Effect of Duration of Exposure to Males on Female Reproductive Performance of the Green Lacewing, Chrysoperla agilis (Neuroptera: Chrysopidae)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Chrysoperla agilis Laboratory Rearing
2.2. Experimental Setup
2.3. Developmental Time and Survival during Juvenile Development
2.4. Female Longevity and Reproduction
2.5. Intrinsic Rate of Population Increase
2.6. Egg Hatchability and Sex Ratio
2.7. Statistical Analysis
3. Results
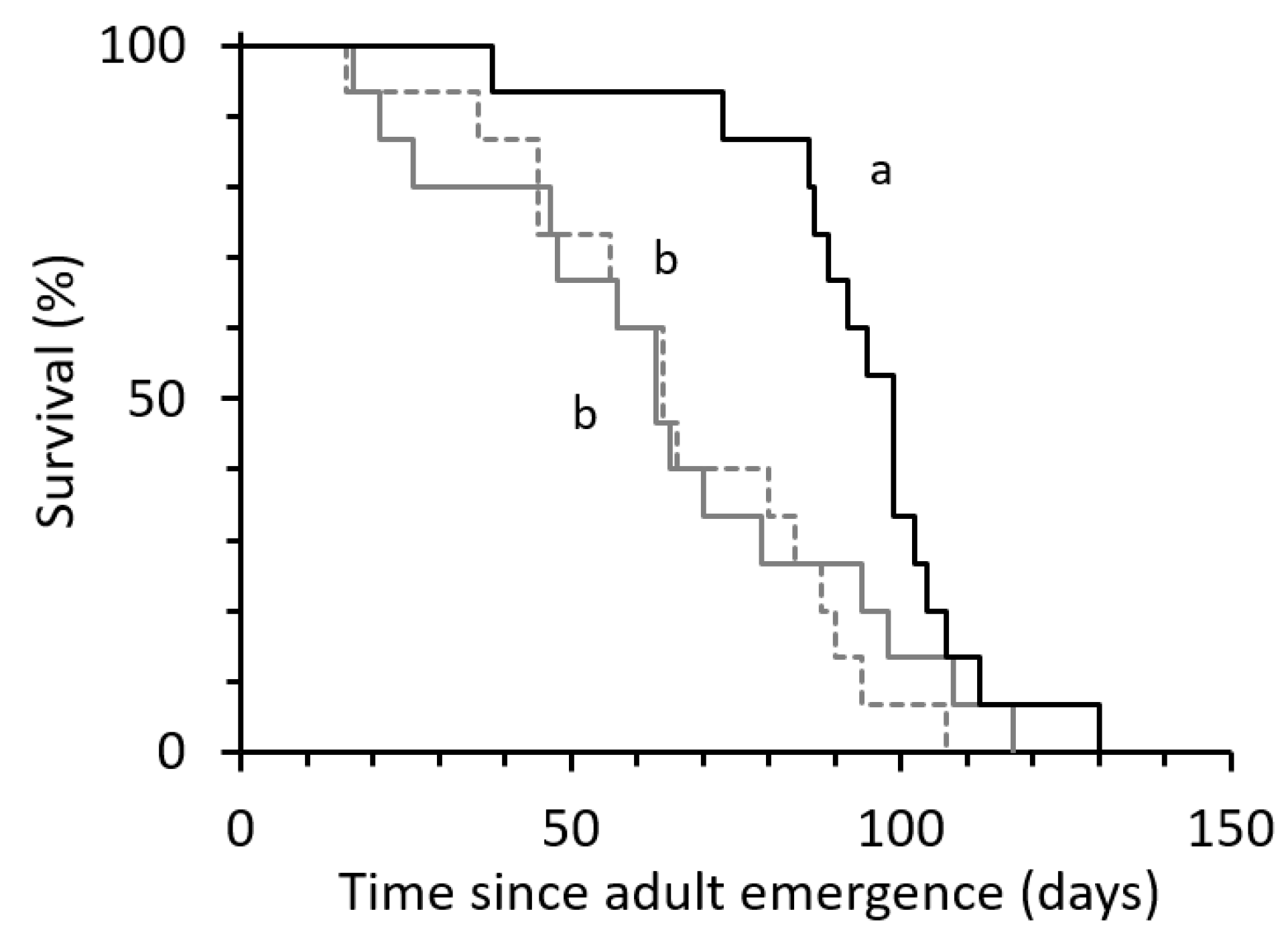
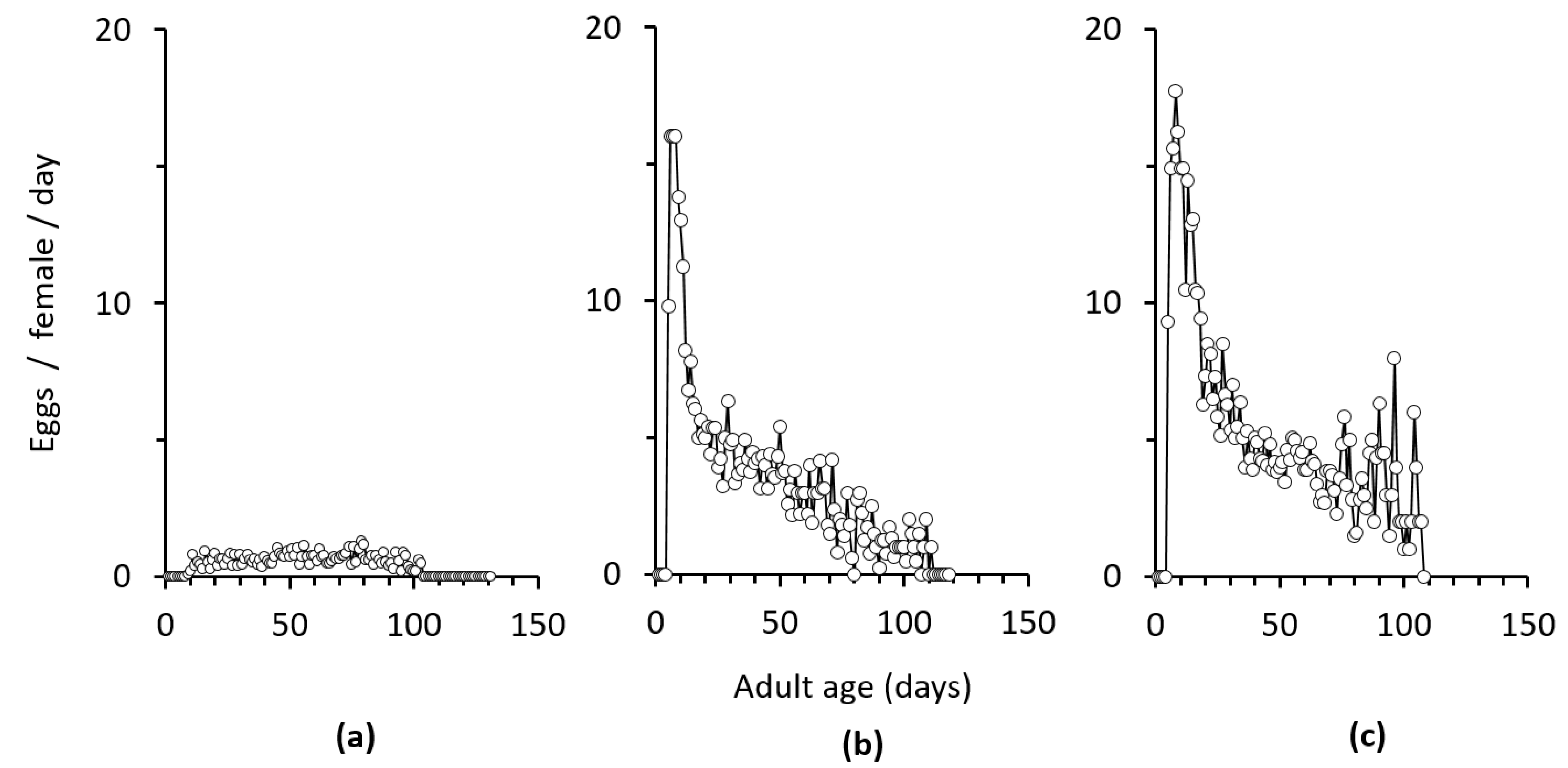
3.1. Effects of Duration of Exposure to Males on Egg Production and Female Longevity
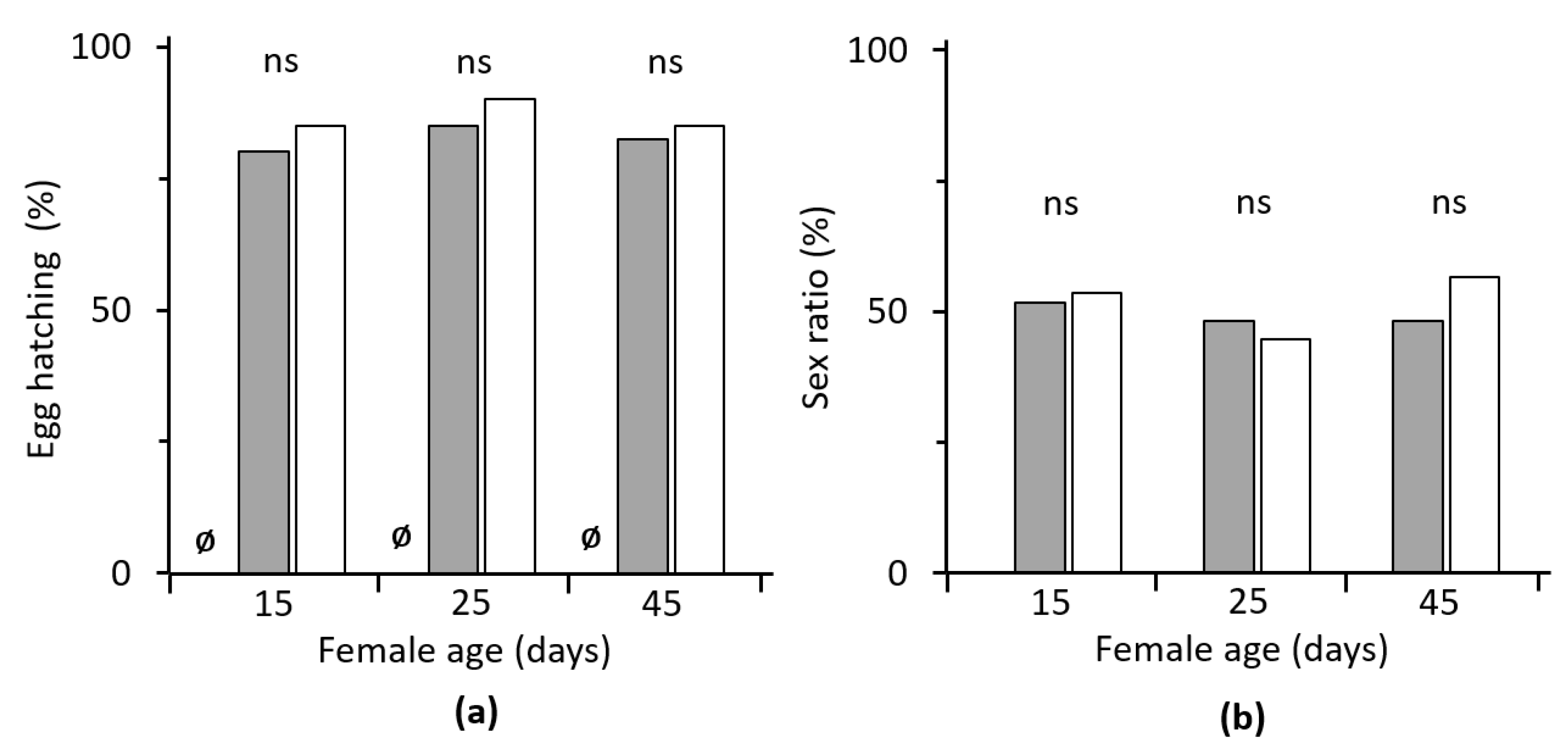
3.2. Egg Hatchability
3.3. Progeny Sex Ratio
3.4. Intrinsic Rate of Population Increase
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Senior, L.J.; McEwen, P.K. The use of lacewings in biological control. In Lacewings in the Crop Environment; Cambridge University Press: Cambridge, UK, 2001; pp. 296–302. [Google Scholar]
- Pappas, M.L.; Broufas, G.D.; Koveos, D.S. Chrysopid predators and their role in biological control. J. Entomol. 2011, 8, 301–326. [Google Scholar] [CrossRef]
- Henry, C.S.; Brooks, S.J.; Duelli, P.; Johnson, J.B.; Wells, M.M.; Mochizuki, A. Parallel evolution in courtship songs of North American and European green lacewings (Neuroptera: Chrysopidae). Biol. J. Linn. Soc. 2012, 105, 776–796. [Google Scholar] [CrossRef]
- Henry, C.S.; Brooks, S.J.; Johnson, J.R.; Wells, M.M.; Duelli, P. Song analysis reveals a permanent population of the mediterranean lacewing Chrysoperla agilis (Neuroptera: Chrysopidae) living in central Alaska. Ann. Entomol. Soc. Am. 2011, 104, 649–657. [Google Scholar] [CrossRef]
- Henry, C.S.; Brooks, S.J.; Thierry, D.; Duelli, P.; Johnson, J.B. The common green lacewing (Chrysoperla carnea s. lat.) and the sibling species problem. In Lacewings in the Crop Environment; Cambridge University Press: Cambridge, UK, 2001; pp. 29–42. [Google Scholar]
- Brooks, S.J. A taxonomic review of the common green lacewing genus Chrysoperla (Neuroptera: Chrysopidae). Bull. Br. Mus. Nat. Hist. 1994, 63, 137–210. [Google Scholar]
- Noh, S.; Henry, C.S. Sexually monomorphic mating preferences contribute to premating isolation based on song in European green lacewings. Evolution 2010, 64, 261–270. [Google Scholar] [CrossRef]
- Henry, C.S.; Brooks, S.J.; Duelli, P.; Johnson, J.B. A lacewing with the wanderlust: The European song species ‘Maltese’, Chrysoperla agilis, sp.n., of the carnea group of Chrysoperla (Neuroptera: Chrysopidae). Syst. Entomol. 2003, 28, 131–148. [Google Scholar] [CrossRef]
- Pappas, M.L.; Karagiorgou, E.; Papaioannou, G.; Koveos, D.S.; Broufas, G.D. Developmental temperature responses of Chrysoperla agilis (Neuroptera: Chrysopidae), a member of the European carnea cryptic species group. Biol. Control 2013, 64, 291–298. [Google Scholar] [CrossRef]
- Henry, C.S.; Brooks, S.J.; Johnson, J.B.; Duelli, P. Chrysoperla lucasina (Lacroix): A distinct species of green lacewing, confirmed by acoustical analysis (Neuroptera: Chrysopidae). Syst. Entomol. 1996, 21, 205–218. [Google Scholar] [CrossRef]
- Henry, C.S.; Busher, C. Patterns of Mating and Fecundity in Several Common Green Lacewings (Neuroptera: Chrysopidae) of Eastern North America. Psyche 1987, 94, 219–244. [Google Scholar] [CrossRef][Green Version]
- Henry, C.S. The sexual behavior of green lacewings. In Biology of Chrysopidae; Springer: Berlin/Heidelberg, Germany, 1984; pp. 101–110. [Google Scholar]
- López-Arroyo, J.I.; Tauber, C.A.; Tauber, M.J. Intermittent oviposition and remating in Ceraeochrysa cincta (Neuroptera: Chrysopidae). Ann. Entomol. Soc. Am. 1999, 92, 587–593. [Google Scholar] [CrossRef]
- Pappas, M.L.; Broufas, G.D.; Koveos, D.S. Effects of various prey species on development, survival and reproduction of the predatory lacewing Dichochrysa prasina (Neuroptera: Chrysopidae). Biol. Control 2007, 43, 163–170. [Google Scholar] [CrossRef]
- Canard, M.; Principi, M.M. Development of Chrysopidae. In Biology of Chrysopidae; Springer: Berlin/Heidelberg, Germany, 1984; pp. 57–75. [Google Scholar]
- Barnard, P.C. Adult morphology related to classification. In Biology of Chrysopidae; Springer: Berlin/Heidelberg, Germany, 1984; pp. 19–29. [Google Scholar]
- Wang, J.J.; Tsai, J.H. Development and functional response of Coelophora inaequalis (Cleoptera: Coccinellidae) feeding on brown citrus aphid, Toxoptera citricida (Homoptera: Aphididae). Agric. For. Entomol. 2001, 3, 65–69. [Google Scholar] [CrossRef]
- Birch, L.C. The intrinsic rate of natural increase of an insect population. J. Anim. Ecol. 1948, 17, 15–26. [Google Scholar] [CrossRef]
- SPSS. IBM SPSS Statistics for Windows, Version 19.0; IBM: New York, NY, USA, 2011. [Google Scholar]
- Arnqvist, G.; Nilsson, T. The evolution of polyandry: Multiple mating and female fitness in insects. Anim. Behav. 2000, 60, 145–164. [Google Scholar] [CrossRef]
- Sirot, L.K.; Brockmann, H.J. Costs of sexual interactions to females in rambur’s forktail damselfly, Ischnura ramburi (Zygoptera: Coenagrionidae). Anim. Behav. 2001, 61, 415–424. [Google Scholar] [CrossRef][Green Version]
- Rousset, A. Reproductive physiology and fecundity. In Biology of Chrysopidae; Springer: Berlin/Heidelberg, Germany, 1984; pp. 116–129. [Google Scholar]
- Dantas, P.C.; Santos, H.C.P.; Bozdoğan, H.; Serrão, J.E.; Carvalho, G.A. Morphology of the male and female reproductive tracts of virgin and mated Chrysoperla externa (Hagen, 1861) (Neuroptera: Chrysopidae). Microsc. Res. Tech. 2021, 84, 860–868. [Google Scholar] [CrossRef]
- Philippe, R. Biologie de la reproduction de Chrysopa perla (L.) (Neuroptera, Chrysopidae) en fonction d’alimentation imaginale. Ann. Zool. Ecol. Anim. 1972, 4, 213–227. [Google Scholar]
- Ribeiro, M.J.; Carvalho, C.F. Aspectos biológicos de Chrysoperla externa (Hagen, 1861) (Neuroptera: Chrysopidae) em diferentes condições de acasalamento. Rev. Bras. Entomol. 1991, 35, 423–427. [Google Scholar]
- Sheldon, J.K.; MacLeod, E.G. Studies on the biology of the Chrysopidae. 4. A field and laboratory study of the seasonal cycle of Chrysopa carnea Stephens in Central Illinois (Neuroptera: Chrysopidae). Trans. Am. Entomol. Soc. 1974, 100, 437–512. [Google Scholar]
- Jones, S.L.; Lingren, P.D.; Bee, M.J. Diel periodicity of feeding, mating, and oviposition of adult Chrysopa carnea. Ann. Entomol. Soc. Am. 1977, 70, 43–47. [Google Scholar] [CrossRef]
- Foster, S.P.; Ayers, R.H. Multiple mating and its effects in the lightbrown apple moth, Epiphyas postvittana (Walker). J. Insect Physiol. 1996, 42, 657–667. [Google Scholar] [CrossRef]
- Gwynne, D.T. The evolution of edible ‘sperm sacs’ and other forms of courtship feeding in crickets, katydids and their kin (Orthoptera: Ensifera). In The Evolution of Mating Systems in Insects and Arachnids; Cambridge University Press: Cambridge, UK, 1997; pp. 110–129. [Google Scholar]
- Madsen, T.; Shine, R.; Loman, J.; Håkansson, T. Why do female adders copulate so frequently? Nature 1992, 355, 440–441. [Google Scholar] [CrossRef]
- Thornhill, R.; Alcock, J. The Evolution of Mating Systems in Insects and Arachnids; Cambridge University Press: Cambridge, UK, 1983. [Google Scholar]
- Pennington, J.T. The ecology of fertilization of echinoid eggs: The consequences of sperm dilution, adult aggregation, and synchronous spawning. Biol. Bull. 1985, 169, 417–430. [Google Scholar] [CrossRef] [PubMed]

| Treatments | rm (day−1) |
|---|---|
| Virgin females | - |
| 1 week mating | 0.1237 ± 0.0004a 1 |
| Lifetime | 0.1321 ± 0.0001b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Athanasiadis, K.; Pappas, M.L.; Broufas, G.D. Effect of Duration of Exposure to Males on Female Reproductive Performance of the Green Lacewing, Chrysoperla agilis (Neuroptera: Chrysopidae). Insects 2021, 12, 560. https://doi.org/10.3390/insects12060560
Athanasiadis K, Pappas ML, Broufas GD. Effect of Duration of Exposure to Males on Female Reproductive Performance of the Green Lacewing, Chrysoperla agilis (Neuroptera: Chrysopidae). Insects. 2021; 12(6):560. https://doi.org/10.3390/insects12060560
Chicago/Turabian StyleAthanasiadis, Konstantinos, Maria L. Pappas, and George D. Broufas. 2021. "Effect of Duration of Exposure to Males on Female Reproductive Performance of the Green Lacewing, Chrysoperla agilis (Neuroptera: Chrysopidae)" Insects 12, no. 6: 560. https://doi.org/10.3390/insects12060560
APA StyleAthanasiadis, K., Pappas, M. L., & Broufas, G. D. (2021). Effect of Duration of Exposure to Males on Female Reproductive Performance of the Green Lacewing, Chrysoperla agilis (Neuroptera: Chrysopidae). Insects, 12(6), 560. https://doi.org/10.3390/insects12060560








