Ecological Preferences of Calliphoridae and Sarcophagidae (Diptera) in the Region Calabria (Southern Italy)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Areas
2.2. Sampling Methods
2.3. Data Analysis
3. Results
3.1. Environmental Data
3.2. Abundances and Diversity
3.3. Seasonal Distribution
3.4. Altitudinal Gradient
3.5. Habitat Preferences
3.6. IndVal
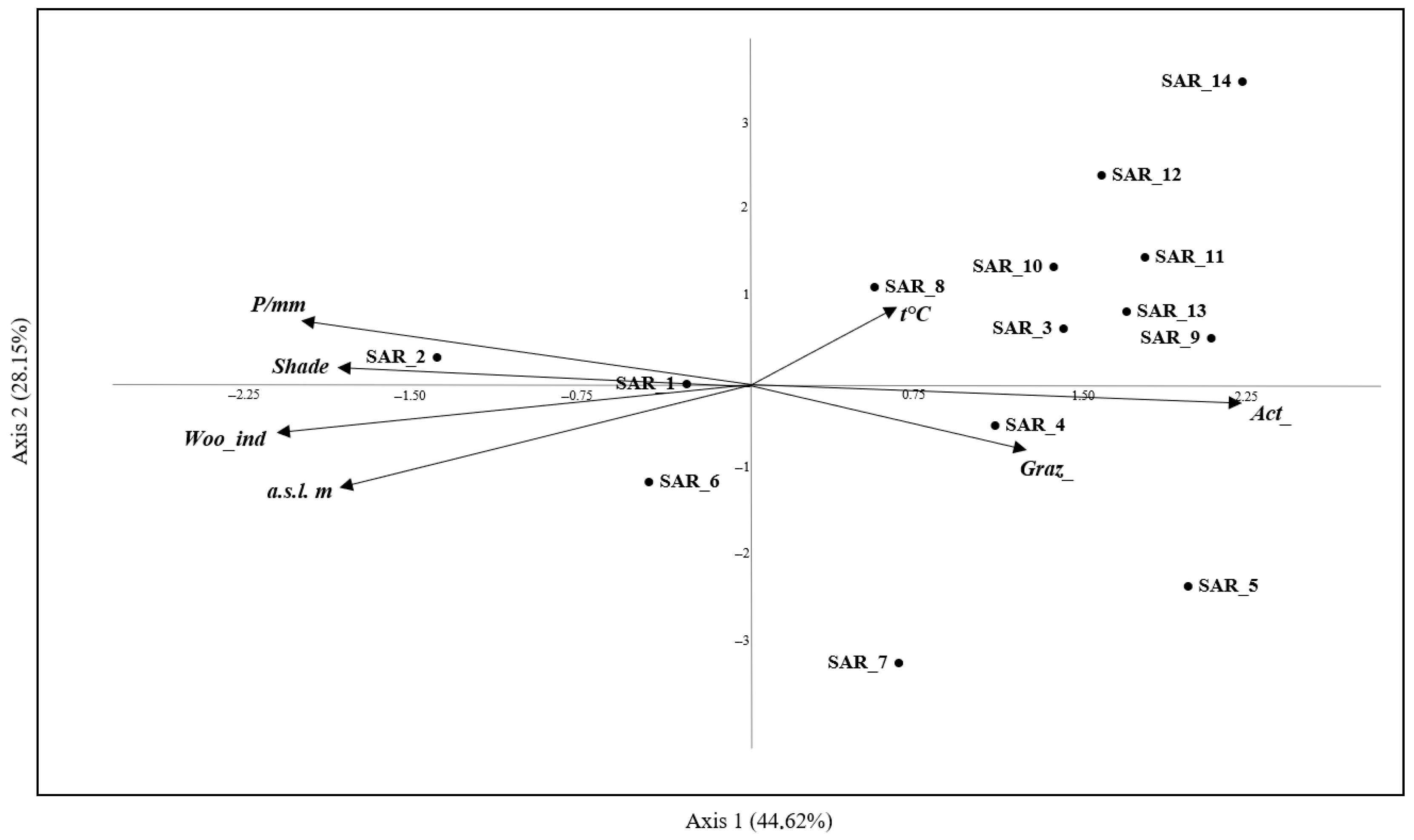
3.7. CCA
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| CCA Code | Species | n | (%) | Vegetation Type | Coverage | ||||
|---|---|---|---|---|---|---|---|---|---|
| WL | PF | BF | SM | C | O | ||||
| CAL_1 | Calliphora vomitoria (Linnaeus, 1758) | 16,422 | 45.50% | 2539 | 6859 | 6895 | 129 | 13754 | 2668 |
| CAL_2 | Calliphora vicina Robineau-Desvoidy, 1830 | 10,856 | 29.94% | 4059 | 2590 | 3040 | 1167 | 5630 | 5226 |
| CAL_3 | Lucilia caesar (Linnaeus, 1758) | 5151 | 14.21% | 776 | 2219 | 1324 | 832 | 3543 | 1608 |
| CAL_4 | Lucilia ampullacea Villeneuve, 1922 | 2353 | 6.49% | 343 | 838 | 445 | 727 | 1283 | 1070 |
| CAL_5 | Lucilia sericata (Meigen, 1826) | 731 | 2.02% | 140 | 11 | 9 | 571 | 20 | 711 |
| CAL_6 | Chrysomya albiceps (Wiedemann, 1819) | 317 | 0.87% | 160 | 78 | 24 | 55 | 102 | 215 |
| CAL_7 | Calliphora rohdendorfi (Grunin, 1966) | 134 | 0.37% | \ | 56 | 78 | \ | 134 | \ |
| CAL_8 | Protocalliphora azurea (Fallén, 1817) | 93 | 0.26% | 68 | 11 | 6 | 8 | 17 | 76 |
| CAL_9 | Melinda gentilis Robineau-Desvoidy, 1830 | 67 | 0.18% | 13 | 24 | 26 | 4 | 50 | 17 |
| CAL_10 | Onesia floralis Robineau-Desvoidy, 1830 | 46 | 0.13% | 23 | 15 | 3 | 5 | 18 | 28 |
| CAL_11 | Eurychaeta muscaria (Meigen, 1826) | 46 | 0.13% | 4 | 25 | 13 | 4 | 38 | 8 |
| \ | Melinda viridicyanea (Robineau-Desvoidy, 1830) | 30 | 0.08% | 9 | 10 | 10 | 1 | 20 | 10 |
| \ | Bellardia viarum (Robineau-Desvoidy, 1830) | 5 | 0.01% | 1 | 2 | 2 | \ | 4 | 1 |
| \ | Protophormia terraenovae (Robineau-Desvoidy, 1830) | 2 | 0.01% | 1 | \ | 1 | \ | 1 | 1 |
| Total | 36,253 | 100% | 8136 | 12,738 | 11,876 | 3503 | 24,614 | 11,639 | |
| N° species→ | 13 | 13 | 14 | 11 | 14 | 13 | |||
| CCA Code | Species | n | (%) | Vegetation Type | Coverage | ||||
|---|---|---|---|---|---|---|---|---|---|
| WL | PF | BF | SM | C | O | ||||
| SAR_1 | Sarcophaga (Sarcophaga) croatica Baranov, 1941 | 1495 | 45.52% | 446 | 508 | 207 | 334 | 715 | 780 |
| SAR_2 | Sarcophaga (Sarcophaga) subvicina Rohdendorf, 1937 | 481 | 14.65% | 66 | 224 | 123 | 68 | 347 | 134 |
| SAR_3 | Sarcophaga (Liosarcophaga) portschinskyi (Rohdendorf, 1937) | 194 | 5.91% | 81 | 23 | 2 | 88 | 25 | 169 |
| SAR_4 | Sarcophaga (Parasarcophaga) albiceps Meigen, 1826 | 179 | 5.45% | 95 | 13 | 16 | 55 | 29 | 150 |
| SAR_5 | Ravinia pernix (Harris, 1780) | 124 | 3.78% | 119 | 2 | \ | 3 | 2 | 122 |
| SAR_6 | Sarcophaga (Robineauella) caerulescens Zetterstedt, 1838 | 103 | 3.14% | 44 | 30 | 23 | 6 | 53 | 50 |
| SAR_7 | Angiometopa falleni Pape, 1986 | 89 | 2.71% | 70 | 19 | \ | \ | 19 | 70 |
| SAR_8 | Sarcophaga (Heteronychia) haemorrhoa Meigen, 1826 | 88 | 2.68% | 38 | 10 | \ | 40 | 10 | 78 |
| SAR_9 | Sarcophaga (Thyrsocnema) incisilobata Pandellé, 1896 | 77 | 2.34% | 56 | 2 | \ | 19 | 1 | 76 |
| SAR_10 | Sarcophaga (Liosarcophaga) teretirostris Pandellé, 1896 | 54 | 1.64% | 17 | 7 | 2 | 28 | 9 | 45 |
| SAR_11 | Sarcophaga (Bercaea) africa (Wiedemann, 1824) | 45 | 1.37% | 14 | \ | \ | 31 | \ | 45 |
| SAR_12 | Sarcophaga (Rosellea) aratrix Pandelle, 1896 | 44 | 1.34% | 9 | \ | \ | 35 | \ | 44 |
| SAR_13 | Sarcophaga (Liopygia) argyrostoma (Robineau-Desvoidy, 1830) | 39 | 1.19% | 16 | \ | \ | 23 | \ | 39 |
| SAR_14 | Sarcophaga (Helicophagella) melanura Meigen, 1826 | 35 | 1.07% | 1 | \ | \ | 34 | \ | 35 |
| \ | Sarcophaga (Liosarcophaga) aegyptica Salem, 1935 | 26 | 0.79% | \ | \ | \ | 26 | \ | 26 |
| \ | Sarcophaga (Heteronychia) filia Rondani, 1860 | 26 | 0.79% | 20 | 6 | \ | \ | 6 | 20 |
| \ | Sarcophila latifrons (Fallén, 1817) | 26 | 0.79% | 11 | 2 | \ | 13 | 2 | 24 |
| \ | Sarcophaga (Heteronychia) vagans Meigen, 1826 | 24 | 0.73% | 10 | 13 | 1 | \ | 14 | 10 |
| \ | Sarcophaga (Myorhina) nigriventris Meigen, 1826 | 23 | 0.70% | 16 | 7 | \ | \ | 7 | 16 |
| \ | Sarcophaga (Liosarcophaga) jacobsoni (Rohdendorf, 1937) | 20 | 0.61% | 1 | \ | 1 | 18 | 1 | 19 |
| \ | Sarcophaga (Helicophagella) agnata Rondani, 1860 | 19 | 0.58% | 4 | 11 | 4 | \ | 15 | 4 |
| \ | Sarcophaga (Heteronychia) consanguinea Rondani, 1860 | 16 | 0.49% | \ | 1 | \ | 15 | 1 | 15 |
| \ | Sarcophaga (Heteronychia) vicina Macquart, 1835 | 12 | 0.37% | 2 | 3 | 2 | 5 | 5 | 7 |
| \ | Sarcophaga (Helicophagella) hirticrus Pandellé, 1896 | 11 | 0.33% | 2 | 1 | \ | 8 | 1 | 10 |
| \ | Sarcophaga (Helicophagella) novercoides Böttcher, 1913 | 11 | 0.33% | 1 | 9 | \ | 1 | 9 | 2 |
| \ | Sarcophaga (Heteronychia) depressifrons Zetterstedt, 1845 | 4 | 0.12% | 4 | \ | \ | \ | \ | 4 |
| \ | Sarcophaga (Liopygia) crassipalpis Macquart, 1839 | 4 | 0.12% | \ | \ | \ | 4 | \ | 4 |
| \ | Sarcophaga (Liosarcophaga) tibialis Macquart, 1851 | 4 | 0.12% | 1 | \ | \ | 3 | \ | 4 |
| \ | Sarcophaga (Heteronychia) siciliana (Enderlein, 1928) | 2 | 0.06% | \ | \ | \ | 2 | \ | 2 |
| \ | Sarcophaga (Myorhina) socrus Rondani, 1860 | 2 | 0.06% | \ | 2 | \ | \ | 2 | \ |
| \ | Sarcophaga (Sarcophaga) lehmanni (Muller, 1922) | 2 | 0.06% | \ | 1 | 1 | \ | 2 | \ |
| \ | Agria affinis (Fallén, 1817) | 2 | 0.06% | 2 | \ | \ | \ | \ | 2 |
| \ | Sarcophaga (Heteronychia) dissimilis Meigen, 1826 | 1 | 0.03% | \ | \ | \ | 1 | \ | 1 |
| \ | Sarcophaga (Pandelleisca) similis Meade, 1876 | 1 | 0.03% | \ | \ | 1 | \ | 1 | 0 |
| \ | Macronychia polyodon (Meigen, 1824) | 1 | 0.03% | 1 | \ | \ | \ | \ | 1 |
| Total | 3284 | 100% | 1147 | 894 | 383 | 860 | 1276 | 2008 | |
| N° species→ | 27 | 21 | 12 | 24 | 23 | 32 | |||
References
- Pape, T.; Blagoderov, V.; Mostovski, M.B. Order Diptera Linnaeus, 1758. In: Zhang, Z.-Q. (Ed.) Animal biodiversity: An outline of higher-level classification and survey of taxonomic richness. Zootaxa 2011, 3148, 222–229. [Google Scholar] [CrossRef]
- Yeates, D.K.; Wiegmann, B.M. Phylogeny and evolution of Diptera: Recent insights and new perspectives. In The Evolutionary Biology of Flies; Yeates, D.K., Wiegmann, B.M., Eds.; Columbia University Press: New York, NY, USA, 2005; pp. 14–44. [Google Scholar]
- Byrd, J.H.; Tomberlin, J.K. Forensic Entomology: The Utility of Arthropods in Legal Investigations, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Skevington, J.H.; Dang, P.T. Exploring the diversity of flies (Diptera). Biodiversity 2002, 3, 3–27. [Google Scholar] [CrossRef]
- Zumpt, F. Myiasis in Man and Animals in the Old World—A Textbook for Physicians, Veterinarians and Zoologists; Butterworth & Co., Ltd.: London, UK, 1965. [Google Scholar]
- Blackith, R.E.; Blackith, R.M. Differential attraction of calyptrate flies (Diptera) to faeces. J. Nat. Hist. 1993, 27, 645–655. [Google Scholar] [CrossRef]
- Scholl, P.J.; Colwell, D.D.; Cepeda-Palacios, R. Myiasis (Muscoidea, Oestroidea). In Medical and Veterinary Entomology, 3rd ed.; Mullen, G.R., Durden, L.A., Eds.; Academic Press: London, UK, 2019; Chapter 19; pp. 383–419. [Google Scholar]
- Nuorteva, P. Synanthropy of blowflies (Dipt., Calliphoridae) in Finland. Ann. Entomol. Fenn. 1963, 29, 1–49. [Google Scholar]
- Linhares, A.X. Synanthropy of Calliphoridae and Sarcophagidae (Diptera) in the city of Campinas, Sao Paulo, Brazil. Rev. Bras. Entomol. 1981, 25, 189–215. [Google Scholar]
- Schnack, J.A.; Mariliuis, J.C.; Centeno, N.; Muzón, J. Composicion específica, ecologia y sinantropia de Calliphoridae (Insecta: Diptera) en el Gran Buenos Aires. Rev. Soc. Entomol. Argent. 1995, 54, 161–171. [Google Scholar]
- Greco, S.; Brandmayr, P.; Bonacci, T. Synanthropy and temporal variability of Calliphoridae living in Cosenza (Calabria, Southern Italy). J. Insect Sci. 2014, 14, 216. [Google Scholar] [CrossRef]
- de Sousa, J.R.P.; Esposito, M.C.; Carvalho Filho, F.D.S.; Juen, L. The potential uses of sarcosaprophagous flesh flies and blowflies for the evaluation of the regeneration and conservation of forest clearings: A case study in the Amazon Forest. J. Insect Sci. 2014, 14, 215. [Google Scholar] [CrossRef]
- Hwang, C.; Turner, B.D. Spatial and temporal variability of necrophagous Diptera from urban to rural areas. Med. Vet. Entomol. 2005, 19, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Martín-Vega, D.; Baz, A. Sarcosaprophagous Diptera assemblages in natural habitats in central Spain: Spatial and seasonal changes in composition. Med. Vet. Entomol. 2013, 27, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Fremdt, H.; Amendt, J. Species composition of forensically important blow flies (Diptera: Calliphoridae) and flesh flies (Diptera: Sarcophagidae) through space and time. Forensic Sci. Int. 2014, 236, 1–9. [Google Scholar] [CrossRef]
- Dufek, M.I.; Larrea, D.D.; Damborsky, M.P.; Mulieri, P.R. The effect of anthropization on Sarcophagidae (Diptera: Calyptratae) community structure: An assessment on different types of habitats in the humid Chaco ecoregion of Argentina. J. Med. Entomol. 2020, 57, 1468–1479. [Google Scholar] [CrossRef]
- Rognes, K. Blowflies (Diptera, Calliphoridae) of Fennoscandia and Denmark (Fauna Entomologica Scandinavica); BRILL: Leiden, The Netherlands, 1991; Volume 24, pp. 1–272. [Google Scholar]
- Pape, T. The Sarcophagidae (Diptera) of Fennoscandia and Denmark (Fauna Entomologica Scandinavica); BRILL: Leiden, The Netherlands, 1987; Volume 19, pp. 1–203. [Google Scholar]
- Povolný, D.; Verves, Y. The Flesh-Flies of Central Europe (Insecta, Diptera, Sarcophagidae); Verlag Dr. Friedrich Pfeil: München, Germany, 1997; Volume 24 Spixiana Supplement, pp. 1–264. [Google Scholar]
- Prado e Castro, C.; García, M.D.; Arnaldos, M.I.; González-Mora, D. Sarcophagidae (Diptera) attracted to piglet carcasses including new records for Portuguese fauna. Graellsia 2010, 66, 285–294. [Google Scholar] [CrossRef]
- Szpila, K.; Mądra, A.; Jarmusz, M.; Matuszewski, S. Flesh flies (Diptera: Sarcophagidae) colonising large carcasses in Central Europe. Parasitol. Res. 2015, 114, 2341–2348. [Google Scholar] [CrossRef]
- Krčmar, S.; Whitmore, D.; Pape, T.; Buenaventura, E. Checklist of the Sarcophagidae (Diptera) of Croatia, with new records from Croatia and other Mediterranean countries. ZooKeys 2019, 831, 95–155. [Google Scholar] [CrossRef] [PubMed]
- Krčmar, S. Diversity of flesh flies (Sarcophagidae, Sarcophaginae) of pond habitats in rural areas in the Croatian part of Baranja. ZooKeys 2023, 1159, 17–36. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sánchez, A.; Rojo, S.; Marcos-García, M.A. Annual and spatial activity of dung flies and carrion in a Mediterranean holm-oak pasture ecosystem. Med. Vet. Entomol. 2000, 14, 56–63. [Google Scholar] [CrossRef]
- Meoli, M.; Cherix, D.; Wyss, C. Contributo alla conoscenza delle mosche necrofaghe (Diptera, Calliphoridae) del sud delle Alpi (Ticino, Svizzera). Boll. Soc. Ticin. Sci. Nat. 2004, 92, 91–96. [Google Scholar]
- Baz, A.; Cifrián, B.; Díaz-äranda, L.M.; Martín-Vega, D. The distribution of adult blow-flies (Diptera: Calliphoridae) along an altitudinal gradient in Central Spain. Ann. Soc. Entomol. Fr. (N.S.) 2007, 43, 289–296. [Google Scholar] [CrossRef]
- Matuszewski, S.; Bajerlein, D.; Konwerski, S.; Szpila, K. Insect succession and carrion decomposition in selected forests of Central Europe. Part 2: Composition and residency patterns of carrion fauna. Forensic Sci. Int. 2010, 195, 42–51. [Google Scholar] [CrossRef]
- Prado e Castro, C.; Serrano, A.; Martins Da Silva, P.; García, M.D. Carrion flies of forensic interest: A study of seasonal community composition and succession in Lisbon, Portugal. Med. Vet. Entomol. 2012, 26, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Feddern, N.; Amendt, J.; Schyma, C.; Jackowski, C.; Tschui, J. A preliminary study about the spatiotemporal distribution of forensically important blow flies (Diptera: Calliphoridae) in the area of Bern, Switzerland. Forensic Sci. Int. 2018, 289, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Arias-Robledo, G.; Stevens, J.R.; Wall, R. Spatial and temporal habitat partitioning by calliphorid blowflies. Med. Vet. Entomol. 2019, 33, 228–237. [Google Scholar] [CrossRef]
- Demirözer, O.; Pekbey, G.; Hayat, R.; Herdoğan, A.; Acanski, J.; Milicic, M.; Uzun, A. First contribution on distribution, abundance, and species richness of blowfly species (Diptera) of Isparta Province with five new records for the Turkish fauna. Turk. J. Zool. 2020, 44, 69–79. [Google Scholar] [CrossRef]
- Ourrad, O.; Ahmed, D.S.A.; Sadou, S.A.; Bouzrarf, K.; Belqat, B. Diversity of flies (Diptera: Brachycera) in breeding farms in the Kabylia region (North-central Algeria), and identification of some myiaseginic species. Biodiversitas J. Biol. Divers. 2022, 23, 2276–2284. [Google Scholar] [CrossRef]
- Hodecek, J.; Jakubec, P. Spatio-temporal distribution and habitat preference of necrophagous Calliphoridae based on 160 real cases from Switzerland. Int. J. Leg. Med. 2022, 136, 923–934. [Google Scholar] [CrossRef]
- Pérez-Marcos, M.; Ruiz-Franco, L.; García, M.D.; López-Gallego, E.; Arnaldos, M.I. The influence of altitude on the abundance and occurrence of species of the family Calliphoridae (Diptera) in corpses in a Mediterranean area. J. Forensic Sci. 2024, 69, 944–952. [Google Scholar] [CrossRef]
- Corotti, S.; Dattrino, F.; Boulkenafet, F.; Lambiase, S. Altitudinal variations in forensically relevant dipterans in Trentino Region (Italy): Implications for PMI estimation and forensic ecology. Forensic Sci. Med. Pathol. 2025, 21, 610–623. [Google Scholar] [CrossRef]
- Gemmellaro, M.D.; Anderson, G.S.; Hamilton, G.C.; Domínguez-Trujillo, M.; Weidner, L.M. Species richness and distribution of Calliphoridae along an elevation gradient in Sicily (Italy) and Ecuador. Insects 2025, 16, 498. [Google Scholar] [CrossRef] [PubMed]
- Bezzi, M. Contribuzioni alla fauna ditterologica italiana. Ditteri della Calabria. Bull. Soc. Entomol. Ital. 1895, 27, 39–78. [Google Scholar]
- Venturi, F. Sistematica e geonemia dei Sarcophagidi (excl. Sarcophaga Meig. s. l.) italiani (Diptera). Frustula Entomol. 1960, 2, 1–124. [Google Scholar]
- Whitmore, D.; Greco, S.; Bonacci, T. Nuovi dati su ditteri del genere Sarcophaga in Calabria (Diptera: Sarcophagidae). In Proceedings of the 11° Convegno Nazionale sulla Biodiversità, Matera, Italy, 9–10 June 2016. [Google Scholar]
- Bonacci, T.; Brandmayr, P.; Greco, S.; Tersaruolo, C.; Vercillo, V.; Zetto Brandmayr, T. A preliminary investigation of insect succession on carrion in Calabria (southern Italy). Terr. Arthropod Rev. 2010, 3, 97–110. [Google Scholar] [CrossRef]
- Bonacci, T.; Zetto Brandmayr, T.; Brandmayr, P.; Vercillo, V.; Porcelli, F. Successional patterns of the insect fauna on a pig carcass in southern Italy and the role of Crematogaster scutellaris (Hymenoptera, Formicidae) as a carrion invader. Entomol. Sci. 2011, 14, 125–132. [Google Scholar] [CrossRef]
- Bonacci, T.; Greco, S.; Cavalcanti, B.; Brandmayr, P.; Vercillo, V. The flesh fly Sarcophaga (Liopygia) crassipalpis Macquart 1839 as an invader of a corpse in Calabria (Southern Italy). J. Forensic Sci. Criminol. 2014, 2, 1–5. [Google Scholar] [CrossRef]
- Bonacci, T.; Brandmayr, P. Primi dati sui ditteri che causano miasi canine in Calabria. In Proceedings of the 25th Italian National Congress of Entomology, Padua, Italy, 20–24 June 2016; p. 315. [Google Scholar]
- Bonacci, T.; Mendicino, F.; Bonelli, D.; Carlomagno, F.; Curia, G.; Scapoli, C.; Pezzi, M. Investigations on arthropods associated with decay stages of buried animals in Italy. Insects 2021, 12, 311. [Google Scholar] [CrossRef]
- Pezzi, M.; Bonelli, D.; Mendicino, F.; Carlomagno, F.; Munari, C.; Mistri, M.; Chicca, M.; Szpila, K.; Bonacci, T. Calliphora rohdendorfi (Grunin, 1966) (Diptera: Calliphoridae): A new blow fly in the Italian fauna detected in Calabrian Apennines. Eur. Zool. J. 2024, 91, 395–405. [Google Scholar] [CrossRef]
- Peris, S.V.; Gonzáles-Mora, D. About Calliphora and its allies (Diptera). Eos 1989, 65, 165–201. [Google Scholar]
- Rognes, K. Blowflies (Diptera, Calliphoridae) of Israel and adjacent areas, including a new species from Tunisia. In Entomologica Scandinavica Supplements; Scandinavian Society of Entomology: Lund, Sweden, 2002; Volume 59, pp. 1–148. [Google Scholar]
- Lehrer, A.Z. Sarcophaginae de l’Afrique (Insecta, Diptera, Sarcophagidae). Entomologica 2003, 37, 5–528. [Google Scholar]
- Whitmore, D. A review of the Sarcophaga (Heteronychia) (Diptera: Sarcophagidae) of Sardinia. Zootaxa 2009, 2318, 566–588. [Google Scholar] [CrossRef]
- Richet, R.; Blackith, R.M.; Pape, T. Sarcophaga of France; Pensoft Publishers: Sofia, Bulgaria, 2011; p. 327. [Google Scholar]
- Falk, S. British Blow Flies (Calliphoridae) and Woodlouse Flies (Rhinophoridae). Draft Key to British Calliphoridae and Rhinophoridae Steven Falk. 2016. Available online: http://www.stevenfalk.co.uk/files/21577/testkeytobritishblowflies132016.pdf (accessed on 15 February 2022).
- Whitmore, D.; Dupont, S.; Falk, S. Key to Adult Flesh Flies (Diptera: Sarcophagidae) of the British Isles. 2020. Available online: https://osf.io/preprints/osf/vf5r6_v1 (accessed on 14 February 2022).
- Szpila, K. Key for identification of European and Mediterranean blowflies (Diptera, Calliphoridae) of medical and veterinary importance—Adult flies. In Forensic Entomology, an Introduction, 2nd ed.; Gennard, D., Ed.; Wiley-Blackwell: Oxford, UK, 2012; pp. 77–81. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 4. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 16 January 2023).
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- ter Braak, C.J.F. Ordination. In Data Analysis in Community and Landscape Ecology; Jongman, R.H.G., ter Braak, C.J.F., van Tongeren, O.F.R., Eds.; Centre for Agricultural Publishing and Documentation: Wageningen, The Netherlands, 1987; pp. 91–173. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology, Developments in Environmental Modelling, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 1998; Volume 20, p. 853. [Google Scholar]
- Dufrêne, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- De Cáceres, M.; Sol, D.; Lapiedra, O.; Legendre, P. A framework for estimating niche metrics using the resemblance between qualitative resources. Oikos 2011, 120, 1341–1350. [Google Scholar] [CrossRef]
- Easterbrook, D.J. Surface Processes and Landforms, 2nd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1999. [Google Scholar]
- Merritt, R.W.; Courtney, G.W.; Keiper, J.B. Diptera. In Encyclopedia of Insects, 2nd ed.; Resh, V.H., Cardé, R.T., Eds.; Academic Press, Elsevier: Amsterdam, The Netherlands, 2009; pp. 284–297. [Google Scholar]
- Greenberg, B. Flies and disease. Sci. Am. 1965, 213, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Amendt, J.; Krettek, R.; Zehner, R. Forensic entomology. Naturwissenschaften 2004, 91, 51–65. [Google Scholar] [CrossRef]
- Cherix, D.; Wyss, C.; Pape, T. Occurrences of flesh flies (Diptera: Sarcophagidae) on human cadavers in Switzerland, and their importance as forensic indicators. Forensic Sci. Int. 2012, 220, 158–163. [Google Scholar] [CrossRef]
- Ren, L.; Shang, Y.; Chen, W.; Meng, F.; Cai, J.; Zhu, G.; Chen, L.; Wang, Y.; Deng, J.; Guo, Y. A brief review of forensically important flesh flies (Diptera: Sarcophagidae). Forensic Sci. Res. 2018, 3, 16–26. [Google Scholar] [CrossRef]
- Pezzi, M.; Bonacci, T.; Leis, M.; Mamolini, E.; Marchetti, M.G.; Krčmar, S.; Chicca, M.; Del Zingaro, C.N.F.; Faucheux, M.J.; Scapoli, C. Myiasis in domestic cats: A global review. Parasites Vectors 2019, 12, 372. [Google Scholar] [CrossRef]
- Pezzi, M.; Scapoli, C.; Chicca, M.; Leis, M.; Marchetti, M.G.; Del Zingaro, C.N.F.; Vicentini, C.B.; Mamolini, E.; Giangaspero, A.; Bonacci, T. Cutaneous myiasis in cats and dogs: Cases, predisposing conditions and risk factors. Vet. Med. Sci. 2021, 7, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Zabala, J.; Díaz, B.; Saloña-Bordas, M.I. Seasonal blowfly distribution and abundance in fragmented landscapes. Is it useful in forensic inference about where a corpse has been decaying? PLoS ONE 2014, 9, e99668. [Google Scholar] [CrossRef]
- MacLeod, J.; Donnelly, J. Some ecological relationships of natural populations of Calliphorine blowflies. J. Anim. Ecol. 1957, 26, 135–170. [Google Scholar] [CrossRef]
- McCain, C.M.; Garfinkel, C.F. Climate change and elevational range shifts in insects. Curr. Opin. Insect Sci. 2021, 47, 111–118. [Google Scholar] [CrossRef]
- Wall, R.; French, N.P.; Morgan, K.L. Blowfly species composition sheep myiasis in Britain. Med. Vet. Entomol. 1992, 6, 177–178. [Google Scholar] [CrossRef]
- Vanin, S.; Tasinato, P.; Ducolin, G.; Terranova, C.; Zancaner, S.; Montisci, M.; Ferrara, S.D.; Turchetto, M. Use of Lucilia species for forensic investigations in Southern Europe. Forensic Sci. Int. 2008, 177, 37–41. [Google Scholar] [CrossRef]
- MacLeod, J. A survey of British sheep blowflies. Bull. Entomol. Res. 1943, 34, 65–88. [Google Scholar] [CrossRef]
- Pinilla Beltran, Y.T.; Segura, N.A.; Bello, F.J. Synanthropy of Calliphoridae and Sarcophagidae (Diptera) in Bogotá, Colombia. Neotrop. Entomol. 2012, 41, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.-S.; Baek, I.-S.; Kang, M.-G.; Park, S.-H. Spatio-temporal influence on the distribution of forensically relevant blowflies (Diptera: Calliphoridae) in Gyeongsangnam-do, South Korea. Insects 2024, 15, 536. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Filho, S.J.M.; dos Santos Lobato, F.; Medeiros de Abreu, C.H.; Rebelo, M.T. Where in Europe is Chrysomya albiceps? Modelling present and future potential distributions. NeoBiota 2023, 85, 81–99. [Google Scholar] [CrossRef]
- de Carvalho Queiroz, M.M. Temperature requirements of Chrysomya albiceps (Wiedemann, 1819) (Diptera, Calliphoridae) under laboratory conditions. Mem. Inst. Oswaldo Cruz 1996, 91, 785–788. [Google Scholar] [CrossRef]
- Grassberger, M.; Friedrich, E.; Reiter, C. The blowfly Chrysomya albiceps (Wiedemann) (Diptera: Calliphoridae) as a new forensic indicator in Central Europe. Int. J. Leg. Med. 2003, 117, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Introna, F., Jr.; Campobasso, C.P.; Di Fazio, A. Three case studies in forensic entomology from Southern Italy. J. Forensic Sci. 1998, 43, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Grassberger, M.; Reiter, C. Effect of temperature on development of the forensically important holarctic blow fly Protophormia terraenovae (Robineau-Desvoidy) (Diptera: Calliphoridae). Forensic Sci. Int. 2002, 128, 177–182. [Google Scholar] [CrossRef]
- Pereira de Sousa, J.R.; Esposito, M.C.; da Silva Carvalho Filho, F. Diversity of Calliphoridae and Sarcophagidae (Diptera, Oestroidea) in continuous forest and gaps at different stages of regeneration in the Urucu oilfield in western Brazilian Amazonia. Rev. Bras. Entomol. 2011, 55, 578–582. [Google Scholar] [CrossRef]
- Mendes, T.P.; Esposito, M.C.; da Silva Carvalho-Filho, F.; Juen, L.; Alvarado, S.T.; Pereira de Sousa, J.R. Necrophagous flies (Diptera: Calliphoridae and Sarcophagidae) as indicators of the conservation or anthropization of environments in eastern Amazonia, Brazil. J. Insect Conserv. 2021, 25, 719–732. [Google Scholar] [CrossRef]
- Mulieri, P.R.; Dufek, M.I.; Dos Santos, J.R.; Torres-Domínguez, D.M.; Patitucci, L.D. Effect of vegetation types on Sarcophagidae (Diptera) in Ciervo de los Pantanos National Park, a natural remnant in a highly endangered landscape. J. Med. Entomol. 2022, 59, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Pezzi, M.; Whitmore, D.; Chicca, M.; Lanfredi, M.; Leis, M. Traumatic myiasis caused by an association of Sarcophaga tibialis (Diptera: Sarcophagidae) and Lucilia sericata (Diptera: Calliphoridae) in a domestic cat in Italy. Korean J. Parasitol. 2015, 53, 471–475. [Google Scholar] [CrossRef] [PubMed]


| Variables | Code | Description |
|---|---|---|
| Altitude (220 m to 1820 m) | a.s.l. m | Altitudinal gradient of the 17 sampling sites. |
| Grazing degree (0 to 2) | Graz_ | Grazing activity was estimated through direct observation and the manipulation of traps at and around the sites. The following scale was used to categorize the level of grazing activity observed: 0 = no activity; 1 = sporadic presence of grazing animals during a few short periods throughout the year; 2 = grazing activity occurring for approximately half of the year. |
| Human activities degree (1 to 4) | Act_ | The presence of human activity, population centers, and primary roads with moderate or intense traffic was observed around sites. This was assessed using satellite imagery with 500 × 500 m grids, as outlined in Hwang and Turner [13]. |
| Shade degree (0 to 4) | Shade | The approximate level of shade around the traps was visually assessed based on tree cover, using the following scale: 0 = fully exposed; 1 = partially shaded; 2 = moderately shaded; 3 = heavily shaded; 4 = fully shaded and constant. |
| Forest cover index (0 to 100%) | Woo_ind | The proportion of the area covered by forest relative to the total area of a given territory, expressed as a percentage. The calculations were performed using the wooded surface layers of the areas under study (Qgis software). |
| Average seasonal temperature (°C) | t °C | The seasonal average temperature in Celsius degrees for each site was obtained through consultations with ARPCAL climate monitoring stations. |
| Average seasonal rainfall (P/mm) | P/mm | The average seasonal rainfall for each site was obtained through consultations with ARPCAL climate monitoring station. |
| Sites | Minimum T (°C) | Maximum T (°C) | Mean T (°C) | Mean P (mm) |
|---|---|---|---|---|
| 2As, 5As | −6.8 | 32.7 | 11.6 | 159.2 |
| 1As, 3As, 4As | −6.6 | 32.2 | 11 | 137.5 |
| 5Si | −12.6 | 35.9 | 9.3 | 76.4 |
| 1Si, 2Si, 3Si | −14.3 | 31.5 | 7.17 | 100.6 |
| 4Si, 6Si | −14.9 | 28 | 6.5 | 101.2 |
| 1Se, 3Se, 5Se | −6.8 | 36.4 | 12 | 108.3 |
| 2Se, 4Se | −5.4 | 37.1 | 12 | 116.9 |
| 1Re | −2.1 | 42.7 | 16 | 79.1 |
| Calliphoridae | Sarcophagidae | |
|---|---|---|
| p-Value | p-Value | |
| Spring vs. Summer | 0.0144 | 0.0006 |
| Spring vs. Autumn | 0.0024 | 1 |
| Spring vs. Winter | 0.0006 | 0.0012 |
| Summer vs. Autumn | 0.0006 | 0.0006 |
| Summer vs. Winter | 0.0006 | 0.0006 |
| Autumn vs. Winter | 0.0384 | 0.0054 |
| Calliphoridae | Sarcophagidae | |
|---|---|---|
| p-Value | p-Value | |
| Hilly vs. Submontane | 0.0081 | 0.1038 |
| Hilly vs. Montane | 0.0009 | 0.0003 |
| Submontane vs. Montane | 0.1149 | 0.0084 |
| Calliphoridae | Sarcophagidae | |
|---|---|---|
| p-Value | p-Value | |
| WL vs. PF | 0.0024 | 0.303 |
| WL vs. BF | 0.0264 | 0.0264 |
| WL vs. SM | 0.0312 | 0.3234 |
| SM vs. PF | 0.0012 | 0.0462 |
| SM vs. BF | 0.0018 | 0.003 |
| BF vs. PF | 1 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonelli, D.; Mendicino, F.; Carlomagno, F.; Luzzi, G.; Siclari, A.; Fumo, F.; Di Biase, E.; Mistri, M.; Whitmore, D.; Munari, C.; et al. Ecological Preferences of Calliphoridae and Sarcophagidae (Diptera) in the Region Calabria (Southern Italy). Insects 2025, 16, 886. https://doi.org/10.3390/insects16090886
Bonelli D, Mendicino F, Carlomagno F, Luzzi G, Siclari A, Fumo F, Di Biase E, Mistri M, Whitmore D, Munari C, et al. Ecological Preferences of Calliphoridae and Sarcophagidae (Diptera) in the Region Calabria (Southern Italy). Insects. 2025; 16(9):886. https://doi.org/10.3390/insects16090886
Chicago/Turabian StyleBonelli, Domenico, Federica Mendicino, Francesco Carlomagno, Giuseppe Luzzi, Antonino Siclari, Federica Fumo, Erica Di Biase, Michele Mistri, Daniel Whitmore, Cristina Munari, and et al. 2025. "Ecological Preferences of Calliphoridae and Sarcophagidae (Diptera) in the Region Calabria (Southern Italy)" Insects 16, no. 9: 886. https://doi.org/10.3390/insects16090886
APA StyleBonelli, D., Mendicino, F., Carlomagno, F., Luzzi, G., Siclari, A., Fumo, F., Di Biase, E., Mistri, M., Whitmore, D., Munari, C., Pezzi, M., & Bonacci, T. (2025). Ecological Preferences of Calliphoridae and Sarcophagidae (Diptera) in the Region Calabria (Southern Italy). Insects, 16(9), 886. https://doi.org/10.3390/insects16090886












