Mechanical Acaricides Active against the Blacklegged Tick, Ixodes scapularis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ticks and Industrial Minerals
2.2. Determining the Time to Mortality of I. scapularis after Exposure to Celite 610
2.3. Determining the Time to Mortality of I. scapularis after Exposure to Imergard™ WP
2.4. Statistical Analysis
2.5. Scanning Electron Microscopy
3. Results
3.1. Efficacy of Celite 610 against I. scapularis
3.2. Efficacy of Imergard™ WP against I. scapularis
3.3. Scanning Electron Microscopy
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Eisen, R.J.; Eisen, L. The blacklegged tick, Ixodes scapularis: An increasing public health concern. Trends Parasitol. 2018, 34, 295–309. [Google Scholar] [CrossRef]
- Lyme Disease|CDC. Available online: https://www.cdc.gov/lyme/index.html (accessed on 20 May 2022).
- Adams, D.A.; Thomas, K.R.; Jajosky, R.A.; Foster, L.; Sharp, P.; Onweh, D.H.; Schley, A.W.; Anderson, W.J.; Arguin, P.M.; Artus, A.; et al. Summary of notifiable infectious diseases and conditions—United States, 2014. MMWR. Morb. Mortal. Wkly. Rep. 2019, 63, 1–152. [Google Scholar] [CrossRef] [PubMed]
- Eisen, R.J.; Eisen, L.; Beard, C.B. County-scale distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the continental United States. J. Med. Entomol. 2016, 53, 349–386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beard, C.B.; Eisen, R.J.; Barker, C.M.; Garofalo, J.F.; Hahn, M.; Hayden, M.; Monaghan, A.J.; Ogden, N.H.; Schramm, P.J. US Global Change Research Program. Washington, DC, USA. 2016; pp. 129–156. Available online: https://health2016.globalchange.gov/low/ClimateHealth2016_05_Vector_small.pdf (accessed on 17 June 2022).
- Piesman, J.; Eisen, L. Prevention of tick-borne diseases. Annu. Rev. Entomol. 2007, 53, 323–343. [Google Scholar] [CrossRef] [PubMed]
- Stafford, K.C.; Williams, S.C.; Molaei, G. Integrated pest management in controlling ticks and tick-associated diseases. J. Integr. Pest Manag. 2017, 8, 28. [Google Scholar] [CrossRef] [Green Version]
- Stafford, K.C. Tick Management Handbook: An Integrated Guide for Homeowners, Pest Control Operators, and Public Health Officials for the Prevention of Tick-Associated Disease. Available online: https://stacks.cdc.gov/view/cdc/11444 (accessed on 23 May 2022).
- Abbas, R.Z.; Zaman, M.A.; Colwell, D.D.; Gilleard, J.; Iqbal, Z. Acaricide resistance in cattle ticks and approaches to its management: The state of play. Vet. Parasitol. 2014, 203, 6–20. [Google Scholar] [CrossRef]
- Willadsen, P. Tick control: Thoughts on a research agenda. Vet. Parasitol. 2006, 138, 161–168. [Google Scholar] [CrossRef]
- George, J.E.; Pound, J.M.; Davey, R.B. Chemical control of ticks on cattle and the resistance of these parasites to acaricides. Parasitology 2004, 129 (Suppl. S1), S353–S366. [Google Scholar] [CrossRef]
- Agwunobi, D.O.; Yu, Z.; Liu, J. A Retrospective review on ixodid tick resistance against synthetic acaricides: Implications and perspectives for future resistance prevention and mitigation. Pestic. Biochem. Physiol. 2021, 173, 104776. [Google Scholar] [CrossRef]
- Eiden, A.L.; Kaufman, P.E.; Oi, F.M.; Allan, S.A.; Miller, R.J. Detection of permethrin resistance and fipronil tolerance in Rhipicephalus sanguineus (Acari: Ixodidae) in the United States. J. Med. Entomol. 2015, 52, 429–436. [Google Scholar] [CrossRef]
- Burtis, J.C.; Poggi, J.D.; Payne, B.; Campbell, S.R.; Harrington, L.C. Susceptibility of Ixodes scapularis (Acari: Ixodidae) to permethrin under a long-term 4-poster deer treatment area on shelter island, NY. J. Med. Entomol. 2021, 58, 1966–1969. [Google Scholar] [CrossRef] [PubMed]
- Çağlan Günal, A.; Kayiran Tunca, S.; Arslan, P.; Gül, G.; Aylin, S.-D. How does sublethal permethrin effect non-target aquatic organisms? Environ. Sci. Pollut. Res. 2021, 28, 52405–52417. [Google Scholar] [CrossRef] [PubMed]
- Deguenon, J.M.; Azondekon, R.; Agossa, F.R.; Padonou, G.G.; Anagonou, R.; Ahoga, J.; N’dombidje, B.; Akinro, B.; Stewart, D.A.; Wang, B.; et al. Imergard TM WP: A non-chemical alternative for an indoor residual spray, effective against pyrethroid-resistant Anopheles gambiae (s.l.) in Africa. Insects 2020, 11, 322. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Deguenon, J.M.; Cave, G.; Denning, S.S.; Reiskind, M.H.; Watson, D.W.; Stewart, D.A.; Gittins, D.; Zheng, Y.; Liu, X.; et al. New Thinking for filth fly control: Residual, non-chemical wall spray from volcanic glass. Med. Vet. Entomol. 2021, 35, 451–461. [Google Scholar] [CrossRef]
- Deguenon, J.M.; Riegel, C.; Cloherty-Duvernay, E.R.; Chen, K.; Stewart, D.A.; Wang, B.; Gittins, D.; Tihomirov, L.; Apperson, C.S.; McCord, M.G.; et al. New mosquitocide derived from volcanic rock. J. Med. Entomol. 2021, 58, 458–464. [Google Scholar] [CrossRef]
- Mitchell, R.D.; Mott, D.W.; Dhammi, A.; Reisig, D.D.; Roe, R.M.; Stewart, D.A. Field evaluation of a new thrips control agent for coton: A mechanical insecticide. In Proceedings of the Beltwide Cotton Conference, San Antonio, TX, USA, 3–5 January 2018; pp. 786–795. [Google Scholar]
- Showler, A.T.; Flores, N.; Caesar, R.M.; Mitchel, R.D.; De León, A.A.P. Lethal effects of a commercial diatomaceous earth dust product on Amblyomma americanum (Ixodida: Ixodidae) larvae and nymphs. J. Med. Entomol. 2020, 57, 1575–1581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korunić, Z.K.; Rozman, V.; Liška, A.; Lucić, P. A review of natural insecticides based on diatomaceous earths. Poljoprivreda 2016, 22, 10–18. [Google Scholar] [CrossRef]
- Islam, M.S.; Rahman, M.M. Diatomaceous earth-induced alterations in the reproductive attributes in the housefly Musca domestica L. (Diptera: Muscidae). Elixir Appl. Zool. 2021, 96, 241–41244. [Google Scholar] [CrossRef]
- Doggett, S.L.; Geary, M.J.; Lilly, D.; Russell, R.C. The Efficacy of Diatomaceous Earth against the Common Bed Bug, Cimex Lectularius; A Report for Mount Sylvia Diatomite, Westmead Hospital, Sydney. 2008. Available online: https://www.researchgate.net/publication/322974799_The_Efficacy_of_Diatomaceous_Earth_against_the_Common_Bed_Bug_Cimex_lectularius (accessed on 17 June 2022).
- Showler, A.T. Effects of kaolin-based particle film application on boll weevil (Coleoptera: Curculionidae) injury to cotton. J. Econ. Entomol. 2002, 95, 754–762. [Google Scholar] [CrossRef]
- Liang, G.; Liu, T.X. Repellency of a kaolin particle film, surround, and a mineral oil, sunspray oil, to silverleaf whitefly (Homoptera: Aleyrodidae) on melon in the laboratory. J. Econ. Entomol. 2002, 95, 317–324. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Bazrafkan, S.; Vatandoost, H.; Abaei, M.R.; Ahmadi, M.S.; Tavassoli, M.; Mansoreh Shayeghi, R. The insecticidal effect of diatomaceous earth against adults and nymphs of Blattella germanica. Asian Pac. J. Trop. Biomed. 2014, 4, S228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maxim, L.D.; Niebo, R.; Mcconnell, E.E. Perlite toxicology and epidemiology—A review. Inhal. Toxicol. 2014, 26, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Joiner, A. Review of the extrinsic stain removal and enamel/dentine abrasion by a calcium carbonate and perlite containing whitening toothpaste. Int. Dent. J. 2006, 56, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jackson, L.S.; Jablonski, J.E. Factors affecting the levels of heavy metals in juices processed with filter aids. J. Food Prot. 2017, 80, 892–902. [Google Scholar] [CrossRef]
- Abas Shah, M.; Ali Khan, A. Use of diatomaceous earth for the management of stored-product pests. Int. J. Pest Manag. 2014, 60, 100–113. [Google Scholar] [CrossRef]
- Pepin, K.M.; Eisen, R.J.; Mead, P.S.; Piesman, J.; Fish, D.; Hoen, A.G.; Barbour, A.G.; Hamer, S.; Diuk-Wasser, M.A. Geographic variation in the relationship between human lyme disease incidence and density of infected host-seeking Ixodes scapularis nymphs in the eastern United States. Am. J. Trop. Med. Hyg. 2012, 86, 1062–1071. [Google Scholar] [CrossRef] [Green Version]
- Guedes, N.M.P.; Braga, L.S.; Rosi-Denadai, C.A.; Guedes, R.N.C. Desiccation resistance and water balance in populations of the maize weevil Sitophilus zeamais. J. Stored Prod. Res. 2015, 64, 146–153. [Google Scholar] [CrossRef]
- Needham, G.R.; Teel, P.D. Off-host physiological ecology of ixodid ticks. Annu. Rev. Entomol. 1991, 36, 659–681. [Google Scholar] [CrossRef] [PubMed]
- Stafford, K.C. Survival of immature Ixodes scapularis (Acari: Ixodidae) at different relative humidities. J. Med. Entomol. 1994, 31, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.F.; Magnarelli, L.A. Biology of Ticks. Infect. Dis. Clin. N. Am. 2008, 22, 195–215. [Google Scholar] [CrossRef] [PubMed]
- Schulze, T.L.; Jordan, R.A.; Hung, R.W. Effects of selected meteorological factors on diurnal questing of Ixodes scapularis and Amblyomma americanum (Acari: Ixodidae). J. Med. Entomol. 2001, 38, 318–324. [Google Scholar] [CrossRef]
- Bertrand, M.R.; Wilson, M.L. Microclimate-dependent survival of unfed adult Ixodes scapularis (Acari: Ixodidae) in nature: Life cycle and study design implications. J. Med. Entomol. 1996, 33, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Hayes, L.E.; Scott, J.A.; Iii, K.C.S. Influences of weather on Ixodes scapularis nymphal densities at long-term study sites in Connecticut. Ticks Tick. Borne. Dis. 2015, 6, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Berger, K.A.; Ginsberg, H.S.; Dugas, K.D.; Hamel, L.H.; Mather, T.N. Adverse moisture events predict seasonal abundance of lyme disease vector ticks (Ixodes scapularis). Parasit. Vectors 2014, 7, 181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonenshine, D.E.; Roe, R.M. Biology of Ticks, 2nd ed.; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Vail, S.G.; Smith, G. Air temperature and relative humidity effects on behavioral activity of blacklegged tick (Acari: Ixodidae) nymphs in New Jersey. J. Med. Entomol. 1998, 35, 1025–1028. [Google Scholar] [CrossRef] [PubMed]
- Athanassiou, C.G.; Kavallieratos, N.G.; Chiriloaie, A.; Vassilakos, T.N.; Fătu, V.; Drosu, S.; Ciobanu, M.; Dudoiu, R. Insecticidal efficacy of natural diatomaceous earth deposits from Greece and Romania against four stored grain beetles: The effect of temperature and relative humidity. Bull. Insectol. 2016, 69, 25–34. [Google Scholar]
- Arthur, F.H. Toxicity of diatomaceous earth to red flour beetles and confused flour beetles (Coleoptera: Tenebrionidae): Effects of temperature and relative humidity. J. Econ. Entomol. 2000, 93, 526–532. [Google Scholar] [CrossRef] [PubMed]
- le Patourel, G.N.J. The effect of grain moisture content on the toxicity of a sorptive silica dust to four species of grain beetle. J. Stored Prod. Res. 1986, 22, 63–69. [Google Scholar] [CrossRef]
- Suppan, J.; Walzl, M.; Klepal, W. The spiracle glands in Ixodes ricinus (Linnaeus, 1758) (Acari: Ixodidae). Acarologia 2013, 53, 221–230. [Google Scholar] [CrossRef] [Green Version]
- Fielden, L.J.; Knolhoff, L.M.; Villarreal, S.M.; Ryan, P. Underwater survival in the dog tick Dermacentor variabilis (Acari:Ixodidae). J. Insect Physiol. 2011, 57, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Pugh, P.J.A.; King, E.P.; Fordy, M.R. Spiracular transpiration in ticks: A passive diffusion barrier in three species of Ixodidae (Metastigmata: Acarina). J. Zool. 1990, 221, 63–75. [Google Scholar] [CrossRef]
- Pugh, P.J.A.; King, P.E.; Fordy, M.R. The spiracle of Ixodes ricinus (L.) (Ixodidae: Metastigmata: Acarina): A passive diffusion barrier for water vapour. Zool. J. Linn. Soc. 1988, 93, 113–131. [Google Scholar] [CrossRef]
- Baker, G.T. Spiracular plate of nymphal and adult hard ticks (Acarina: Ixodidae): Morphology and cuticular ultrastructure. Biology 1997, 116, 341–347. [Google Scholar] [CrossRef]

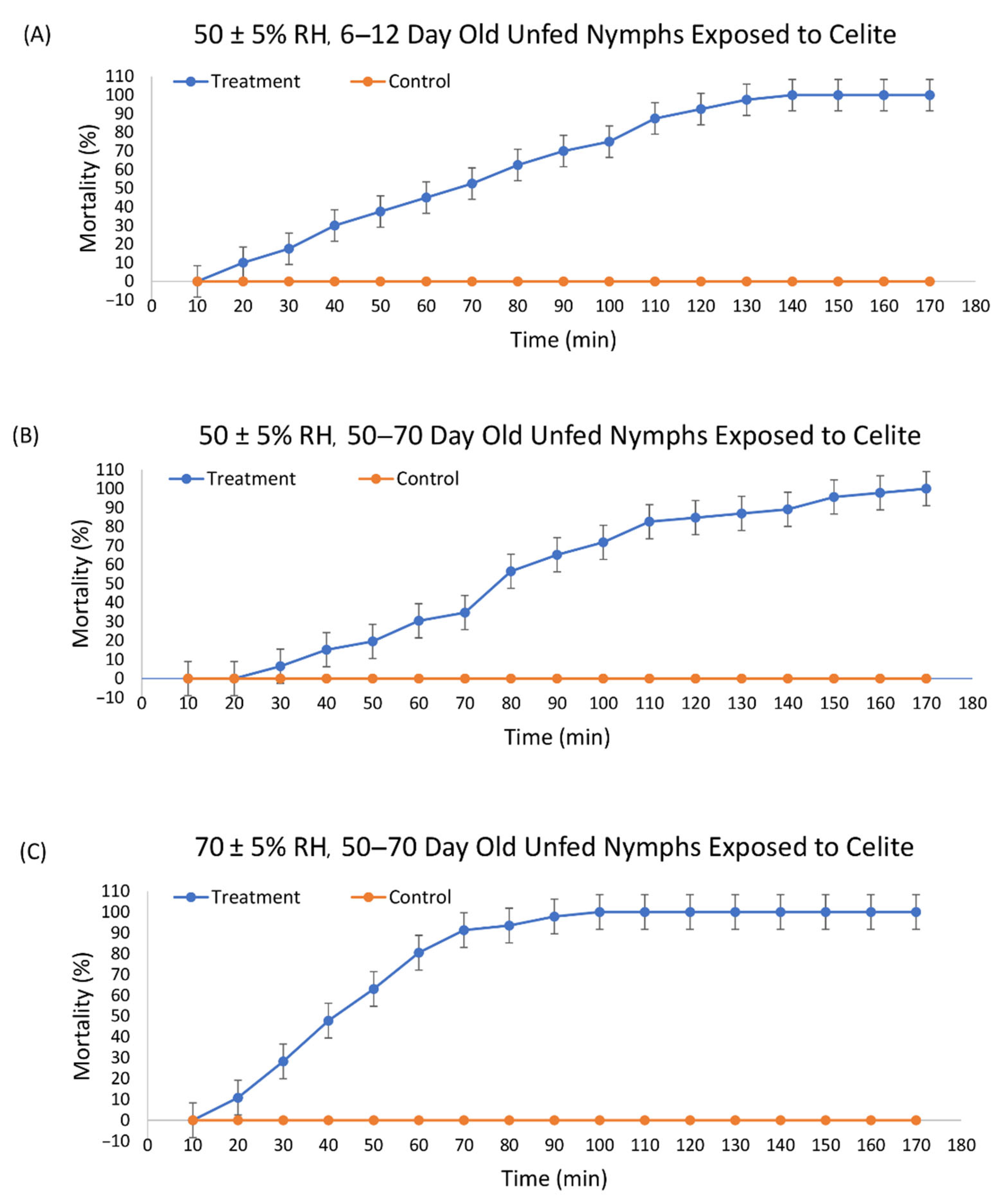
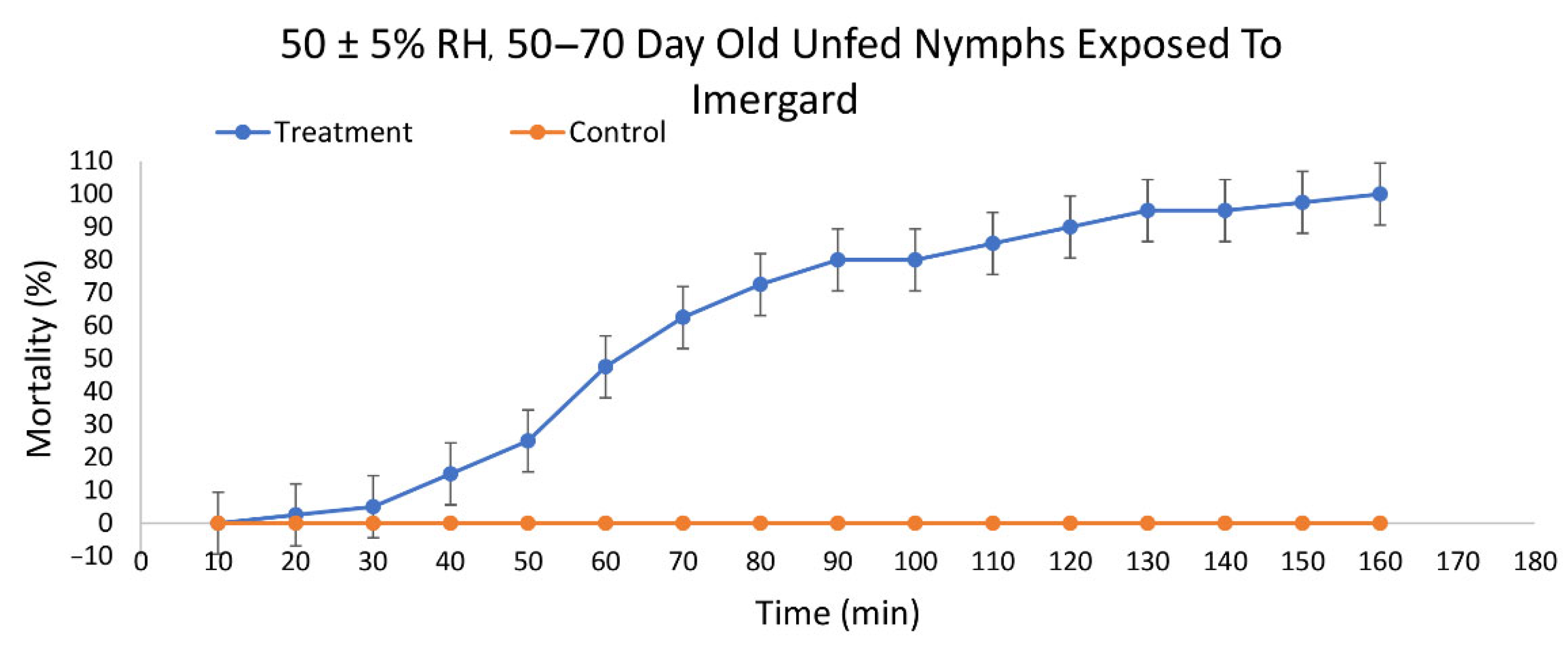


| Mineral | RH * | Last Molt (Days) | N | Slope (SE) | LT50 † (95% CI ‡) | LT90 † (95% CI ‡) | χ2 |
|---|---|---|---|---|---|---|---|
| Celite 610 | 50 ± 5% | 6–10 | 40 | 0.028 | 66.84A (62.12, 71.43) | 113.42A (106.49 122.12) | 7.20 |
| Celite 610 | 50 ± 5% | 50–70 | 46 | 0.027 | 81.68B (77.44, 85.86) | 128.31B (122.26, 135.53) | 10.90 |
| Celite 610 | 70 ± 5% | 50–70 | 46 | 0.050 | 43.84C (40.61, 46.93) | 69.46C (65.18, 74.89) | 5.12 |
| Imergard™ WP | 50 ± 5% | 50–70 | 40 | 0.030 | 70.40A (65.99, 74.67) | 112.56A (106.61 119.78) | 17.464 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Richardson, E.A.; Ponnusamy, L.; Roe, R.M. Mechanical Acaricides Active against the Blacklegged Tick, Ixodes scapularis. Insects 2022, 13, 672. https://doi.org/10.3390/insects13080672
Richardson EA, Ponnusamy L, Roe RM. Mechanical Acaricides Active against the Blacklegged Tick, Ixodes scapularis. Insects. 2022; 13(8):672. https://doi.org/10.3390/insects13080672
Chicago/Turabian StyleRichardson, Elise A., Loganathan Ponnusamy, and R. Michael Roe. 2022. "Mechanical Acaricides Active against the Blacklegged Tick, Ixodes scapularis" Insects 13, no. 8: 672. https://doi.org/10.3390/insects13080672
APA StyleRichardson, E. A., Ponnusamy, L., & Roe, R. M. (2022). Mechanical Acaricides Active against the Blacklegged Tick, Ixodes scapularis. Insects, 13(8), 672. https://doi.org/10.3390/insects13080672







