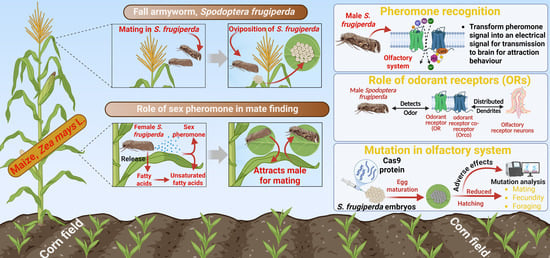
Exploring the Role of Pheromones and CRISPR/Cas9 in the Behavioral and Olfactory Mechanisms of Spodoptera frugiperda
Simple Summary
Abstract
1. Introduction
2. Pheromone Dynamics and Recognition in S. frugiperda
2.1. Biosynthesis of Sexual Pheromone in S. frugiperda
2.2. Factors Affecting Sex Pheromone Release from Female S. frugiperda

2.2.1. Adult Female Age
2.2.2. Mating Status
2.3. Mechanism of Pheromone Recognition by Male S. frugiperda
Dose-Dependent Response of Male S. frugiperda to Sex Pheromones

3. Mechanisms of CRISPR/CAS Editing in Insects
3.1. Basic Mechanism of CRISPR/Cas System
3.2. Application in Insects
4. CRISPR/Cas-Mediated Disruption of the Olfactory System in Spodoptera frugiperda
4.1. Identification and Function of Key Olfactory Genes in Spodoptera frugiperda
4.2. Practice and Effects of CRISPR/Cas9-Mediated Orco Knockout


5. Effects of Orco Knockout on Host Plant Volatile Perception in Spodoptera frugiperda
6. Self-Limiting Spodoptera frugiperda: A Genetic Control Strategy
7. Practical Advantages and Limitations of CRISPR/Cas9
8. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAT | Alcohol acetyltransferase |
| ACC | Acetyl-CoA carboxylase |
| BE | Base editors |
| C16:0-CoA | Saturated 16-carbon fatty acyl-CoA |
| Cas12a | CRISPR-associated protein 12a |
| Cas12b | CRISPR-associated protein 12b |
| Cas13 | CRISPR-associated protein 13 |
| Cas7–11 | CRISPR-associated protein 7–11 |
| Cas9 | CRISPR-associated protein 9 |
| CRISPR-Cas9 | Clustered Regularly Interspaced Short Palindromic Repeats |
| CTNBio | Comissão Técnica Nacional de Biossegurança |
| CTNBio | Comissão Técnica Nacional de Biossegurança |
| DNA | Deoxyribonucleic acid |
| DsRed2 | Discosoma red fluorescent protein 2 |
| E7–12:OAc | (E)-7-dodecenyl acetate |
| FARs | Fatty acyl CoA reductases |
| FAS | Fatty acid synthase |
| HDR | Homology-directed repair |
| MAG | Male’s accessory gland |
| MNs | Meganucleases |
| mRNA | Messenger RNA |
| NHEJ | Non-homologous end joining |
| Orco | Odorant receptor co-receptor |
| ORNs | Olfactory receptor neurons |
| ORs | Odorant receptors |
| PAM | Protospacer adjacent motif |
| PBAN | Pheromone biosynthesis-activating neuropeptide |
| PRs | Pheromone receptors |
| qPCR | Quantitative polymerase chain reaction |
| RNA | Ribonucleic acid |
| SDNs | Site-directed nucleases |
| Sfabd-A | Spodoptera frugiperda abdominal-A homeotic |
| SfHsp20.15 | Spodoptera frugiperda heatshock protein 20.15 |
| SfHsp20.71 | Spodoptera frugiperda heatshock protein 20.71 |
| SfHsp20D | Spodoptera frugiperda heatshock protein 20D |
| SfHsp70D | Spodoptera frugiperda heatshock protein 70D |
| SfruOR11 | Spodoptera frugiperda odorant receptor 11 |
| SfruOR13 | Spodoptera frugiperda odorant receptor 13 |
| SfruOR16 | Spodoptera frugiperda odorant receptor 16 |
| SfruOR18 | Spodoptera frugiperda odorant receptor 18 |
| SfruOR22 | Spodoptera frugiperda odorant receptor 22 |
| SfruOR23 | Spodoptera frugiperda odorant receptor 23 |
| SfruOR30 | Spodoptera frugiperda odorant receptor 30 |
| SfruOR36 | Spodoptera frugiperda odorant receptor 36 |
| SfruOR38 | Spodoptera frugiperda odorant receptor 38 |
| SfruOR41 | Spodoptera frugiperda odorant receptor 41 |
| SfruOR49 | Spodoptera frugiperda odorant receptor 49 |
| SfruOR5 | Spodoptera frugiperda odorant receptor 5 |
| SfruOR52 | Spodoptera frugiperda odorant receptor 52 |
| SfruOR56 | Spodoptera frugiperda odorant receptor 56 |
| SfruOR6 | Spodoptera frugiperda odorant receptor 6 |
| SfruOR62 | Spodoptera frugiperda odorant receptor 62 |
| sgRNA | Single-guide RNA |
| SpCas9-HF1 | Streptococcus pyogenes Cas9-High-fidelity variant 1 |
| TALENs | Transcription activator-like effector nucleases |
| tTAV | Tetracycline repressor protein |
| Z11–16-CoA | (Z)-11-hexadecenoyl-CoA |
| Z7–12:CoA | (Z)-7-dodecenyl acetate |
| Z7–12:OAc | (Z)-7-dodecenyl acetate |
| Z9–12:OAc | (Z)-9-dodecenyl acetate |
| Z9–14:OAc | (Z)-9-tetradecenyl acetate |
| Z9–14:OAc | (Z)-9-tetradecenyl acetate |
| ZFNs | Zinc finger nucleases |
References
- Maeder, M.L.; Gersbach, C.A. Genome-editing technologies for gene and cell therapy. Mol. Ther. 2016, 24, 430–446. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.M. The genome editing revolution. J. Genet. Eng. Biotechnol. 2020, 18, 68. [Google Scholar] [CrossRef]
- Li, J.; Wu, S.; Zhang, K.; Sun, X.; Lin, W.; Wang, C.; Lin, S. Clustered regularly interspaced short palindromic repeat/crispr-associated protein and its utility all at sea: Status, challenges, and prospects. Microorganisms 2024, 12, 118. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Zafar, M.M.; Farooq, Z.; Ahmed, S.R.; Ijaz, A.; Anwar, Z.; Abbas, H.; Tariq, M.S.; Tariq, H.; Mustafa, M.; et al. Breakthrough in CRISPR/Cas system: Current and future directions and challenges. Biotechnol. J. 2023, 18, 2200642. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Bhattacharjee, O.; Mandal, D.; Sen, M.K.; Dey, D.; Dasgupta, A.; Kazi, T.A.; Gupta, R.; Sinharoy, S.; Acharya, K.; et al. CRISPR-Cas9 system: A new-fangled dawn in gene editing. Life Sci. 2019, 232, 116636. [Google Scholar] [CrossRef]
- Asmamaw, M.; Zawdie, B. Mechanism and applications of CRISPR/Cas-9-mediated genome editing. Biol. Targets Ther. 2021, 15, 353–361. [Google Scholar]
- Komal, J.; Desai, H.R.; Samal, I.; Mastinu, A.; Patel, R.D.; Kumar, P.D.; Majhi, P.K.; Mahanta, D.K.; Bhoi, T.K. Unveiling the genetic symphony: Harnessing CRISPR-Cas genome editing for effective insect pest management. Plants 2023, 12, 3961. [Google Scholar] [CrossRef]
- Singh, S.; Rahangdale, S.; Pandita, S.; Saxena, G.; Upadhyay, S.K.; Mishra, G.; Verma, P.C. CRISPR/Cas9 for insect pests management: A comprehensive review of advances and applications. Agriculture 2022, 12, 1896. [Google Scholar] [CrossRef]
- Sun, D.; Guo, Z.; Liu, Y.; Zhang, Y. Progress and prospects of CRISPR/Cas systems in insects and other arthropods. Front. Physiol. 2017, 8, 608. [Google Scholar] [CrossRef]
- Li, J.J.; Shi, Y.; Wu, J.N.; Li, H.; Smagghe, G.; Liu, T.X. CRISPR/Cas9 in lepidopteran insects: Progress, application and prospects. J. Insect Physiol. 2021, 135, 104325. [Google Scholar] [CrossRef]
- Ashok, K.; Bhargava, C.N.; Asokan, R.; Pradeep, C.; Kennedy, J.S.; Manamohan, M.; Rai, A. CRISPR/Cas9 mediated mutagenesis of the major sex pheromone gene, acyl-CoA delta-9 desaturase (DES9) in fall armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae). Int. J. Biol. Macromol. 2023, 253, 126557. [Google Scholar] [CrossRef]
- Ashok, K.; Bhargava, C.N.; Asokan, R.; Pradeep, C.; Pradhan, S.K.; Kennedy, J.S.; Balasubramani, V.; Murugan, M.; Jayakanthan, M.; Geethalakshmi, V.; et al. CRISPR/Cas9 mediated editing of pheromone biosynthesis activating neuropeptide (PBAN) gene disrupts mating in the Fall armyworm, Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae). 3 Biotech 2023, 13, 370. [Google Scholar] [CrossRef]
- Durand, K.; An, H.; Nam, K. Invasive fall armyworms are corn strain. Sci. Rep. 2024, 14, 5696. [Google Scholar] [CrossRef]
- Nurkomar, I.; Putra, I.L.; Buchori, D.; Setiawan, F. Association of a global invasive pest Spodoptera frugiperda (Lepidoptera: Noctuidae) with local parasitoids: Prospects for a new approach in selecting biological control agents. Insects 2024, 15, 205. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Soja, E.; Sosa-Gomez, D.R.; Roque-Specht, V.F.; Malaquias, J.V.; Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Biotic potential and reproductive parameters of Spodoptera frugiperda (JE Smith, 1797) (Lepidoptera: Noctuidae). J. Agric. Sci. 2019, 11, 240. [Google Scholar]
- Abro, Z.; Kimathi, E.; De Groote, H.; Tefera, T.; Sevgan, S.; Niassy, S.; Kassie, M. Socioeconomic and health impacts of fall armyworm in Ethiopia. PLoS ONE 2021, 16, e0257736. [Google Scholar] [CrossRef]
- Zhang, D.D. Insecticide resistance monitoring for the invasive populations of fall armyworm, Spodoptera frugiperda in China. J. Integr. Agric. 2021, 20, 783–791. [Google Scholar] [CrossRef]
- Sánchez-Bayo, F. Insecticides mode of action in relation to their toxicity to non-target organisms. J. Environ. Anal. Toxicol. 2012, S4, 002. [Google Scholar]
- Reisenman, C.E.; Lei, H.; Guerenstein, P.G. Neuroethology of olfactory-guided behavior and its potential application in the control of harmful insects. Front. Physiol. 2016, 7, 271. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Liu, X.; Liu, H.; Shan, S.; Shen, S.; Bai, M.; Lan, H.; Khashaveh, A.; Gu, S.; Zhang, Y. Knockout of the delta11-desaturase Sfrudes1 disrupts sex pheromone biosynthesis, mating and oviposition in the fall armyworm, Spodoptera frugiperda. Pestic. Biochem. Physiol. 2024, 200, 105832. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, A.; López, S.; Bernabé, A.; Guerrero, Á.; Quero, C. Influence of age, host plant and mating status in pheromone production and new insights on perception plasticity in Tuta absoluta. Insects 2019, 10, 256. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, J.; Krieger, J. Insect pheromone receptors–key elements in sensing intraspecific chemical signals. Front. Cell. Neurosci. 2018, 12, 425. [Google Scholar] [CrossRef]
- Bratovich, C.; Saluso, A.; Murua, M.G.; Guerenstein, P.G. Evaluation of sex pheromone formulations to attract Spodoptera frugiperda (Lepidoptera: Noctuidae) adult males in Argentina. Rev. De La Soc. Entomológica Argent. 2019, 78, 7–14. [Google Scholar] [CrossRef]
- Qu, C.; Kang, Z.; Zhang, B.; Fang, Y.; Wang, R.; Li, F.; Zhao, H.; Luo, C. Genome-wide identification and expression profiling of candidate sex pheromone biosynthesis genes in the fall armyworm (Spodoptera frugiperda). Insects 2022, 13, 1078. [Google Scholar] [CrossRef]
- Muthukumar, M.; Ashok, K.; Kennedy, J.S. Study of sexual behaviour and characterization of female sex pheromone of fall armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) collected from different locations of Tamil Nadu, India. J. Asia-Pac. Entomol. 2023, 26, 102150. [Google Scholar] [CrossRef]
- Phuong, T.T.; Fujii, T.; Ishikawa, Y.; Tung, N.D.; Giang, H.T. Female sex pheromone of Spodoptera frugiperda Vietnam population and a selected lure for adult monitoring in maize fields. J. Asia-Pac. Entomol. 2024, 27, 102343. [Google Scholar] [CrossRef]
- Renou, M. Pheromones and general odor perception in insects. Neurobiol. Chem. Commun. 2014, 1, 23–56. [Google Scholar]
- Stengl, M.; Funk, N.W. The role of the coreceptor Orco in insect olfactory transduction. J. Comp. Physiol. A 2013, 199, 897–909. [Google Scholar] [CrossRef]
- Yan, H.; Jafari, S.; Pask, G.; Zhou, X.; Reinberg, D.; Desplan, C. Evolution, developmental expression and function of odorant receptors in insects. J. Exp. Biol. 2020, 223, jeb208215. [Google Scholar] [CrossRef]
- Wicher, D.; Miazzi, F. Functional properties of insect olfactory receptors: Ionotropic receptors and odorant receptors. Cell Tissue Res. 2021, 383, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Wicher, D. Olfactory signaling in insects. Prog. Mol. Biol. Transl. Sci. 2015, 130, 37–54. [Google Scholar]
- Zufall, F.; Domingos, A.I. The structure of Orco and its impact on our understanding of olfaction. J. Gen. Physiol. 2018, 150, 1602–1605. [Google Scholar] [CrossRef]
- Paulo, D.F.; Junqueira, A.C.; Arp, A.P.; Vieira, A.S.; Ceballos, J.; Skoda, S.R.; Pérez-de-León, A.A.; Sagel, A.; McMillan, W.O.; Scott, M.J.; et al. Disruption of the odorant coreceptor Orco impairs foraging and host finding behaviors in the New World screwworm fly. Sci. Rep. 2021, 11, 11379. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.B.; Mo, B.T.; Li, G.C.; Huang, L.Q.; Guo, H.; Gong, X.L.; Wang, C.Z. Mutagenesis of the odorant receptor co-receptor (Orco) reveals severe olfactory defects in the crop pest moth Helicoverpa armigera. BMC Biol. 2022, 20, 214. [Google Scholar] [CrossRef]
- Fandino, R.A.; Haverkamp, A.; Bisch-Knaden, S.; Zhang, J.; Bucks, S.; Nguyen, T.A.; Schröder, K.; Werckenthin, A.; Rybak, J.; Stengl, M.; et al. Mutagenesis of odorant coreceptor Orco fully disrupts foraging but not oviposition behaviors in the hawkmoth Manduca sexta. Proc. Natl. Acad. Sci. USA 2019, 116, 15677–15685. [Google Scholar] [CrossRef] [PubMed]
- Saveer, A.M.; Hatano, E.; Wada-Katsumata, A.; Meagher, R.L.; Schal, C. Nonanal, a new fall armyworm sex pheromone component, significantly increases the efficacy of pheromone lures. Pest Manag. Sci. 2023, 79, 2831–2839. [Google Scholar] [CrossRef] [PubMed]
- Alabaster, A.; Isoe, J.; Zhou, G.; Lee, A.; Murphy, A.; Day, W.A.; Miesfeld, R.L. Deficiencies in acetyl-CoA carboxylase and fatty acid synthase 1 differentially affect eggshell formation and blood meal digestion in Aedes aegypti. Insect Biochem. Mol. Biol. 2011, 41, 946–955. [Google Scholar] [CrossRef]
- Choi, M.Y.; Jurenka, R.A. C75, a fatty acid synthase inhibitor, inhibits feeding activity and pheromone production in a moth, Helicoverpa zea. J. Asia-Pac. Entomol. 2006, 9, 43–48. [Google Scholar] [CrossRef]
- Groot, A.T.; Marr, M.; Schöfl, G.; Lorenz, S.; Svatos, A.; Heckel, D.G. Host strain specific sex pheromone variation in Spodoptera frugiperda. Front. Zool. 2008, 5, 20. [Google Scholar] [CrossRef]
- Ma, P.W.; Ramaswamy, S.B. Biology and ultrastructure of sex pheromone-producing tissue. In Insect Pheromone Biochemistry and Molecular Biology; Academic Press: Cambridge, MA, USA, 2003; pp. 19–51. [Google Scholar]
- Akter, S.; Hossain, M.S.; Ali, R.; Regmi, R.; Park, S.J.; Mainali, B. Variations in Sex Pheromone of the Australian Population of Fall Armyworm: Influence of Age and Mating Status. J. Chem. Ecol. 2025, 51, 55. [Google Scholar] [CrossRef]
- Smith, A.F.; Schal, C. Corpus allatum control of sex pheromone production and calling in the female brown-banded cockroach, Supella longipalpa (F.) (Dictyoptera: Blattellidae). J. Insect Physiol. 1990, 36, 251–257. [Google Scholar] [CrossRef]
- Toivonen, J.M.; Partridge, L. Endocrine regulation of aging and reproduction in Drosophila. Mol. Cell. Endocrinol. 2009, 299, 39–50. [Google Scholar] [CrossRef]
- Force, E.; Debernard, S. Endocrine regulation of reproductive behaviors in insects: A comprehensive review. Curr. Opin. Insect Sci. 2025, 69, 101360. [Google Scholar] [CrossRef]
- Liu, Y.; Zou, K.; Wang, T.; Guan, M.; Duan, H.; Yu, H.; Wu, D.; Du, J. Genome-Wide Identification and Analysis of Family Members with Juvenile Hormone Binding Protein Domains in Spodoptera frugiperda. Insects 2024, 15, 573. [Google Scholar] [CrossRef] [PubMed]
- Hassanien, I.T.; Grötzner, M.; Meyering-Vos, M.; Hoffmann, K.H. Neuropeptides affecting the transfer of juvenile hormones from males to females during mating in Spodoptera frugiperda. J. Insect Physiol. 2014, 66, 45–52. [Google Scholar] [CrossRef]
- Li, C.; Yu, J.F.; Lu, Q.; Xu, J.; Liu, J.H.; Ye, H. Molecular characterization and functional analysis of a putative sex-peptide receptor in the tobacco cutworm Spodoptera litura (F abricius, 1775) (Lepidoptera: N octuidae). Austral Entomol. 2014, 53, 424–431. [Google Scholar] [CrossRef]
- Comte, A.; Delarue, A.; François, M.C.; Monsempes, C.; Meslin, C.; Montagné, N.; Jacquin-Joly, E. Functional Investigation of the Receptor to the Major Pheromone Component in the C-Strain and the R-Strain of the Fall Armyworm Spodoptera frugiperda. Insects 2025, 16, 304. [Google Scholar] [CrossRef]
- Wu, H.; Xu, M.; Hou, C.; Huang, L.Q.; Dong, J.F.; Wang, C.Z. Specific olfactory neurons and glomeruli are associated to differences in behavioral responses to pheromone components between two Helicoverpa species. Front. Behav. Neurosci. 2015, 9, 206. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Gong, X.L.; Li, G.C.; Mo, B.T.; Jiang, N.J.; Huang, L.Q.; Wang, C.Z. Functional analysis of pheromone receptor repertoire in the fall armyworm, Spodoptera frugiperda. Pest Manag. Sci. 2022, 78, 2052–2064. [Google Scholar] [CrossRef]
- Qiu, L.; He, L.; Tan, X.; Zhang, Z.; Wang, Y.; Li, X.; He, H.; Ding, W.; Li, Y. Identification and phylogenetics of Spodoptera frugiperda chemosensory proteins based on antennal transcriptome data. Comp. Biochem. Physiol. Part D Genom. Proteom. 2020, 34, 100680. [Google Scholar] [CrossRef]
- Liu, X.; Shi, L.; Khashaveh, A.; Shan, S.; Lv, B.; Gu, S.; Zhang, Y. Loss of binding capabilities in an ecologically important odorant receptor of the fall armyworm, Spodoptera frugiperda, by a single point mutation. J. Agric. Food Chem. 2023, 71, 13003–13013. [Google Scholar] [CrossRef]
- Wang, Y.; Qiu, L.; Wang, B.; Guan, Z.; Dong, Z.; Zhang, J.; Cao, S.; Yang, L.; Wang, B.; Gong, Z.; et al. Structural basis for odorant recognition of the insect odorant receptor OR-Orco heterocomplex. Science 2024, 384, 1453–1460. [Google Scholar] [CrossRef]
- Ray, I.; Mohanty, S. Pheromone-binding proteins in pest control: From molecular insights to real-world applications. J. Agric. Food Chem. 2025, 73, 21701–21727. [Google Scholar] [CrossRef]
- Zhang, D.D.; Löfstedt, C. Moth pheromone receptors: Gene sequences, function, and evolution. Front. Ecol. Evol. 2015, 3, 105. [Google Scholar] [CrossRef]
- Guo, J.M.; Liu, X.L.; Liu, S.R.; Wei, Z.Q.; Han, W.K.; Guo, Y.; Dong, S.L. Functional characterization of sex pheromone receptors in the fall armyworm (Spodoptera frugiperda). Insects 2020, 11, 193. [Google Scholar] [CrossRef] [PubMed]
- Malo, E.A.; Castrejón-Gómez, V.R.; Cruz-López, L.; Rojas, J.C. Antennal sensilla and electrophysiological response of male and female Spodoptera frugiperda (Lepidoptera: Noctuidae) to conspecific sex pheromone and plant odors. Ann. Entomol. Soc. Am. 2004, 97, 1273–1284. [Google Scholar] [CrossRef]
- Malzahn, A.; Lowder, L.; Qi, Y. Plant genome editing with TALEN and CRISPR. Cell Biosci. 2017, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Cardi, T.; Murovec, J.; Bakhsh, A.; Boniecka, J.; Bruegmann, T.; Bull, S.E.; Eeckhaut, T.; Fladung, M.; Galovic, V.; Linkiewicz, A.; et al. CRISPR/Cas-mediated plant genome editing: Outstanding challenges a decade after implementation. Trends Plant Sci. 2023, 28, 1144–1165. [Google Scholar] [CrossRef]
- Redman, M.; King, A.; Watson, C.; King, D. What is CRISPR/Cas9? Arch. Dis. Child. Educ. Pract. 2016, 101, 213–215. [Google Scholar] [CrossRef] [PubMed]
- Drury, D.W.; Dapper, A.L.; Siniard, D.J.; Zentner, G.E.; Wade, M.J. CRISPR/Cas9 gene drives in genetically variable and nonrandomly mating wild populations. Sci. Adv. 2017, 3, e1601910. [Google Scholar] [CrossRef]
- Kim, H.; Ishidate, T.; Ghanta, K.S.; Seth, M.; Conte, D., Jr.; Shirayama, M.; Mello, C.C. A co-CRISPR strategy for efficient genome editing in Caenorhabditis elegans. Genetics 2014, 197, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Salum, Y.M.; Yin, A.; Zaheer, U.; Liu, Y.; Guo, Y.; He, W. CRISPR/Cas9-based genome editing of fall armyworm (Spodoptera frugiperda): Progress and prospects. Biomolecules 2024, 14, 1074. [Google Scholar] [CrossRef]
- Wu, K.; Shirk, P.D.; Taylor, C.E.; Furlong, R.B.; Shirk, B.D.; Pinheiro, D.H.; Siegfried, B.D. CRISPR/Cas9 mediated knockout of the abdominal-A homeotic gene in fall armyworm moth (Spodoptera frugiperda). PLoS ONE 2018, 13, e0208647. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, Z. CRISPR-Cas systems: Overview, innovations and applications in human disease research and gene therapy. Comput. Struct. Biotechnol. J. 2020, 18, 2401–2415. [Google Scholar] [CrossRef] [PubMed]
- Kalajdzic, P.; Schetelig, M.F. CRISPR/Cas-mediated gene editing using purified protein in Drosophila suzukii. Entomol. Exp. Appl. 2017, 164, 350–362. [Google Scholar] [CrossRef]
- Lamb, A.M.; Walker, E.A.; Wittkopp, P.J. Tools and strategies for scarless allele replacement in Drosophila using CRISPR/Cas9. Fly 2017, 11, 53–64. [Google Scholar] [CrossRef]
- El-Awaad, E.; Merzendorfer, H. CRISPR/Cas: An emerging molecular technology for biological control of fall armyworm. New Plant Prot. 2025, 2, e26. [Google Scholar] [CrossRef]
- Bi, H.L.; Xu, J.; Tan, A.J.; Huang, Y.P. CRISPR/Cas9-mediated targeted gene mutagenesis in Spodoptera litura. Insect Sci. 2016, 23, 469–477. [Google Scholar] [CrossRef]
- Zuo, Y.; Wang, H.; Xu, Y.; Huang, J.; Wu, S.; Wu, Y.; Yang, Y. CRISPR/Cas9 mediated G4946E substitution in the ryanodine receptor of Spodoptera exigua confers high levels of resistance to diamide insecticides. Insect Biochem. Mol. Biol. 2017, 89, 79–85. [Google Scholar] [CrossRef]
- Zhang, S.; Jacquin-Joly, E.; Montagné, N.; Liu, F.; Liu, Y.; Wang, G. Identification of an odorant receptor responding to sex pheromones in Spodoptera frugiperda extends the novel type-I PR lineage in moths. Insect Sci. 2024, 31, 489–502. [Google Scholar] [CrossRef]
- Zhang, S.; Yan, S.; Mei, X.; Wang, G.; Liu, Y. Identification of a new lineage of pheromone receptors in mirid bugs (Heteroptera: Miridae). Pestic. Biochem. Physiol. 2025, 208, 106277. [Google Scholar] [CrossRef] [PubMed]
- Holdcraft, R.; Rodriguez-Saona, C.; Stelinski, L.L. Pheromone autodetection: Evidence and implications. Insects 2016, 7, 17. [Google Scholar] [CrossRef]
- Costa, C.G.; Rodrigues, S.R.; Fuhrmann, J. Morphology of the antennal sensilla of two species of Hoplopyga Thomson, 1880 (Coleoptera, Scarabaeidae, Cetoniinae). Rev. Bras. De Entomol. 2021, 65, e20200078. [Google Scholar] [CrossRef]
- Wang, W.; He, P.; Liu, T.; Jing, X.; Zhang, S. Morphology and distribution of antennal sensilla on Spodoptera frugiperda (Lepidoptera: Noctuidae) larvae and adults. Diversity 2023, 15, 992. [Google Scholar] [CrossRef]
- Benton, R.; Vannice, K.S.; Gomez-Diaz, C.; Vosshall, L.B. Variant ionotropic glutamate receptors as chemosensory receptors in Drosophila. Cell 2009, 136, 149–162. [Google Scholar] [CrossRef]
- Sun, Y.L.; Dong, J.F.; Yang, H.B.; Li, D.X.; Tian, C.H. Identification and characterization of chemosensory receptors in the pheromone gland-ovipositor of Spodoptera frugiperda (JE Smith). Insects 2022, 13, 481. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.L.; Yan, Q.; Yang, Y.L.; Hou, W.; Miao, C.L.; Peng, Y.C.; Dong, S.L. A gustatory receptor GR8 tunes specifically to D-fructose in the common cutworm Spodoptera litura. Insects 2019, 10, 272. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.L.; Jiang, P.S.; Dong, B.X.; Tian, C.H.; Dong, J.F. Candidate chemosensory receptors in the antennae and maxillae of Spodoptera frugiperda (JE Smith) larvae. Front. Physiol. 2022, 13, 970915. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, B.; Grossi, G.; Falabella, P.; Liu, Y.; Yan, S.; Lu, J.; Xi, J.; Wang, G. Molecular basis of alarm pheromone detection in aphids. Curr. Biol. 2017, 27, 55–61. [Google Scholar] [CrossRef]
- Pelosi, P.; Zhou, J.J.; Ban, L.P.; Calvello, M. Soluble proteins in insect chemical communication. Cell. Mol. Life Sci. CMLS 2006, 63, 1658–1676. [Google Scholar] [CrossRef]
- Liu, X.L.; Wu, Z.R.; Liao, W.; Zhang, X.Q.; Pei, Y.W.; Lu, M. The binding affinity of two general odorant binding proteins in Spodoptera frugiperda to general volatiles and insecticides. Int. J. Biol. Macromol. 2023, 252, 126338. [Google Scholar] [CrossRef] [PubMed]
- Han, W.K.; Tang, F.X.; Yu, N.; Zhang, Y.X.; Liu, Z.W. A nonsensory odorant-binding protein plays an important role in the larval development and adult mating of Spodoptera frugiperda. Insect Sci. 2023, 30, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, A.; Spinelli, S.; Tegoni, M.; He, X.; Field, L.; Zhou, J.J.; Cambillau, C. The crystal structure of odorant binding protein 7 from Anopheles gambiae exhibits an outstanding adaptability of its binding site. J. Mol. Biol. 2011, 414, 401–412. [Google Scholar] [CrossRef]
- Leal, W.S.; Nikonova, L.; Peng, G. Disulfide structure of the pheromone binding protein from the silkworm moth, Bombyx mori. FEBS Lett. 1999, 464, 85–90. [Google Scholar] [CrossRef]
- Tsouri, A.; Douris, V. The Role of Chemosensory Proteins in Insecticide Resistance: A Review. Insects 2025, 16, 496. [Google Scholar] [CrossRef]
- Jia, C.; Mohamed, A.; Cattaneo, A.M.; Huang, X.; Keyhani, N.O.; Gu, M.; Zang, L.; Zhang, W. Odorant-binding proteins and chemosensory proteins in Spodoptera frugiperda: From genome-wide identification and developmental stage-related expression analysis to the perception of host plant odors, sex pheromones, and insecticides. Int. J. Mol. Sci. 2023, 24, 5595. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, R.; Gao, J.; Xiao, X.; Yin, X.; Hu, S.; Zhang, Y.; Liang, P.; Gu, S. Two cuticle-enriched chemosensory proteins confer multi-insecticide resistance in Spodoptera frugiperda. Int. J. Biol. Macromol. 2024, 266, 130941. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, X.; Cao, S.; Ma, B.; Guo, M.; Shen, J.; Wang, G. Fine structure and olfactory reception of the labial palps of Spodoptera frugiperda. Front. Physiol. 2021, 12, 680697. [Google Scholar] [CrossRef]
- Peter, E.; Tamiru, A.; Sevgan, S.; Dubois, T.; Kelemu, S.; Kruger, K.; Torto, B.; Yusuf, A. Companion crops alter olfactory responses of the fall armyworm (Spodoptera frugiperda) and its larval endoparasitoid (Cotesia icipe). Chem. Biol. Technol. Agric. 2023, 10, 61. [Google Scholar] [CrossRef]
- Chen, X.; Yao, S.; Xie, L.; Li, J.; Xiong, L.; Yang, X.; Chen, Y.; Cao, F.; Hou, Q.; You, M.; et al. Disruption of the odorant receptor co-receptor (Orco) reveals its critical role in multiple olfactory behaviors of a cosmopolitan pest. Insect Biochem. Mol. Biol. 2025, 177, 104248. [Google Scholar] [CrossRef]
- Boronat-Garcia, A.; Iben, J.; Dominguez-Martin, E.; Stopfer, M. Identification and analysis of odorant receptors expressed in the two main olfactory organs, antennae and palps, of Schistocerca americana. Sci. Rep. 2022, 12, 22628. [Google Scholar] [CrossRef] [PubMed]
- Mukunda, L.; Lavista-Llanos, S.; Hansson, B.S.; Wicher, D. Dimerisation of the Drosophila odorant coreceptor Orco. Front. Cell. Neurosci. 2014, 8, 261. [Google Scholar] [CrossRef]
- Tiwari, V.; Sowdhamini, R. Structure modelling of odorant receptor from Aedes aegypti and identification of potential repellent molecules. Comput. Struct. Biotechnol. J. 2023, 21, 2204–2214. [Google Scholar] [CrossRef]
- Sun, H.; Bu, L.A.; Su, S.C.; Guo, D.; Gao, C.F.; Wu, S.F. Knockout of the odorant receptor co-receptor, orco, impairs feeding, mating and egg-laying behavior in the fall armyworm Spodoptera frugiperda. Insect Biochem. Mol. Biol. 2023, 152, 103889. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, I. Attracting carnivorous arthropods with plant volatiles: The future of biocontrol or playing with fire? Biol. Control 2012, 60, 77–89. [Google Scholar] [CrossRef]
- Darathi, D.; Santa, K. Volatile mediated plant-insect interactions: A review. Int. J. Zool. Investig. 2022, 8, 291–304. [Google Scholar] [CrossRef]
- Jones, W.D.; Nguyen, T.A.; Kloss, B.; Lee, K.J.; Vosshall, L.B. Functional conservation of an insect odorant receptor gene across 250 million years of evolution. Curr. Biol. 2005, 15, R119–R121. [Google Scholar] [CrossRef]
- Wicher, D.; Schäfer, R.; Bauernfeind, R.; Stensmyr, M.C.; Heller, R.; Heinemann, S.H.; Hansson, B.S. Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature 2008, 452, 1007–1011. [Google Scholar] [CrossRef]
- Wang, Q.; Jia, Y.; Smid, H.M.; Weldegergis, B.T.; Greenberg, L.O.; Jongsma, M.; Dicke, M.; Haverkamp, A. Loss of olfaction reduces caterpillar performance and increases susceptibility to a natural enemy. elife 2025, 14, RP105585. [Google Scholar] [CrossRef]
- Li, H.; Wan, P.; Zhu, Z.; Xu, D.; Cong, S.; Xu, M.; Yin, H. Spodoptera frugiperda Uses Specific Volatiles to Assess Maize Development for Optimal Offspring Survival. Insects 2025, 16, 592. [Google Scholar] [CrossRef]
- Zhang, C.; Tang, B.; Tan, H.; Wang, X.; Dai, W. The Orco gene involved in recognition of host plant volatiles and sex pheromone in the chive maggot Bradysia odoriphaga. Pestic. Biochem. Physiol. 2023, 197, 105709. [Google Scholar] [CrossRef] [PubMed]
- Song, C.A.; Yang, L.I.; Qing, Y.A.; Gui-rong, W.A. Mutagenesis of odorant coreceptor Orco reveals the distinct role of olfaction between sexes in Spodoptera frugiperda. J. Integr. Agric. 2023, 22, 2162–2172. [Google Scholar]
- Zhao, Y.; Li, L.; Wei, L.; Wang, Y.; Han, Z. Advancements and Future Prospects of CRISPR-Cas-Based Population Replacement Strategies in Insect Pest Management. Insects 2024, 15, 653. [Google Scholar] [CrossRef]
- Yadav, S.; Sarangi, S.; Parmar, S.; Thodusu, M.; Soni, S.; Pandey, K. Genetic engineering in insect management: New frontiers in pest control. Microbiol. Res. J. Int. 2024, 34, 106–121. [Google Scholar] [CrossRef]
- Zhou, L.; Alphey, N.; Walker, A.S.; Travers, L.M.; Morrison, N.I.; Bonsall, M.B.; Raymond, B. The application of self-limiting transgenic insects in managing resistance in experimental metapopulations. J. Appl. Ecol. 2019, 56, 688–698. [Google Scholar] [CrossRef]
- Reavey, C.E.; Domingues, F.A.; Ercit, K.; Pinto, R.L.; Naish, N.; Yadav, M.; Frazon, T.; Cabala, K.; Sulston, E.; Pickl-Herk, A.; et al. Field Performance of a Self-Limiting, Genetically Engineered Fall Armyworm for Biological Pest Management. Front. Agron. 2025, 7, 1613689. [Google Scholar] [CrossRef]
- Reavey, C.E.; Walker, A.S.; Joyce, S.P.; Broom, L.; Willse, A.; Ercit, K.; Poletto, M.; Barnes, Z.H.; Marubbi, T.; Troczka, B.J.; et al. Self-limiting fall armyworm: A new approach in development for sustainable crop protection and resistance management. BMC Biotechnol. 2022, 22, 5. [Google Scholar] [CrossRef]
- Reeves, R.G.; Phillipson, M. Mass releases of genetically modified insects in area-wide pest control programs and their impact on organic farmers. Sustainability 2017, 9, 59. [Google Scholar] [CrossRef]
- Shelton, A.M.; Long, S.J.; Walker, A.S.; Bolton, M.; Collins, H.L.; Revuelta, L.; Johnson, L.M.; Morrison, N.I. First field release of a genetically engineered, self-limiting agricultural pest insect: Evaluating its potential for future crop protection. Front. Bioeng. Biotechnol. 2020, 7, 482. [Google Scholar] [CrossRef] [PubMed]
- Grilli, S.; Galizi, R.; Taxiarchi, C. Genetic technologies for sustainable management of insect pests and disease vectors. Sustainability 2021, 13, 5653. [Google Scholar] [CrossRef]
- Burt, A. Heritable strategies for controlling insect vectors of disease. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130432. [Google Scholar] [CrossRef]
- Khalil, A.M. Traditional Strategies and Cutting-Edge Technologies Used for Animal Breeding: A Comprehensive Review. Sustain. Food Connect 2025, 1, 2025.0002. [Google Scholar] [CrossRef]
- Lim, L.W. CRISPR-Cas application in insect: Food, medicine, pest control, disease resistance and textile. Food Biosci. 2025, 171, 07292. [Google Scholar] [CrossRef]
- Gu, J.; Wang, J.; Bi, H.; Li, X.; Merchant, A.; Zhang, P.; Zhang, Q.; Zhou, X. CRISPR/Cas9-mediated mutagenesis of sex-specific doublesex splicing variants leads to sterility in Spodoptera frugiperda, a global invasive pest. Cells 2022, 11, 3557. [Google Scholar] [CrossRef]
- Yogi, D.; Ashok, K.; Anu, C.N.; Shashikala, T.; Pradeep, C.; Bhargava, C.N.; Parvathy, M.S.; Jithesh, M.N.; Manamohan, M.; Jha, G.K.; et al. CRISPR/Cas12a ribonucleoprotein mediated editing of tryptophan 2, 3-dioxygenase of Spodoptera frugiperda. Transgenic Res. 2024, 33, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.H.; Chereddy, S.C.; Howell, J.L.; Palli, S.R. Genome editing in the fall armyworm, Spodoptera frugiperda: Multiple sgRNA/Cas9 method for identification of knockouts in one generation. Insect Biochem. Mol. Biol. 2020, 122, 103373. [Google Scholar] [CrossRef] [PubMed]
- Basit, A.; Mobarak, S.H.; Liu, T.X. CRISPR/Cas9-based strategies for effective control of Spodoptera frugiperda in maize: A review of precision gene editing approaches. Entomol. Gen. 2025, 45, 651–666. [Google Scholar] [CrossRef]
- Tanny, T.; Sallam, M.; Soda, N.; Nguyen, N.T.; Alam, M.; Shiddiky, M.J. CRISPR/Cas-based diagnostics in agricultural applications. J. Agric. Food Chem. 2023, 71, 11765–11788. [Google Scholar] [CrossRef]
- Koonin, E.V.; Gootenberg, J.S.; Abudayyeh, O.O. Discovery of diverse CRISPR-Cas systems and expansion of the genome engineering toolbox. Biochemistry 2023, 62, 3465–3487. [Google Scholar] [CrossRef] [PubMed]
- Bryson, J.W.; Auxillos, J.Y.; Rosser, S.J. Multiplexed activation in mammalian cells using a split-intein CRISPR/Cas12a based synthetic transcription factor. Nucleic Acids Res. 2022, 50, 549–560. [Google Scholar] [CrossRef]
- Abudayyeh, O.O.; Gootenberg, J.S.; Konermann, S.; Joung, J.; Slaymaker, I.M.; Cox, D.B.; Shmakov, S.; Makarova, K.S.; Semenova, E.; Minakhin, L.; et al. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science 2016, 353, aaf5573. [Google Scholar] [CrossRef]
- Chen, X.; Tan, A.; Palli, S.R. Identification and functional analysis of promoters of heat-shock genes from the fall armyworm, Spodoptera frugiperda. Sci. Rep. 2020, 10, 2363. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y. Advances in CRISPR/Cas9. BioMed Res. Int. 2022, 2022, 9978571. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, D.; Matsugi, E.; Kishi, K.; Inoue, Y.; Nigorikawa, K.; Nomura, W. SpCas9-HF1 enhances accuracy of cell cycle-dependent genome editing by increasing HDR efficiency, and by reducing off-target effects and indel rates. Mol. Ther. -Nucleic Acids 2024, 35, 102124. [Google Scholar] [CrossRef] [PubMed]
- Dhandapani, R.K.; Gurusamy, D.; Palli, S.R. Protamine–lipid–dsRNA nanoparticles improve RNAi efficiency in the fall armyworm, Spodoptera frugiperda. J. Agric. Food Chem. 2022, 70, 6634–6643. [Google Scholar] [CrossRef]
- Aghamiri, S.; Talaei, S.; Ghavidel, A.A.; Zandsalimi, F.; Masoumi, S.; Hafshejani, N.H.; Jajarmi, V. Nanoparticles-mediated CRISPR/Cas9 delivery: Recent advances in cancer treatment. J. Drug Deliv. Sci. Technol. 2020, 56, 101533. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, Y.; Zhang, C.; Li, M.-J.; Iqbal, A.; Ahmed, K.S.; Idrees, A.; Habiba; Yang, B.-M.; Jiang, L. Exploring the Role of Pheromones and CRISPR/Cas9 in the Behavioral and Olfactory Mechanisms of Spodoptera frugiperda. Insects 2026, 17, 35. https://doi.org/10.3390/insects17010035
Wang Y, Zhang C, Li M-J, Iqbal A, Ahmed KS, Idrees A, Habiba, Yang B-M, Jiang L. Exploring the Role of Pheromones and CRISPR/Cas9 in the Behavioral and Olfactory Mechanisms of Spodoptera frugiperda. Insects. 2026; 17(1):35. https://doi.org/10.3390/insects17010035
Chicago/Turabian StyleWang, Yu, Chen Zhang, Mei-Jun Li, Asim Iqbal, Kanwer Shahzad Ahmed, Atif Idrees, Habiba, Bai-Ming Yang, and Long Jiang. 2026. "Exploring the Role of Pheromones and CRISPR/Cas9 in the Behavioral and Olfactory Mechanisms of Spodoptera frugiperda" Insects 17, no. 1: 35. https://doi.org/10.3390/insects17010035
APA StyleWang, Y., Zhang, C., Li, M.-J., Iqbal, A., Ahmed, K. S., Idrees, A., Habiba, Yang, B.-M., & Jiang, L. (2026). Exploring the Role of Pheromones and CRISPR/Cas9 in the Behavioral and Olfactory Mechanisms of Spodoptera frugiperda. Insects, 17(1), 35. https://doi.org/10.3390/insects17010035