Eugenol and Thymol Derivatives as Antifeedant Agents against Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Dryophthoridae) Larvae
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Pheromone Mass Trapping
2.2. Laboratory Rearing of RPW
2.3. Source of Eugenol and Thymol Derivatives
2.4. Bioassay
A = Weight of the larva after the experiment
B = Weight of the larva before the experiment
D = Weight of the food consumed by the larva
C = Weight of the food consumed in control
T = Weight of the food consumed in treatment
2.5. Data Analysis
3. Results
3.1. Eugenol Derivatives
3.2. Thymol Derivatives
3.3. Comparison of Eugenol and Thymol Zerivatives
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wahizatul, A.A.; Zazali, C.; Abdul Rahman, A.R.; Nurul Izzah, A.G. A new invasive coconut pest in Malaysia: The red palm weevil (Curculionidae: Rhynchophorus ferrugineus). Planter 2013, 89, 97–110. [Google Scholar]
- DOA (Department of Agriculture). Report on Current Status of Attack of the Red Palm Weevil, Rhynchophorus Ferrugineus in Terengganu; Biosecurity Division, Department of Agriculture: Kuala Lumpur, Malaysia, 2011. [Google Scholar]
- DOA (Department of Agriculture). Report on Current Status of Attack of the Red Palm Weevil, Rhynchophorus Ferrugineus in Terengganu; Biosecurity Division, Department of Agriculture, Government Press: Kuala Lumpur, Malaysia, 2016. [Google Scholar]
- Revival of Coconut Industry in Malaysia Available online:. Available online: Ap.fftc.agnet.org (accessed on 8 August 2020).
- Oil Palm Planted Area. 2017. Available online: http://bepi.mpob.gov.my/index.php/en/statistics/area/188-area2017.html (accessed on 8 August 2020).
- Yong, K.W.; Aisyah, A.B.; Wahizatul, A.A. Fecundity, fertility and survival of Red Palm Weevil (Rhynchophorus ferrugineus) larvae reared on sago palm. Sains Malays. 2015, 44, 1371–1375. [Google Scholar] [CrossRef]
- Abbas, T.S.M. IPM of the red palm weevil, Rhynchophorus ferrugineus. Integr. Manag. Plant Pest Dis. 2010, 5, 209–233. [Google Scholar]
- Faleiro, J.R. A review of the issues and management of the red palm weevil Rhynchophorus ferrugineus (Coleoptera: Rhynchophoridae) in coconut and date palm during the last one hundred years. Int. J. Trop. Insect Sci. 2006, 26, 135–154. [Google Scholar]
- Ishak, I.; Ng, L.C. , Haris-Hussain, M.; Jalinas, J., Idris, A.B.; Azlina, Z.; Samsudin, A.; Wahizatul, A.A. Pathogenicity of an indigenous strain of the Entomopathogenic fungus Metarhizium anisopliae (Hypocreales: Clavicipitaceae) (MET-GRA4 strain) as a potential biological control agent against the red palm weevil (Coleoptera: Dryophthoridae). J. Econ. Entomol. 2020, 113, 43–49. [Google Scholar]
- Aktar, M.W.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: Their benefits and hazards. Interdiscip. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef]
- Essumang, D.K.; Togoh, G.K.; Chokky, L. Pesticide residues in the water and fish (Lagoon Tilapia) samples from Lagoons in Ghana. B. Chem. Soc. Ethiop. 2009, 23, 19–27. [Google Scholar] [CrossRef]
- Dankyi, E.; Gordon, C.; Carboo, D.; Fomsgaard, I.S. Quantification of neonicotinoid insecticide residues in soils from cocoa plantations using a QuEChERS extraction procedure and LC-MS/MS. Sci. Total Environ. 2014, 499, 276–283. [Google Scholar] [CrossRef]
- Wawrzyniak, M. The effect of selected plant extracts on the cabbage butterfly, Pieris brassicae L. (Lepidoptera). Pol. J. Entomol. 1996, 65, 93–99. [Google Scholar]
- Taghizadeh Saroukolai, A.; Nouri-Ganbalani, G.; Rafiee-Dastjerdi, H.; Hadian, J. Antifeedant activity and toxicity of some essential oils to Colorado Potato Beetle, Leptinotarsa decemlineata Say (Coleoptera: Chrysomelidae). Plant Prot. Sci. 2014, 50, 207–216. [Google Scholar] [CrossRef]
- Shukla, P.; Vidyasagar, P.S.P.V.; Aldosari, S.A.; Abdel-Azim, M. Antifeedant activity of three essential oils against the red palm weevil, Rhynchophorus ferrugineus. B. Insectol. 2012, 65, 71–76. [Google Scholar]
- Ali, M.A.; Mohanny, K.M.; Mohamed, G.S.; Allam, R.O.H. Efficacy of some promising plant essential oils to control the red palm weevil Rhynchophorus ferrugineus (coleoptera: Curculionidae) under laboratory condition. Int. J. Agric. Sci. 2019, 1, 12–45. [Google Scholar]
- Mohamed, A.; Alghamdi, H.A.; Reyad, N.F. Nano essential oils against the red palm weevil, Rhynchophorus ferrugineus Olivier (Coleoptera: Curculionidae). Entomol. Res. 2020, 50, 215–220. [Google Scholar]
- Wahizatul, A.A.; Shahrol, N.D.; Mohd. Haris, H.; Yong, K.W.; Zazali, C.; Ahmad, S.S. Field trapping of adult red palm weevil Rhynchophorus ferrugineus Olivier (Coleoptera: Curculionidae) with food baits and synthetic pheromone lure in a coconut plantation. Philipp Agric. Sci. 2014, 97, 409–415. [Google Scholar]
- Zulkifli, A.N.; Zakeri, H.A.; Wahizatul, A.A. Food consumption, developmental time, and protein profile of the digestive system of the red palm weevil, Rhychrophorus ferrugineus (Coleoptera: Dryophthoridae) larvae reared on three different diets. J. Insect Sci. 2018, 18, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, D.; Abad, R.F.P.; Rashidi, M.R.; Mohammadi, S.A. Study og cotton bollworm, Helicoverpa armigera Hubner (Lepidoptera: Noctuidae) using Dyar’s Rule. Mun. Ent. Zool. 2010, 5, 216–224. [Google Scholar]
- Nurul Hazwani, C.A.R.; Asnuzilawati, A.; Noraznawati, I.; Hasnah, O. Synthesis and antibacterial study of eugenol derivatives. Asian J. Chem. 2017, 29, 22–26. [Google Scholar]
- Nurul Hazwani, C.A.R.; Asnuzilawati, A.; Noraznawati, I.; Fauziah, A.; Hasnah, O.; Soraya, S.M.T. Synthesis and antibacterial study of thymol derivatives. Asian J. Chem. 2018, 30, 126–128. [Google Scholar]
- El-Mergawy, R.A.A.M.; Al-Ajlan, A.M. Red palm weevil, Rhynchophorus ferrugineus (Olivier): Economic importance, biology, biogeography and integrated pest management. J. Agric. Sci. Tech. 2011, 1, 1–23. [Google Scholar]
- Price, D.N.; Berry, M.S. Comparison of effects of octopamine and insecticidal essential oils on activity in the nerve cord, foregut, and dorsal unpaired median neurons of cockroaches. J. Insect Physiol. 2006, 52, 309–319. [Google Scholar] [CrossRef]
- Kim, S.I.; Lee, D.W. Toxicity of basil and orange essential oils and their components against two coleopteran stored products insect pests. J. Asia-Pac. Entomol. 2014, 17, 13–17. [Google Scholar] [CrossRef]
- Renou, M.; Lucas, P.; Malo, E.; Quero, C.; Guerrero, A. Effects of trifluoromethyl ketones and related compounds on the EAG and behavioural responses to pheromones in male moths. Chem. Senses. 1997, 22, 407–416. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bau, J.; Martínez, D.; Renou, M.; Guerrero, A. Pheromone-triggered orientation flight of male moths can be disrupted by trifluoromethyl ketones. Chem. Senses. 1999, 24, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Traboulsi, A.F.; Taoubi, K.; El-Haj, S.; Bessiere, J.M.; Rammal, S. Insecticidal properties of essential plant oils against the mosquito Culex pipiens molestus (Diptera: Culicidae). Pest Manag. Sci. 2002, 58, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Waliwitiya, R.; Kennedy, C.J.; Lowenberger, C.A. Larvicidal and oviposition-altering activity of monoterpenoids, trans-anithole and rosemary oil to the yellow fever mosquito Aedes aegypti (Diptera: Culicidae). Pest Manag. Sci. 2009, 65, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Ziaee, M. The effects of topical application of two essential oils against Sitophilus granaries (Coleoptera: Curculionidae) and Tribolium confusum (Coleoptera: Tenebrionidae). J. Crop Prot. 2014, 3, 589–595. [Google Scholar]
- Oliveira, A.P.; Santos, A.A.; Santana, A.S.; Lima, A.P.S.; Melo, C.R.; Santana, E.D.; Sampaio, T.S.; Blank, A.F.; Araujo, A.P.A.; Cristaldo, P.F.; et al. Essential oil of Lippia sidoides and its major compound thymol: Toxicity and walking response of populations of Sitophilus zeamais (Coleoptera: Curculionidae). Crop Prot. 2018, 112, 33–38. [Google Scholar] [CrossRef]
- Persinger, R.L.; Poynter, M.E.; Ckless, K.; Janssen-Heininger, Y.M. Molecular mechanisms of nitrogen dioxide induced epithelial injury in the lung. Mol. Cell. Biochem. 2002, 234, 71–80. [Google Scholar] [CrossRef]
- Al-Jabr, A.M.; Hussain, A.; Rizwan-ul-Haq, M.; Al-Ayedh, H. Toxicity of plant secondary metabolites modulating detoxification genes expression for nature red palm weevil pesticide development. Molecules. 2017, 22, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Koul, O. Phytochemicals and insect control: An antifeedant approach. Crit. Rev. Plant Sci. 2008, 27, 1–24. [Google Scholar] [CrossRef]
- Jacobson, M.; Reed, D.K.; Crystal, M.M.; Moreno, D.S.; Soderstrom, E.L. Chemistry and biological activity of insect feeding deterrents from certain weed and crop plants. Entomol. Exp. Appl. 1978, 24, 448–457. [Google Scholar] [CrossRef]
- Isman, M.B.; Machial, C.M.; Miresmailli, S.; Bainard, L.D. Essential Oil Based Pesticides: New Insights from Old Chemistry; Wiley-VCH: Weinheim, Germany, 2017. [Google Scholar]
- Hosozawa, S.; Kato, N.; Munakata, K.; Chen, Y.L. Antifeeding active substances for insect in plant. Agric. Biol. Chem. 1974, 38, 1045–1048. [Google Scholar] [CrossRef]
- Ribes, S.; Ruiz-Rico, M.; Pérez-Esteve, É.; Fuentes, A.; Talens, P.; Martínez-Máñez, R.; Barat, J.M. Eugenol and thymol immobilized on mesoporous silica based material as an innovative antifungal system: Application in strawberry jam. Food Control. 2017, 81, 181–188. [Google Scholar] [CrossRef]
- Yousefi, M.; Hoseini, S.M.; Vatnikov, Y.A.; Nikishov, A.A.; Kulikov, E.V. Thymol as a new anesthetic in common carp (Cyprinus carpio): Efficacy and physiological effects in comparison with eugenol. Aquaculture 2018, 495, 376–383. [Google Scholar] [CrossRef]
- Denniston, K.; Topping, J.; Caret, R. General, Organic, and Biochemistry, 6th ed.; McGraw-Hill: New York, NY, USA, 2008. [Google Scholar]
- Wang, L.; Zhang, X.W.; Pan, L.L.; Liu, W.F.; Wang, D.P.; Zhang, G.Y.; Yin, Y.X.; Yin, A.; Jia, S.G.; Yu, X.G.; et al. A large-scale gene discovery for the red palm weevil Rhynchophorus ferrugineus (Coleoptera: Curculionidae). Insect Sci. 2013, 20, 689–702. [Google Scholar] [CrossRef]
- Park, N.J.; Kamble, S.T. Distribution and inhibition of esterases in various body tissues of susceptible and resistant German cockroaches (Dictyoptera: Blattellidae). Ann. Entomol. Soc. Am. 1999, 92, 556–562. [Google Scholar] [CrossRef]
- Kamita, S.G.; Hammock, B.D. Juvenile hormone esterase: Biochemistry and structure. J. Pest. Sci. 2010, 35, 265–274. [Google Scholar] [CrossRef]
- Bamidele, O.S.; Ajele, J.O.; Olajuyigbe, F.M. An evaluation of glutathione transferase associated with Dichlorvos degradation in African palm weevil (Rynchophorus phoenicis) larva. Cogent Biol. 2017, 3, 1286764. [Google Scholar] [CrossRef]
| Series | Code | Name | Class | Molecular Structure | Appearance |
|---|---|---|---|---|---|
| Eugenol derivative | WN11 | 4-allyl-2-methoxy-1-(4-nitrobenzyloxy)-benzene | Ether |  | Yellow solid |
| WN16 | 4-allyl-2-methoxy-1-(4-trifluoromethyl-benzyloxy)-benzene | Ether | 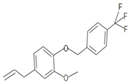 | White solid | |
| WN14 | 4-allyl-2methoxyphenyl 4-ethylbenzoate | Ester | 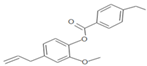 | White solid | |
| WN15 | 4-allyl-2-methoxyphenyl 4-bromobenzoate | Ester |  | White solid | |
| Thymol derivative | WNT1 | 2-(benzyloxy)-1-isopropyl-4-methylbenzene | Ether |  | Oily solution |
| WNT2 | 2-isopropyl-4-methyl-2-((4-nitrobenzyl) oxy) benzene | Ether | 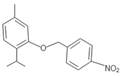 | Yellowish orange solid | |
| WNT4 | 2-isopropyl-5-methylphenyl 4-bromobenzoate | Ester |  | Oily yellow solution | |
| WNT5 | 2-isopropyl-5-methylphenyl 4-ethylbenzoate | Ester |  | Oily yellow solution |
| Treatment | Concentration (ppm) | Consumption (g/day) | RGR (g/g day) | RCR (g/g day) | ECI | FDI (%) |
|---|---|---|---|---|---|---|
| Control | 0 | 0.386 ± 0.013 * | −0.011 ± 0.01 | 0.316 ± 0.039 a | −9.156 ±1.8 ab | - |
| WN11 | 200 | 0.215 ± 0.003 a,A | −0.008 ± 0.00 | 0.090 ± 0.005 b | −2.120 ± 4.38 ab | 44.22 ab,A |
| 400 | 0.207 ± 0.002 b,A | −0.016 ± 0.01 | 0.095 ± 0.007 b | −22.78 ± 7.98 a | 46.37 b,A | |
| 600 | 0.228 ± 0.0004 a,B | −0.027 ± 0.02 | 0.159 ± 0.041 b | −14.93 ± 12.9 ab | 40.73 a,A | |
| WN14 | 200 | 0.240 ± 0.001 a,AB | −0.001 ± 0.01 | 0.1692 ± 0.024 b | 2.899 ± 8.21 ab | 37.74 a,A |
| 400 | 0.241 ± 0.002 d,B | −0.006 ± 0.02 | 0.214 ± 0.029 ab | 0.733 ± 18.15 ab | 37.43 a,A | |
| 600 | 0.235 ± 0.0006 ab,A | 0.019 ±0.01 | 0.212 ± 0.058 ab | 19.15 ± 11.18 ab | 39.13 a,A | |
| WN15 | 200 | 0.237 ± 0.002 a,C | 0.017 ± 0.001 | 0.096 ± 0.003 b | 25.72 ± 1.51 b | 38.47 ab,A |
| 400 | 0.218 ± 0.001 c,A | −0.023 ± 0.01 | 0.129 ± 0.014 b | −7.499 ± 7.25 ab | 43.49 ab,A | |
| 600 | 0.225 ± 0.0008 a,B | −0.007 ± 0.01 | 0.112 ± 0.013 b | 2.10 ± 11.04 ab | 41.71 a,A | |
| WN16 | 200 | 0.210 ± 0.014 a,AB | −0.005 ± 0.01 | 0.099 ± 0.013 b | −3.127 ± 7.51 ab | 45.74 b,B |
| 400 | 0.177 ± 0.004 a,A | −0.009 ± 0.02 | 0.093 ± 0.021 b | −8.855 ± 12.0 ab | 54.14 c,C | |
| 600 | 0.245 ± 0.006 b,B | −0.022 ± 0.00 | 0.143 ± 0.023 b | −15.16 ± 2.27 ab | 36.59 a,A |
| Functional Group | N | Mean | Standard Error |
|---|---|---|---|
| Ether | 18 | 44.63 * | 1.40 |
| Ester | 18 | 39.66 | 0.81 |
| Treatment | Concentration (ppm) | Consumption (g/day) | RGR (g/g day) | RCR (g/g day) | ECI | FDI (%) |
|---|---|---|---|---|---|---|
| Control | 0 | 0.386 ± 0.013 * | −0.011 ± 0.01 | 0.316 ± 0.039 a | −9.156 ±1.8 ab | - |
| WNT1 | 200 | 0.213 ± 0.001 b,B | −0.002 ± 0.01 | 0.075 ± 0.010 b | −5.213 ± 7.496 | 44.72 a,A |
| 400 | 0.221 ± 0.001 b,C | −0.012 ± 0.01 | 0.089 ± 0.005 b | −12.146 ± 8.568 | 42.64 a,A | |
| 600 | 0.206 ± 0.0006 a,A | −0.027 ± 0.01 | 0.101 ± 0.015 b | −27.446 ± 4.889 | 46.52 b,A | |
| WNT2 | 200 | 0.178 ± 0.003 a,A | −0.005 ± 0.00 | 0.096 ± 0.015 b | 0.489 ± 5.774 | 53.88 b,B |
| 400 | 0.189 ± 0.004 a,B | −0.011 ± 0.02 | 0.121 ± 0.043 b | 1.459 ± 14.451 | 50.91 b,B | |
| 600 | 0.227 ± 0.001 b,C | 0.001 ± 0.02 | 0.116 ± 0.007 b | 3.523 ± 16.979 | 41.14 ab,A | |
| WNT4 | 200 | 0.222 ± 0.002 c,A | −0.024 ± 0.01 | 0.109 ± 0.024 b | −21.626 ± 1.375 | 42.49 a,A |
| 400 | 0.240 ± 0.002 c,B | −0.009 ± 0.00 | 0.081 ± 0.009 b | −11.349 ± 3.949 | 37.64 a,A | |
| 600 | 0.227 ± 0.0005 b,A | −0.009 ± 0.00 | 0.073 ± 0.004 b | −11.906 ± 1.095 | 41.10 ab,A | |
| WNT5 | 200 | 0.229 ± 0.001 c,A | 0.005 ± 0.00 | 0.092 ± 0.003 b | 0.175 ± 2.723 | 40.62 a,A |
| 400 | 0.237 ± 0.003 c,AB | −0.014 ± 0.01 | 0.102 ± 0.015 b | −11.607 ± 2.622 | 38.58 a,A | |
| 600 | 0.238 ± 0.0007 c,B | −0.003 ± 0.01 | 0.110 ± 0.008 b | −3.595 ± 4.371 | 38.13 a,A |
| Functional Group | N | Mean | Standard Error |
|---|---|---|---|
| Ether | 18 | 46.64 * | 1.40 |
| Ester | 18 | 39.76 | 0.81 |
| Functional Group | N | Mean | Standard Error |
|---|---|---|---|
| Eugenol | 36 | 42.15 | 0.8993 |
| Thymol | 36 | 43.20 | 0.9046 |
| Functional Group | N | Mean | Standard Error |
|---|---|---|---|
| Ether | 36 | 45.63 * | 0.92 |
| Ester | 36 | 39.71 | 0.54 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, T.K.; Asari, A.; Salleh, S.A.; Azmi, W.A. Eugenol and Thymol Derivatives as Antifeedant Agents against Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Dryophthoridae) Larvae. Insects 2021, 12, 551. https://doi.org/10.3390/insects12060551
Yan TK, Asari A, Salleh SA, Azmi WA. Eugenol and Thymol Derivatives as Antifeedant Agents against Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Dryophthoridae) Larvae. Insects. 2021; 12(6):551. https://doi.org/10.3390/insects12060551
Chicago/Turabian StyleYan, Tay Karh, Asnuzilawati Asari, Siti Aishah Salleh, and Wahizatul Afzan Azmi. 2021. "Eugenol and Thymol Derivatives as Antifeedant Agents against Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Dryophthoridae) Larvae" Insects 12, no. 6: 551. https://doi.org/10.3390/insects12060551
APA StyleYan, T. K., Asari, A., Salleh, S. A., & Azmi, W. A. (2021). Eugenol and Thymol Derivatives as Antifeedant Agents against Red Palm Weevil, Rhynchophorus ferrugineus (Coleoptera: Dryophthoridae) Larvae. Insects, 12(6), 551. https://doi.org/10.3390/insects12060551






