More Power with Flower for the Pupal Parasitoid Trichopria drosophilae: A Candidate for Biological Control of the Spotted Wing Drosophila
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Rearing of Insects and Preparation of Plants
2.2. Evaluation of Floral Diet on Trichopria drosophilae Performance
2.3. Evaluation of Floral Diet on Drosophila suzukii
2.4. Data Analysis
3. Results
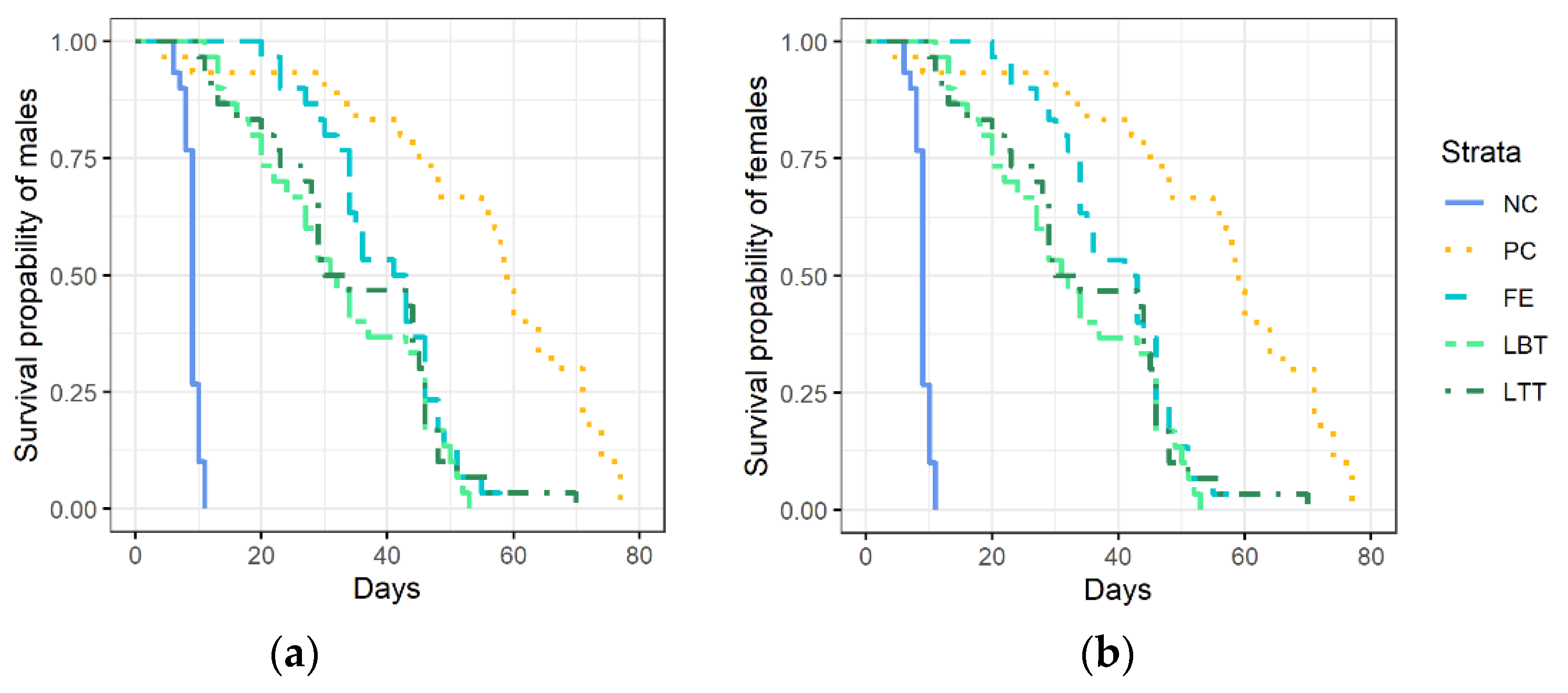
3.1. Survival of Trichopria drosophilae at Different Diets
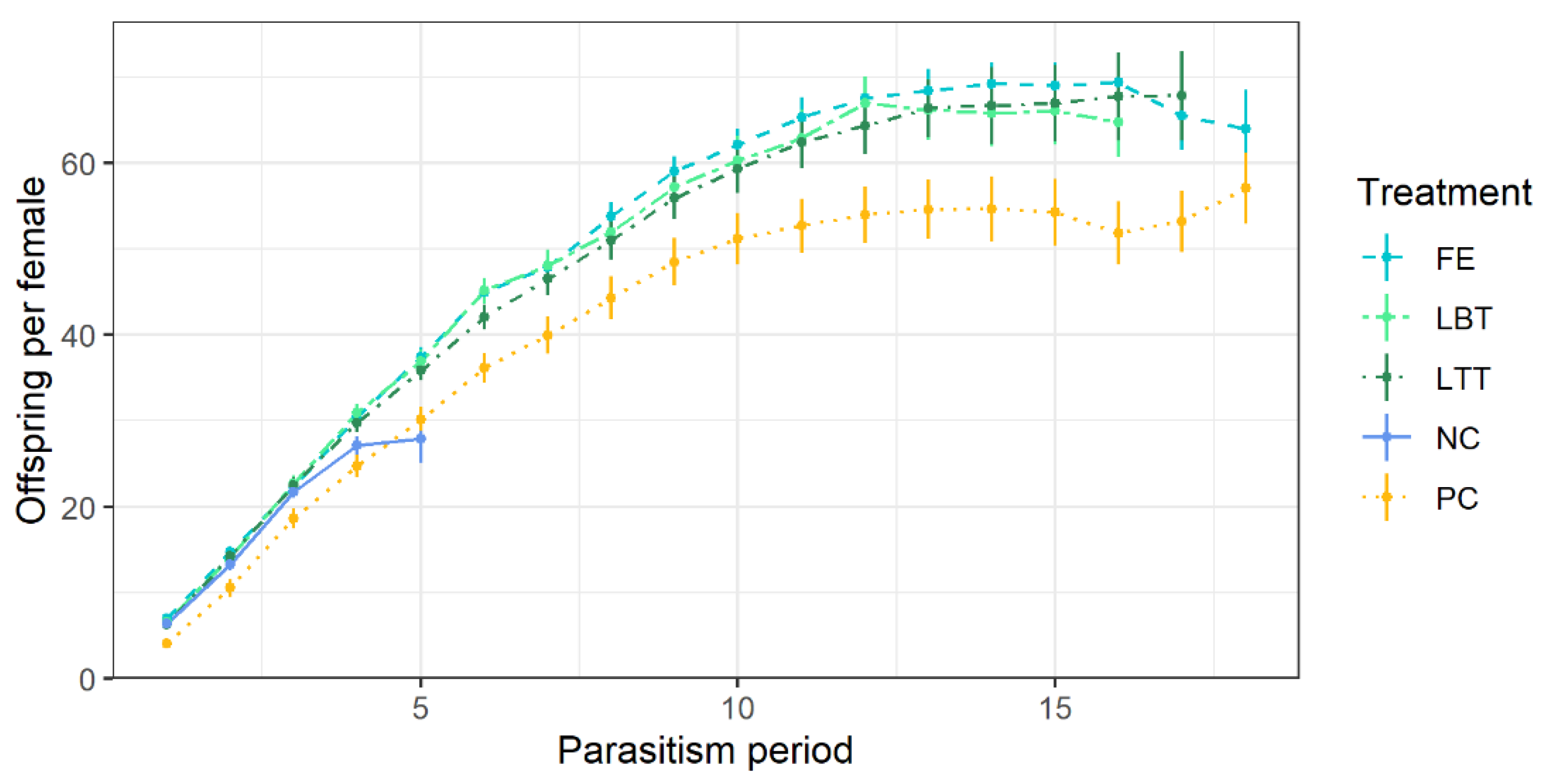
3.2. Parasitism Performance of Trichopria drosophilae at Different Diets
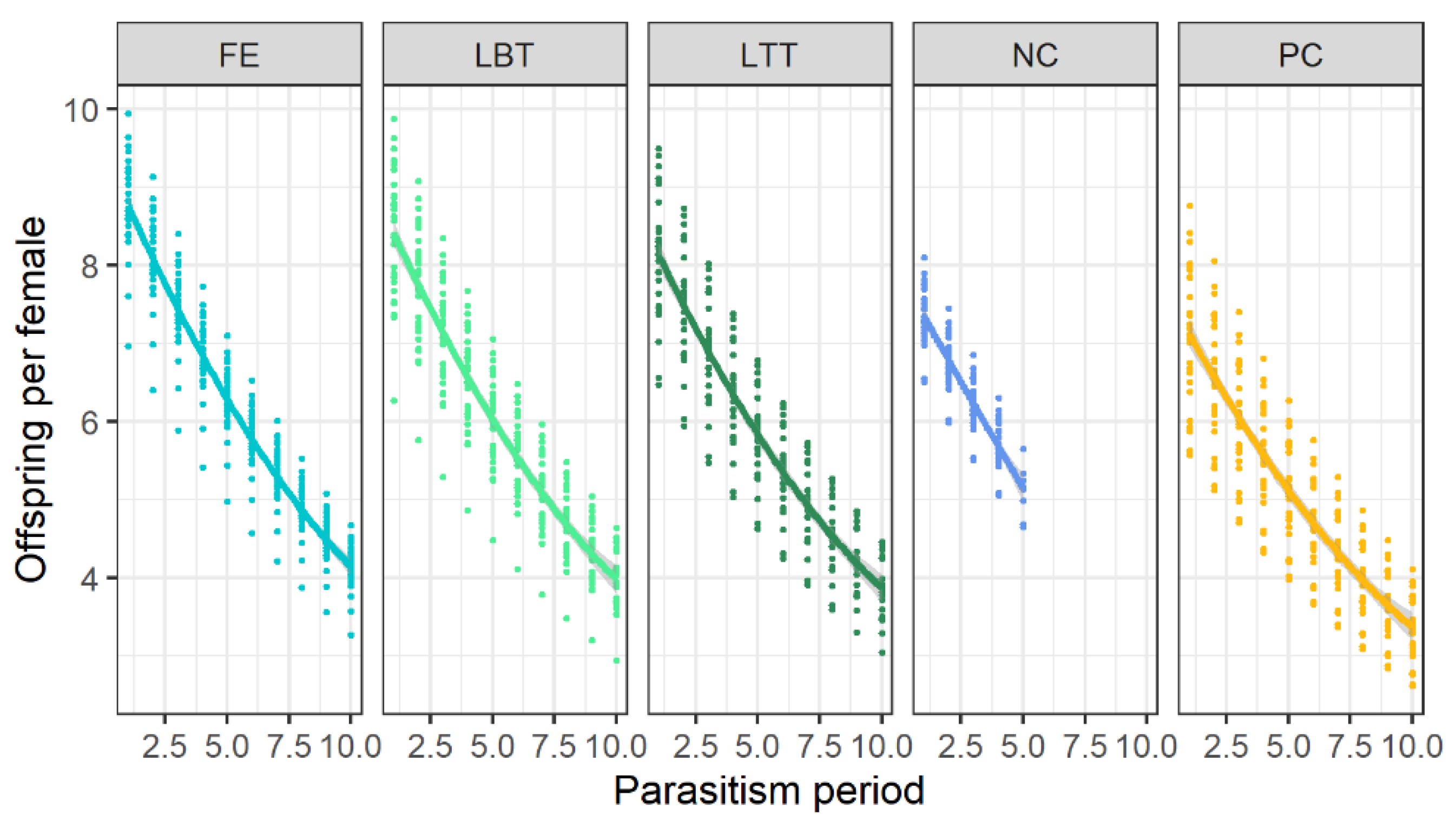
3.3. Effects on F1-Progeny of Trichopria drosophilae at Different Diets
3.4. Life Table Parameters of Trichopria drosophilae at Different Diets
3.5. Effects of Different Diets on Drosophila suzukii Performance
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Benelli, G.; Giunti, G.; Tena, A.; Desneux, N.; Caselli, A.; Canale, A. The impact of adult diet on parasitoid reproductive performance. J. Pest Sci. 2017, 90, 807–823. [Google Scholar] [CrossRef]
- Zhu, P.; Zheng, X.; Xie, G.; Chen, G.; Lu, Z.; Gurr, G. Relevance of the ecological traits of parasitoid wasps and nectariferous plants for conservation biological control: A hybrid meta-analysis. Pest Manag. Sci. 2020, 76, 1881–1892. [Google Scholar] [CrossRef] [PubMed]
- Deguine, J.-P.; Muru, D.; Plessix, S. Interactions Between Flowering Plants and Arthropods in Organic Agroecosystems. In Organic Farming; Elsevier: Amsterdam, The Netherlands, 2019; pp. 213–249. ISBN 9780128132722. [Google Scholar]
- Falagiarda, M.; Schmidt, S. Parasitization effectiveness of Trichopria drosophilae against Drosophila suzukii in release trials in the field. ATTI Giornate Fitopatol. 2018, 211–216. [Google Scholar]
- Garcia-Cancino, M.D.; Gonzalez-Cabrera, J.; Sanchez-Gonzalez, J.A.; Arredondo-Bernal, H.C. Biological and Population Parameters, as well as Oviposition Preference, of Two Pupal Parasitoids of Drosophila suzukii (Diptera: Drosophilidae) in Mexico. J. Entomol. Sci. 2020, 55, 87–97. [Google Scholar] [CrossRef]
- Gonzalez-Cabrera, J.; Moreno-Carrillo, G.; Sanchez-Gonzalez, J.; Mendoza-Ceballos, M.; Arredono-Bernal, H. Single and Combined Release of Trichopria drosophilae (Hymenoptera: Diapriidae) to Control Drosophila suzukii (Diptera: Drosophilidae. Neotrop. Entomol. 2019, 48, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Pfab, F.; Rossi Stacconi, M.V.; Anfora, G.; Grassi, A.; Walton, V.; Pugliese, A. Optimized timing of parasitoid release: A mathematical model for biological control of Drosophila suzukii. Theor. Ecol. 2018, 11, 489–501. [Google Scholar] [CrossRef]
- Lee, J.C.; Wang, X.; Daane, K.M.; Hoelmer, K.A.; Isaacs, R.; Sial, A.A.; Walton, V.M. Biological Control of Spotted-Wing Drosophila (Diptera: Drosophilidae)-Current and Pending Tactics. J. Integr. Pest Manag. 2019, 10. [Google Scholar] [CrossRef]
- Rossi Stacconi, M.V.; Grassi, A.; Ioriatti, C.; Anfora, G. Augmentative releases of Trichopria drosophilae for the suppression of early season Drosophila suzukii populations. Biol. Control 2019, 64, 9–19. [Google Scholar] [CrossRef]
- Rossi Stacconi, M.V.; Amiresmaeili, N.; Biondi, A.; Carli, C.; Caruso, S.; Dindo, M.L.; Francati, S.; Gottardello, A.; Grassi, A.; Lupi, D.; et al. Host location and dispersal ability of the cosmopolitan parasitoid Trichopria drosophilae released to control the invasive spotted wing Drosophila. Biol. Control 2018, 117, 188–196. [Google Scholar] [CrossRef]
- Wang, X.-G.; Kacar, G.; Biondi, A.; Daane, K.M. Life-history and host preference of Trichopria drosophilae, a pupal parasitoid of spotted wing drosophila. Biol. Control 2016, 61, 387–397. [Google Scholar] [CrossRef]
- Collatz, J.; Romeis, J. Flowers and fruits prolong survival of drosophila pupal parasitoids. J. Appl. Entomol. 2021, 145, 629–634. [Google Scholar] [CrossRef]
- Ellers, J.; Ruhe, B.; Visser, B. Discriminating between energetic content and dietary composition as an explanation for dietary restriction effects. J. Insect. Physiol. 2011, 57, 1670–1676. [Google Scholar] [CrossRef] [PubMed]
- Carton, Y.; Bouletreau, M.; Van Alphen, J.J.M.; van Lenteren, J.C. The Drosophila Parasitic Wasps. In The Genetics and Biology of Drosophila; Ashburner, M., Carson, H.L., Thompson, J.N., Jr., Eds.; Academic Press: Cambridge, MA, USA, 1986; pp. 347–394. [Google Scholar]
- Jervis, M.; Kidd, N.A.C.; Fitton, M.G.; Huddleston, T.; Dawah, H.A. Flower-visiting by hymenopteran parasitoids. J. Nat. Hist. 1993, 27, 67–105. [Google Scholar] [CrossRef]
- Russell, M. A meta-analysis of physiological and behavioral responses of parasitoid wasps to flowers of individual plant species. Biol. Control 2015, 82, 96–103. [Google Scholar] [CrossRef]
- Bertazzini, M.; Forlani, G. Intraspecific Variability of Floral Nectar Volume and Composition in Rapeseed (Brassica napus L. var. oleifera). Front. Plant. Sci. 2016, 7, 288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Englert, C.; Herz, A. Native predators and parasitoids for biological regulation of Drosophila suzukii in Germany. In Proceedings of the 17th International Conference on Organic Fruit-Growing—Ecofruit, Hohenheim, Germany, 15–17 February 2016; Foerdergemeinschaft Oekologischer Obstbau e.V., Ed.; pp. 284–285. [Google Scholar]
- Englert, C.; Dumath, E.; Herz, A. Trichopria drosophilae—A potential candidate to control Drosophila suzukii in Germany? Berichte aus dem Julius Kühn-Institut 2017, 192, 19. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Birch, L.C. The intrinsic rate of natural increase of an insect population. J. Anim. Ecol. 1948, 17, 15–26. [Google Scholar] [CrossRef]
- Yongfu, L. Constructing Life Tables with R. Available online: https://yongfu.name/2017/12/11/Life_Tables.html#comment-4929152334 (accessed on 5 March 2021).
- Rossi Stacconi, M.V.; Panel, A.; Baser, N.; Ioriatti, C.; Pantezzi, T.; Anfora, G. Comparative life history traits of indigenous Italian parasitoids of Drosophila suzukii and their effectiveness at different temperatures. Biol. Control 2017, 112, 20–27. [Google Scholar] [CrossRef]
- Jervis, M.A.; Ellers, J.; Harvey, J.A. Resource acquisition, allocation, and utilization in parasitoid reproductive strategies. Annu. Rev. Entomol. 2008, 53, 361–385. [Google Scholar] [CrossRef] [Green Version]
- Chappell, C.R.; Fukami, T. Nectar yeasts: A natural microcosm for ecology. Yeast 2018, 35, 417–423. [Google Scholar] [CrossRef] [Green Version]
- Schaeffer, R.N.; Mei, Y.Z.; Andicoechea, J.; Manson, J.S.; Irwin, R.E. Consequences of a nectar yeast for pollinator preference and performance. Funct. Ecol. 2017, 31, 613–621. [Google Scholar] [CrossRef]
- Gamez-Virues, S.; Gurr, G.M.; Raman, A.; La Salle, J.; Nicol, H. Effects of flowering groundcover vegetation on diversity and activity of wasps in a farm shelterbelt in temperate Australia. Biol. Control 2009, 54, 211–218. [Google Scholar] [CrossRef]
- Li, J.; Wang, Y.; Zhu, C.-J.; Zhang, M.; Hu, H.-Y. Offspring sex ratio shifts of the solitary parasitoid wasp, Trichopria drosophilae (Hymenoptera: Diapriidae), under local mate competition. Entomol. Fenn. 2018, 29, 97–104. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Li, M.-Y.; Xiong, Y.; LIu, S.-N.; Xiao, C.; Tang, G.-W. Mating behavior of Trichopria drosophilae and the effect of male mating frequency on the production of female offspring. Chin. J. Appl. Entomol. 2017, 54, 749–754. [Google Scholar]
- Woltering, S.B.; Romeis, J.; Collatz, J. Influence of the Rearing Host on Biological Parameters of Trichopria drosophilae, a Potential Biological Control Agent of Drosophila suzukii. Insects 2019, 10, 12. [Google Scholar]
- Chen, J.; Zhou, S.; Wang, Y.; Shi, M.; Chen, X.; Huang, J. Biocontrol characteristics of the fruit fly pupal parasitoid Trichopria drosophilae (Hymenoptera: Diapriidae) emerging from different hosts. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Häussling, B.J.M.; Lienenlüke, J.; Stökl, J. The preference of Trichopria drosophilae for pupae of Drosophila suzukii is independent of host size. Sci. Rep. 2021, 11, 995. [Google Scholar] [CrossRef]
- Araj, S.-E.; Shields, M.W.; Wratten, S.D. Weed floral resources and commonly used insectary plants to increase the efficacy of a whitefly parasitoid. Biol. Control 2019, 64, 553–561. [Google Scholar] [CrossRef]
- Araj, S.-E.; Wratten, S.D. Comparing existing weeds and commonly used insectary plants as floral resources for a parasitoid. Biol. Control 2015, 81, 15–20. [Google Scholar] [CrossRef]
- Arno, J.; Oveja, M.F.; Gabarra, R. Selection of flowering plants to enhance the biological control of Tuta absoluta using parasitoids. Biol. Control 2018, 122, 41–50. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Mao, J.; Reynolds, O.L.; Chen, W.; He, W.; You, M.; Gurr, G.M. Alyssum (Lobularia maritima) selectively attracts and enhances the performance of Cotesia vestalis, a parasitoid of Plutella xylostella. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef] [Green Version]
- Munir, S.; Dosdall, L.M.; Keddie, A. Selective effects of floral food sources and honey on life-history traits of a pest-parasitoid system. Entomol. Exp. Appl. 2018, 166, 500–507. [Google Scholar] [CrossRef] [Green Version]
- Johanowicz, D.L.; Mitchell, E.R. Effects of sweet alyssum flowers on the longevity of the parasitoid wasps Cotesia marginiventris (Hymenoptera: Braconidae) and Diadegma insulare (Hymenoptera: Ichneumonidae). Fla. Entomol. 2000, 83, 41–47. [Google Scholar] [CrossRef]
- Berndt, L.A.; Wratten, S.D. Effects of alyssum flowers on the longevity, fecundity, and sex ratio of the leafroller parasitoid Dolichogenidea tasmanica. Biol. Control 2005, 32, 65–69. [Google Scholar] [CrossRef]
- McIntosh, H.R.; Skillman, V.P.; Galindo, G.; Lee, J.C. Floral Resources for Trissolcus japonicus, a Parasitoid of Halyomorpha halys. Insects 2020, 11, 413. [Google Scholar] [CrossRef] [PubMed]
- Barbir, J.; Badenes-Perez, F.R.; Fernandez-Quintanilla, C.; Dorado, J. The attractiveness of flowering herbaceous plants to bees (Hymenoptera: Apoidea) and hoverflies (Diptera: Syrphidae) in agro-ecosystems of Central Spain. Agric. For. Entomol. 2015, 17, 20–28. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, A.L.; Gontijo, L.M. Alyssum flowers promote biological control of collard pests. Biol. Control 2017, 62, 185–196. [Google Scholar] [CrossRef]
- Amorós-Jiménez, R.; Pineda, A.; Fereres, A.; Marcos-García, M.Á. Feeding preferences of the aphidophagous hoverfly Sphaerophoria rueppellii affect the performance of its offspring. Biol. Control 2014, 59, 427–435. [Google Scholar] [CrossRef]
- Aparicio, Y.; Gabarra, R.; Arno, J. Attraction of Aphidius ervi (Hymenoptera: Braconidae) and Aphidoletes aphidimyza (Diptera: Cecidomyiidae) to Sweet Alyssum and Assessment of Plant Resources Effects on their Fitness. J. Econ. Entomol. 2018, 111, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Rohrig, E.; Sivinski, J.; Teal, P.; Stuhl, C.; Aluja, M. A floral-derived compound attractive to the tephritid fruit fly parasitoid Diachasmimorpha longicaudata (Hymenoptera: Braconidae). J. Chem. Ecol. 2008, 34, 549–557. [Google Scholar] [CrossRef]
- Rohrig, E.; Sivinski, J.; Wharton, R. Comparison of parasitic Hymenoptera captured in Malaise traps baited with two flowering plants, Lobularia maritima (Brassicales: Brassicaceae) and Spermacoce verticillata (Gentianales: Rubiaceae). Fla. Entomol. 2008, 91, 621–627. [Google Scholar]
- Woltz, J.M.; Lee, J.C. Pupation behavior and larval and pupal biocontrol of Drosophila suzukii in the field. Biol. Control 2017, 110, 62–69. [Google Scholar] [CrossRef] [Green Version]
- Tochen, S.; Walton, V.M.; Lee, J.C. Impact of floral feeding on adult Drosophila suzukii survival and nutrient status. J. Pest Sci. 2016, 89, 793–802. [Google Scholar] [CrossRef]
- Nicolson, S.W. Pollen feeding in the Eucalypt Nectar Fly, Drosophila flavohirta. Physiol. Entomol. 1994, 19, 58–60. [Google Scholar] [CrossRef]
- Sivinski, J.; Aluja, M.; Holler, T. Food sources for adult Diachasmimorpha longicaudata, a parasitoid of tephritid fruit flies: Effects on longevity and fecundity. Entomol. Exp. Appl. 2006, 118, 193–202. [Google Scholar] [CrossRef]
- Eijs, I.E.M.; Ellers, J.; van Duinen, G.J. Feeding strategies in drosophilid parasitoids: The impact of natural food resources on energy reserves in females. Ecol. Entomol. 1998, 23, 133–138. [Google Scholar] [CrossRef]


| Demographic parameters | NC | PC | FE | LBT | LTT |
|---|---|---|---|---|---|
| Net reproduction rate R0 | 18.0 | 29.3 | 42.8 | 37.5 | 32.3 |
| Mean generation time T | 24.5 | 20.1 | 30.5 | 28.9 | 28.6 |
| Doubling time DT | 5.8 | 5.8 | 5.2 | 5.2 | 5.3 |
| Intrinsic rate of increase r | 0.119 | 0.119 | 0.134 | 0.133 | 0.129 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herz, A.; Dingeldey, E.; Englert, C. More Power with Flower for the Pupal Parasitoid Trichopria drosophilae: A Candidate for Biological Control of the Spotted Wing Drosophila. Insects 2021, 12, 628. https://doi.org/10.3390/insects12070628
Herz A, Dingeldey E, Englert C. More Power with Flower for the Pupal Parasitoid Trichopria drosophilae: A Candidate for Biological Control of the Spotted Wing Drosophila. Insects. 2021; 12(7):628. https://doi.org/10.3390/insects12070628
Chicago/Turabian StyleHerz, Annette, Eva Dingeldey, and Camilla Englert. 2021. "More Power with Flower for the Pupal Parasitoid Trichopria drosophilae: A Candidate for Biological Control of the Spotted Wing Drosophila" Insects 12, no. 7: 628. https://doi.org/10.3390/insects12070628
APA StyleHerz, A., Dingeldey, E., & Englert, C. (2021). More Power with Flower for the Pupal Parasitoid Trichopria drosophilae: A Candidate for Biological Control of the Spotted Wing Drosophila. Insects, 12(7), 628. https://doi.org/10.3390/insects12070628







