Mosquitoes Larvicidal Activity of Ocimum kilimandscharicum Oil Formulation under Laboratory and Field-Simulated Conditions
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Collection
2.2. Extraction of Essential Oil of O. kilimandscharicum
2.3. Identification of Constituents of O. kilimandscharicum Essential Oil
2.4. O. kilimandscharicum Essential Oil Formulations
2.4.1. Emulsified O. kilimandscharicum Oil Formulation
2.4.2. O. kilimanscharicum Essential Oil in a Solvent
2.5. Sources of Mosquito Larvae
2.6. Larvicidal Activity of O. kilimandscharicum Oil
2.7. Effects of Sublethal Doses of Emulsified O. kilimandscharicam Oil Formulation
2.8. Larvicidal Activity under Field-Simulated Conditions
2.9. Data Analysis
3. Results
3.1. Oil Yield of O. kilimandscharicum
3.2. Chemical Composition of the Essential Oil
3.3. Laboratory Larvicidal Bioassay
3.4. Effects of Sublethal Doses of the Emulsified O. kilimandscharicum Oil Formulation
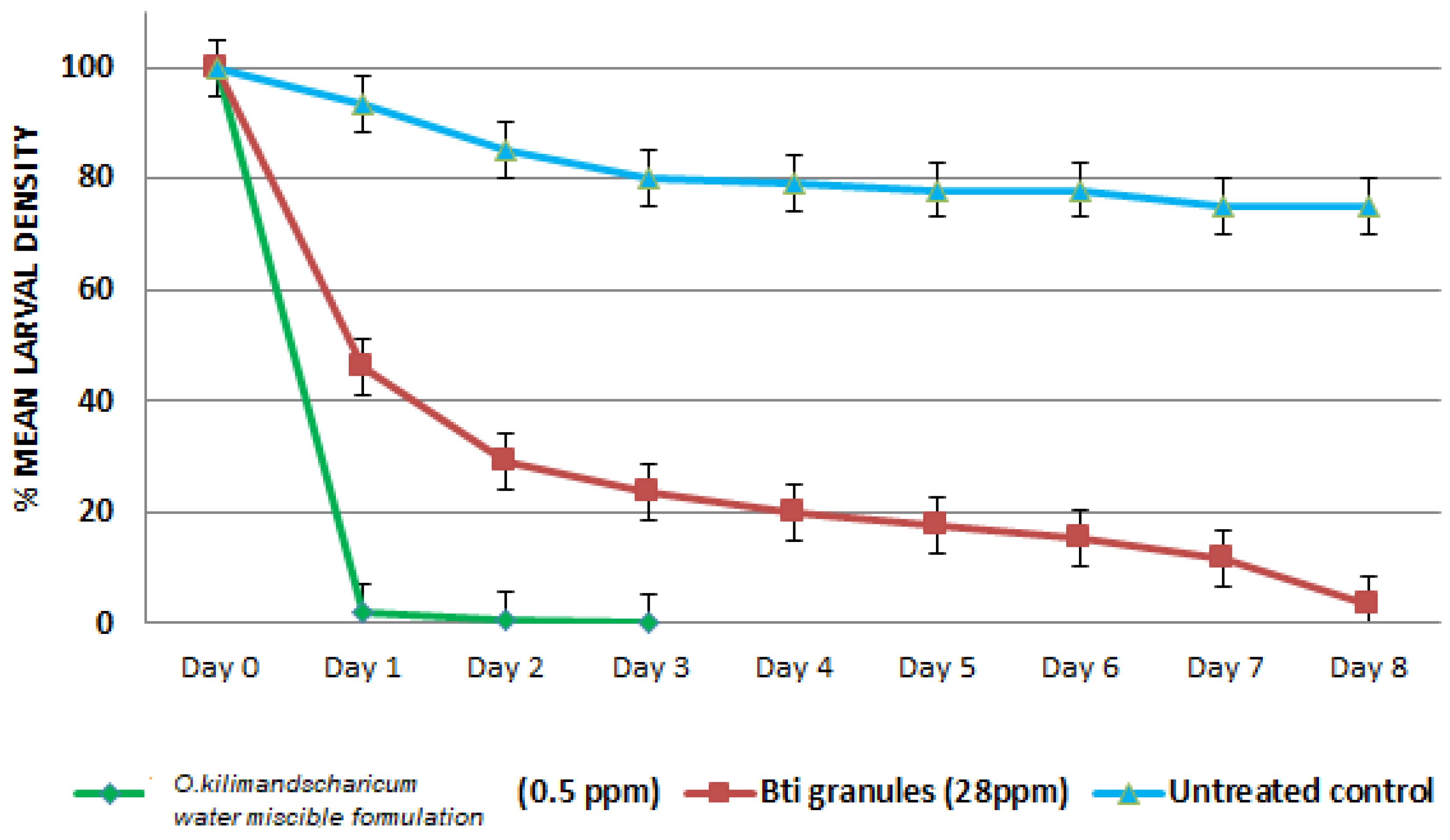
3.5. Field-Simulated Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rueda, L.M. Global diversity of mosquitoes (Insecta: Diptera: Culicidae) in freshwater. Hydrobiologia 2008, 595, 477–487. [Google Scholar] [CrossRef]
- WHO. World Malaria Report; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Paixão, E.S.; Teixeira, M.G.; Rodrigues, L.C. Zika, chikungunya and dengue: The causes and threats of new and reemerging arboviral diseases. BMJ Glob. Health 2018, 3, e000530. [Google Scholar] [CrossRef]
- Brown, A.W.A. Insecticide resistance in mosquitoes: A pragmatic review. J. Am. Mosq. Control Assoc. 1986, 2, 123. [Google Scholar]
- Liu, H.; Xu, Q.; Zhang, L.; Liu, N. Chlorpyrifos Resistance in Mosquito Culex quinquefasciatus. J. Med. Entomol. 2005, 42, 815–820. [Google Scholar] [CrossRef]
- Ghebreyesus, T.A. World Malaria Report 2005; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Couston, M. L’Assemblee mondiale de l’oms. In Proceedings of the Sixty-Eighth World Health Assembly, Geneva, Switzerland, 18–26 May 2015. [Google Scholar]
- Lee, S.C.; Kim, J.H.; Lee, S.J. Floating of the lobes of mosquito (Aedes togoi) larva for respiration. Sci. Rep. 2017, 7, 43050. [Google Scholar] [CrossRef]
- Pavel, R. Acute toxicity and synergistic and antagonistic effects of the aromatic compounds of some essential oils against Culex quinquefasciatus Say larvae. Parasitol. Res. 2015, 114, 3835–3853. [Google Scholar] [CrossRef]
- Arnason, J.T.; Philogene, B.J.R.; Morand, P. ChemInform Abstract: Insecticides of Plant Origin (ACS Symposium Series No. 387). ChemInform 1990, 21. [Google Scholar] [CrossRef]
- Hedlin, P.A.; Hollingworth, R.M.; Masler, E.P.; Miyamoto, J.; Thompson, D.G. Phytochemicals for Pests Control. J. Am. Chem. Soc. 1997, 658, 388. [Google Scholar]
- Pavela, R.; Vrchotová, N. Insecticidal effect of furanocoumarins from fruits of Angelica archangelica L. against larvae Spodoptera littoralis Bois. Ind. Crops Prod. 2013, 43, 33–39. [Google Scholar] [CrossRef]
- Pavel, R. Insecticidal properties of Pimpinella anisum essential oils against the Culex quinquefasciatus and the non-target organism Daphnia magna. J. Asia-Pac. Entomol. 2014, 17, 287–293. [Google Scholar] [CrossRef]
- Bouzid, M.; Colón-González, F.J.; Lung, T.; Lake, I.R.; Hunter, P.R. Climate change and the emergence of vector-borne diseases in Europe: Case study of dengue fever. BMC Public Health 2014, 14, 781. [Google Scholar] [CrossRef]
- Kangethe, L.N.; Ahmed, H.; Omar, S.; Gathirwa, J.; Kirra, P.; Kaniaru, S.; Kamau, T.; Kimani, F.; Nganga, J.K.; Irungu, L. Synergistic Antiplasmodial Activity of Artemisia annua fractions against in vitro cultures of Plasmodium falciparum. Afr. J. Pharmacol. Ther. 2016, 5, 201–205. [Google Scholar]
- Seyoum, A.; Killeen, G.F.; Kabiru, E.W.; Knols, B.G.J.; Hassan, A. Field efficacy of thermally expelled or live potted repellent plants against African malaria vectors in western Kenya. Trop. Med. Int. Heath. 2003, 8, 1005–1011. [Google Scholar] [CrossRef]
- Santana-Méridas, O.; González-Coloma, A.; Sánchez-Vioque, R. Agricultural residues as a source of bioactive natural products. Phytochem. Rev. 2012, 11, 447–466. [Google Scholar] [CrossRef]
- Walker, K.; Lynch, M. Contributions of Anopheles larval control to malaria suppression in tropical Africa: Review of achievements and potential. Med Vet.-Èntomol. 2007, 21, 2–21. [Google Scholar] [CrossRef]
- Fillinger, U.; Lindsay, S.W. Larval source management for malaria control in Africa: Myths and reality. Malar. J. 2011, 10, 353. [Google Scholar] [CrossRef]
- Junqueira, J.; Mendes, J. Susceptibility of Aedes aegypti (L) to the insect growth regulators diflubenzuron and methoprene in Uberlândia, State of Minas Gerais. Rev. Soc. Bras. Med. Trop. 2007, 40, 612–616. [Google Scholar] [CrossRef]
- Marcombe, S.; Darriet, F.; Agnew, P.; Etienne, M.; Yp-Tcha, M.M.; Yébakima, A.; Corbel, V. Field efficacy of new larvicide products for control of multi-resistant Aedes aegypti populations in Martinique (French West Indies). Am. J. Trop. Med. Hyg. 2011, 84, 118–126. [Google Scholar] [CrossRef]
- World Health Organization. Whopes-recommended compounds and formulations for control of mosquito larvae. PLoS Negl. Trop. Dis. 2016, 10, e0005235. [Google Scholar]
- Khan, G.Z.; Khan, I.; Khan, I.; Ali, A.; Salman, M.; Ullah, K. Evaluation of different formulations of IGRs against Aedes albopictus and Culex quinquefasciatus (Diptera: Culicidae). Asian Pac. J. Trop. Biomed. 2016, 6, 485–491. [Google Scholar] [CrossRef]
- Imbahale, S.S.; Mukabana, W.R. Efficacy of neem chippings for mosquito larval control under field conditions. BMC Ecol. 2015, 15, 8. [Google Scholar] [CrossRef]
- Sukumar, K.; Perich, M.J.; Booba, L.R. Botanical derivatives in mosquito control: A review. J. Am. Mosq. Control Assoc. 1991, 7, 210–237. [Google Scholar]
- Said-Al, H.A.H.; Said-Al, A.H.L.; Hussein, A.H.; Hikal, W.M.; Tkachenko, K.G. Essential Oils with Potential as Insecticidal Agents: A Review. Int. J. Environ. Plan. Manag. 2017, 3, 23–33. [Google Scholar]
- Gajger, I.T.; Dar, S.A. Plant Allelochemicals as Sources of Insecticides. Insects 2021, 12, 189. [Google Scholar] [CrossRef]
- Pavel, R. Essential oils for the development of eco-friendly mosquito larvicides: A review. Ind. Crops Prod. 2015, 76, 174–187. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Stahl-Biskup, E.; Sáez, F. Thyme: The Genus Thymus; CRC Press: Hamburg, Germany, 2003; pp. 1–333. [Google Scholar]
- Miresmailli, S.; Isman, M.B. Botanical insecticides inspired by plant-herbivore chemical interactions. Trends Plant Sci. 2014, 19, 29–35. [Google Scholar] [CrossRef]
- Ghosh, A.; Chowdhury, N.; Chandra, G. Plant extracts as potential mosquito larvicides. Indian J. Med. Res. 2012, 135, 581–598. [Google Scholar]
- Soni, N.; Gill, D.; Sagar, B.S.; Raheja, S.; Agrawal, S. Ocimum kilimandscharicum: A Systematic Review. J. Drug Deliv. Ther. 2012, 2, 45–52. [Google Scholar] [CrossRef]
- Njoroge, G.N.; Bussmann, R.W.; Gemmill, B.; Newton, L.E.; Ngumi, V.W. Utilisation of weed species as sources of traditional medicines in central Kenya. Lyonia 2004, 7, 71–87. [Google Scholar]
- Kashyap, C.P.; Ranjeet, K.; Vikrant, A.; Vipin, K. Therapeutic Potency of Ocimum kilimandscharicumguerke—A Review. Glob. J. Pharmacol. 2011, 5, 191–200. [Google Scholar]
- Sarin, R.V.; Narwal, S.; Bafna, P.A. Anti-diarrhoeal activity of aqueous extract of Ocimum kilimandscharicum. J. Ethnopharmacol. 2013, 148, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Mukungu, N.; Abuga, K.; Okalebo, F.; Ingwela, R.; Mwangi, J. Medicinal plants used for management of malaria among the Luhya community of Kakamega East sub-County, Kenya. J. Ethnopharmacol. 2016, 194, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Singh, D. Advances in Plant Biopesticides; Springer: New Delhi, India, 2014; pp. 1–401. [Google Scholar] [CrossRef]
- Ostertag, F.; Weiss, J.; McClements, D.J. Low-energy formation of edible nanoemulsions: Factors influencing droplet size produced by emulsion phase inversion. J. Colloid Interface Sci. 2012, 388, 95–102. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines for Laboratory and Field Testing of Mosquito Larvicides; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Am. Mosq. Control Assoc. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- WHO. Larval Source Management—A Supplementary Measure for Malaria Vector Control: An Operational Manual; World Health Organization: Geneva, Switzerland, 2013; p. 128. [Google Scholar]
- Singh, K.; Singh, D.K. Toxicity to the snail Limnaea acuminata of plant-derived molluscicides in combination with synergists. Pest Manag. Sci. 2000, 56, 889–898. [Google Scholar] [CrossRef]
- Wei, X.; Koo, I.; Kim, S.; Zhang, X. Compound identification in GC-MS by simultaneously evaluating the mass spectrum and retention index. Analyst 2014, 139, 2507–2514. [Google Scholar] [CrossRef]
- Wassmer, C.S.; Grau, G.E.R. Severe malaria: What’s new on the pathogenesis front? Int. J. Parasitol. 2017, 47, 145–152. [Google Scholar] [CrossRef]
- Ohia, C.M.D.; Ana, G.R.E.E. Bio-insecticides: The one-health response to mosquitoborne diseases of public health importance. J. Biol. Agric. Healthc. 2015, 5, 22–26. [Google Scholar]
- Bekele, J.; Hassanali, A. Blend effects in the toxicity of the essential oil constituents of Ocimum kilimandscharicum and Ocimum kenyense (Labiateae) on two post-harvest insect pests. Phytochemistry 2001, 57, 385–391. [Google Scholar] [CrossRef]
- Dua, V.K.; Pandey, A.C.; Raghavendra, K.; Gupta, A.; Sharma, T.; Dash, A.P. Larvicidal activity of neem oil (Azadirachta indica) formulation against mosquitoes. Malar. J. 2009, 8, 124–126. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, A.K.; Olorunfemi, O.S. Investigation of changes in the levels of some tissue biochemicals in Culex quinquefasciatus (Diptera: Culicidae) exposed to sublethal toxicity of Cypermethrin. Sci. Afr. 2019, 4, e00109. [Google Scholar] [CrossRef]
- Balandrin, M.F.; Klocke, J.A. Medicinal, Aromatic, and Industrial Materials from Plants. In Medicinal and Aromatic Plants I. Biotechnology in Agriculture and Forestry; Bajaj, Y.P.S., Ed.; Springer: Berlin/Heidelberg, Germany, 1988; Volume 4. [Google Scholar] [CrossRef]
- Aygun, D.; Doganay, Z.; Altintop, L.; Guven, H.; Onar, M.; Deniz, T.; Sunter, T. Serum acetylcholinesterase and prognosis of acute organophosphate poisoning. J. Toxicol.—Clin. Toxicol. 2002, 40, 903–910. [Google Scholar] [CrossRef]
- Enan, E. Insecticidal activity of essential oils: Octopaminergic sites of action. Comp. Biochem. Physiol.—Part C Toxicol. Pharmacol. 2001, 130, 325–337. [Google Scholar] [CrossRef]
- Obeng-Ofori, D.; Reichmuth, C.H.; Bekele, A.J.; Hassanali, A. Toxicity and protectant potential of camphor, a major component of essential oil of Ocimum kilimandscharicum, against four stored product beetles. Int. J. Pest Manag. 1998, 44, 203–209. [Google Scholar] [CrossRef]
- Fournier, D.; Mutero, A. Modification of acetylcholinesterase as a mechanism of resistance to insecticides. Comp. Biochem. Physiol.—Part C Toxicol. Pharmacol. 1994, 108, 19–31. [Google Scholar] [CrossRef]
- Kostyukovsky, M.; Rafaeli, A.; Gileadi, C.; Demchenko, N.; Shaaya, E. Activation of octopaminergic receptors by essential oil constituents isolated from aromatic plants: Possible mode of action against insect pests. Pest Manag. Sci. 2002, 58, 1101–1106. [Google Scholar] [CrossRef]
- Miyazawa, M.; Watanabe, H.; Kameoka, H. Inhibition of acetylcholinesterase activity by monoterpenoids with a methane skeleton. J. Agric. Food Chem. 1997, 45, 677–679. [Google Scholar] [CrossRef]
- Mordue, A.J.; Blackwell, A. Azadirachtin: An update. J. Insect Physiol. 1993, 39, 903–924. [Google Scholar] [CrossRef]
- Grundy, D.L.; Still, C.C. Inhibition of acetylcholinesterases by pulegone1,2-epoxide. Pestic. Biochem. Physiol. 1985, 23, 383–388. [Google Scholar] [CrossRef]
- Schmutterer, H. Properties and potential of natural pesticides from the neem tree, Azadirachta indica. Annu. Rev. Entomol. 1990, 35, 271–297. [Google Scholar] [CrossRef] [PubMed]
- Coats, J.R.; Karr, L.L.; Drewes, C.D. Toxicity and Neurotoxic Effects of Monoterpenoids. In Insects and Earthworms; ACS Symposium Series; Department of Entomology and Department of Zoology; Iowa State University: Ames, IA, USA, 1991; pp. 305–316. [Google Scholar]
- Ryan, M.F.; Byrne, O. Plant-insect coevolution and inhibition of acetylcholinesterase. J. Chem. Ecol. 1988, 14, 1965–1975. [Google Scholar] [CrossRef] [PubMed]
- Rajamma, A.; Dubey, S.; Sateesha, S.; Tiwari, S.; Ghosh, S. Comparative larvicidal activity of different species of Ocimum against Culex quinquefasciatus. Nat. Prod. Res. 2011, 25, 1916–1922. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Jayaramaiah, R.H.; Sarate, P.; Thulasiram, H.V.; Kulkarni, M.J.; Giri, A.P. Insecticidal potential of defense metabolites from Ocimum kilimandscharicum against Helicoverpa armigera. PLoS ONE 2014, 9, e104377. [Google Scholar] [CrossRef]
- Chaves, R.D.S.B.; Martins, R.L.; Rodrigues, A.B.L.; Érica, D.M.R.; Farias, A.L.F.; Brandão, L.B.; Santos, L.L.; Galardo, A.K.R.; de Almeida, S.S.M.D.S. Evaluation of larvicidal potential against larvae of Aedes aegypti (Linnaeus, 1762) and of the antimicrobial activity of essential oil obtained from the leaves of Origanum majorana L. PLoS ONE 2020, 15, e0235740. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.P.; Haddi, K.; Corrêa, R.F.T.; Zapata, V.L.; Piau, T.B.; Souza, L.F.; Santos, S.M.G.; Oliveira, E.E.; Jumbo, L.O.; Ribeiro, B.M.; et al. Prolonged mosquitocidal activity of Siparuna guianensis essential oil encapsulated in chitosan nanoparticles. PLoS Negl. Trop. Dis. 2019, 13, e0007624. [Google Scholar] [CrossRef]
- Asawalam, E.F.; Emosairue, S.O.; Hassanali, A. Contribution of different constituents to the toxicity of the essential oil constituents of Vernonia amygdalina (Compositae) and Xylopia aetiopica (Annonaceae) on maize weevil, Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae). Afr. J. Biotechnol. 2008, 7, 2957–2962. [Google Scholar] [CrossRef]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef]
- Kishore, N.; Mishra, B.B.; Tiwari, V.K.; Tripathi, V. A Review on Natural Products with Mosquitocidal Potentials. In Opportunity, Challenge, and Scope of Natural Products in Medicinal Chemistry; ScienceOpen, Inc.: Burlington, MA, USA, 2011. [Google Scholar]

| RI | Compounds | Relative Content (%) |
|---|---|---|
| 923 | Tricyclene | 0.71 |
| 931 | α-Pinene | 1.94 |
| 946 | Camphene | 7.14 |
| 989 | Myrcene | 1.88 |
| 1003 | α-Phellandrene | 0.76 |
| 1008 | δ-2-Carene | 0.68 |
| 1024 | p-Cymene | 0.23 |
| 1029 | Limonene | 18.61 |
| 1046 | (E)-β-Ocimene | 0.92 |
| 1059 | γ-Terpinene | 1.05 |
| 1085 | α-Terpinolene | 2.23 |
| 1093 | 6-Camphenol | 0.6 |
| 1098 | Trans-sabinene hydrate | 0.86 |
| 1100 | Linalool | 4.32 |
| 1143 | Camphor | 36.58 |
| 1158 | Isoborneol | 0.14 |
| 1165 | Borneol | 0.97 |
| 1167 | α-Terpineol | 3.6 |
| 1180 | Terpinen-4-ol | 3.9 |
| 1247 | Geranial | 0.21 |
| 1286 | (E)-Linalool oxide acetate | 1.4 |
| 1351 | α-Cubebene | 0.08 |
| 1357 | Eugenol | 0.14 |
| 1378 | α-Copaene | 0.5 |
| 1419 | (E)-β-Caryophyllene | 1.94 |
| 1433 | β-Copaene | 1.07 |
| 1450 | trans-Muurola-3,5-diene | 0.26 |
| 1455 | α-Humulene | 0.38 |
| 1470 | Bicyclogermacrene | 0.47 |
| 1476 | Geranyl propanoate | 1.28 |
| 1480 | γ-Muurolene | 0.12 |
| 1482 | Germacrene D | 1.17 |
| 1497 | Viridiflorene | 0.46 |
| 1503 | α-Bisabolene | 0.12 |
| 1541 | Sesquisabinene hydrate, cis- | 2.12 |
| 1601 | Humulene epoxide II | 0.08 |
| 1700 | Geranyl propanoate | 0.17 |
| 1470 | Dauca-5,8-diene | 0.23 |
| Treatment | Mosquito Species | Time (h) | Concentrations (ppm)/% Mortality | Lethal Concentration | Treatment Response | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control (-ve)-Water | 0.1 | 0.25 | 0.5 | 1 | LC50 (95%CL) | LC90 (95%CL) | R2 | X2, df, p-Value | |||
| O. kilimandscharicum oil in acetone | An. gambiae | 24 | 0.0 | 4.7 | 18.2 | 37.1 | 60.0 | 0.74 (0.07–0.97) | 3.45 (2.13–8.73) | 0.9929 | X2 = 1954.00 df = 18 p < 0.0001 |
| 48 | 0.0 | 20.4 | 43.6 | 63.4 | 100.0 | 0.31 (0.11–0.87) | 1.82 (0.06–51.63) | 0.9786 | X2 = 2025.06 df = 4, p < 0.0001 | ||
| O. kilimandscharicum water-emulsion | An. gambiae | 24 | 0.0 | 34.2 | 67.2 | 75.9 | 99.4 | 0.07 (0.61–0.13) | 2.39 (1.08–19.70) | 0.794 | X2 = 1923.69 d 8, p< 0.0001 |
| 48 | 0.0 | 21.0 | 94.0 | 99.9 | 100.0 | 0.14 (0.09–0.20) | 0.22 (0.10–0.49) | 0.9394 | X2 = 794.00, Df = 4, p < 0.0001 | ||
| An. arabiensis | 24 | 0.0 | 29.9 | 46.2 | 59.0 | 71.0 | 0.31 (0.18–0.50) | 4.72 (1.74–117.50) | 0.87635 | X2 = 4688.80 df = 18, p < 0.0001 | |
| 48 | 0.0 | 43.2 | 62.1 | 74.8 | 84.8 | 0.14 (0.07–0.20) | 1.63 (0.86–7.52) | 0.8394 | X2 = 3052.08 d 8, p < 0.0001 | ||
| Emulsion/surfactant (Tween 80) | An. arabiensis | 24 | 0.0 | 0.4 | 1.1 | 2.3 | 4.3 | 75.28 (10.70) | 1877.55 (69.99) | 0.9978 | X2 = 47.97 df = 18, p < 0.0001 |
| 48 | 0.0 | 0.9 | 2.3 | 4.3 | 7.7 | 30.162 (8.63–1145.80) | 648.88 (68.05–475,024.38) | 0.9973 | X2 = 75.11 df = 18, p < 0.0001 | ||
| An. gambiae | 24 | 0.0 | 1.3 | 2.6 | 4.1 | 6.3 | 172.57 (14.78–0.00) | 12,795.98 (172.20 | 0.9694 | X2 = 111.91 df = 18, p < 0.0001 | |
| 48 | 0.0 | 1.6 | 3.6 | 6.1 | 9.9 | 31.99 (8.13–615.21) | 1011.90 (78.57–3,997,163.24) | 0.9847 | X2 = 142.31 df = 18, p < 0.0001 | ||
| Treatment (ppm) | An. gambiae | An. arabiensis | ||
|---|---|---|---|---|
| Mean No of Pupae (M ± SE) | Mean No of Adults (M ± SE) | Mean No of Pupae (M ± SE) | Mean No of Adults (M ± SE) | |
| LC70 | 31.3 ± 1.68 a | 29.0 ± 2.3 a | 32.0 ± 1.15 a | 22.3 ± 1.28 a |
| LC50 | 49.7 ±1.68 b | 44.7 ± 2.3 b | 45.0 ± 1.15 b | 39.0 ± 1.28 b |
| LC20 | 71.3 ± 1.68 c | 67.0 ± 2.3 c | 65.0 ± 1.15 c | 62.3 ± 1.28 c |
| Control | 79.0 ± 1.68 d | 75.3 ± 2.3 d | 73.0 ± 1.15 d | 68.0 ± 1.28 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ochola, J.B.; Mutero, C.M.; Marubu, R.M.; Haller, B.F.; Hassanali, A.; Lwande, W. Mosquitoes Larvicidal Activity of Ocimum kilimandscharicum Oil Formulation under Laboratory and Field-Simulated Conditions. Insects 2022, 13, 203. https://doi.org/10.3390/insects13020203
Ochola JB, Mutero CM, Marubu RM, Haller BF, Hassanali A, Lwande W. Mosquitoes Larvicidal Activity of Ocimum kilimandscharicum Oil Formulation under Laboratory and Field-Simulated Conditions. Insects. 2022; 13(2):203. https://doi.org/10.3390/insects13020203
Chicago/Turabian StyleOchola, John Bwire, Clifford Maina Mutero, Rose Muthoni Marubu, Barbara Frei Haller, Ahmed Hassanali, and Wilber Lwande. 2022. "Mosquitoes Larvicidal Activity of Ocimum kilimandscharicum Oil Formulation under Laboratory and Field-Simulated Conditions" Insects 13, no. 2: 203. https://doi.org/10.3390/insects13020203
APA StyleOchola, J. B., Mutero, C. M., Marubu, R. M., Haller, B. F., Hassanali, A., & Lwande, W. (2022). Mosquitoes Larvicidal Activity of Ocimum kilimandscharicum Oil Formulation under Laboratory and Field-Simulated Conditions. Insects, 13(2), 203. https://doi.org/10.3390/insects13020203






