Simple Summary
Different isolates of Cydia pomonella granulovirus (CpGV) have been formulated into different biological control agents that are applied to reduce the infestation and losses induced by codling moth in organic and integrated apple and pear orchards. Cyd-X® is the most widely applied CpGV product in apple orchards in the USA and contains a single active ingredient, the isolate CpGV-M (GV-0001). Aiming to investigate the susceptibility of codling moth to GV-0001, we used a discriminating virus concentration to screen five codling moth populations derived from Washington State (USA), which were reared in the laboratory (colony WA1-WA5). The results indicate that colony WA3 had a significantly reduced susceptibility to GV-0001 and survivors from the bioassay can produce offspring. WA3 represents the first documented case of CpGV resistance in the USA. In further tests, WA3 showed resistance to all commercial CpGV agents currently registered in the USA. Nonetheless, the resistant WA3 colony showed high susceptibility to three newly developed CpGV preparations. Therefore, the introduction of the novel CpGV formulations may allow the maintenance of sustainable management programs for codling moth in the USA.
Abstract
Codling moth (Cydia pomonella L.) is a very important pest in apple, pear, and walnut orchards worldwide, including the USA. Cydia pomonella granulovirus (CpGV) is used to control codling moth in organic and conventional production. Due to increasing codling moth infestations from organic apple orchards in Washington State, USA, five codling moth colonies (WA1-WA5) were screened for their susceptibility relative to the isolate GV-0001, the main active ingredient of Cyd-X®, using a discriminating concentration of 6 × 104 OB/mL. Compared to a susceptible laboratory colony, the observed results indicated that GV-0001 lacked efficacy against codling moth colony WA3. It was confirmed that WA3 was the first case of codling moth resistance to CpGV in the USA. Further testing of WA3 was performed on a range of CpGV isolates and a lack of efficacy was observed against additional isolates. However, three newly developed CpGV preparations can efficiently infect larvae from the resistant colony WA3. Our results suggest that there is an urgent need to monitor the situation in the USA, aiming to prevent the emergence or spread of additional codling moth populations with CpGV resistance. Strategies to sustain the efficacy of codling moth control using novel CpGV formulations need to be developed.
1. Introduction
Codling moth (Cydia pomonella L.) (Lepidoptera: Tortricidae) is a temperate insect species, and during the larval stage, it can damage pome fruit (apple and pear), stone fruit (apricot, plum), and walnut, etc., through internal feeding. Codling moth invaded North America over 200 years ago and has since become a very serious pest in apple, pear, and walnut production [1]. Codling moth larvae bore deep into the core of unripe and ripening apples, called the “worm in the apple”, resulting in direct yield loss when it is not well controlled under the economic threshold [1,2]. In the USA, there are two to three annual generations of codling moth [3]. Overwintering diapausing larvae pupate in early spring and emerging moths mate and begin to lay eggs right after fruit trees bloom, typically during petal fall, at which time the first instars will bore into the developing fruit. During feeding, frass pushed out through the entrance hole (or “sting”) becomes an obvious sign of an infested fruit [4,5]. The larvae leave the fruit at the fifth instar to pupate under the bark of tree trunk or other protected locations in the orchard. The successive generations continue feeding on developing fruit, although depending on harvest times, infested fruit is not always readily observable and may result in contaminated fruit. Although chemical pesticides can control codling moth, there are non-target risks and documented resistance to pyrethroids and other chemicals [6,7,8]. In addition, the consumers’ demand for organic fruit directly pushes pest control strategies towards biological methods. For example, mating disruption using phermones, spinosad, and commercial formulations of Cydia pomonella granulovirus (CpGV) have been applied to reduce codling moth population in organic and conventional orchards [9,10,11,12].
CpGV is a double-strand DNA virus belonging to the genus Betabaculovirus of the family Baculoviridae [13]. The occlusion body (OB) of CpGV with an average size of 360 × 210 nm (length × width) contains a single virion [14]. CpGV is very specific and highly virulent to codling moth larvae. The median lethal dose (LD50) is 1.37~28 OBs for first instars (L1) and 10~92 OBs for fifth instars (L5) [15], depending on the experimental setup. CpGV was developed as a biological control agent in Europe and North America in the 1970s and 1980s and was first registered in the late 1980s [16]. Viral OBs are ingested by codling moth larva, resulting in the initial infection of the midgut from where infection spreads to other tissues. At the end of infection, the larval body is full of newly produced Obs and the larvae turn whitish or are milky in color. CpGV-infected codling moth larvae will succumb within 3–7 days post-infection but delayed lethality occurs in some populations, taking up to 14 days until the larvae die. When CpGV products are applied for control of codling moth, the efficacy rates of 75% to 90% can be achieved in the field [17].
CpGV agents have been developed into the most broadly applied biological control products for managing codling moth in organic and integrated pest management (IPM) orchards around the world, due to its host specific, highly effective, and environmentally sound characteristics [9,18,19]. Since CpGV was first isolated from an orchard in Mexico, termed CpGV-M [20], more than forty CpGV isolates have been collected and grouped into seven phylogenetic lineages, namely genome group A to G [20,21]. Isolates from genome groups A, E, and B, such as CpGV-M, CpGV-S, and CpGV-E2, have been developed into different commercial formulations (Table 1). CpGV was further isolated, selected, and passaged on oriental fruit moth, Grapholita molesta Busck (Lepidoptera: Tortricidae), for many generations with high virulence against both codling moth and oriental fruit moth.

Table 1.
Commercial and experimental CpGV products/formulations in the USA and Europe. Preparations used in this study are marked with an asterisk *.
After many years of successful application of commercial products that were based on CpGV-M, codling moth populations in southern Germany and France were identified to be 1000- to 100,000-fold less susceptible to CpGV [22,23]. These findings were the first documented cases of host resistance to commercial baculovirus biocontrol products. The resistance of codling moth field populations to products containing CpGV-M was termed type I and was further documented in other European countries, e.g., the Netherlands, Switzerland, Austria, Italy, and Czech Republic, threatening local organic apple production [23,24,25]. Meanwhile, extensive resistance testing and genetic studies revealed three types (I to III) of field resistance to CpGV with distinguishable patterns of inheritance and differing susceptibility to CpGV isolates belonging to different genome groups [23,26]. Beyond that, newly discovered types of resistance have occurred in France and Italy [27]. The most common type I resistance has been characterized as dominant and Z chromosomal inherited, highly stable, and probably with no fitness costs [23,28]. Laboratory-selected and naturally collected CpGV isolates can overcome current resistance types [23,26,29,30].
Apple is one of the most valuable pome fruit varieties in the USA and more than 90% of USA organic apple are produced in Washington State [31]. In these orchards, CpGV products are an important component of the pest management program and are often paired with mating disruption. Several CpGV products have been used for decades on 8100–12,200 ha in North America [9]. Currently, CpGV products of Cyd-X®, Cyd-X HP®, Madex HP® and ViroSoft CP4® are being used in the USA. The frequent application of CpGV per generation for successive years and an increase in infested fruit leads to the question of CpGV resistance in the USA.
Some organic apple orchards in Washington State noted high codling moth infestation despite frequent CpGV application. We collected overwintering larvae from five of these orchards in Washington State to test their susceptibility to CpGV products. We applied a protocol using a discriminating CpGV concentration to differentiate between susceptible and resistant codling moth larvae [23]. We further aimed to determine the resistance status of these colonies against different CpGV products and formulations and provided possible solutions to overcome CpGV resistance using newly developed CpGV products.
2. Materials and Methods
2.1. Insects and Viruses
A susceptible laboratory colony (LabS) was derived from Benzon Research (Carlisle, PA, USA) insect rearing company. This colony was established more than 20 years ago at the Yakima Agricultural Research Laboratory in Washington State. Five populations (WA1-WA5) collected as diapausing larvae from organic apple orchards in eastern Washington State in the winter of 2018–2019 were used. Diapausing larvae were kept under dark conditions at 10 °C until pupation. Adults and progeny larvae were reared in a climate chamber at 25 °C under the photoperiod of 16/8 h (light/dark) at Rutgers Agricultural Research and Extension Center in Bridgeton, NJ, USA. All codling moth colonies were maintained on an artificial diet that is composed of corn meal, wheat germ, beer yeast, agar, ascorbic acid, two preservatives of Nipagin and Benzoic acid, and water [32].
Virus stocks of different commercial products and test formulations (Table 1) were stored at −20 °C. The OB concentration was counted using a Petroff-Hausser chamber (depth 0.02 mm) under the dark field of a light microscope (Nikon ECLIPSE E600).
2.2. Activity of CpGV Formulations on Codling Moth Larvae
For resistance testing, virus OBs were suspended in molecular grade water to a concentration of 3 × 106 OB/mL, and a total volume of 1 mL was incorporated into 49 mL of semi-artificial diet [32] once it had cooled down to 45 °C, resulting in a final concentration of 6 × 104 OB/mL derived from the LC95 value of the susceptible codling moth colony against CpGV-M in laboratory bioassays [23,28]. At this so-called discriminating concentration, the mortality of fully susceptible neonate larvae (L1) is expected to be >95% after 7 days post infection (dpi) and up to 100% after 14 dpi, whereas the average mortality of CpGV-resistant codling moth (CpR) is significantly decreased [23]. Therefore, this discriminating concentration was applied to monitor the susceptibility of L1 larvae of LabS and WA1 to WA5. After mixing the diet with OBs, 50 mL of diet was poured into an autoclavable 50-well tray (LICEFA, Bad Salzuflen, Germany) and allowed to dry at room temperature for 1 day. The dimension of each well is 1.5 × 1.5 × 1.5 cm (length × width × height). One freshly hatched (0–12 h old) codling moth larva (L1) was transferred to each well. The same procedure was performed for the untreated control, with 1 mL of molecular grade water incorporated into the diet. Larvae of the LabS colony and WA1-WA5 were all evaluated using the same protocol. Thirty to fifty larvae were evaluated for each replicate in resistance test, and three replicates of each isolate were evaluated if not otherwise indicated. Any colony indicating less than 30% mortality to GV-0001 (Cyd-X®) at 7 dpi was further bioassayed against seven other CpGV isolates/products (Table 1) at the discriminating OB concentration [23]. Dead individuals were recorded at 1, 7, 14, and 21 dpi. Larvae that died on day 1 because of handling were excluded from the experiment. If needed, the mean mortality at 7, 14, and 21 dpi was corrected for control mortality according to Abbott’s formula [33]. Data were analyzed with t-test and one-way ANOVA followed by multiple comparisons using Tukey–Kramer HSD for significant difference analysis in JMP PRO 15. The results were plotted in GraphPad Prism 9.1 (GraphPad Software, San Diego, CA, USA).
3. Results and Discussion
3.1. Resistance Test of WA Colonies
When the larvae of the LabS colony and five WA colonies were exposed to GV-0001 at the discriminating concentration of 6 × 104 OB/mL, the mortalities of LabS (n = 3 replicates, N = 146 larvae), WA1 (n = 1, N = 36), WA2 (n = 1, N = 29), WA3 (n = 3, N = 143), WA4 (n = 1, N = 35), and WA5 (n = 2, N = 65) were 95.4%, 100.0%, 100.0%, 15.9%, 41.8%, and 100% at 7 dpi, respectively (Figure 1). Compared to the larvicidal activity of GV-0001 on the internal reference LabS, colonies WA3 and WA4 showed a reduced susceptibility. On the other hand, WA3 had 41.8% and 67.3% mortality at 14 and 21 dpi, respectively, and the WA4 colony showed a mortality of 67.7% and 100% at 14 and 21 dpi, respectively. Taken together, the resistance test showed that WA3 was less susceptible than the other colonies and is considered to be partially resistant to GV-0001 (Cyd-X®), which is composed of CpGV-M (genome group A) (Table 1). It is likely that also WA4 shows partial resistance to GV-001 because mortality at day 7 and day 14 was also considerably reduced when compared to LabS or WA1, WA2, and WA5, which showed typical >95% mortality of a susceptible colony when challenged with the discriminating concentration. However, the number of reared larvae was not sufficient for performing additional replicates for WA1, WA2, WA4, and WA5 (Figure 1). Statistical analyses were only conducted for LabS and WA3 colonies, showing that the mortalities of LabS and WA3 induced by GV-0001 were significantly different at 7, 14, and 21 dpi, respectively (p < 0.05) (Figure 1).
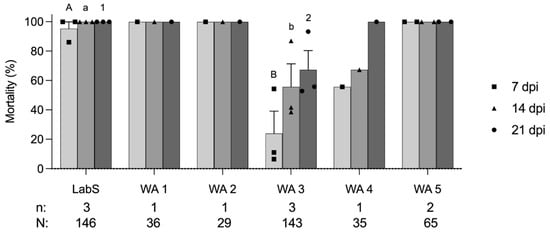
Figure 1.
Mortality (mean ± standard error) at 7, 14, and 21 days post-infection (dpi) of six codling moth colonies LabS, WA1 to WA5 exposed to 6 × 104 OB/mL GV-0001 (Cyd-X®). Each data point representing mortality at 7, 14, and 21 dpi was plotted as a square, triangle, and circle, respectively. Data were analyzed with t-test at p < 0.05. Different capital letters, lowercase letters, and numbers represent the significant differences of mortality at 7, 14, and 21 dpi, respectively. The number of replicates (n) and the total number of tested individuals (N) of each codling moth colony are shown below the chart.
According to product specifications, CpGV products should be applied every 7–8 sunny days, which may easily exceed 10 sprays during a single apple growing season in some regions in Washington State. Comparing the CpGV application history in Europe and the current result, a lack of efficacy of GV-0001 has occurred in Washington State. CpGV-exposed larvae from the WA3 colony have successfully pupated and emerged into adults (data not shown), indicating that resistance to GV-0001 can be passed to the next generation. Laboratory resistance testing using artificial diet mixed with CpGV OBs can achieve reliable results to recognize CpGV-resistant codling moth populations. It is, therefore, a good indicator if a given codling moth population is resistant to CpGV agents and products [34,35].
3.2. Activity Test of CpGV Formulations to WA3
After identifying that GV-0001 had reduced efficacy on the WA3 colony, we tested seven other available CpGV preparations containing different CpGV isolates (Figure 1). All CpGV preparations showed a high virulence to LabS colony (Figure S1), while the WA3 colony had a significantly different susceptibility against CpGV preparations in the tests at 7 dpi (F = 7.64, df = 7, p < 0.05), at 14 dpi (F = 7.51, df = 7, p < 0.05), and at 21 dpi (F = 6.02, df = 7, p < 0.05) (Figure 2). At 7 dpi, the mortality caused by GV-0001, GV-0014 (Madex HP®), and CpGV-S (Virosoft CP4®) was significantly lower than that induced by GV-0015, GV-0013, and GV-0017, respectively (p < 0.05). At 14 dpi, the mortality caused by GV-0014 and CpGV-S was significantly lower than that induced by GV-0015, GV-0013, and GV-0017, respectively (p < 0.05). At 21 dpi, only CpGV-S induced mortality was significantly lower than that induced by GV-0015, GV-0013, and GV-0017, respectively (p < 0.05). The WA3 colony demonstrated low susceptibility to CpGV isolates of the genome groups A and E but not relative to genome group B. This pattern of susceptibility is similar to type II or type III resistance [26]. All newly developed isolates of GV-0015, GV-0013, and GV-0017 consisting of or containing a portion of CpGV belonging to genome group B appear to be more virulent to WA3 larvae than the other CpGV preparation in all tests (Figure 2). These findings are akin to previous reports of resistance tests, and CpGV isolates belonging to genome group B were able to overcome all current types of resistance [21,26]. After 21 dpi, the surviving larvae completed development into adults. When the adults were single-pair crossed, they laid viable eggs, which were used to establish a genetic homogeneous WA3 colony (data not shown), indicating that gene responses to CpGV-resistance were present in the wild WA3 population. Similar characteristics of field codling moth populations containing resistant and susceptible individuals have been observed in European countries [26,36].
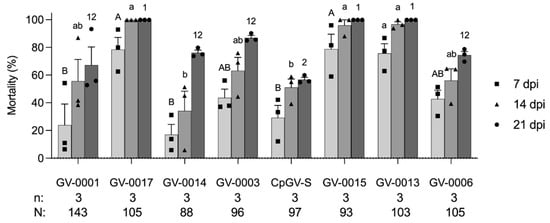
Figure 2.
Mortality (mean ± standard error) of first instars of codling moth from WA3 colony exposed to eight CpGV formulations at a concentration of 6 × 104 OBs/mL. Mortality was recorded at 7, 14, and 21 days post infection (dpi). Each data point representing the mortality at 7, 14, and 21 dpi was plotted as a square, triangle, and circle, respectively. Data were analyzed with one-way ANOVA followed by Tukey–Kramer HSD comparison at p < 0.05. Different capital letters, lowercase letters, and numbers represent the significant differences of mortality at 7, 14, and 21 dpi, respectively. All tested individuals (N) and replicates (n) are shown below the chart.
Here, for the first time in the USA, we document the resistance of codling moth to CpGV-M (= GV-0001) in Washington State, USA, indicating that the genetic background to establish CpGV resistance is not restricted to codling moth populations in Europe. To further investigate the resistance observed in the WA3 colony, the mechanism and inheritance of resistance require further investigation. As a first step, inbreeding and further selection is necessary to establish a genetically homogenous line. Single-pair crossing experiments and resistance tests will then be needed to identify the mode of inheritance and to evaluate if the noted CpGV resistance of WA3 is indeed similar to type II or type III resistance [23,26]. From our assays, it can be concluded that it does not follow the resistance pattern of type I resistance given the lack of susceptibility to CpGV variants belonging to genome group A or E (GV-0001, GV-0003, GV-0014, GV-0006, and CpGV-S) [23,26]. Learning from the lessons on CpGV resistance in Europe, it is important to carefully monitor the performance of currently used CpGV products in the USA and to conduct more resistance testing of codling moth populations where high infestations and control failures of CpGV products are noted [22,25,27]. On the other hand, CpGV application in the USA and elsewhere needs to be carefully and properly carried out in accordance to the appropriate degree-day timing and life stage target and weather conditions [9,19,37], aiming to use CpGV products in the most sustainable way possible. With the identification of CpGV preparations causing sufficient mortality of WA3, novel CpGV products could be made available to growers to overcome high infestations rates caused by codling moth.
4. Conclusions
The results indicated that GV-0001, the main isolate of Cyd-X®, lacked efficacy against WA3 colony and a third of inoculated WA3 larvae survived until the end of the bioassay. One other colony, WA4, has become less susceptible to GV-0001, suggesting that resistance may be more widespread than indicated by our study. It was further confirmed that colony WA3 was the first reported case of CpGV resistance in the USA and WA3 shows resistance to all commercial CpGV products available on the US market. Most importantly, three newly developed CpGV formulations showed similar efficacy as Cyd-X®, in susceptible codling moth populations offering new possibilities to improve control efficacy and successfully kill CpGV-resistant CM populations in the USA.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/insects13060533/s1, Figure S1: Susceptibility (mean ± standard error) of eight CpGV products to LabS colony.
Author Contributions
Conceptualization, J.A.J. and A.L.N.; colony maintenance A.R. and J.F., methodology, J.A.J. and J.F.; data curation, J.F., A.R. and A.L.N.; formal analysis, J.F. and A.L.N.; writing—original draft preparation, J.F.; writing—review and editing, J.A.J. and A.L.N.; funding acquisition, A.L.N. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported in part by a Certis USA and Andermatt BioControl gift and specific cooperative agreement with Rutgers, the State University of New Jersey. Mention of specific products or active ingredients in this publication is solely for the purpose of providing information and does not imply recommendation or endorsement by Rutgers.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
Special thanks to the staff at JKI in Darmstadt, Germany for their assistance in establishing protocols.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Balaško, K.M.; Bažok, R.; Mikac, K.M.; Lemic, D.; Živković, P.I. Pest management challenges and control practices in codling moth: A review. Insects 2020, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Jumean, Z.; Wood, C.; Gries, G. Frequency distribution of larval codling moth, Cydia pomonella L. aggregations on trees in unmanaged apple orchards of the Pacific Northwest. Environ. Entomol. 2009, 38, 1395–1399. [Google Scholar] [CrossRef] [PubMed]
- Lacey, L.A.; Shapiro-Ilan, D.I.; Glenn, G.M. Post-application of anti-desiccant agents improves efficacy of entomopathogenic nematodes in formulated host cadavers or aqueous suspension against diapausing codling moth larvae (Lepidoptera: Tortricidae). Biocontrol. Sci. Technol. 2010, 20, 909–921. [Google Scholar] [CrossRef]
- Witzgall, P.; Stelinski, L.; Gut, L.; Thomson, D. Codling moth management and chemical ecology. Annu. Rev. Entomol. 2007, 53, 503–522. [Google Scholar] [CrossRef]
- Willett, M.J.; Neven, L.; Miller, C.E. The occurrence of codling moth in low latitude countries: Validation of pest distribution reports. Hort. Technol. 2009, 19, 633–637. [Google Scholar] [CrossRef]
- Pajač, I.; Pejić, I.; Barić, B. Codling Moth, Cydia pomonella (Lepidoptera: Tortricidae)—Major pest in apple production: An overview of its biology, resistance, genetic structure and control strategies. Agric. Conspec. Sci. 2011, 76, 87–92. [Google Scholar]
- Reyes, M.; Franck, P.; Charmillot, P.-J.; Ioriatti, C.; Olivares, J.; Pasqualini, E.; Sauphanor, B. Diversity of insecticide resistance mechanisms and spectrum in European populations of the codling moth, Cydia pomonella. Pest Manag. Sci. 2007, 63, 890–902. [Google Scholar] [CrossRef]
- Reyes, M.; Franck, P.; Olivares, J.; Margaritopoulos, J.; Knight, A.; Sauphanor, B. Worldwide variability of insecticide resistance mechanisms in the codling moth, Cydia pomonella L. (Lepidoptera: Tortricidae). Bull. Entomol. Res. 2009, 99, 359–369. [Google Scholar] [CrossRef]
- Lacey, L.A.; Thomson, D.; Vincent, C.; Arthurs, S.P. Codling moth granulovirus: A comprehensive review. Biocontrol. Sci. Technol. 2008, 18, 639–663. [Google Scholar] [CrossRef]
- Pringle, K.L.; Eyles, D.K.; Brown, L. Trends in codling moth activity in apple orchards under mating disruption using pheromones in the Elgin area, Western Cape Province, South Africa. Afr. Entomol. 2003, 11, 65–75. [Google Scholar]
- Arthurs, S.P.; Lacey, L.A.; Miliczky, E.R. Evaluation of the codling moth granulovirus and spinosad for codling moth control and impact on non-target species in pear orchards. Biol. Control. 2007, 41, 99–109. [Google Scholar] [CrossRef]
- Liu, W.; Xu, J.; Zhang, R. The optimal sex pheromone release rate for trapping the codling moth Cydia pomonella (Lepidoptera: Tortricidae) in the field. Sci. Rep. 2016, 6, 21081. [Google Scholar] [CrossRef] [PubMed]
- Harrison, R.L.; Herniou, E.A.; Jehle, J.A.; Theilmann, D.A.; Burand, J.P.; Becnel, J.J.; Krell, P.J.; van Oers, M.M.; Mowery, J.D.; Bauchan, G.R. ICTV Report Consortium, ICTV virus taxonomy profile: Baculoviridae. J. Gen. Virol. 2018, 99, 1185–1186. [Google Scholar] [CrossRef] [PubMed]
- Hess, R.T.; Falcon, L.A. Temporal events in the invasion of the codling moth, Cydia pomonella, by a granulosis virus: An electron microscope study. J. Invertebr. Pathol. 1987, 50, 85–105. [Google Scholar] [CrossRef]
- Steineke, S.B.; Jehle, J.A. Mathematical Modeling of the Population Dynamics of Genetically Modified Microorganisms Using Baculoviruses as an Example; Umweltbundesamt: Berlin, Germany, 2002; pp. 35–37. [Google Scholar]
- Huber, J. Western Europe. In Insect Viruses and Pest Management; Hunter-Fujita, F.R., Entwistle, P.F., Evans, H.F., Crook, N.E., Eds.; John Wiley & Sons, Inc.: New York, NY, USA; Chichester, UK, 1998; pp. 201–215. [Google Scholar]
- Huber, J.; Dickler, E. Codling moth granulosis virus: Its efficiency in the field in comparison with organophosphorus insecticides. J. Econ. Entomol. 1977, 70, 557–561. [Google Scholar] [CrossRef]
- Arthurs, S.; Dara, S.K. Microbial biopesticides for invertebrate pests and their markets in the United States. J. Invertebr. Pathol. 2019, 165, 13–21. [Google Scholar] [CrossRef]
- Arthurs, S.P.; Lacey, L.A. Field evaluation of commercial formulations of the codling moth granulovirus: Persistence of activity and success of seasonal applications against natural infestations of codling moth in Pacific Northwest apple orchards. Biol. Control. 2004, 31, 388–397. [Google Scholar] [CrossRef]
- Tanada, Y.A. Granulosis virus of codling moth, Carpocapsa pomenella (Linnaeus) (Olethreutidae, Lepidoptera). J. Insect Pathol. 1964, 6, 39–47. [Google Scholar]
- Fan, J.; Jehle, J.A.; Wennmann, J.T. Population structure of Cydia pomonella granulovirus isolates revealed by quantitative analysis of genetic variation. Virus Evol. 2021, 7, veaa073. [Google Scholar] [CrossRef]
- Fritsch, E.; Undorf-Spahn, K.; Kienzle, J.; Zebitz, C.P.W.; Huber, J. Apfelwickler-Granulovirus: Erste Hinweise auf Unterschiede in der Empfindlichkeit lokaler Apfelwickler-Populationen. Nachrbl. Deut Pflanzenschutzd 2005, 57, 29–34. [Google Scholar]
- Asser-Kaiser, S.; Fritsch, E.; Undorf-Spahn, K.; Kienzle, J.; Eberle, K.E.; Gund, N.A.; Reineke, A.; Zebitz, C.P.; Heckel, D.G.; Huber, J.; et al. Rapid emergence of baculovirus resistance in codling moth due to dominant, sex-linked inheritance. Science 2007, 317, 1916–1918. [Google Scholar] [CrossRef] [PubMed]
- Berling, M.; Blachere-Lopez, C.; Soubabere, O.; Lery, X.; Bonhomme, A.; Sauphanor, B.; Lopez-Ferber, M. Cydia pomonella granulovirus genotypes overcome virus resistance in the codling moth and improve virus efficiency by selection against resistant hosts. Appl. Environ. Microbiol. 2009, 75, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Zichová, T.; Falta, V.; Kocourek, F.; Stará, J. Difference in the susceptibility of codling moth populations to Cydia pomonella granulovirus in the Czech Republic. Horti. Sci. 2011, 38, 21–26. [Google Scholar] [CrossRef]
- Sauer, A.J.; Schulze-Bopp, S.; Fritsch, E.; Undorf-Spahn, K.; Jehle, J.A. A third type of resistance to Cydia pomonella granulovirus in codling moths shows a mixed Z-Linked and autosomal inheritance pattern. Appl. Environ. Microbiol. 2017, 83, e01036-17. [Google Scholar] [CrossRef] [PubMed]
- Siegwart, M.; Maugin, S.; Besse, S.; Lopez-Ferber, M.; Hinsberger, A.; Gauffre, B. Le carpocapse des pommes résiste au virus de la granulose. Phytoma 2020, 738, 45–50. [Google Scholar]
- Undorf-Spahn, K.; Fritsch, E.; Huber, J.; Kienzle, J.; Zebitz, C.P.; Jehle, J.A. High stability and no fitness costs of the resistance of codling moth to Cydia pomonella granulovirus (CpGV-M). J. Invertebr. Pathol. 2012, 111, 136–142. [Google Scholar] [CrossRef]
- Graillot, B.; Berling, M.; Blachere-Lopez, C.; Siegwart, M.; Besse, S.; Lopez-Ferber, M. Progressive adaptation of a CpGV isolate to codling moth populations resistant to CpGV-M. Viruses 2014, 6, 5135–5144. [Google Scholar] [CrossRef]
- Berling, M.; Rey, J.B.; Ondet, S.J.; Tallot, Y.; Soubabère, O.; Bonhomme, A.; Sauphanor, B.; Lopez-Ferber, M. Field trials of CpGV virus isolates overcoming resistance to CpGV-M. Virol. Sin. 2009, 24, 470. [Google Scholar] [CrossRef]
- 2019 Organic Survey-USDA. Available online: https://www.nass.usda.gov/Publications/AgCensus/2017/Online_Resources/Organics/ORGANICS.pdf (accessed on 22 October 2020).
- Ivaldi-Sender, C. Techniques simples pour un elevage permanent de la Tordeuse orientale, Grapholita molesta (Lepidoptera, Tortricidae) sur milieu artificiel. Ann. Zool. Ecol. Anim. 1974, 6, 337–343. [Google Scholar]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Schmitt, A.; Bisutti, I.L.; Ladurner, E.; Benuzzi, M.; Sauphanor, B.; Kienzle, J.; Zingg, D.; Undorf-Spahn, K.; Fritsch, E.; Huber, J.; et al. The occurrence and distribution of resistance of codling moth to Cydia pomonella granulovirus in Europe. J. Appl. Entomol. 2013, 137, 641–649. [Google Scholar] [CrossRef]
- Zingg, D.; Züger, M.; Bollhalder, F.; Andermatt, M. Use of resistance overcoming CpGV isolates and CpGV resistance situation of the codling moth in Europe seven years after the first discovery of resistance to CpGV-M. IOBC-WPRS Bull. 2011, 66, 401–404. [Google Scholar]
- Graillot, B.; Blachere-López, C.; Besse, S.; Siegwart, M.; López-Ferber, M. Importance of the host phenotype on the preservation of the genetic diversity in codling moth granulovirus. Viruses 2019, 11, 621. [Google Scholar] [CrossRef] [PubMed]
- Pitcairn, M.J.; Zalom, F.G.; Rice, R.E. Degree-day forecasting of generation time of Cydia pomonella (Lepidoptera: Tortricidae) populations in California. Environ. Entomol. 1992, 21, 441. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).