Genome-Wide Identification of the Gustatory Receptor Gene Family of the Invasive Pest, Red Palm Weevil, Rhynchophorus ferrugineus (Olivier, 1790)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. R. ferrugineus Genetic Data
2.2. Gene Identification
2.3. Phylogenetic Analysis
3. Results
3.1. Antennal and Mouthpart Transcriptomes
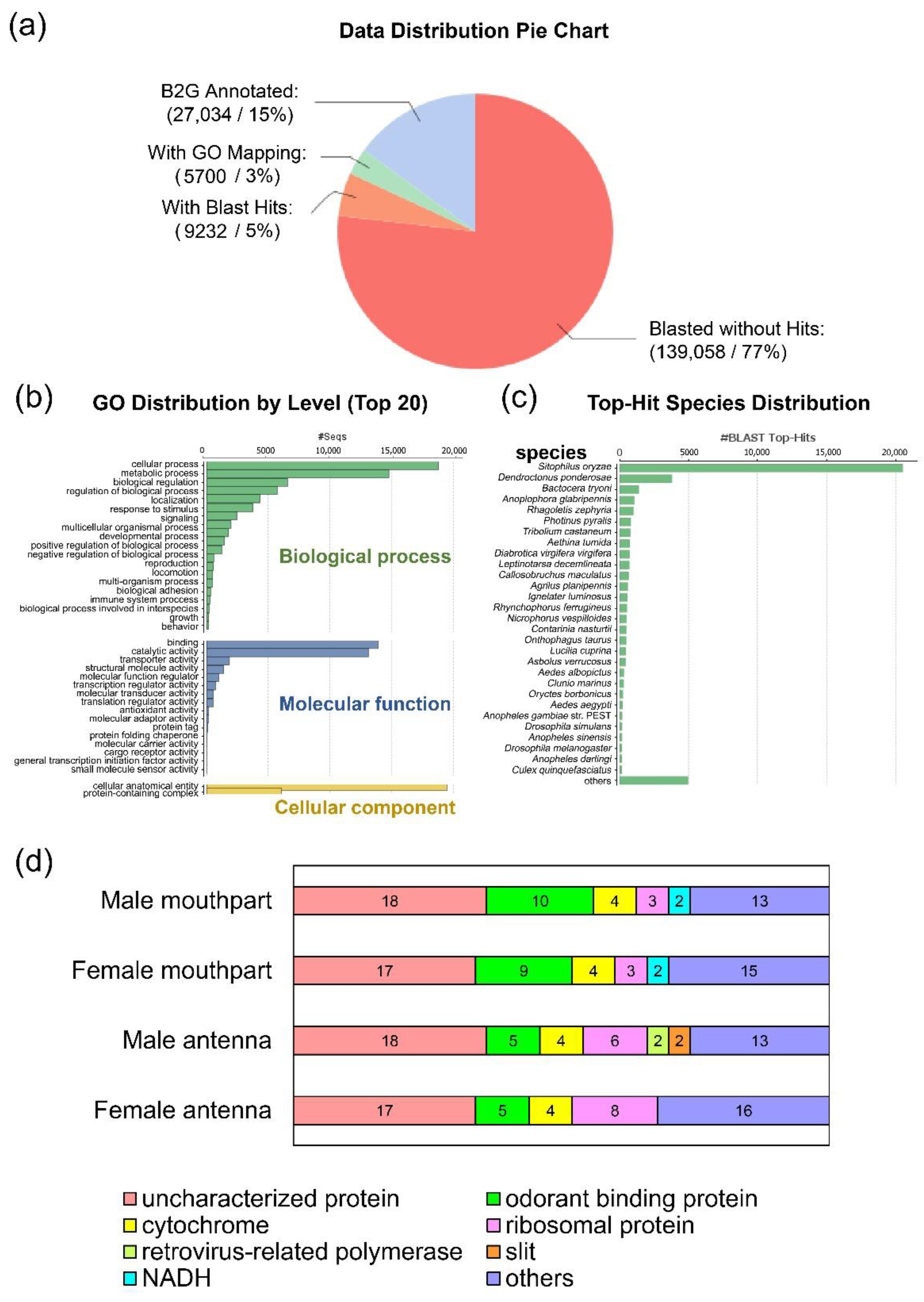
3.1.1. Transcriptome Data
3.1.2. Gene Annotation and Expression Level
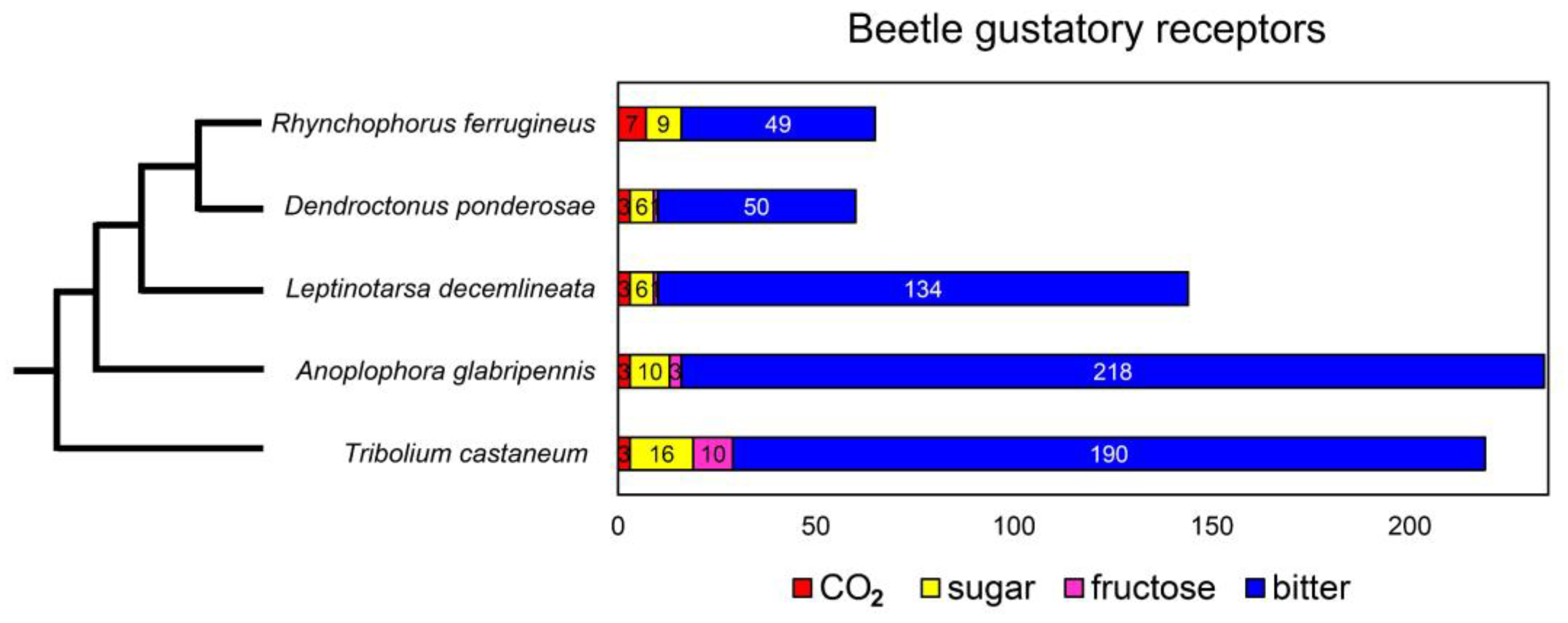
3.2. R. ferrugineus Gustatory Receptor Gene Family and Evolution
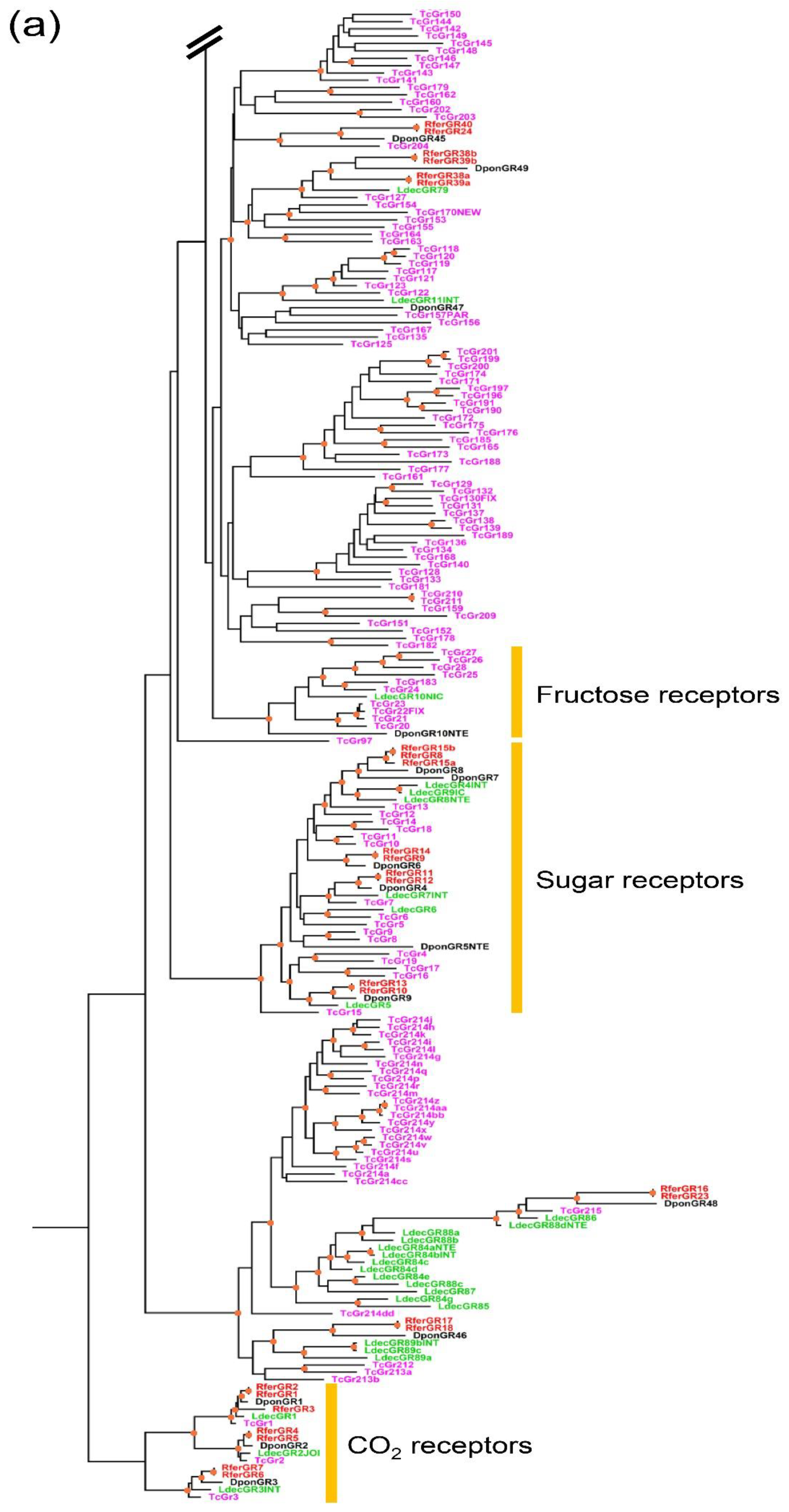
3.2.1. Gustatory Receptor Gene Family
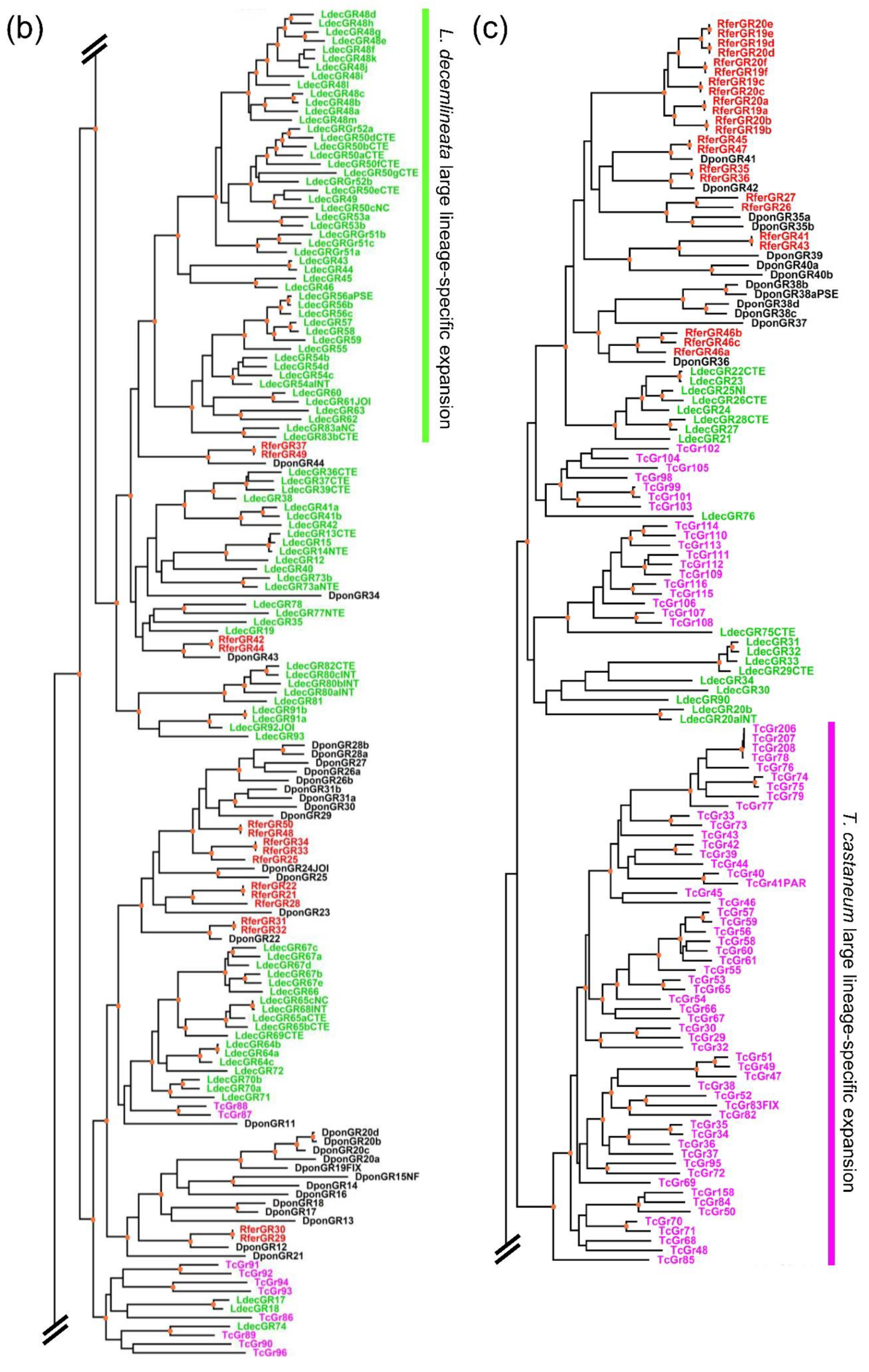
3.2.2. Phylogenetic Analysis
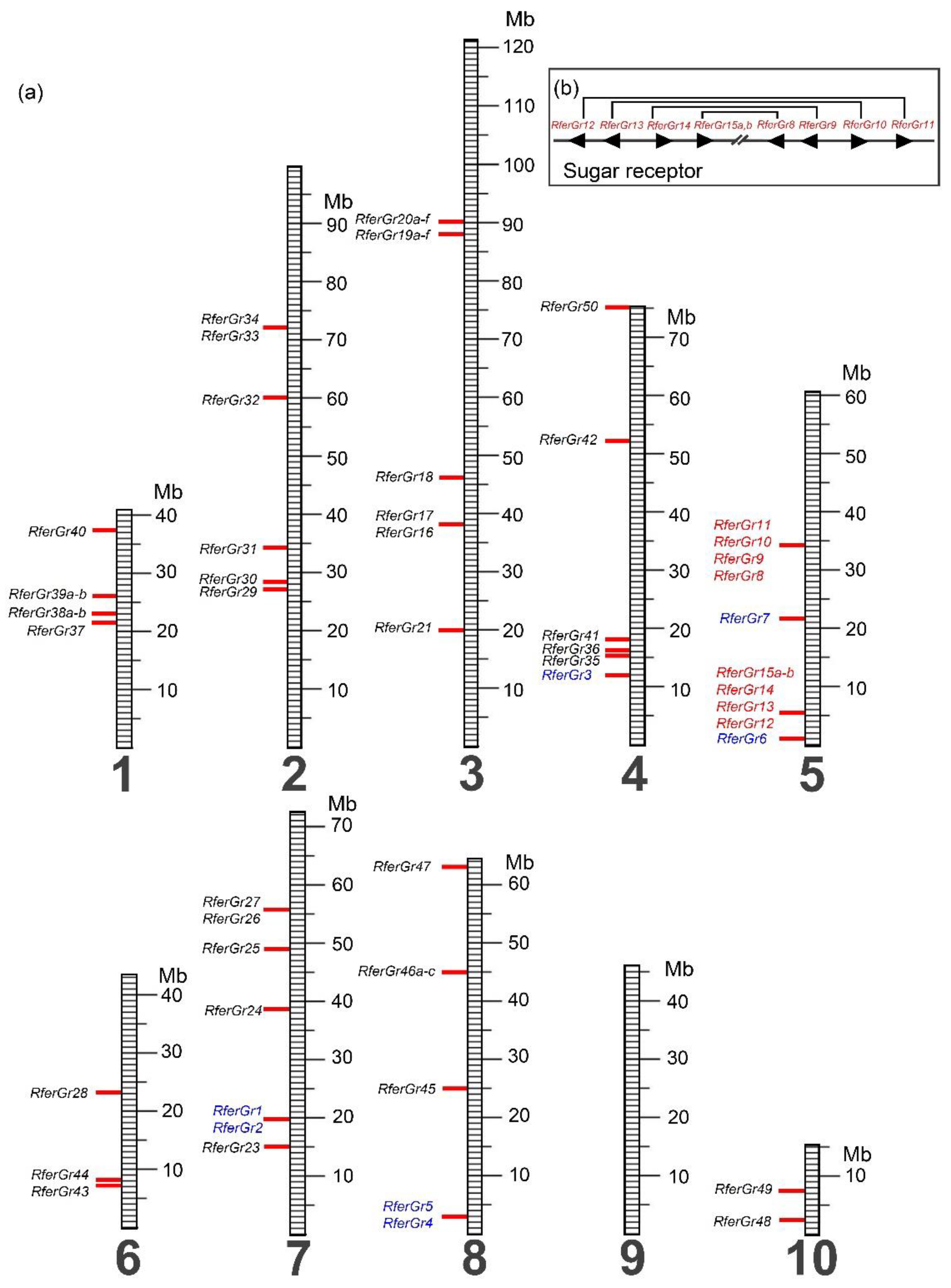
3.2.3. Chromosomal Location
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El-Shafie, H.A.F.; Faleiro, J.R. Red Palm Weevil Rhynchophorus ferrugineus (Coleoptera: Curculionidae): Global Invasion, Current Management Options, Challenges and Future Prospects; IntechOpen: London, UK, 2020; ISBN 978-1-78985-850-1. [Google Scholar]
- FAO. Red Palm Weevil: Guidelines on Management Practices; FAO: Rome, Italy, 2020; ISBN 978-92-5-132189-8. [Google Scholar]
- Ferry, M.; Gómez, S. The Red Palm Weevil in the Mediterranean Area. Palms 2002, 46, 172–178. [Google Scholar]
- FAO. Framework Strategy for Eradication of Red Palm Weevil. Scientific Consultation and High-Level Meeting on Red Palm Weevil Management; FAO: Rome, Italy, 2017; p. 32. Available online: http://www.fao.org/3/a-bu018e.pdf (accessed on 27 May 2021).
- Hallett, R.H.; Gries, G.; Gries, R.; Borden, J.; Czyzewska, E.; Oehlschlager, C.; Pierce, H.D.; Angerilli, N.; Rauf, A. Aggregation Pheromones of Two Asian Palm Weevils, Rhynchophorus ferrugineus and R. vulneratus. Naturwissenschaften 1993, 80, 328–331. [Google Scholar] [CrossRef]
- Hallett, R.H.; Oehlschlager, A.C.; Borden, J.H. Pheromone Trapping Protocols for the Asian Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Curculionidae). Int. J. Pest Manag. 1999, 45, 231–237. [Google Scholar] [CrossRef]
- Oehlschlager, A.C.; Prior, R.N.; Perez, A.L.; Gries, R.; Gries, G.; Pierce, H.D.; Laup, S. Structure, Chirality, and Field Testing of a Male-Produced Aggregation Pheromone of Asian Palm Weevil Rhynchophorus bilineatus (Montr.) (Coleoptera: Curculionidae). J. Chem. Ecol. 1995, 21, 1619–1629. [Google Scholar] [CrossRef]
- Oehlschlager, A.C. Palm Weevil Pheromones–Discovery and Use. J. Chem. Ecol. 2016, 42, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Al-Dawsary, M.M.S. Functional Compounds from the Integument of Adult Red Palm Weevil Rhynchophorus Ferrugineus. Saudi J. Biol. Sci. 2014, 21, 275–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ando, T.; Inomata, S.-I.; Yamamoto, M. Lepidopteran Sex Pheromones. Top. Curr. Chem. 2004, 239, 51–96. [Google Scholar] [CrossRef] [PubMed]
- El-Shafie, H.A.F.; Faleiro, J.R. Optimizing Components of Pheromone-Baited Trap for the Management of Red Palm Weevil, Rhynchophorus Ferrugineus (Coleoptera: Curculionidae) in Date Palm Agro-Ecosystem. J. Plant Dis. Prot. 2017, 124, 279–287. [Google Scholar] [CrossRef]
- Benton, R.; Sachse, S.; Michnick, S.W.; Vosshall, L.B. Atypical Membrane Topology and Heteromeric Function of Drosophila Odorant Receptors In Vivo. PLoS Biol. 2006, 4, e20. [Google Scholar] [CrossRef] [Green Version]
- Lundin, C.; Käll, L.; Kreher, S.A.; Kapp, K.; Sonnhammer, E.L.; Carlson, J.R.; von Heijne, G.; Nilsson, I. Membrane Topology of the Drosophila OR83b Odorant Receptor. FEBS Lett. 2007, 581, 5601–5604. [Google Scholar] [CrossRef] [Green Version]
- Nei, M.; Niimura, Y.; Nozawa, M. The Evolution of Animal Chemosensory Receptor Gene Repertoires: Roles of Chance and Necessity. Nat. Rev. Genet. 2008, 9, 951–963. [Google Scholar] [CrossRef]
- Hallem, E.A.; Carlson, J.R. Coding of Odors by a Receptor Repertoire. Cell 2006, 125, 143–160. [Google Scholar] [CrossRef] [Green Version]
- Carey, A.F.; Wang, G.; Su, C.-Y.; Zwiebel, L.J.; Carlson, J.R. Odorant Reception in the Malaria Mosquito Anopheles gambiae. Nature 2010, 464, 66–71. [Google Scholar] [CrossRef] [Green Version]
- Missbach, C.; Dweck, H.K.; Vogel, H.; Vilcinskas, A.; Stensmyr, M.C.; Hansson, B.S.; Grosse-Wilde, E. Evolution of Insect Olfactory Receptors. Elife 2014, 3, e02115. [Google Scholar] [CrossRef] [PubMed]
- Larsson, M.C.; Domingos, A.I.; Jones, W.D.; Chiappe, M.E.; Amrein, H.; Vosshall, L.B. Or83b Encodes a Broadly Expressed Odorant Receptor Essential for Drosophila Olfaction. Neuron 2004, 43, 703–714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wicher, D.; Schäfer, R.; Bauernfeind, R.; Stensmyr, M.C.; Heller, R.; Heinemann, S.H.; Hansson, B.S. Drosophila Odorant Receptors Are Both Ligand-Gated and Cyclic-Nucleotide-Activated Cation Channels. Nature 2008, 452, 1007–1011. [Google Scholar] [CrossRef]
- Jones, P.L.; Pask, G.M.; Rinker, D.C.; Zwiebel, L.J. Functional Agonism of Insect Odorant Receptor Ion Channels. Proc. Natl. Acad. Sci. USA 2011, 108, 8821–8825. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, T.; Pellegrino, M.; Sato, K.; Vosshall, L.B.; Touhara, K. Amino Acid Residues Contributing to Function of the Heteromeric Insect Olfactory Receptor Complex. PLoS ONE 2012, 7, e32372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hopf, T.A.; Morinaga, S.; Ihara, S.; Touhara, K.; Marks, D.S.; Benton, R. Amino Acid Coevolution Reveals Three-Dimensional Structure and Functional Domains of Insect Odorant Receptors. Nat. Commun. 2015, 6, 6077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engsontia, P.; Sanderson, A.P.; Cobb, M.; Walden, K.K.O.; Robertson, H.M.; Brown, S. The Red Flour Beetle’s Large Nose: An Expanded Odorant Receptor Gene Family in Tribolium castaneum. Insect Biochem. Mol. Biol. 2008, 38, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Schoville, S.D.; Chen, Y.H.; Andersson, M.N.; Benoit, J.B.; Bhandari, A.; Bowsher, J.H.; Brevik, K.; Cappelle, K.; Chen, M.-J.M.; Childers, A.K.; et al. A Model Species for Agricultural Pest Genomics: The Genome of the Colorado Potato Beetle, Leptinotarsa decemlineata (Coleoptera: Chrysomelidae). Sci. Rep. 2018, 8, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andersson, M.N.; Keeling, C.I.; Mitchell, R.F. Genomic Content of Chemosensory Genes Correlates with Host Range in Wood-Boring Beetles (Dendroctonus ponderosae, Agrilus planipennis, and Anoplophora glabripennis). BMC Genom. 2019, 20, 690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKenna, D.D.; Scully, E.D.; Pauchet, Y.; Hoover, K.; Kirsch, R.; Geib, S.M.; Mitchell, R.F.; Waterhouse, R.M.; Ahn, S.-J.; Arsala, D.; et al. Genome of the Asian Longhorned Beetle (Anoplophora glabripennis), a Globally Significant Invasive Species, Reveals Key Functional and Evolutionary Innovations at the Beetle–Plant Interface. Genome Biol. 2016, 17, 227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, W.; Papanicolaou, A.; Zhang, H.-J.; Anderson, A. Expansion of a Bitter Taste Receptor Family in a Polyphagous Insect Herbivore. Sci. Rep. 2016, 6, 23666. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.-J.; Anderson, A.R.; Trowell, S.C.; Luo, A.-R.; Xiang, Z.-H.; Xia, Q.-Y. Topological and Functional Characterization of an Insect Gustatory Receptor. PLoS ONE 2011, 6, e24111. [Google Scholar] [CrossRef] [Green Version]
- Isono, K.; Morita, H. Molecular and Cellular Designs of Insect Taste Receptor System. Front. Cell. Neurosci. 2010, 4, 20. [Google Scholar] [CrossRef] [Green Version]
- Robertson, H.M. The Insect Chemoreceptor Superfamily Is Ancient in Animals. Chem. Senses 2015, 40, 609–614. [Google Scholar] [CrossRef]
- Sato, K.; Tanaka, K.; Touhara, K. Sugar-Regulated Cation Channel Formed by an Insect Gustatory Receptor. Proc. Natl. Acad. Sci. USA 2011, 108, 11680–11685. [Google Scholar] [CrossRef] [Green Version]
- Miyamoto, T.; Slone, J.; Song, X.; Amrein, H. A Fructose Receptor Functions as a Nutrient Sensor in the Drosophila Brain. Cell 2012, 151, 1113–1125. [Google Scholar] [CrossRef] [Green Version]
- Kain, P.; Badsha, F.; Hussain, S.M.; Nair, A.; Hasan, G.; Rodrigues, V. Mutants in Phospholipid Signaling Attenuate the Behavioral Response of Adult Drosophila to Trehalose. Chem. Senses 2010, 35, 663–673. [Google Scholar] [CrossRef] [Green Version]
- Bredendiek, N.; Hütte, J.; Steingräber, A.; Hatt, H.; Gisselmann, G.; Neuhaus, E.M. Go α Is Involved in Sugar Perception in Drosophila. Chem. Senses 2011, 36, 69–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Gracia, A.; Vieira, F.G.; Almeida, F.C.; Rozas, J. Comparative Genomics of the Major Chemosensory Gene Families in Arthropods. In eLS; American Cancer Society: Atlanta, GA, USA, 2011; ISBN 978-0-470-01590-2. [Google Scholar]
- Moon, S.J.; Lee, Y.; Jiao, Y.; Montell, C. A Drosophila Gustatory Receptor Essential for Aversive Taste and Inhibiting Male-to-Male Courtship. Curr. Biol. 2009, 19, 1623–1627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, Y.; Moon, S.J.; Montell, C. Multiple Gustatory Receptors Required for the Caffeine Response in Drosophila. Proc. Natl. Acad. Sci. USA 2009, 106, 4495–4500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shim, J.; Lee, Y.; Jeong, Y.T.; Kim, Y.; Lee, M.G.; Montell, C.; Moon, S.J. The Full Repertoire of Drosophila Gustatory Receptors for Detecting an Aversive Compound. Nat. Commun. 2015, 6, 8867. [Google Scholar] [CrossRef] [Green Version]
- Jiao, Y.; Moon, S.J.; Montell, C. A Drosophila Gustatory Receptor Required for the Responses to Sucrose, Glucose, and Maltose Identified by mRNA Tagging. Proc. Natl. Acad. Sci. USA 2007, 104, 14110–14115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiao, Y.; Moon, S.J.; Wang, X.; Ren, Q.; Montell, C. Gr64f Is Required in Combination with Other Gustatory Receptors for Sugar Detection in Drosophila. Curr. Biol. 2008, 18, 1797–1801. [Google Scholar] [CrossRef] [Green Version]
- Dahanukar, A.; Lei, Y.-T.; Kwon, J.Y.; Carlson, J.R. Two Gr Genes Underlie Sugar Reception in Drosophila. Neuron 2007, 56, 503–516. [Google Scholar] [CrossRef] [Green Version]
- Miyamoto, T.; Amrein, H. Suppression of Male Courtship by a Drosophila Pheromone Receptor. Nat. Neurosci. 2008, 11, 874–876. [Google Scholar] [CrossRef] [Green Version]
- Wisotsky, Z.; Medina, A.; Freeman, E.; Dahanukar, A. Evolutionary Differences in Food Preference Rely on Gr64e, a Receptor for Glycerol. Nat. Neurosci. 2011, 14, 1534–1541. [Google Scholar] [CrossRef]
- Jones, W.D.; Cayirlioglu, P.; Kadow, I.G.; Vosshall, L.B. Two Chemosensory Receptors Together Mediate Carbon Dioxide Detection in Drosophila. Nature 2007, 445, 86–90. [Google Scholar] [CrossRef]
- Kwon, J.Y.; Dahanukar, A.; Weiss, L.A.; Carlson, J.R. The Molecular Basis of CO2 Reception in Drosophila. Proc. Natl. Acad. Sci. USA 2007, 104, 3574–3578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liman, E.R.; Zhang, Y.V.; Montell, C. Peripheral Coding of Taste. Neuron 2014, 81, 984–1000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bray, S.; Amrein, H. A Putative Drosophila Pheromone Receptor Expressed in Male-Specific Taste Neurons Is Required for Efficient Courtship. Neuron 2003, 39, 1019–1029. [Google Scholar] [CrossRef] [Green Version]
- Koganezawa, M.; Haba, D.; Matsuo, T.; Yamamoto, D. The Shaping of Male Courtship Posture by Lateralized Gustatory Inputs to Male-Specific Interneurons. Curr. Biol. 2010, 20, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erdelyan, C.N.G.; Mahood, T.H.; Bader, T.S.Y.; Whyard, S. Functional Validation of the Carbon Dioxide Receptor Genes in Aedes aegypti Mosquitoes Using RNA Interference. Insect Mol. Biol. 2012, 21, 119–127. [Google Scholar] [CrossRef]
- Tribolium Genome Sequencing Consortium; Richards, S.; Gibbs, R.; Weinstock, G.; Brown, S.J.; Denell, R.; Beeman, R.; Gibbs, R.; Bucher, G.; Friedrich, M.; et al. The Genome of the Model Beetle and Pest Tribolium castaneum. Nature 2008, 452, 949. [Google Scholar]
- Leal, W.S. Odorant Reception in Insects: Roles of Receptors, Binding Proteins, and Degrading Enzymes. Annu. Rev. Entomol. 2013, 58, 373–391. [Google Scholar] [CrossRef]
- Pelosi, P.; Iovinella, I.; Felicioli, A.; Dani, F.R. Soluble Proteins of Chemical Communication: An Overview across Arthropods. Front. Physiol. 2014, 5, 320. [Google Scholar] [CrossRef] [Green Version]
- Vieira, F.G.; Rozas, J. Comparative Genomics of the Odorant-Binding and Chemosensory Protein Gene Families across the Arthropoda: Origin and Evolutionary History of the Chemosensory System. Genome Biol. Evol. 2011, 3, 476–490. [Google Scholar] [CrossRef]
- Kim, M.-S.; Repp, A.; Smith, D.P. LUSH Odorant-Binding Protein Mediates Chemosensory Responses to Alcohols in Drosophila Melanogaster. Genetics 1998, 150, 711–721. [Google Scholar] [CrossRef]
- Xu, P.; Atkinson, R.; Jones, D.N.M.; Smith, D.P. Drosophila OBP LUSH Is Required for Activity of Pheromone-Sensitive Neurons. Neuron 2005, 45, 193–200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graham, L.A.; Brewer, D.; Lajoie, G.; Davies, P.L. Characterization of a Subfamily of Beetle Odorant-Binding Proteins Found in Hemolymph*S. Mol. Cell. Proteom. 2003, 2, 541–549. [Google Scholar] [CrossRef] [Green Version]
- Pelosi, P.; Zhou, J.-J.; Ban, L.P.; Calvello, M. Soluble Proteins in Insect Chemical Communication. Cell. Mol. Life Sci. 2006, 63, 1658–1676. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Liu, L.; Qin, W.Q.; Li, C.X.; Peng, Z.Q. Transcriptomic Identification of Chemoreceptor Genes in the Red Palm Weevil Rhynchophorus ferrugineus. Genet. Mol. Res. 2015, 14, 7469–7480. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Liu, L.; Qin, W.; Luo, Y.; Ma, X.; Haider, N.; Inayeh, M. Identification and Tissue Expression Profiling of Odorant Binding Protein Genes in the Red Palm Weevil, Rhynchophorus ferrugineus. Springerplus 2016, 5, 1542. [Google Scholar] [CrossRef] [Green Version]
- Antony, B.; Soffan, A.; Jakše, J.; Abdelazim, M.M.; Aldosari, S.A.; Aldawood, A.S.; Pain, A. Identification of the Genes Involved in Odorant Reception and Detection in the Palm Weevil Rhynchophorus ferrugineus, an Important Quarantine Pest, by Antennal Transcriptome Analysis. BMC Genom. 2016, 17, 69. [Google Scholar] [CrossRef] [Green Version]
- Soffan, A.; Antony, B.; Abdelazim, M.; Shukla, P.; Witjaksono, W.; Aldosari, S.A.; Aldawood, A.S. Silencing the Olfactory Co-Receptor RferOrco Reduces the Response to Pheromones in the Red Palm Weevil, Rhynchophorus ferrugineus. PLoS ONE 2016, 11, e0162203. [Google Scholar] [CrossRef] [PubMed]
- Antony, B.; Johny, J.; Aldosari, S.A. Silencing the Odorant Binding Protein RferOBP1768 Reduces the Strong Preference of Palm Weevil for the Major Aggregation Pheromone Compound Ferrugineol. Front. Physiol. 2018, 9, 252. [Google Scholar] [CrossRef]
- Antony, B.; Johny, J.; Montagné, N.; Jacquin-Joly, E.; Capoduro, R.; Cali, K.; Persaud, K.; Al-Saleh, M.A.; Pain, A. Pheromone Receptor of the Globally Invasive Quarantine Pest of the Palm Tree, the Red Palm Weevil (Rhynchophorus ferrugineus). Mol. Ecol. 2021, 30, 2025–2039. [Google Scholar] [CrossRef]
- Hazzouri, K.M.; Sudalaimuthuasari, N.; Kundu, B.; Nelson, D.; Al-Deeb, M.A.; Le Mansour, A.; Spencer, J.J.; Desplan, C.; Amiri, K.M.A. The Genome of Pest Rhynchophorus ferrugineus Reveals Gene Families Important at the Plant-Beetle Interface. Commun. Biol. 2020, 3, 1–14. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [Green Version]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; Couger, M.B.; Eccles, D.; Li, B.; Lieber, M.; et al. De Novo Transcript Sequence Reconstruction from RNA-Seq Using the Trinity Platform for Reference Generation and Analysis. Nat. Protoc. 2013, 8, 1494–1512. [Google Scholar] [CrossRef] [PubMed]
- Simão, F.A.; Waterhouse, R.M.; Ioannidis, P.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO: Assessing Genome Assembly and Annotation Completeness with Single-Copy Orthologs. Bioinformatics 2015, 31, 3210–3212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, B.; Dewey, C.N. RSEM: Accurate Transcript Quantification from RNA-Seq Data with or without a Reference Genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef] [Green Version]
- Engsontia, P.; Sangket, U.; Chotigeat, W.; Satasook, C. Molecular Evolution of the Odorant and Gustatory Receptor Genes in Lepidopteran Insects: Implications for Their Adaptation and Speciation. J. Mol. Evol. 2014, 79, 21–39. [Google Scholar] [CrossRef]
- Birney, E.; Clamp, M.; Durbin, R. GeneWise and Genomewise. Genome Res. 2004, 14, 988–995. [Google Scholar] [CrossRef] [Green Version]
- Bernsel, A.; Viklund, H.; Hennerdal, A.; Elofsson, A. TOPCONS: Consensus Prediction of Membrane Protein Topology. Nucleic Acids Res. 2009, 37, W465–W468. [Google Scholar] [CrossRef] [Green Version]
- Katoh, K.; Misawa, K.; Kuma, K.; Miyata, T. MAFFT: A Novel Method for Rapid Multiple Sequence Alignment Based on Fast Fourier Transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef] [Green Version]
- Capella-Gutiérrez, S.; Silla-Martínez, J.M.; Gabaldón, T. TrimAl: A Tool for Automated Alignment Trimming in Large-Scale Phylogenetic Analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [Green Version]
- Ozaki, M. Odorant-Binding Proteins in Taste System: Putative Roles in Taste Sensation and Behavior. In Olfactory Concepts of Insect Control-Alternative to insecticides; Picimbon, J.-F., Ed.; Springer International Publishing: Cham, Switzerland, 2019; Volume 2, pp. 187–204. ISBN 978-3-030-05165-5. [Google Scholar]
- Robertson, H.M.; Warr, C.G.; Carlson, J.R. Molecular Evolution of the Insect Chemoreceptor Gene Superfamily in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2003, 100, 14537–14542. [Google Scholar] [CrossRef] [Green Version]
- Wanner, K.W.; Robertson, H.M. The Gustatory Receptor Family in the Silkworm Moth Bombyx mori Is Characterized by a Large Expansion of a Single Lineage of Putative Bitter Receptors. Insect Mol. Biol. 2008, 17, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, F.; Johny, J.; Walker, W.B.; Guan, Q.; Mfarrej, S.; Jakše, J.; Montagné, N.; Jacquin-Joly, E.; Alqarni, A.A.; Al-Saleh, M.A.; et al. Antennal Transcriptome Sequencing and Identification of Candidate Chemoreceptor Proteins from an Invasive Pest, the American Palm Weevil, Rhynchophorus palmarum. Sci. Rep. 2021, 11, 8334. [Google Scholar] [CrossRef] [PubMed]
- Benton, R. Multigene Family Evolution: Perspectives from Insect Chemoreceptors. Trends Ecol. Evol. 2015, 30, 590–600. [Google Scholar] [CrossRef]
- McBride, C.S.; Arguello, J.R.; O’Meara, B.C. Five Drosophila Genomes Reveal Nonneutral Evolution and the Signature of Host Specialization in the Chemoreceptor Superfamily. Genetics 2007, 177, 1395–1416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engsontia, P.; Sangket, U.; Robertson, H.M.; Satasook, C. Diversification of the Ant Odorant Receptor Gene Family and Positive Selection on Candidate Cuticular Hydrocarbon Receptors. BMC Res. Notes 2015, 8, 380. [Google Scholar] [CrossRef] [Green Version]
- Dembilio, Ó.; Jacas, J.A. Bio-Ecology and Integrated Management of the Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Curculionidae), in the Region of Valencia (Spain). Hell. Plant Prot. J. 2012, 5, 1–12. [Google Scholar]
- Kojima, W. Attraction to Carbon Dioxide from Feeding Resources and Conspecific Neighbours in Larvae of the Rhinoceros Beetle Trypoxylus dichotomus. PLoS ONE 2015, 10, e0141733. [Google Scholar] [CrossRef]
- Freeman, E.G.; Wisotsky, Z.; Dahanukar, A. Detection of Sweet Tastants by a Conserved Group of Insect Gustatory Receptors. Proc. Natl. Acad. Sci. USA 2014, 111, 1598–1603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, L.A.; Dahanukar, A.; Kwon, J.Y.; Banerjee, D.; Carlson, J.R. The Molecular and Cellular Basis of Bitter Taste in Drosophila. Neuron 2011, 69, 258–272. [Google Scholar] [CrossRef] [Green Version]
- Sang, J.; Rimal, S.; Lee, Y. Gustatory Receptor 28b Is Necessary for Avoiding Saponin in Drosophila melanogaster. EMBO Rep. 2019, 20, e47328. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Gong, X.-L.; Li, G.-C.; Huang, L.-Q.; Ning, C.; Wang, C.-Z. A Gustatory Receptor Tuned to the Steroid Plant Hormone Brassinolide in Plutella xylostella (Lepidoptera: Plutellidae). Elife 2020, 9, e64114. [Google Scholar] [CrossRef] [PubMed]

| Libraries | Details | Numbers |
|---|---|---|
| Male antenna | Total reads | 42,872,182 |
| %GC | 38 | |
| Female antenna | Total reads | 38,301,355 |
| %GC | 36 | |
| Male mouthparts | Total reads | 40,701,754 |
| %GC | 38 | |
| Female mouthparts | Total reads | 45,615,562 |
| %GC | 37 | |
| Pooled assembly | Min contig length | 101 |
| Max contig length | 27,357 | |
| N50 | 953 | |
| Mean contig length | 381.73 | |
| Total bases (bp) | 69,037,346 | |
| Unique contigs (Unigenes) | 181,024 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Engsontia, P.; Satasook, C. Genome-Wide Identification of the Gustatory Receptor Gene Family of the Invasive Pest, Red Palm Weevil, Rhynchophorus ferrugineus (Olivier, 1790). Insects 2021, 12, 611. https://doi.org/10.3390/insects12070611
Engsontia P, Satasook C. Genome-Wide Identification of the Gustatory Receptor Gene Family of the Invasive Pest, Red Palm Weevil, Rhynchophorus ferrugineus (Olivier, 1790). Insects. 2021; 12(7):611. https://doi.org/10.3390/insects12070611
Chicago/Turabian StyleEngsontia, Patamarerk, and Chutamas Satasook. 2021. "Genome-Wide Identification of the Gustatory Receptor Gene Family of the Invasive Pest, Red Palm Weevil, Rhynchophorus ferrugineus (Olivier, 1790)" Insects 12, no. 7: 611. https://doi.org/10.3390/insects12070611
APA StyleEngsontia, P., & Satasook, C. (2021). Genome-Wide Identification of the Gustatory Receptor Gene Family of the Invasive Pest, Red Palm Weevil, Rhynchophorus ferrugineus (Olivier, 1790). Insects, 12(7), 611. https://doi.org/10.3390/insects12070611








