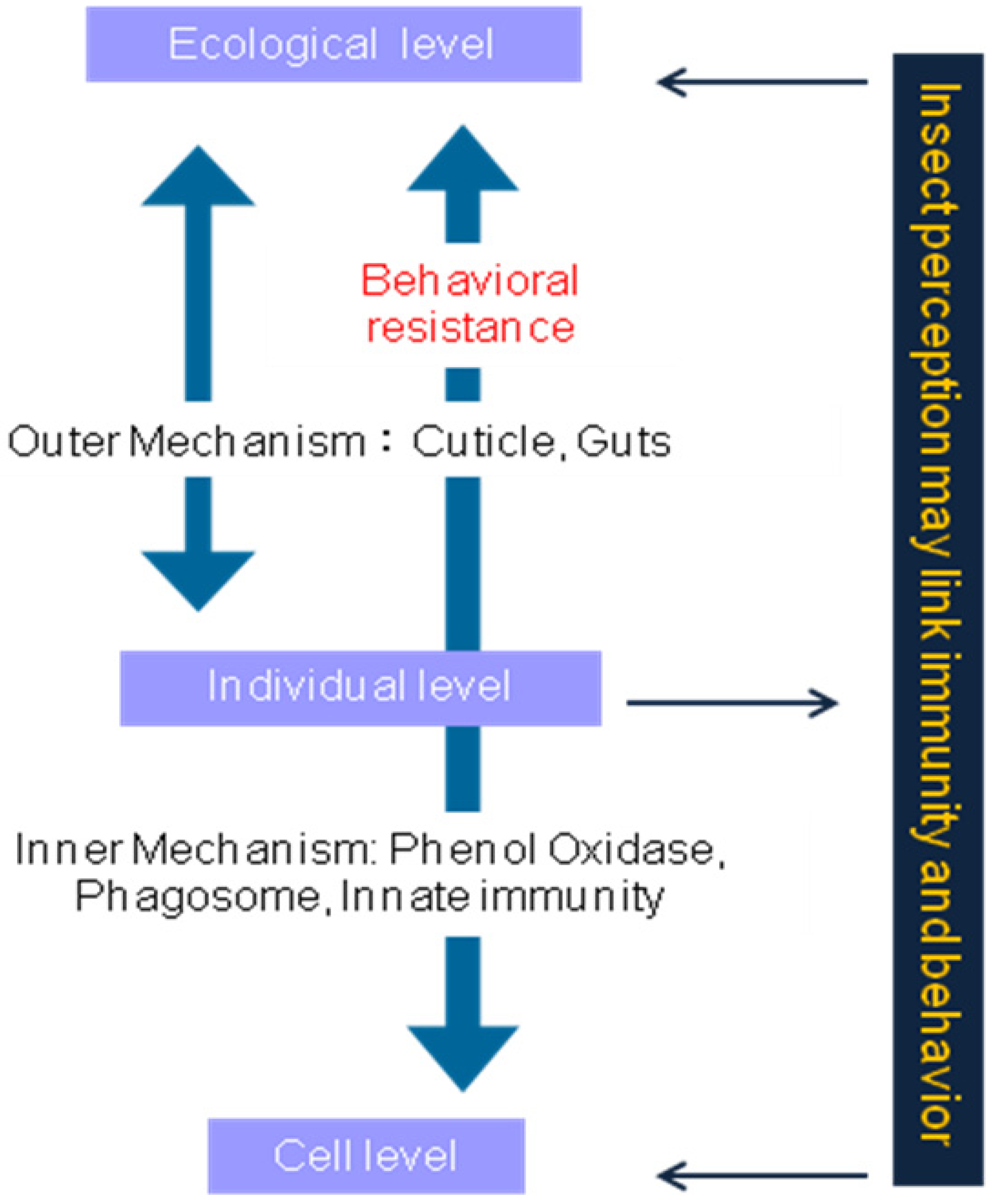
7. Discussion and Conclusions
Acknowledgments
Conflicts of Interest
References
- Mooring, M.S.; Blumstein, D.T.; Stoner, C.J. The evolution of parasite-defence grooming in ungulates. Biol. J. Linn. Soc. 2004, 81, 17–37. [Google Scholar] [CrossRef]
- Sachs, B.D. The development of grooming and its expression in adult animals. Ann. N. Y. Acad. Sci. 1988, 525, 1–17. [Google Scholar] [CrossRef]
- Borchelt, P.L. Care of the Body Surface (COBS). In Comparative Psychology: An Evolutionary Analysis of Animal Behavior; Denny, M.R., Ed.; John Wiley & Sons Inc.: New York, NY, USA, 1980; pp. 363–384. [Google Scholar]
- Kalueff, A.V.; Tuohimaa, P. Grooming analysis algorithm for neurobehavioural stress research. Brain Res. Prot. 2004, 13, 151–158. [Google Scholar] [CrossRef]
- Szebenyi, A.L. Cleaning behaviour in Drosophila melanogaster. Anim. Behav. 1969, 17, 641–651. [Google Scholar] [CrossRef]
- Valentine, B.D. Grooming behavior in Coleoptera. Coleopts. Bull. 1973, 27, 63–73. [Google Scholar]
- Goldman, L.J.; Callahan, P.S.; Carlysle, T.C. Tibial combs and proboscis cleaning in mosquitoes. Annu. Rev. Entomol. Soc. Am. 1972, 65, 1299–1302. [Google Scholar]
- Newland, P.L. Avoidance reflexes mediated by contact chemoreceptors on the legs of locusts. J. Comp. Physiol. A 1998, 183, 313–324. [Google Scholar] [CrossRef]
- Newland, P.L.; Rogers, S.M.; Gaaboub, I.; Matheson, T. Parallel somatotopic maps of gustatory and mechanosensory neurons in the central nervous system of an insect. J. Comp. Neurol. 2000, 425, 82–96. [Google Scholar] [CrossRef]
- Rogers, S.M.; Newland, P.L. Local movements evoked by chemical stimulation of the hind leg in the locust Schistocerca gregaria. J. Exp. Biol. 2000, 203, 423–433. [Google Scholar]
- Dürr, V.; Matheson, T. Graded limb targeting in an insect is caused by the shift of a single movement pattern. J. Neurophysiol. 2003, 90, 1754–1765. [Google Scholar] [CrossRef]
- Page, K.L.; Matheson, T. Wing hair sensilla underlying aimed hindleg scratching of the locust. J. Exp. Biol. 2004, 207, 2691–2703. [Google Scholar] [CrossRef]
- Hlavac, T.F. Grooming systems of insects: Structure, mechanics. Ann. Entomol. Soc. Am. 1975, 68, 823–826. [Google Scholar]
- Schönitzer, K.; Renner, M. The function of the antenna cleaner of the honeybee (Apis mellifica). Apidologie 1984, 15, 23–32. [Google Scholar] [CrossRef]
- Walker, E.D.; Archer, W.E. Sequential organization of grooming behaviors of the mosquito Aedes triseriatus. J. Insect Behav. 1988, 1, 97–109. [Google Scholar] [CrossRef]
- Basibuyuk, H.H.; Quicke, D.L.J. Morphology of the antenna cleaner in the Hymenoptera with particular reference to non-aculeate families (Insecta). Zool. Scr. 1995, 28, 152–177. [Google Scholar]
- Basibuyuk, H.H.; Qjuicke, D.L.J. Grooming behaviours in the Hymenoptera (Insecta): Potential phylogenetic significance. Zool. J. Linn. Soc. 1999, 125, 349–382. [Google Scholar] [CrossRef]
- Hosoda, N.; Gorb, S.N. Friction force reduction triggers feet grooming behavior in beetles. Proc. R. Soc. B 2011, 278, 1748–1752. [Google Scholar] [CrossRef]
- Farish, D.J. The evolutionary implications of qualitative variation in the grooming behavior of the Hymenoptera (Insecta). Anim. Behav. 1972, 20, 662–676. [Google Scholar] [CrossRef]
- Thelen, E.; Farish, D.J. Analysis of grooming behaviour of wild and mutant strains of Brucon hebefor (Braconidae-Hymenoptera). Behaviour 1977, 62, 70–102. [Google Scholar] [CrossRef]
- Valentine, B.D.; Glorioso, M.J. Grooming behavior in Diplura (Insecta: Apterygota). Psyche 1978, 85, 191–200. [Google Scholar] [CrossRef]
- Lefebvre, L. Grooming in crickets: Timing and hierarchical organization. Anim. Behav. 1981, 29, 973–984. [Google Scholar] [CrossRef]
- Smolinsky, A.N.; Bergner, C.L.; LaPorte, J.L.; Kalueff, A.V. Analysis of grooming behavior and its utility in studying animal stress, anxiety, and depression. Neuromethods 2009, 42, 21–36. [Google Scholar] [CrossRef]
- Roy, H.E.; Steinkraus, D.C.; Eilenberg, J.; Hajek, A.E.; Pell, J.K. Bizarre interactions and endgames: Entomopathogenic fungi and their arthropod hosts. Annu. Rev. Entomol. 2006, 51, 331–357. [Google Scholar] [CrossRef]
- Valentine, B.D. Mutual grooming in cucujoid beetles (Coleoptera: Silvanidae). Insecta Mundi. 2007. Paper 54. Available online: http://digitalcommons.unl.edu/insectamundi/54/ (accessed on 8 April 2013).
- Hefetz, A.; Soroker, V.; Dahbi, A.; Malherbe, M.C.; Fresneau, D. The front basitarsal brush in Pachycondyla apicalis and its role in hydrocarbon circulation. Chemoecology 2001, 11, 17–24. [Google Scholar] [CrossRef]
- Root-Bernstein, M. Displacement activities during the honeybee transition from waggle dance to foraging. Anim. Behav. 2010, 79, 935–938. [Google Scholar] [CrossRef]
- Kovac, D.; Maschwitz, U. Secretion-Grooming in aquatic beetles (Hydradephaga): A chemical protection against contamination of the hydrofuge respiratory region. Chemoecology 1990, 1, 131–138. [Google Scholar] [CrossRef]
- Yanagawa, A.; Shimizu, S. Resistance of the termite, Coptotermes formosanus Shiraki to Metarhizium anisopliae due to grooming. BioControl 2007, 52, 75–85. [Google Scholar] [CrossRef]
- Lusebrink, I.; Dettner, K.; Seifert, K. Stenusine, an antimicrobial agent in the rove beetle genus Stenus (Coleoptera, Staphylinidae). Naturwissenschaften 2008, 95, 751–755. [Google Scholar] [CrossRef]
- Peng, Y.S.; Fang, Y.; Xu, S.; Ge, L.; Nasr, M.E. The resistance mechanism of the Asian honey bee, Apis cerana Fabr, to an ectoparasitic mite Varroa jacobsoni Oudemans. J. Invertebr. Pathol. 1987, 49, 54–60. [Google Scholar] [CrossRef]
- Vincent, C.M.; Bertram, S.M. Crickets groom to avoid lethal parasitoids. Anim. Behav. 2010, 79, 51–56. [Google Scholar] [CrossRef]
- Elder, W.H. The oil gland of birds. Wilson Bull. 1954, 66, 6–31. [Google Scholar]
- Jacob, J.; Ziswiler, V. The uropygial gland. J. Avian Biol. 1982, 6, 199–324. [Google Scholar]
- Moyer, B.; Rock, A.N.; Clayton, D.H. Experimental test of the importance of preen oil in rock doves (Columba livia). Auk 2003, 120, 490–496. [Google Scholar] [CrossRef]
- Graystock, P.; Hughes, W.O.H. Disease resistance in a weaver ant, Polyrhachis dives, and the role of antibiotic-producing glands. Behav. Ecol. Sociobiol. 2011, 65, 2319–2327. [Google Scholar] [CrossRef]
- Baracchi, D.; Mazza, G.; Turillazzi, S. From individual to collective immunity: The role of the venom as antimicrobial agent in the Stenogastrinae wasp societies. J. Insect Physiol. 2012, 58, 188–193. [Google Scholar] [CrossRef]
- Gratwick, M. The contamination of insects of different species exposed to dust deposits. Bull. Entomol. Res. 1957, 48, 741–753. [Google Scholar] [CrossRef]
- Reingold, S.C.; Camhi, J.M. Abdominal grooming in the cockroach: Development of an adult behavior. J. Insect Physiol. 1978, 24, 101–110. [Google Scholar] [CrossRef]
- Vandervorst, P.; Ghysen, A. Genetic control of sensory connections in Drosophila. Nature 1980, 86, 65–67. [Google Scholar] [CrossRef]
- El-Awami, I.O.; Dent, D.R. The interaction of surface and dust particle size on the pick-up and grooming behaviour of the German cockroach Blattella germanica. Entomol. Exp. Appl. 1995, 77, 81–87. [Google Scholar] [CrossRef]
- Matheson, T. Hindleg targeting during scratching in the locust. J. Exp. Biol. 1997, 200, 93–100. [Google Scholar]
- Hay, D.A. Genetical and maternal determinants of the activity and preening behaviour of Drosophila melanogaster reared in different environments. Heredity 1972, 28, 311–336. [Google Scholar] [CrossRef]
- Ashton, K.; Wagoner, A.P.; Carrillo, R.; Gibson, G. Quantitative trait loci for the monoamine-related traits heart rate and headless behavior in Drosophila melanogaster. Genetics 2001, 157, 283–294. [Google Scholar]
- Spruijt, B.; van Hooff, J.; Gispen, W. Ethology and neurobiology of grooming behavior. Physiol. Rev. 1992, 72, 825–852. [Google Scholar]
- Eaton, R.C.; Farley, R.D. The neural control of cercal grooming behaviour in the cockroach, Periplaneta americana. J. Insect Physiol. 1969, 15, 1047–1065. [Google Scholar] [CrossRef]
- Berkowitz, A.; Laurent, G. Local Control of Leg Movements and motor patterns during grooming in locusts. J. Neurosci. 1996, 16, 8067–8078. [Google Scholar]
- Canal, I.; Acebes, A.; Ferrus, A. Single neuron mosaics of the Drosophila gigas mutant project beyond normal targets and modify behavior. J. Neurosci. 1998, 18, 999–1008. [Google Scholar]
- Stamp, N.E. Interactions of parasitoids and checkerspot caterpillars Euphydryas spp. (Nymphalidae). J. Res. Lepid. 1984, 23, 2–18. [Google Scholar]
- Hays, D.B.; Vinson, S.B. Acceptance of Heliothis virescens (F.) as a host by the parasite Cardiochiles nigriceps viereck (Hymenoptera, Braconidae). Anim. Behav. 1971, 19, 3–52. [Google Scholar]
- Gross, P. Insect behavioral and morphological defenses against parasitoids. Annu. Rev. Entomol. 1993, 38, 251–273. [Google Scholar] [CrossRef]
- Henderson, A.E.; Hallett, R.H.; Soroka, J.J. Prefeeding behavior of the crucifer flea beetle, Phyllotreta cruciferae, on host and nonhost crucifers. J. Insect Behav. 2004, 17, 17–39. [Google Scholar] [CrossRef]
- Qiu, Y.; van Loon, J.J.A.; Roessingh, P. Chemoreception of oviposition inhibiting terpenoids in the diamondback moth Plutella xylostella. Entomol. Exp. Appl. 1998, 87, 143–155. [Google Scholar]
- Yang, C.-H.; Belawat, P.; Hafen, E.; Jan, L.Y.; Jan, Y.-N. Drosophila egg-laying site selection as a system to study simple decision-making processes. Science 2008, 319, 1679–1683. [Google Scholar] [CrossRef]
- Wuellner, C.T.; Porter, S.D.; Gilbert, L.E. Eclosion, mating, and grooming behavior of the parasitoid fly Pseudacteon curvatus (Diptera: Phoridae). Fla. Entomol. 2002, 85, 563–566. [Google Scholar] [CrossRef]
- Böröczky, K.; Wada-Katsumata, A.; Batchelor, D.; Zhukovskaya, M.; Schal, C. Insects groom their antennae to enhance olfactory acuity. Proc. Natl. Acad. Sci. USA 2013, 110, 3615–3620. [Google Scholar] [CrossRef]
- Jacquet, M.; Lebon, C.; Lemperiere, G.; Boyer, S. Behavioural functions of grooming in male Aedes albopictus (Diptera: Culicidae), the Asian tiger mosquito. Appl. Entomol. Zool. 2012, 47, 359–363. [Google Scholar] [CrossRef]
- Bozic, J.; Valentincic, T. Quantitative analysis of social grooming behavior of the honey bee Apis mellifera carnica. Apidologie 1995, 26, 141–147. [Google Scholar] [CrossRef]
- Robinson, W.H. Antennal Grooming and Movement Behavior in the German Cockroach, Blattella germanica (L.). In Proceedings of the Second International Conference on Urban Pests, Edinburgh, UK, July 1996; pp. 361–369.
- Phillis, R.W.; Bramlage, A.T.; Wotus, C.; Whittaker, A.; Gramates, L.S.; Seppala, D.; Farahanchi, F.; Caruccio, P.; Murphey, R.K. Isolation of mutations affecting neural circuitry required for grooming behavior in Drosophila melanogaster. Genetics 1993, 133, 581–592. [Google Scholar]
- Carlin, N.F.; Holldobler, B.; Gladstein, D.S. The kin recognition system of carpenter ants (Camponotus spp.). Behav. Ecol. Sociobiol. 1986, 20, 219–227. [Google Scholar]
- Ozaki, M.; Wada-Katsumata, A.; Fujikawa, K.; Iwasaki, M.; Yokohari, F.; Satoji, Y.; Nisimura, T.; Yamaoka, R. Ant nestmate and non-nestmate discrimination by a chemosensory sensillum. Science 2005, 309, 311–314. [Google Scholar] [CrossRef]
- Dettner, K.; Liepert, C. Chemical mimicry and camouflage. Ann. Rev. Entomol. 1994, 39, 129–154. [Google Scholar] [CrossRef]
- Seid, M.A.; Brown, B.V. A new host association of Commoptera solenopsidis (Diptera: Phoridae) with the ant Pheidole dentata (Hymenoptera: Formicidae) and behavioral observations. Fla. Entomol. 2009, 92, 309–313. [Google Scholar] [CrossRef]
- Rath, W. Co-Adaptation of Apis cerana Fabr and Varroa jacobsoni Oud. Apidologie 1999, 30, 97–110. [Google Scholar] [CrossRef]
- Boucias, D.G.; Pendland, J.C. Principles of Insect Pathology; Kluwer Academic Publisher: Boston, MA, USA, 1998; p. 537. [Google Scholar]
- Elphick, C.; Dunning, J.B.; John, B. Behaviour. In The Sibley Guide to Bird Life & Behaviour; Elphick, C., Dunning, J.B., Sibley, D., Eds.; Christopher Helm: London, UK, 2001; pp. 58–59. [Google Scholar]
- Honegger, H.-W.; Reif, H.; Müller, W. Sensory mechanisms of eye cleaning behavior in the cricket Gryllus campestris. J. Comp. Physiol. 1979, 129, 247–256. [Google Scholar] [CrossRef]
- Fujimura, K.; Yokohari, F.; Tateda, H. Classification of antennal olfactory receptors of the cockroach, Periplaneta americana L. Zool. Sci. 1991, 8, 243–255. [Google Scholar]
- Zhukovskaya, M.I. Modulation by octopamine of olfactory responses to nonpheromone odorants in the cockroach, Periplaneta americana L. Chem. Senses 2012, 37, 421–429. [Google Scholar] [CrossRef]
- Tinbergen, N. The Study of Instinct; Clarendon: Oxford, UK, 1951; p. 228. [Google Scholar]
- Wilz, K.J. The disinhibition interpretation of the “displacement” activities during courtship in the three-spined stickleback, Gasterosteus aculeatus. Anim. Behav. 1970, 18, 682–687. [Google Scholar] [CrossRef]
- Anselme, P. Abnormal patterns of displacement activities: A review and reinterpretation. Behav. Process. 2008, 79, 48–58. [Google Scholar] [CrossRef]
- File, S.E.; Mabbutt, P.S.; Walker, J.H. Comparison of adaptive responses in familiar and novel environments: Odulatory factors. Ann. N. Y. Acad. Sci. 1988, 525, 69–79. [Google Scholar] [CrossRef]
- David, J.P.; Boyer, S.; Mesneau, A.; Ball, A.; Ranson, H.; Dauphin-Villemant, C. Involvement of cytochrome P450 monooxygenases in the response of mosquito larvae to dietary plant xenobiotics. Insect Biochem. Mol. Biol. 2006, 36, 410–420. [Google Scholar] [CrossRef]
- King-Jones, K.; Horner, M.A.; Lam, G.; Thummel, C.S. The DHR96 nuclear receptor regulates xenobiotic responses in Drosophila. Cell Metab. 2006, 4, 37–48. [Google Scholar] [CrossRef]
- Davenport, A.; Evans, P.D. Stress-Induced changes in the octopamine levels of insect haemo-lymph. Insect Biochem. 1984, 14, 135–143. [Google Scholar] [CrossRef]
- Woodring, J.P.; Meier, O.W.; Rose, R. Effect of development, photoperiod, and stress on octopamine levels in the house cricket, Acheta domesticus. J. Insect Physiol. 1988, 34, 759–765. [Google Scholar] [CrossRef]
- Hirashima, A.; Nagano, T.; Takeya, R.; Eto, M. Effect of larval density on whole-body biogenic amine levels of Tribolium freemani Hinton. Comp. Biochem. Physiol. 1993, 106, 457–461. [Google Scholar]
- Libersat, F.; Pflueger, H.J. Monoamines and the orchestration of behavior. BioScience 2004, 54, 17–25. [Google Scholar] [CrossRef]
- Cox, R.L.; Wilson, W.T. Effects of permethrin on the behavior of individually tagged honey bees, Apis mellifera L. (Hymenoptera, Apidae). Environ. Entomol. 1984, 13, 375–378. [Google Scholar]
- Wiles, J.A.; Jepson, P.C. Sub-Lethal effects of deltamethrin residues on the within-crop behaviour and distribution of Coccinella septempunctata Entomol. Exp. Appl. 1994, 72, 33–45. [Google Scholar] [CrossRef]
- Longley, M.; Jepson, P.C. Effects of honeydew and insecticide residues on the distribution of foraging aphid parasitoids under glasshouse and field conditions. Entomol. Exp. Appl. 1996, 81, 189–198. [Google Scholar] [CrossRef]
- Boucias, D.G.; Stokes, C.; Storey, G.; Pendland, J.C. The effects of imidacloprid on the termite Reticulitermes flavipes and its interaction with the mycopathogen Beauveria bassiana. Pflanzensch. Nachr. Bayer 1996, 49, 103–144. [Google Scholar]
- Neves, P.M.; Alves, S.B. Grooming capacity inhibition in Cornitermes cumulans (Kollar) (Isoptera: Termitidae) inoculated with entomopathogenic fungi and treated with imidacloprid. An. Soc. Entomol. Bras. 2000, 29, 537–545. [Google Scholar] [CrossRef]
- James, R.R.; Xu, J. Mechanisms by which pesticides affect insect immunity. J. Invertebr. Pathol. 2012, 109, 175–182. [Google Scholar] [CrossRef]
- Golenda, C.F.; Forgash, A.J. Grooming behavior in response to fenvalerate treatment in pyrethroid-resistant house flies. Entomol. Exp. Appl. 1986, 40, 169–175. [Google Scholar] [CrossRef]
- Koppenhöfer, A.M.; Grewal, P.S.; Kaya, H.K. Synergism of imidacloprid and entomopathogenic nematodes against white grubs: The mechanism. Entomol. Exp. Appl. 2000, 94, 283–293. [Google Scholar]
- Rosengaus, R.; Traniello, J. Disease susceptibility and the adaptive nature of colony demography in the dampwood termite Zootermopsis angusticollis. Behav. Ecol. Sociobiol. 2001, 50, 546–556. [Google Scholar] [CrossRef]
- Hughes, W.O.H.; Eilenberg, J.; Boomsma, J.J. Trade-Offs in group living: Transmission and disease resistance in leaf-cutting ants. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2002, 269, 1811–1819. [Google Scholar] [CrossRef]
- Hensler, K. Intracellular recordings of neck muscle motoneurones during eye cleaning behaviour of the cricket. J. Exp. Biol. 1986, 120, 153–172. [Google Scholar]
- Phillis, R.; Statton, D.; Caruccio, P.; Murphey, R.K. Mutations in the 8 kDa dynein light chain gene disrupt sensory axon projections in the Drosophila imaginal CNS. Development 1996, 122, 2955–2963. [Google Scholar]
- Reingold, S.C.; Camhi, J.M. A quantitative analysis of rhythmic leg movements during three different behaviors in the cockroach, Periplaneta americana. J. Insect Physiol. 1977, 23, 1407–1420. [Google Scholar] [CrossRef]
- strand, F.; Anderbrant, O.; Jönsson, P. Behaviour of male pine sawflies, Neodiprion sertifer, released downwind from pheromone sources. Entomol. Exp. Appl. 2000, 95, 119–128. [Google Scholar]
- Yanagawa, A.; Yokohari, F.; Shimizu, S. The role of antennae in removing entomopathogenic fungi from cuticle of the termite, Coptotermes formosanus. J. Insect Sci. 2009, 9, 1–9. [Google Scholar] [CrossRef]
- Zhukovskaya, M.I. Odorant-Dependent changes of the antennal surface secretions in the cockroach, Periplaneta americana. Sensornye. Syst. 2011, 25, 78–86. (in Russian). [Google Scholar]
- Eisner, T.; Deyrup, M.; Jacobs, R.; Meinwald, J. Necrodols: Anti-Insectan terpenes from defensive secretion of carrion beetle (Necrodes surinamensis). J. Chem. Ecol. 1986, 12, 1407–1415. [Google Scholar] [CrossRef]
- Dethier, V.G. Sensitivity of the contact chemoreceptors of the blowfly to vapors. Proc. Nat. Acad. Sci. (Wash.) 1972, 69, 2189–2192. [Google Scholar] [CrossRef]
- Städler, E.; Hanson, F.E. Olfactory capabilities of the “gustatory” chemoreceptors of the tobacco hornworm larvae. J. Comp. Physiol. 1975, 104, 97–102. [Google Scholar] [CrossRef]
- Maldonado, H.; Levin, L. Distance estimation and the monocular cleaning reflex in praying mantis. Z. Vergl. Physiol. 1967, 56, 258–267. [Google Scholar] [CrossRef]
- Page, K.L.; Zakotnik, J.; Dürr, V.; Matheson, T. Motor control of aimed limb movements in an insect. J. Neurophysiol. 2008, 99, 484–499. [Google Scholar] [CrossRef]
- Torres, G.; Horowitz, J.M. Activating properties of cocaine and cocaethylene in a behavioral preparation of Drosophila melanogaster. Synapse 1998, 29, 148–161. [Google Scholar] [CrossRef]
- Yellman, C.; Tao, H.; He, B.; Hirsh, J. Conserved and sexually dimorphic behavioral responses to biogenic amines in decapitated Drosophila. Proc. Natl. Acad. Sci. USA 1997, 94, 4131–4136. [Google Scholar] [CrossRef]
- Schaefer, P.L.; Ritzmann, R.E. Descending influences on escape behavior and motor pattern in the cockroach. J. Neurobiol. 2001, 49, 9–28. [Google Scholar] [CrossRef]
- Corfas, G.; Dudai, Y. Habituation and dishabituation of a cleaning reflex in normal and mutant Drosophila. J. Neurosci. 1989, 9, 56–62. [Google Scholar]
- Weisel-Eichler, A.; Haspel, G.; Libersat, F. Venom of a parasitoid wasp induces prolonged grooming in the cockroach. J. Exp. Biol. 1999, 202, 957–964. [Google Scholar]
- Gal, R.; Rosenberg, L.A.; Libersat, F. Parasitoid wasp uses a venom cocktail injected into the brain to manipulate the behavior and metabolism of its cockroach prey. Arch. Insect Biochem. Physiol. 2005, 60, 198–208. [Google Scholar]
- Zack, S. The effects of foreleg amputation on head grooming behaviour in the praying mantis, Sphodromantis lineola. J. Comp. Physiol. 1978, 125, 253–258. [Google Scholar] [CrossRef]
- Campbell, F.L. A new antennal sensillum of Blattella germanica (Dictyoptera: Blattellidae) and its presence in other Blattaria. Ann. Entomol. Soc. Am. 1972, 65, 888–892. [Google Scholar]
- Frings, H.; Frings, M. The loci of contact chemoreceptors in insects. Am. Mid. Nat. 1949, 41, 602–658. [Google Scholar] [CrossRef]
- Mustard, J.A.; Pham, P.M.; Smith, B.H. Modulation of motor behavior by dopamine and the D1-like dopamine receptor AmDOP2 in the honey bee. J. Insect Physiol. 2010, 56, 422–430. [Google Scholar] [CrossRef]
- Fussnecker, B.L.; Smith, B.H.; Mustard, J.A. Octopamine and tyramine influence the behavioral profile of locomotor activity in the honey bee (Apis mellifera). J. Insect Physiol. 2006, 52, 1083–1092. [Google Scholar] [CrossRef]
- Schröder, R.; Hilker, M. The relevance of background odor in resource location by insects: A behavioral approach. BioScience 2008, 58, 308–316. [Google Scholar] [CrossRef]
- Webb, B. Cognition in insects. Phil. Trans. R. Soc. B 2012, 367, 2715–2722. [Google Scholar] [CrossRef]
- Teichert, H.; Dötterl, S.; Frame, D.; Kirejtshuk, A.; Gottsberger, G. A novel pollination mode, saprocantharophily, in Duguetia cadaverica (Annonaceae): A stinkhorn (Phallales) flower mimie. Flora 2012, 207, 522–529. [Google Scholar] [CrossRef]
- Bruce, T.J.A.; Pickett, J.A. Perception of plant volatile blends by herbivorous insects—Finding the right mix. Phytochemistry 2011, 72, 1605–1611. [Google Scholar] [CrossRef]
- Clarkson, J.M.; Charnley, A.K. New insights into the mechanisms of fungal pathogenesis in insects. Trends Microbiol. 1996, 4, 197–203. [Google Scholar] [CrossRef]
- Gripenberg, S.; Mayhew, P.J.; Parnell, M.; Roslin, T. A meta-analysis of preference-performance relationships in phytophagous insects. Ecol. Lett. 2010, 13, 383–393. [Google Scholar]
- Cunningham, J.P. Can mechanism help explain insect host choice? J. Evol. Biol. 2012, 25, 244–251. [Google Scholar] [CrossRef]
- Floyd, M.; Evans, D.A.; Howse, P.E. Electrophysiological and behavioural studies on naturally occurring repellents to Reticultermes lucifugus. J. Insect Physiol. 1976, 22, 697–701. [Google Scholar] [CrossRef]
- Dong, C.; Zhang, J.; Chen, W.; Huang, H.; Hu, Y. Characterization of a newly discovered China variety of Metarhizium anisopliae (M. anisopliae var. dcjhyium) for virulence to termites, isoenzyme, and phylogenic analysis. Microbiol. Res. 2007, 162, 53–61. [Google Scholar] [CrossRef]
- Sun, J.; Fuxa, J.R.; Richter, A.; Ring, D. Interactions of Metarhizium anisopliae and tree-based mulches in repellence and mycoses against Coptotermes formosanus (Isoptera: Rhinotermitidae). Environ. Entomol. 2008, 37, 755–763. [Google Scholar] [CrossRef]
- Carey, A.F.; Carlson, J.R. Insect olfaction from model system to disease control. Proc. Natl. Acad. Sci. USA 2011, 108, 12987–12995. [Google Scholar] [CrossRef]
- Pinho, P.G.D.; Ribeiro, B.; Gonçalves, R.F.; Baptista, P.; Valentão, P.; Seabra, R.M.; Andrade, P.B. Correlation between the pattern volatiles and the overall aroma of wild edible mushrooms. J. Agric. Food Chem. 2008, 56, 1704–1712. [Google Scholar] [CrossRef]
- Mburu, D.M.; Maniania, N.K.; Hassanali, A. Comparison of volatile blends and nucleotides sequences of two Beauveria bassiana isolates of different virulence and repellency towards the termite Macrotermes michealseni. J. Chem. Ecol. 2013, 39, 101–108. [Google Scholar] [CrossRef]
- Pasquinelli, A.E.; Hunter, S.; Bracht, J. MicroRNAs: A developing story. Curr. Opin. Gen. Dev. 2005, 15, 200–205. [Google Scholar] [CrossRef]
- Vreugdenhil, E.; Berezikov, E. Fine-Tuning the brain: microRNAs. Front. Neuroendocrinol. 2010, 31, 128–133. [Google Scholar] [CrossRef]
- Abbott, A.L. Uncovering new functions for MiroRNAs in Caenorhabditis elegans. Curr. Biol. 2011, 21, 668–671. [Google Scholar] [CrossRef]
- Pradel, E.; Zhang, Y.; Pujol, N.; Matsuyama, T.; Bargmann, C.I.; Ewbank, J.J. Detection and avoidance of a natural product from the pathogenic bacterium Serratia marcescens by Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 2007, 104, 2295–2300. [Google Scholar]
- Zhang, Y.; Lu, H.; Bargmann, C.I. Pathogenic bacteria induce aversive olfactory learning in Caenorhabditis elegans. Nature 2005, 438, 179–184. [Google Scholar] [CrossRef]
- Zhang, Y. Neuronal mechanisms of Caenorhabditis elegans and pathogenic bacteria interactions. Curr. Opin. Microbiol. 2008, 11, 257–261. [Google Scholar]
- Song, B.-M.; Faumont, S.; Lackery, S.; Avery, L. Recognition of familiar food activities feeding via an endocrine serotonin signal in Caenorhabditis elegans. eLife 2013, 2, e00329. [Google Scholar] [CrossRef]
- Hendricks, S.J.; Sollars, S.I.; Hill, D.L. Injury-Induced functional plasticity in the peripheral gustatory system. J. Neurosci. 2002, 22, 8607–8613. [Google Scholar]
- Cross-Mellor, S.K.; Hoshooley, J.S.; Kavaliers, M.; Ossenkopp, K.P. Immune activation paired with intraoral sucrose conditions oral rejection. Neuroreport 2004, 15, 2287–2291. [Google Scholar] [CrossRef]
- Cross-Mellor, S.K.; Kavaliers, M.; Ossenkopp, K.-P. Comparing immune activation (lipopolysaccharide) and toxin (lithium chloride)-induced gustatory conditioning: Lipopolysaccharide produces conditioned taste avoidance but not aversion. Behav. Brain Res. 2004, 148, 11–19. [Google Scholar] [CrossRef]
- Cross-Mellor, S.K.; Kavaliers, M.; Ossenkopp, K.-P. The effects of lipopolysaccharide and lithium chloride on the ingestion of a bitter-sweet taste: Comparing intake and palatability. Brain Behav. Immun. 2005, 19, 564–573. [Google Scholar] [CrossRef]
- Pacheco-Lopez, G.; Niemi, M.-B.; Kou, W.; Harting, M.; Fandrey, J.; Schedlowski, M. Neural substrates for behaviorally conditioned immunosuppression in the rat. J. Neurosci. 2005, 25, 2330–2337. [Google Scholar]
- Niemi, M.-B.; Harting, M.; Kou, W.; del Rey, A.; Besedovsky, H.O.; Schedlowski, M.; Pacheco-Lopez, G. Taste-Immunosuppression engram: Reinforcement and extinction. J. Neuroimmunol. 2007, 188, 74–79. [Google Scholar] [CrossRef]
- Pacheco-Lopez, G.; Niemi, M.B.; Engler, H.; Engler, A.; Riether, C.; Doenlen, R.; Espinosa, E.; Oberbeck, R.; Schedlowski, M. Weaken taste-LPS association during endotoxin tolerance. Physiol. Behav. 2008, 93, 261–266. [Google Scholar]
- Vega, F.E.; Kaya, H.K. Insect Pathology, 2nd ed.; Academic Press: San Diego, CA, USA, 2012; p. 508. [Google Scholar]
- Tanada, Y.; Kaya, H.K. Insect Pathology; Academic Press: San Diego, CA, USA, 1993; p. 666. [Google Scholar]
- Lemaitre, B.; Hoffmann, J. The host defense of Drosophila melanogaster. Annu. Rev. Immunol. 2007, 25, 697–743. [Google Scholar] [CrossRef]
- Fouks, B.; Michae, H.; Lattorff, G. Recognition and avoidance of contaminated flowers by foraging bumblebees (Bombus terrestris). PLoS One 2011, 6, e26328. [Google Scholar] [CrossRef]
- Swanson, J.A.I.; Torto, B.; Kells, S.A.; Mesce, K.A.; Tumlinson, J.H.; Spivak, M. Odorants that induce hygenic behaviour in honeybees: Identification of volatile compounds in chalkbrood-infected honeybee larvae. J. Chem. Ecol. 2009, 35, 1108–1116. [Google Scholar] [CrossRef]
- Ugelvig, L.V.; Kronauer, D.J.C.; Schrempf, A.; Heinze, J.; Cremer, S. Rapid anti-pathogen response in ant societies relies on high genetic diversity. Proc. R. Soc. B 2010, 277, 2821–2828. [Google Scholar] [CrossRef]
- Scharf, I.; Modlmeier, A.P.; Beros, S.; Foitzik, S. Ant societies buffer individual-level effects of parasite infections. Am. Nat. 2012, 180, 671–683. [Google Scholar] [CrossRef]
- Tragust, S.; Mitteregger, B.; Barone, V.; Konrad, M.; Ugelvig, L.V.; Cremer, S. Ants disinfect fungus-exposed brood by oral uptake and spread of their poison. Curr. Biol. 2013, 23, 76–82. [Google Scholar] [CrossRef]
- Nielsen, C.; Anurag, A.; Agrawal, A.A.; Hajek, A.E. Ants defend aphids against lethal disease. Biol. Lett. 2010, 23, 205–208. [Google Scholar]
- Schoonhoven, L.M.; van Loon, J.J.A. An inventory of taste in caterpillars: Each species its own key. Acta Zool. Acad. Sci. Hung. 2002, 48, 215–263. [Google Scholar]
- Chapman, R.F. Contact chemoreception in feeding by phytophagous insects. Annu. Rev. Entomol. 2003, 48, 455–484. [Google Scholar] [CrossRef]
- Dahanukar, A.; Hallem, E.A.; Carlson, J.R. Insect chemoreception. Curr. Opin. Neurobiol. 2005, 15, 423–430. [Google Scholar] [CrossRef]
- Hallem, E.A.; Dahanukar, A.; Carlson, J.R. Insect odor and taste receptors. Annu. Rev. Entomol. 2006, 51, 113–135. [Google Scholar] [CrossRef]
- Clark, A.G.; Eisen, M.B.; Smith, D.R.; Bergman, C.M.; Oliver, B.; Markow, T.A.; Kaufman, T.C.; Kellis, M.; Gelbart, W.; Iyer, V.N.; et al. Evolution of genes and genomes on the Drosophila phylogeny. Nature 2007, 450, 203–218. [Google Scholar] [CrossRef]
- McBride, C.S. Rapid evolution of smell and taste receptor genes during host specialization in Drosophila sechellia. Proc. Natl. Acad. Sci. USA 2007, 104, 4996–5001. [Google Scholar] [CrossRef]
- McBride, C.S.; Arguello, J.R. Five drosophila genomes reveal nonneutral evolution and the signature of host specialization in the chemoreceptor superfamily. Genetics 2007, 177, 1395–1416. [Google Scholar] [CrossRef]
- Steiner, S.; Erdmann, D.; Steidle, J.; Ruther, J. Host habitat assessment by a parasitoid using fungal volatiles. Front. Zool. 2007, 4, 3. [Google Scholar] [CrossRef]
- De Bruyne, M.; Baker, T. Odor detection in insects: Volatile codes. J. Chem. Ecol. 2008, 34, 882–897. [Google Scholar] [CrossRef]
- Bartholomay, L.C.; Cho, W.L.; Rocheleau, T.A.; Boyle, J.P.; Beck, E.T.; Fuchs, J.F.; Liss, P.; Rusch, M.; Butler, K.M.; Wu, R.C.C.; et al. escription of the transcriptomes of immune response-activated Hemocytes from the mosquito vectors Aedes aegypti and Armigeres subalbatus. Infect. Immun. 2004, 72, 4114–4126. [Google Scholar] [CrossRef]
- Aguilar, R.; Jedlicka, A.E.; Mintz, M.; Mahairaki, V.; Scott, A.L.; Dimopoulos, G. Global gene expression analysis of Anopheles gambiae responses to microbial challenge. Insect Biochem. Mol. Biol. 2005, 35, 709–719. [Google Scholar] [CrossRef]
- Myles, T.G. Alarm, aggregation, and defense by Reticulitermes flavipes in response to a naturally occurring isolate of Metarhizium anisopliae. Sociobiology 2002, 40, 243–255. [Google Scholar]
- Mburu, D.M.; Ochola, L.; Maniania, N.K.; Njagi, P.G.N.; Gitonga, L.M.; Ndung’u, M.W.; Wanjoya, A.K.; Hassanali, A. Relationship between virulence and repellency of entomopathogenic isolates of Metarhizium anisopliae and Beauveria bassiana to the termite Macrotermes michaelseni. J. Insect Physiol. 2009, 55, 774–780. [Google Scholar] [CrossRef]
- Ennis, D.E.; Dillon, A.B.; Griffin, C.T. Pine weevils modulate defensive behavior in response to parasites of different virulence. Anim. Behav. 2010, 80, 283–288. [Google Scholar] [CrossRef]
- Yanagawa, A.; Yokohari, F.; Shimizu, S. Influence of fungal odor on grooming behavior of the termite, Coptotermes formosanus Shiraki. J. Insect Sci. 2010, 10, 141. [Google Scholar]
- Yanagawa, A.; Fujiwara-Tsujii, N.; Akino, T.; Yoshimura, T.; Yanagawa, T.; Shimizu, S. Behavioral changes in the termite, Coptotermes formosanus (Isoptera), inoculated with six fungal isolates. J. Invertebr. Pathol. 2011, 107, 100–106. [Google Scholar] [CrossRef]
- Yanagawa, A.; Fujiwara-Tsujii, N.; Akino, T.; Yoshimura, T.; Yanagawa, T.; Shimizu, S. Musty odor of entomopathogens enhances disease-prevention behaviors in the termite Coptotermes formosanus. J. Invertebr. Pathol. 2011, 108, 1–6. [Google Scholar]
- Hesketh, H.; Roy, H.E.; Eilenberg, J.; Pell, J.K.; Hails, R.S. Challenges in modelling complexity of fungal entomopathogens in semi-natural populations of insects. BioControl 2010, 55, 55–73. [Google Scholar] [CrossRef]
- Jackson, M.A.; Dunlop, C.A.; Jaronski, A.T. Ecological considerations in producing and formulating fungal entomopathogen for use in insect biocontrol. BioControl 2010, 55, 129–145. [Google Scholar] [CrossRef]
- Gendrin, M.; Welchman, D.P.; Poidevin, M.; Herve, M.; Lemaitre, B. Long-Range activation of systemic immunity through peptidoglycan diffusion in Drosophila. PLoS Pathog. 2009, 5, e1000694. [Google Scholar] [CrossRef]
- Bulmer, M.S.; Bachelet, I.; Raman, R.; Rosengaus, R.B.; Sasisekharan, R. Targeting an antimicrobial effector function in insect immunity as a pest control strategy. Proc. Natl. Acad. Sci. USA 2009, 106, 12652–12657. [Google Scholar]
- Hauton, C.; Smith, V.J. Adaptive immunity in invertebrates: A straw house without a mechanistic foundation. BioEssays 2007, 29, 1138–1146. [Google Scholar] [CrossRef]
- Chou, P.-H.; Chang, H.-S.; Chen, I.-T.; Lin, H.-Y.; Chen, Y.-M.; Yang, H.-L.; Wang, K.C.H.-C. The putative invertebrate adaptive immune protein Litopenaeus vannamei Dscam (LvDscam) is the first reported Dscam to lack a transmembrane domain and cytoplasmic tail. Dev. Comp. Immunol. 2009, 33, 1258–1267. [Google Scholar] [CrossRef]
- Arala-Chaves, M.; Sequeira, T. Is there any kind of adaptive immunity in invertebrates? Aquaculture 2000, 191, 247–258. [Google Scholar] [CrossRef]
- Kurtz, J.; Armitage, S.A.O. Alternative adaptive immunity in invertebrates. Trends Immunol. 2006, 27, 493–496. [Google Scholar] [CrossRef]
- Walker, T.N.; Hughes, W.O.H. Adaptive social immunity in leaf-cutting ants. Biol. Lett. 2009, 5, 446–448. [Google Scholar] [CrossRef]
- Yek, S.H.; Boomsma, J.J.; Schiøtt, M. Differential gene expression in Acromyrmex leaf-cutting ants after challenges with two fungal pathogens. Mol. Ecol. 2013, 22, 2173–2187. [Google Scholar] [CrossRef]
- Cotter, S.C.; Kilner, R.M. Personal immunity versus social immunity. Behav. Ecol. 2010, 21, 663–668. [Google Scholar] [CrossRef]
- Cremer, S.; Armitage, S.A.O.; Schmid-Hempel, P. Social immunity. Curr. Biol. 2007, 17, R693–R702. [Google Scholar] [CrossRef]
- Cremer, S.; Sixt, M. Analogies in the evolution of individual and social immunity. Phil. Trans. R. Soc. B 2009, 364, 129–142. [Google Scholar] [CrossRef]
- Oi, D.H.; Pereira, R.M. Ant behaviour and microbial pathogens (Hymenoptera: Formicidae). Fla. Entomol. 1993, 76, 63–75. [Google Scholar] [CrossRef]
- Yanagawa, A.; Shimizu, S. Defense strategy of the termite, Coptotermes formosanus Shiraki to entomopathogenic fungi. Jpn. J. Environ. Entomol. Zool. 2005, 16, 17–22. [Google Scholar]
- Galvanho, J.P.; Carrera, M.P.; Moreira, D.O.; Erthal, M., Jr.; Silva, C.P.; Samuels, R.I. Imidacloprid inhibits behavioral defences of the leaf-cutting ant Acromyrmex subterraneus subterraneus (Hymenoptera: Formicidae). J. Insect. Behav. 2013, 26, 1–13. [Google Scholar] [CrossRef]
- Kramm, K.R.; West, D.F. Termite pathogens: Effects of ingested Metarhizium, Beauveria, and Gliocladium conidia on worker termite (Reticulitermes sp.). J. Invertebr. Pathol. 1982, 40, 7–11. [Google Scholar] [CrossRef]
- Shimizu, S.; Yamaji, M. Effect of density of the termite, Reticulitermes speratus Kolbe (Isoptera: Rhinotermitidae), on the susceptibilities to Metarhizium anisoplia. Appl. Entomol. Zool. 2003, 38, 125–130. [Google Scholar] [CrossRef]
- Rohlfs, M. Clash of kingdoms or why Drosophila larvae positively respond to fungal competitors. Front. Zool. 2005, 2. [Google Scholar] [CrossRef]
- Aubert, A.; Richard, F.J. Social management of LPS-induced inflammation in Formica polyctena ants. Brain Behav. Immun. 2008, 22, 833–837. [Google Scholar] [CrossRef]
- Wilson-Rich, N.; Spivak, M.; Fefferman, N.H.; Starks, P.T. Genetic, individual, and group facilitation of disease resistance in insect societies. Annu. Rev. Entomol. 2009, 54, 405–423. [Google Scholar] [CrossRef]
- Libersat, F.; Delago, A.; Gal, R. Manipulation of host behavior by parasitic insects and insect parasites. Annu. Rev. Entomol. 2009, 54, 189–207. [Google Scholar] [CrossRef]
- Rohlfs, M.; Obmann, B.; Petersen, R. Competition with filamentous fungi and its implication for a gregarious lifestyle in insects living on ephemeral resources. Ecol. Entomol. 2005, 30, 556–563. [Google Scholar] [CrossRef]
- Hodson, A.K.; Friedman, M.L.; Wu, L.N.; Lewis, E.E. European earwig (Forficula auricularia) as a novel host for the entomopathogenic nematode Steinernema carpocapsae. J. Invertebr. Pathol. 2011, 107, 60–64. [Google Scholar] [CrossRef]
- Wang, Y.; Campbell, J.F.; Gaugler, R. Infection of entomopathogenic nematodes Steinernema glaseri and Heterorhabditis bacteriophora against Popillia japonica (Coleoptera: Scarabaeidae) larvae. J. Invertebr. Pathol. 1995, 66, 178–184. [Google Scholar] [CrossRef]
- Rieger, D.; Fraunholz, C.; Popp, J.; Bichler, D.; Dittmann, R.; Helfrich-Forster, C. The fruit fly Drosophila melanogaster favors dim light and times its activity peaks to early dawn and late dusk. J. Biol. Rhythm. 2007, 22, 387–399. [Google Scholar] [CrossRef]
- Thompson, G.J.; Crozier, Y.C.; Crozier, R.H. Isolation and characterization of a termite transferrin gene up-regulated on infection. Insect Mol. Biol. 2003, 12, 1–7. [Google Scholar] [CrossRef]
- Lemaitre, B.; Reichhart, J.M.; Hoffmann, J.A. Drosophila host defense: Differential induction of antimicrobial peptide genes after infection by various classes of microorganisms. Proc. Natl. Acad. Sci. USA 1997, 94, 14614–14619. [Google Scholar] [CrossRef]
- Gottar, M.; Gobert, V.; Matskevich, A.A.; Reichhart, J.M.; Wang, C.S.; Buft, T.M.; BeIvin, M.; Hoffmann, J.A.; Ferrandon, D. Dual detection of fungal infections in Drosophila via recognition of glucans and sensing of virulence factors. Cell 2006, 127, 1425–1437. [Google Scholar] [CrossRef]
- Alexander, R.D. The evolution of social behavior. Annu. Rev. Ecol. Syst. 1974, 5, 324–383. [Google Scholar]
- Yoshimura, T.; Takahashi, M. Termiticidal performance of an entomogenous fungus, Beauveria brongniartii (Saccardo) Petch in laboratory tests. Jpn. J. Environ. Entomol. Zool. 1998, 9, 16–22. [Google Scholar]
- Wilson, K.; Knell, R.; Boots, M.; Koch-Osborne, J. Group living and investment in immune defense: An interspecific analysis. J. Anim. Ecol. 2003, 72, 133–143. [Google Scholar] [CrossRef]
- Rosengaus, R.B.; Jordan, C.; Lefebvre, M.L.; Traniello, J.F.A. Pathogen alarm behavior in termite: A new form of communication in social insects. Naturwissenschaften 1999, 86, 544–548. [Google Scholar] [CrossRef]
- Ugelvig, L.V.; Cremer, S. Social prophylaxis: Group interaction promotes collective immunity in ant colonies. Curr. Biol. 2007, 17, 1967–1971. [Google Scholar] [CrossRef]
- Fefferman, N.H.; Traniello, J.F.A.; Rosengaus, R.B.; Calleri, D.V., II. Disease prevention and resistance in social insects: Modeling the survival consequences of immunity, hygienic behavior, and colony organization. Behav. Ecol. Sociobiol. 2007, 61, 565–577. [Google Scholar] [CrossRef]
- Radford, A.N. Post-Allogrooming reductions in self-directed behaviour are affect by role and status in the green woodhoopoe. Biol. Lett. 2012, 8, 24–27. [Google Scholar] [CrossRef]
- Rosengaus, R.B.; Maxmen, A.B.; Coates, L.E.; Traniello, J.F.A. Disease resistance: A benefit of sociality in the dampwood termite Zootermopsis angusticollis (Isoptera: Termopsidae). Behav. Ecol. Sociobiol. 1998, 44, 125–134. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.-Y.; Robert, A. Inhibition of Metarhizium anisopliae in the alimentary tract of the eastern subterranean termite Reticulitermes flavipes. J. Invertebr. Pathol. 2009, 101, 130–136. [Google Scholar] [CrossRef]
- Okuno, M.; Tsuji, K.; Sato, H.; Fujisaki, K. Plasticity of grooming behavior against entomopathogenic fungus Metarhizium anisopliae in the ant Lasius japonicas. J. Ethol. 2012, 30, 23–27. [Google Scholar] [CrossRef]
- Little, A.E.F.; Murakami, T.; Mueller, U.G.; Currie, C.R. Defending against parasites: Fungus-Growing ants combine specialized behaviours and microbial symbionts to protect their fungus garden. Biol. Lett. 2006, 2, 12–16. [Google Scholar] [CrossRef]
- Fernandez-Marin, H.; Zimmerman, J.; Rehner, S.; Wcislo, W. Active use of the metapleural glands by ants in controlling fungal infection. Proc. R. Soc. Lond. B 2006, 273, 1689–1695. [Google Scholar] [CrossRef]
- Beattie, A.J.; Turnbull, C.L.; Hough, T.; Knox, R.B. Antibiotic production—A possible function for the metapleural glands of ants (Hymenoptera, Formicidae). Ann. Entomol. Soc. Am. 1986, 79, 448–450. [Google Scholar]
- Bot, A.N.M.; Obermayer, M.L.; Holldobler, B.; Boomsma, J.J. Functional morphology of the metapeural gland in the leaf-cutting ant Acromyrmex octospinosus. Insect Soc. 2001, 48, 63–66. [Google Scholar] [CrossRef]
- Hölldobler, B.; Wilson, E.O. The Ants; Belknap Press: Cambridge, MA, USA, 1990; p. 746. [Google Scholar]
- Mackintosh, J.A.; Trimble, J.E.; Jones, M.K.; Karuso, P.H.; Beattie, A.J.; Veal, D.A. Antimicrobial mode of action of secretions from the metapleural gland of Myrmecia gulosa (Australian bull ants). Can. J. Microbiol. 1995, 41, 136–144. [Google Scholar] [CrossRef]
- Schlüns, H.; Crozier, R.H. Molecular and chemical immune defenses in ants (Hymenoptera: Formicidae). Myrmecol. News 2009, 12, 237–249. [Google Scholar]
- Veal, D.A.; Trimble, J.E.; Beattie, A.J. Antimicrobial properties of secretions from the metapleural glands of Myrmecia gulosa (The Australian bull ants). J. Appl. Bacteriol. 1992, 72, 188–194. [Google Scholar] [CrossRef]
- Moretto, G.; Gonçalves, L.S.; de Jong, D. Heritability Africanized and European honey bee defensive behavior against the mite Varroa jacobsoni. Braz. J. Genet. 1993, 16, 71–77. [Google Scholar]
- Büchler, R. Rate of damaged mites in natural mite fall with regard to seasonal effects and infestation development. Apidologie 1993, 24, 492–493. [Google Scholar]
- Büchler, R. Design and success of a German breeding program for Varroa tolerance. Am. Bee J. 2000, 140, 662–665. [Google Scholar]
- Bienefeld, K.; Zautkea, F.; Proninb, D.; Mazeedc, A. Recording the proportion of damaged Varroa jacobsoni Oud. in the debris of honey bee colonies (Apis mellifera). Apidologie 1999, 30, 249–256. [Google Scholar] [CrossRef]
- Hoffman, S. The occurrence of damaged mites in cage test and under field conditions in hybrids of different carniolan lines. Apidologie 1993, 24, 493–495. [Google Scholar]
- Rosenkranz, P.; Fries, I.; Boecking, O.; Stürmer, M. Damaged Varroa mites in the debris of honey bee (Apis mellifera L.) colonies with and without hatching brood. Apidologie 1997, 28, 427–437. [Google Scholar] [CrossRef]
- Arechavaleta-Velasco, M.E.; Guzman-Novoa, E. Relative effect of four characteristics that restrain the population growth of the mite Varroa destructor in honey bee (Apis mellifera) colonies. Apidologie 2001, 32, 157–174. [Google Scholar] [CrossRef]
- Stanimirovic, Z.; Stevanovic, J.; Cirkovic, D. Behavioural defenses of the honey bee ecotype from Sjenica-Pester against Varroa destructor. Acta Vet. 2005, 55, 69–82. [Google Scholar] [CrossRef]
- Roode, J.C.; Lefévre, T. Behavioral immunity in insects. Insects 2012, 3, 789–820. [Google Scholar] [CrossRef]
- Clemente, C.J.; Bullock, J.M.R.; Beale, A.; Federle, W. Evidence-Self-Cleaning in fluid-based smooth and hairy adhesive systems of insects. J. Exp. Biol. 2009, 213, 635–642. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhukovskaya, M.; Yanagawa, A.; Forschler, B.T. Grooming Behavior as a Mechanism of Insect Disease Defense. Insects 2013, 4, 609-630. https://doi.org/10.3390/insects4040609
Zhukovskaya M, Yanagawa A, Forschler BT. Grooming Behavior as a Mechanism of Insect Disease Defense. Insects. 2013; 4(4):609-630. https://doi.org/10.3390/insects4040609
Chicago/Turabian StyleZhukovskaya, Marianna, Aya Yanagawa, and Brian T. Forschler. 2013. "Grooming Behavior as a Mechanism of Insect Disease Defense" Insects 4, no. 4: 609-630. https://doi.org/10.3390/insects4040609
APA StyleZhukovskaya, M., Yanagawa, A., & Forschler, B. T. (2013). Grooming Behavior as a Mechanism of Insect Disease Defense. Insects, 4(4), 609-630. https://doi.org/10.3390/insects4040609