Chemical Composition of Four Industrial Hemp (Cannabis sativa L.) Pollen and Bee Preference
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
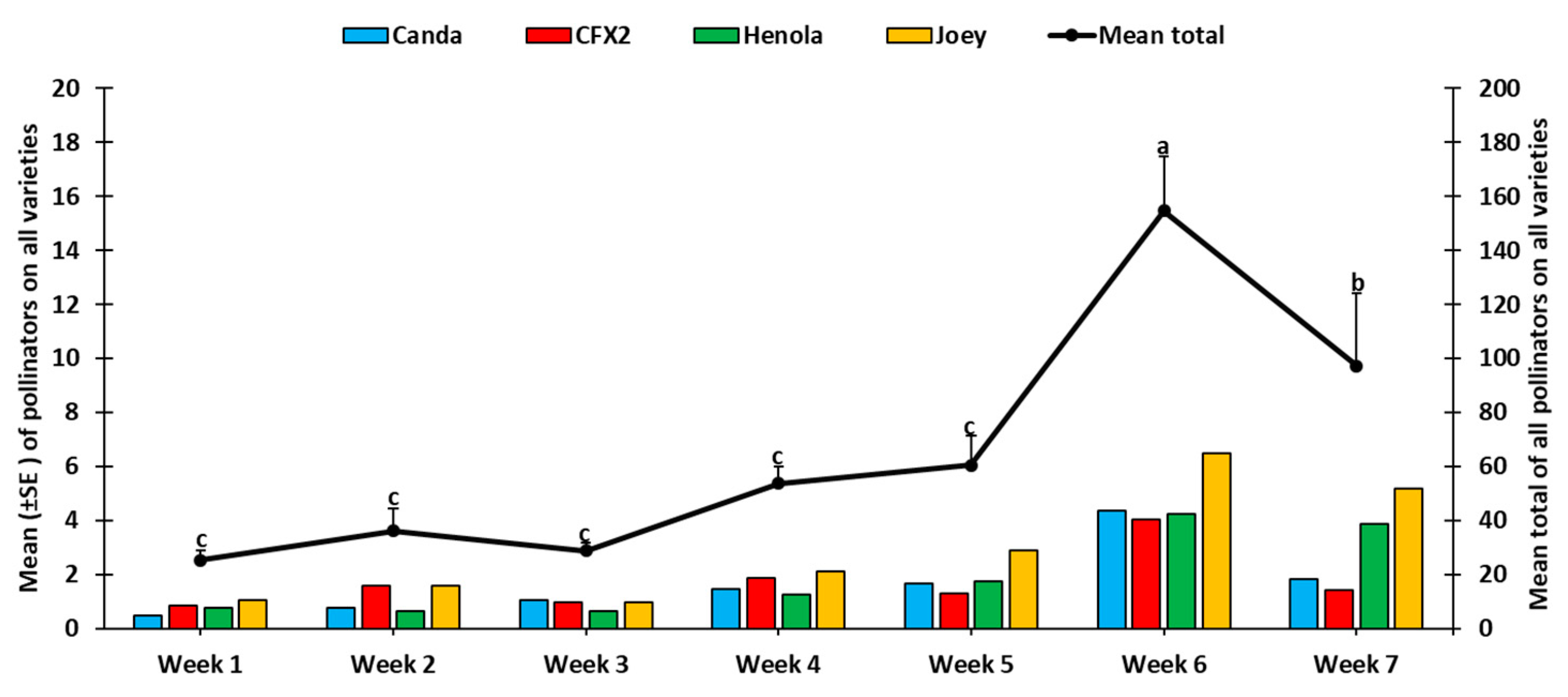
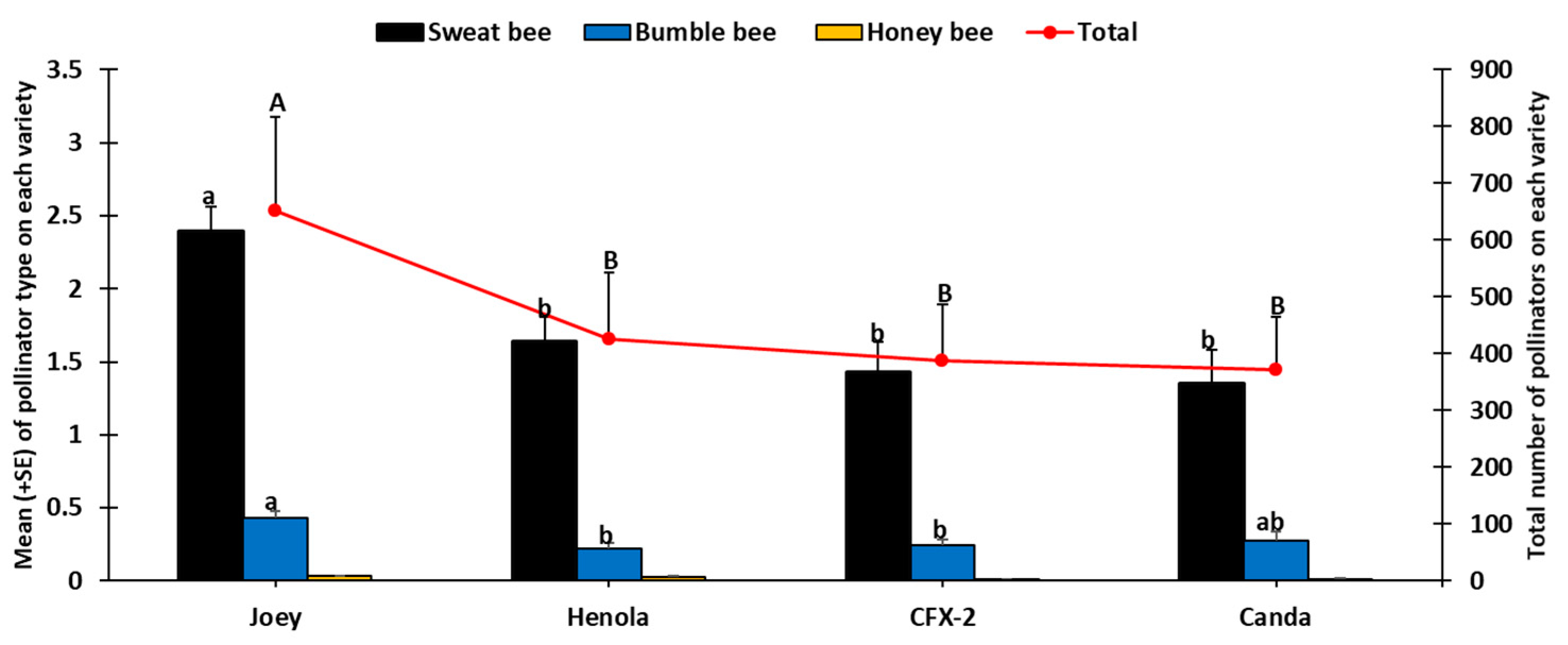
2.2. Insect Sampling
2.2.1. Direct Visual Counts
2.2.2. Assessment of Pollinators Using Pan Traps
2.3. Pollen Sample Collection and Analysis
2.3.1. Pollen Collection
2.3.2. Chemical Analysis
- 1
- Proximate chemical composition
- 2
- Mineral content
- 3
- Amino acid composition
- 4
- Fatty acid composition
2.4. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kraenzel, D.G.; Petry, T.; Nelson, B.; Anderson, M.J.; Mathern, D.; Todd, R. Industrial Hemp as an Alternative Crop in North Dakota: A White Paper Study of the Markets, Profitability, Processing, Agronomics and History; Agricultural Economics Report No. 402; The Institute for Natural Resources and Economic Development, North Dakota State University: Fargo, ND, USA, 1998. [Google Scholar]
- Johnson, R. Hemp as an Agricultural Commodity. Congressional Research Service. Available online: http://nationalaglawcenter.org/wp-content/uploads/assets/crs/RL32725.pdf (accessed on 3 March 2023).
- Burton, R.A.; Andres, M.; Cole, M.; Cowley, J.M.; Augustin, M.A. Industrial hemp seed: From the field to value-added food ingredients. J. Cannabis Res. 2022, 4, 45. [Google Scholar] [CrossRef]
- Small, E.; Marcus, D. Hemp—A new crop with new uses for North America. In Trends in New Crops and New Uses; Janick, J., Whipkey, A., Eds.; ASHS Press: Alexandria, Egypt, 2002; pp. 284–326. [Google Scholar]
- Cerino, P.; Buonerba, C.; Cannazza, G.; D’Auria, J.; Ottoni, E.; Fulgione, A.; Di Stasio, A.; Pierri, B.; Gallo, A. A review of hemp as food and nutritional supplement. Cannabis Cannabinoid Res. 2021, 6, 19–27. [Google Scholar] [CrossRef] [PubMed]
- USDA-NASS. Agricultural Statistics 2022; National Hemp Report; USDA-NASS: Washington, DC, USA, 2022. [Google Scholar]
- O’Brien, C.; Arathi, H.S. Bee diversity and abundance in flowering hemp (Cannabis sativa L.). Biomass Bioenergy 2019, 122, 331–335. [Google Scholar]
- Nicolson, S.W.; Nepi, M.; Pacini, E.; Nicolson, S.W.; Nepi, M.; Pacini, E. Nectaries and Nectar; Springer Science and Business Media: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Nicolson, S.W. Bee food: The chemistry and nutritional value of nectar, pollen and mixtures of the two. Afr. Zool. 2011, 46, 197–204. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Tooker, J.F.; Grozinger, C.M.; Patch, H.M. Bee nutrition and floral resource restoration. Curr. Opin. Insect Sci. 2015, 10, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, M.T.O.; Marquina, A.D.; Serrano, R.B.; Abell, N.G.B. The importance of bee-collected pollen in the diet: A study of its composition. Int. J. Food Sci. 2002, 53, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Margaoan, R.; Marghiatas, L.A.; Dezmirean, D.; Mihai, C.M.; Bobis, O. Bee collected pollen general aspects and chemical composition. Anim. Sci. Biotechnol. 2010, 67, 254–259. [Google Scholar]
- Cane, J.H. Adult pollen diet essential for egg maturation by a solitary Osmia bee. J. Insect Physiol. 2016, 95, 105–109. [Google Scholar]
- Cane, J.H.; Dobson, H.E.M.; Boyer, B. Timing and size of daily pollen meals eaten by adult females of a solitary bee (Nomia melanderi) (Apiformes: Halictidae). Apidologie 2016, 48, 17–30. [Google Scholar] [CrossRef]
- Roulston, T.H.; Cane, J.H. Pollen nutritional content and digestibility for animals. Plant Syst. Evol. 2000, 222, 187–209. [Google Scholar] [CrossRef]
- Weiner, C.N.; Hilpert, A.; Werner, M.; Linsenmair, K.E.; Blüthgen, N. Pollen amino acids and flower specialisation in solitary bees. Apidologie 2010, 41, 476–487. [Google Scholar] [CrossRef]
- Arien, Y.; Dag, A.; Zarchin, S.; Masci, T.; Shafir, S. Omega-3 deficiency impairs honey bee learning. Proc. Natl. Acad. Sci. USA 2015, 112, 15761–15766. [Google Scholar] [CrossRef] [PubMed]
- Vaudo, A.D.; Patch, H.M.; Mortensen, D.A.; Tooker, J.F.; Grozinger, C.M. Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proc. Natl. Acad. Sci. USA 2016, 113, E4035–E4042. [Google Scholar] [CrossRef] [PubMed]
- Russo, L.; Vaudo, A.D.; Fisher, C.J.; Grozinger, C.M.; Shea, K. Bee community preference for an invasive thistle associated with higher pollen protein content. Oecologia 2019, 190, 901–912. [Google Scholar] [CrossRef] [PubMed]
- Treanore, E.D.; Vaudo, A.D.; Grozinger, C.M.; Fleischer, S.J. Examining the nutritional value and effects of different floral resources in pumpkin agroecosystems on Bombus impatiens worker physiology. Apidologie 2019, 50, 542–552. [Google Scholar] [CrossRef]
- Buchmann, S.L. Vibratile pollination in Solanum and Lycopersicon: A look at pollen chemistry. In Solanaceae II: Biology and Systematics; D’Arcy, W.G., Ed.; Columbia University Press: New York, NY, USA, 1986; pp. 237–252. [Google Scholar]
- Ghazoul, J. Buzziness as usual? Questioning the global pollination crisis. Trends Ecol. Evol. 2005, 20, 367–373. [Google Scholar] [CrossRef]
- Steffan-Dewenter, I.; Potts, S.G.; Packer, L.J.T. Pollinator diversity and crop pollination services are at risk. Trends Ecol. Evol. 2005, 20, 651–652. [Google Scholar] [CrossRef]
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemuller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef]
- Williams, P.; Osborne, J. Bumblebee vulnerability and conservation world-wide. Apidologie 2009, 40, 367–387. [Google Scholar] [CrossRef]
- Cameron, S.A.; Lozier, J.D.; Strange, J.P.; Koch, J.B.; Cordes, N.; Solder, L.F.; Griswold, T.L. Patterns of widespread decline in North American bumble bees. Proc. Natl. Acad. Sci. USA 2011, 108, 662–667. [Google Scholar] [CrossRef]
- FDA—Food and Drug Administration. Helping Agriculture’s Helpful Honey Bees; FDA: Silver Spring, MD, USA, 2018; p. 11. [Google Scholar]
- Kremen, C.; Williams, N.M.; Thorp, R.W. Crop pollination from native bees at risk from agricultural intensification. Proc. Natl. Acad. Sci. USA 2002, 99, 16812–16816. [Google Scholar] [CrossRef]
- Kleijn, D.; Raemakers, I. A retrospective analysis of pollen host plant use by stable and declining bumble bee species. Ecology 2008, 89, 1811–1823. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [PubMed]
- Desneux, N.; Decourtye, A.; Delpuech, J.-M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Winfree, R.; Aguilar, R.; Vázquez, D.; LeBuhn, G.; Aizen, M.A. Meta-analysis of bees’ response to anthropogenic disturbance. Ecology 2009, 90, 2068–2076. [Google Scholar] [CrossRef]
- Cane, J.H. Habitat fragmentation and native bees: A premature verdict? Conser. Ecol. 2001, 5, 3. [Google Scholar]
- Schweiger, O.; Biesmeijer, J.C.; Bommarco, R.; Hickler, T.; Hulme, P.E.; Klotz, S.; Kühn, I.; Moora, M.; Nielsen, A.; Ohlemüller, R.; et al. Multiple stressors on biotic interactions: How climate change and alien species interact to affect pollination. Biol. Rev. 2010, 85, 777–795. [Google Scholar] [CrossRef] [PubMed]
- González-Varo, J.P.; Biesmeijer, J.C.; Bommarco, R.; Potts, S.G.; Schweiger, O.; Smith, H.G.; Steffan-Dewenter, I.; Szentgyörgyi, H.; Woyciechowski, M.; Vilà, M. Combined effects of global change pressures on animal-mediated pollination. Trends Ecol. Evol. 2013, 28, 524–530. [Google Scholar]
- Olsen, D.W.; Thornsbury, S.D.; Scott, S. Hope for Hemp: New Opportunities and Challenges for an Old Crop. In Amber Waves: The Economics of Food, Farming, Natural Resources, and Rural America; United States Department of Agriculture, Economic Research Service: Washington, DC, USA, 2020. [Google Scholar]
- Dingha, B.N.; Jackai, L.E.; Amoah, B.A.; Akotsen-Mensah, C. Pollinators on cowpea Vigna unguiculata: Implications for intercropping to enhance biodiversity. Insects 2021, 12, 54. [Google Scholar] [CrossRef]
- Dingha, B.N.; Omaliko, P.C.; Amoah, B.A.; Jackai, L.E.; Shrestha, D. Evaluation of cowpea (Vigna unguiculata) in an intercropping system as pollinator enhancer for increased crop yield. Sustainability 2021, 13, 9612. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemist. Methods 925.09 and 926.08. In Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Association of Official Analytical Chemist. Method 923.03. In Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Association of Official Analytical Chemist. Protein (Crude) in Animal Feed (Dumas Method), Method 968.06. In Official Methods of Analysis of AOAC International; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Association of Official Analytical Chemist. Methods 962.09. In Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Association of Official Analytical Chemist. Method 984.27, 985.01, and 2011.14. In Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Henderson, J.W.; Brooks, A. Improved Amino Acid Methods Using Agilent Zorbax Eclipse Plus C18 Columns for a Variety of Agilent LC Instrumentation and Separation Goals; Agilent Application Note 5990-4547; Angilent Technologist, Inc.: Wilmington, DE, USA, 2010. [Google Scholar]
- Association of Official Analytical Chemist. Method No. 996.06. In Official Methods of Analysis of AOAC International, 19th ed.; AOAC International: Gaithersburg, MD, USA, 2012. [Google Scholar]
- Association of Official Analytical Chemist. Methods Ce 2b-11 (2011), Ce 1h-05 (2009), Ce 1j-07 (2013), Ce 2-66 (2009). In Official Methods and Recommended Practices of the AOCS International; The American Oil Chemists’ Society: Champaign, IL, USA, 2020. [Google Scholar]
- Lago, P.K.; Stanford, D.F. Phytophagous insects associated with cultivated marijuana (Cannabis sativa L.) in northern Mississippi. J. Entomol. Sci. 1989, 24, 437–445. [Google Scholar]
- Flicker, N.R.; Poveda, K.; Grab, H. The bee community of Cannabis sativa and corresponding effects of landscape composition. Environ. Entomol. 2020, 49, 197–202. [Google Scholar]
- Roulston, T.H.; Smith, S.A.; Brewster, A.L. A comparison of pan trap and intensive net sampling techniques for documenting a bee (Hymenoptera: Apiformes) Fauna. J. Kans. Entomol. Soc. 2007, 80, 179–181. [Google Scholar]
- Larsen, N.J.; Minor, M.A.; Cruickshank, R.H.; Robertson, A.W. Optimising methods for collecting Hymenoptera, including parasitoids and Halictidae bees, in New Zealand apple orchards. J. Asia Pac. Entomol. 2014, 17, 375–381. [Google Scholar]
- Wheelock, M.J.; O’Neal, M.E. Insect pollinators in Iowa cornfields: Community identification and trapping method analysis. PLoS ONE 2016, 11, e0143479. [Google Scholar]
- Adamson, N.L.; Roulston, T.H.; Fell, R.D.; Mullins, D.E. From April to August—Wild bees pollinating crops through the growing season in Virginia, USA. Environ. Entomol. 2012, 41, 813–821. [Google Scholar]
- Gollan, R.J.; Ashcroft, M.B.; Batley, M. Comparison of yellow and white pan traps in surveys of bee fauna in New South Wales, Australia (Hymenoptera: Apoidea: Anthophila). Aust. J. Entomol. 2011, 50, 174–178. [Google Scholar]
- Garibaldi, L.A.; Steffan-Dewenter, I.; Winfree, R.; Aizen, M.A.; Bommarco, R.; Cunningham, S.A.; Kremen, C.; Carvalheiro, L.G.; Harder, L.D.; Afik, O.; et al. Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 2013, 339, 1608–1611. [Google Scholar] [PubMed]
- Chagnon, M.; Gingras, J.; De Oliveira, D. Complementary aspects of strawberry pollination by honey and indigenous bees (Hymenoptera). J. Econ. Entomol. 1993, 86, 416–420. [Google Scholar]
- Fründ, J.; Dormann, C.F.; Holzschuh, A.; Tscharntke, T. Bee diversity effects on pollination depend on functional complementarity and niche shifts. Ecology 2013, 94, 2042–2054. [Google Scholar] [PubMed]
- Roulston, T.H.; Cane, J.H.; Buchmann, S.L. What governs protein content of pollen: Pollinator preferences, pollen-pistil interactions, or phylogeny? Ecol. Monogr. 2000, 70, 617–643. [Google Scholar]
- Rasheed, S.A.; Harder, L.D. Economic motivation for plant species preference of pollen-collecting bumble bees. Ecol. Entomol. 1997, 22, 209–219. [Google Scholar]
- Robertson, A.W.; Mountjoy, C.; Faulkner, B.E.; Roberts, M.V.; Macnair, M.R. Bumble bee selection of Mimulus guttatus flowers: The effects of pollen quality and reward depletion. Ecology 1999, 80, 2594–2606. [Google Scholar]
- Hanley, M.E.; Franco, M.; Pichon, S.; Darvill, B.; Goulson, D. Breeding system, pollinator choice and variation in pollen quality in British herbaceous plants. Funct. Ecol. 2008, 22, 592–598. [Google Scholar]
- Kitaoka, T.K.; Nieh, J.C. Bumble bee pollen foraging regulation: Role of pollen quality, storage levels, and odor. Behav. Ecol. Sociobiol. 2009, 63, 625. [Google Scholar]
- Leonhardt, S.D.; Blüthgen, N. The same, but different: Pollen foraging in honeybee and bumblebee colonies. Apidologie (Celle) 2012, 43, 449–464. [Google Scholar]
- Laurent Somme, L.; Vanderplanck, M.; Michez, D.; Lombaerde, I.; Moerman, R.; Wathelet, B.; Wattiez, R.; Lognay, G.; Jacquemart, A.L. Pollen and nectar quality drive the major and minor floral choices of bumble bees. Apidologie (Celle) 2015, 46, 92–106. [Google Scholar]
- Ghosh, S.; Jeon, H.; Jung, C. Foraging behaviour and preference of pollen sources by honey bee (Apis mellifera) relative to protein contents. J. Ecol. Environ. 2020, 44, 4. [Google Scholar]
- Moezel, P.G.; Delfs, J.C.; Pate, J.S.; Loneragan, W.A.; Bell, D.T. Pollen selection by honeybees in shrublands of the Northern Sandplains of Western Australia. J. Apic. Res. 1987, 26, 224–232. [Google Scholar]
- Pernal, S.F.; Currie, R.C. Discrimination and preferences for pollen-based cues by foraging honeybees, Apis mellifera L. Anim. Behav. 2002, 63, 369–390. [Google Scholar]
- Roulston, T.H.; Cane, J.H. The effect of pollen protein concentration on body size in the sweat bee Lasioglossum zephyrum (Hymenoptera: Apiformes). Evol. Ecol. 2002, 16, 49–65. [Google Scholar]
- Cook, S.M.; Awmack, C.S.; Murray, D.A.; Williams, I.H. Are honeybees’ foraging preferences affected by pollen amino acid composition? Ecol. Entomol. 2003, 28, 622–627. [Google Scholar]
- Nicholls, E.; Hempel de Ibarra, N. Assessment of pollen rewards by foraging bees. Funct. Ecol. 2017, 31, 76–87. [Google Scholar]
- Herbert, E.W. Honey bee nutrition. In The Hive and the Honey Bee; Graham, J.M., Ed.; Dadant and Sons: Hamilton, IL, USA, 1997; pp. 197–233. [Google Scholar]
- Somerville, D.; Nicol, H.I. Mineral content of honeybee-collected pollen from southern New South Wales. Aust. J. Exp. Agric. 2002, 42, 1131–1136. [Google Scholar]
- Szczęsna, T. Concentration of selected elements in honeybee-collected pollen. J. Apic. Sci. 2007, 51, 5–13. [Google Scholar]
- Manning, R. Pollen analysis of six species of eucalypt in Western Australia; Rural Industries Research and Development Corporation: Canberra, Australia, 2000. [Google Scholar]
- Szczesna, T.; Rybak-Chmielewska, H.; Was, E.; Kachaniuk, K.; Teper, D. Characteristics of Polish unifloral honeys. I. Rape Honey (Brassica napus L. var. oleifera Metzger). J. Appl. Sci. 2011, 55, 111–119. [Google Scholar]
- Herbert, E.W.; Shimanuki, H. Mineral requirements for brood-rearing by honeybees fed a synthetic diet. J. Apic. Res. 1978, 17, 118–122. [Google Scholar]
- De Groot, A.P. Protein and amino acid requirements of the honeybee (Apis mellifca L.). Physiol. Comp. Oecol. 1953, 3, 1–83. [Google Scholar]
- Neff, J.L.; Simpson, B.B. The roles of phenology and reward structure in the pollination biology of wild sunflower (Helianthus annuus L., Asteraceae). Isr. J. Bot. 1990, 39, 197–216. [Google Scholar]
- Minckley, R.L.; Wcislo, W.T.; Yanega, D.; Buchmann, S.L. Behavior and phenology of a specialist bee (Dieunomia) and sunflower (Helianthus) pollen availability. Ecology 1994, 75, 1406–1419. [Google Scholar]
- Schmidt, L.S.; Schmidt, J.O.; Rao, H.; Wang, W.; Xu, L. Feeding preference and survival of young worker honey bees (Hymenoptera: Apidae) fed rape, sesame, and sunflower pollen. J. Econ. Entomol. 1995, 88, 1591–1595. [Google Scholar]
- Tasei, J.N.; Aupinel, P. Nutritive value of 15 single pollens and pollen mixes tested on larvae produced by bumblebee workers (Bombus terrestris, Hymenoptera: Apidae). Apidologie 2008, 39, 397–409. [Google Scholar]
- Singh, S.; Saini, K.; Jain, K. Quantitative comparison of lipids in some pollens and their phagostimulatory effects in honey bees. J. Apic. Res. 1999, 38, 87–92. [Google Scholar]
- Schmidt, J.O.; Hanna, A. Chemical nature of phagostimulants in pollen attractive to honeybees. J. Insect Behav. 2006, 19, 521–532. [Google Scholar]
- Avni, D.; Hendriksma, H.P.; Dag, A.; Uni, Z.; Shafir, S. Nutritional aspects of honey bee-collected pollen and constraints on colony development in the eastern Mediterranean. J. Insect Physiol. 2014, 69, 65–73. [Google Scholar] [PubMed]
- Nicolson, S.W.; Human, H. Chemical composition of the ‘low quality’ pollen of sunfower (Helianthus annuus, Asteraceae). Apidologie 2012, 44, 144–152. [Google Scholar]
- Brodschneider, R.; Crailsheim, K. Nutrition and health in honey bees. Apidologie 2010, 41, 278–294. [Google Scholar]
- Manning, R. Fatty acids in pollen: A review of their importance for honey bees. Bee World 2001, 82, 60–75. [Google Scholar]
- Canavoso, L.E.; Jouni, Z.E.; Karnas, K.J.; Pennington, J.E.; Wells, M.A. Fat metabolism in insects. Ann. Rev. Nutr. 2001, 21, 23–46. [Google Scholar]
- Cohen, A.C. Insect Diets Science and Technology; CRC Press: Boca Raton, FL, USA, 2004; p. 473. [Google Scholar]
- Nurullahoglu, Z.U.; Uckan, F.; Sak, O.; Ergin, E. Total lipid and fatty acid composition of Apanteles galleriae and its parasitized host. Ann. Entomol. Soc. Am. 2004, 97, 1000–1006. [Google Scholar]
- Wang, Y.M.; Lin, D.S.; Bolewicz, L.; Connor, W.E. The predominance of polyunsaturated fatty acids in the butterfly Morpho peleides before and after metamorphosis. J. Lipid Res. 2006, 47, 530–536. [Google Scholar]
- Khani, A.; Moharramipour, S.; Barzegar, M.; Naderi-Manesh, H. Comparison of fatty acid composition in total lipid of diapause and non-diapause larvae of Cydia pomonella (Lepidoptera: Tortricidae). Insect Sci. 2007, 14, 125–131. [Google Scholar]
- Yang, K.; Wu, D.; Ye, X.; Liu, D.; Chen, J.; Sun, P. Characterization of chemical composition of bee pollen in China. J. Agric. Food Chem. 2013, 61, 708–718. [Google Scholar] [PubMed]
- Giacomini, J.J.; Leslie, J.; Tarpy, D.R.; Palmer-Young, E.C.; Irwin, R.E.; Adler, L.S. Medicinal value of sunflower pollen against bee pathogens. Sci. Rep. 2018, 8, 14394. [Google Scholar]
- Schmidt, J.O. Pollen foraging preferences of honey bees (Hymenoptera, Apidae). Southwest. Entomol. 1982, 7, 255–259. [Google Scholar]
- Moffett, J.O.; Stith, L.S.; Burkhart, C.C.; Shipman, C.W. Honey bee visits to cotton flowers. Environ. Entomol. 1975, 4, 203–206. [Google Scholar]
- Schmidt, J.O. Phagostimulants in pollen. J. Apic. Res. 1985, 24, 107–114. [Google Scholar]
- Schmidt, J.O.; Buchmann, S.L.; Glaiim, M. The nutritional value of Typha latifolia for bees. J. Apic. Res. 1989, 28, 155–165. [Google Scholar]
- Chittka, L.; Raine, N.E. Recognition of flowers by pollinators. Curr. Opin. Plant Biol. 2006, 9, 428–435. [Google Scholar] [PubMed]
- Effah, E.; Holopainen, J.K.; McCormick, A.C. Potential roles of volatile organic compounds in plant competition. Perspect. Plant Ecol. Evol. Syst. 2019, 38, 58–63. [Google Scholar]
- Kumeroa, F.; Komahan, S.; Sofkova-Bobcheva, S.; McCormick, A.C. Characterization of the volatile profiles of six industrial hemp (Cannabis sativa L.) cultivars. Agronomy 2022, 12, 2651. [Google Scholar]
- Farré-Armengol, G.; Filella, I.; Llusià, J.; Peñuelas, J. β-Ocimene, a key floral and foliar volatile involved in multiple interactions between plants and other organisms. Molecules 2017, 22, 1148. [Google Scholar] [PubMed]
- Byers, K.J.; Vela, J.P.; Peng, F.; Riffell, J.A.; Bradshaw, H.D., Jr. Floral volatile alleles can contribute to pollinator-mediated reproductive isolation in monkeyflowers (Mimulus). Plant J. 2014, 80, 1031–1042. [Google Scholar] [PubMed]



| Proximate Content (%) | ||||
|---|---|---|---|---|
| Moisture | Ash | Crude Fiber | Protein | |
| Canda | 81.6 | 1.55 | 1.50 | 6.41 |
| CFX-2 | 80.0 | 1.87 | 2.03 | 6.10 |
| Henola | 76.8 | 1.86 | 2.67 | 6.89 |
| Joey | 80.9 | 1.63 | 1.50 | 6.05 |
| (mg/g) | ||||||
|---|---|---|---|---|---|---|
| Amino Acids | Canda | CFX-2 | Henola | Joey | ||
| Essential amino acids | Most essential | Isoleucine | 2.46 | 2.39 | 2.73 | 2.33 |
| Leucine | 4.00 | 3.87 | 4.54 | 3.84 | ||
| Valine | 3.06 | 2.94 | 3.29 | 2.88 | ||
| Intermediate essential | Arginine | 2.90 | 2.76 | 3.27 | 2.80 | |
| Lysine | 3.53 | 3.31 | 3.96 | 3.38 | ||
| Phenylalanine | 2.34 | 2.33 | 2.69 | 2.29 | ||
| Threonine | 2.32 | 2.30 | 3.59 | 2.25 | ||
| Least essential | Histidine | 1.15 | 1.09 | 1.22 | 1.09 | |
| Methionine | 1.08 | 1.06 | 1.19 | 1.06 | ||
| Non-essential amino acids | Non-essential | Alanine | 3.29 | 3.10 | 3.18 | 3.02 |
| Aspartic Acid | 8.32 | 7.09 | 7.97 | 7.81 | ||
| Cystine | 0.658 | 0.607 | 0.667 | 0.632 | ||
| Glutamic Acid | 5.54 | 5.35 | 6.55 | 5.74 | ||
| Glycine | 2.50 | 2.41 | 2.72 | 2.41 | ||
| Proline | 3.87 | 3.79 | 3.65 | 3.71 | ||
| Serine | 2.72 | 2.64 | 3.01 | 2.68 | ||
| Tyrosine | 1.78 | 1.69 | 2.04 | 1.75 | ||
| (%) | |||||
|---|---|---|---|---|---|
| Fatty Acids | Canda | CFX-2 | Henola | Joey | |
| Saturated | 16:0 Palmitic acid | 0.125 | 0.127 | 0.135 | 0.125 |
| 18:0 Steric acid | 0.014 | 0.015 | 0.022 | 0.014 | |
| 20:0 Arachidic acid | 0.008 | 0.010 | 0.016 | 0.009 | |
| 22:0 Behenic acid | 0.009 | 0.011 | 0.019 | 0.010 | |
| 24:0 Lignoceric acid | 0.008 | 0.009 | 0.013 | 0.008 | |
| Mono-saturated | 9c 18:1 Oleic acid | 0.019 | 0.020 | 0.034 | 0.020 |
| Polyunsaturated | 18:2 Linoleic acid | 0.137 | 0.138 | 0.152 | 0.171 |
| 18:3 Alpha Linolenic acid | 0.109 | 0.122 | 0.133 | 0.141 | |
| Omega 3 | 0.109 | 0.122 | 0.133 | 0.141 | |
| Omega 6 | 0.137 | 0.138 | 0.152 | 0.171 | |
| Omega 9 | 0.019 | 0.020 | 0.034 | 0.020 | |
| Fatty acid types | Saturated | 0.157 | 0.164 | 0.195 | 0.158 |
| Monounsaturated | 0.027 | 0.028 | 0.045 | 0.029 | |
| Polyunsaturated | 0.236 | 0.249 | 0.272 | 0.298 | |
| Total | Total 18:1 cis | 0.028 | 0.029 | 0.047 | 0.030 |
| Total Cis Unsaturated Fatty Acids | 0.263 | 0.277 | 0.317 | 0.327 | |
| Total Fatty Acids | 0.438 | 0.461 | 0.537 | 0.508 | |
| Variety | Minerals (mg) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| B | Ca | Cu | Fe | Mg | Mn | P | K | Zn | |
| Canda | 0.006 | 2.04 | 0.003 | 0.016 | 0.770 | 0.006 | 1.25 | 5.53 | 0.012 |
| CFX-2 | 0.008 | 2.83 | 0.002 | 0.019 | 0.832 | 0.009 | 1.06 | 5.85 | 0.012 |
| Henola | 0.008 | 2.74 | 0.006 | 0.003 | 0.955 | 0.013 | 1.11 | 5.57 | 0.013 |
| Joey | 0.006 | 2.00 | 0.002 | 0.002 | 0.711 | 0.007 | 1.11 | 5.40 | 0.013 |
| Variance | ||||
|---|---|---|---|---|
| Component | PC | Eigenvalue | Percent | Cumulative Percent |
| Proximate analysis | 1 | 3.23 | 80.9 | 80.9 |
| 2 | 0.72 | 17.9 | 98.8 | |
| 3 | 0.05 | 1.2 | 100.0 | |
| Amino acids | 1 | 13.9 | 81.9 | 81.9 |
| 2 | 2.3 | 13.5 | 95.4 | |
| 3 | 0.79 | 4.60 | 100.0 | |
| Fatty acids | 1 | 11.70 | 68.8 | 68.8 |
| 2 | 5.13 | 30.2 | 99.0 | |
| 3 | 0.17 | 1.0 | 100.0 | |
| Minerals | 1 | 6.03 | 67.0 | 67.0 |
| 2 | 1.84 | 20.4 | 87.4 | |
| 3 | 1.13 | 12.6 | 100.0 | |
| Component | Variable | PC1 | PC2 | PC3 |
|---|---|---|---|---|
| Proximate analysis | Moisture | −0.547 | 0.043 | 0.806 |
| Ash | 0.456 | 0.670 | 0.393 | |
| Crude fiber | 0.555 | 0.066 | 0.148 | |
| Protein | 0.431 | −0.738 | 0.417 | |
| Amino acids | Isoleucine | 0.262 | 0.004 | 0.230 |
| Leucine | 0.266 | −0.037 | 0.118 | |
| Valine | 0.260 | 0.898 | 0.224 | |
| Arginine | 0.268 | −0.015 | −0.003 | |
| Lysine | 0.268 | 0.024 | −0.035 | |
| Phenylalanine | 0.260 | −0.125 | 0.174 | |
| Threonine | 0.260 | −0.154 | 0.111 | |
| Histidine | 0.261 | 0.135 | 0.072 | |
| Methionine | 0.266 | −0.077 | 0.076 | |
| Alanine | 0.110 | 0.568 | 0.341 | |
| Aspartic Acid | 0.124 | 0.462 | −0.613 | |
| Cystine | 0.219 | 0.312 | −0.368 | |
| Glutamic Acid | 0.251 | −0.172 | −0.267 | |
| Glycine | 0.267 | 0.017 | 0.075 | |
| Proline | −0.153 | 0.506 | 0.333 | |
| Serine | 0.267 | −0.065 | −0.033 | |
| Tyrosine | 0.266 | −0.054 | −0.103 | |
| Fatty acids | 16:0 Palmitic acid | 0.269 | −0.171 | −0.132 |
| 18:0 Steric acid | 0.272 | −0.161 | 0.044 | |
| 20:0 Arachidic acid | 0.279 | −0.127 | −0.223 | |
| 22:0 Behenic acid | 0.279 | −0.130 | −0.107 | |
| 24:0 Lignoceric acid | 0.269 | −0.171 | −0.132 | |
| 9c 18:1 Oleic acid | 0.279 | −0.125 | 0.199 | |
| 18:2 Linoleic acid | 0.117 | 0.399 | 0.373 | |
| 18:3 Alpha Linolenic acid | 0.184 | 0.333 | −0.455 | |
| Omega 3 fatty acid | 0.184 | 0.333 | −0.455 | |
| Omega 6 fatty acid | 0.117 | 0.399 | 0.373 | |
| Omega 9 fatty acid | 0.279 | −0.125 | 0.199 | |
| Saturated fatty acids | 0.273 | −0.159 | −0.088 | |
| Monounsaturated fatty acids | 0.283 | −0.104 | 0.240 | |
| Polyunsaturated fatty acids | 0.149 | 0.380 | −0.012 | |
| Total 18:1 cis | 0.282 | −0.106 | 0.245 | |
| Total cis unsaturated fatty acids | 0.210 | 0.307 | 0.056 | |
| Total fatty acids | 0.273 | 0.159 | −0.006 | |
| Minerals | Boron (B) | 0.374 | −0.230 | 0.226 |
| Calcium (Ca) | 0.356 | −0.357 | −0.163 | |
| Copper (Cu) | 0.341 | 0.355 | 0.244 | |
| Iron (Fe) | 0.386 | 0.218 | 0.110 | |
| Magnesium (Mg) | 0.388 | 0.024 | 0.288 | |
| Manganese (Mn) | 0.392 | 0.192 | −0.055 | |
| Phosphorus (P) | −0.227 | 0.247 | 0.714 | |
| Potassium (K) | 0.172 | −0.664 | 0.010 | |
| Zinc (Zn) | 0.287 | 0.327 | −0.521 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dingha, B.N.; Jackai, L.E. Chemical Composition of Four Industrial Hemp (Cannabis sativa L.) Pollen and Bee Preference. Insects 2023, 14, 668. https://doi.org/10.3390/insects14080668
Dingha BN, Jackai LE. Chemical Composition of Four Industrial Hemp (Cannabis sativa L.) Pollen and Bee Preference. Insects. 2023; 14(8):668. https://doi.org/10.3390/insects14080668
Chicago/Turabian StyleDingha, Beatrice N., and Louis E. Jackai. 2023. "Chemical Composition of Four Industrial Hemp (Cannabis sativa L.) Pollen and Bee Preference" Insects 14, no. 8: 668. https://doi.org/10.3390/insects14080668
APA StyleDingha, B. N., & Jackai, L. E. (2023). Chemical Composition of Four Industrial Hemp (Cannabis sativa L.) Pollen and Bee Preference. Insects, 14(8), 668. https://doi.org/10.3390/insects14080668







