Identifying Molecular-Based Trophic Interactions as a Resource for Advanced Integrated Pest Management
Abstract
Simple Summary
Abstract
1. Introduction
- MGCA—Molecular gut content analysis is the process of using florescent dye, protein marker or DNA based tools to detect prey remains with the gut contents of predators.
- ELISA—Enzyme-linked immunosorbent assay, which is a protein detection system de-signed to mark prey or detect specific proteins within the gut contents of a predator (e.g., Hagler and Durand 1994).
- PCR or Diagnostic PCR—Polymerase chain reaction. Technique used to create copies of target DNA and in this application, used for diagnostic assessment of prey recently consumed by predators [9]. Or, for detecting parasitoids within hosts [10].
- NGS—Next generation sequencing. Massively parallel sequencing technology capable of producing millions of sequences of DNA or RNA in mixed samples.
- DNA metabarcoding or molecular barcoding—An application of NGS where commonly 6–8 bp MID tags (molecular identification tags or codes) are added to the PCR primers to mark each sample and allows for tracking samples that are combined to form libraries of samples that are then run together. Sequences, reads recovered from samples, are later used to determine the species in samples by the MID tags for each sample to assemble trophic links using available sequence data in GenBank and BOLD [11].
- eDNA—Environmental DNA from any sample source. These could be from soil or in-sect or plant, pollen on legs of bees, arthropod exuviae, swabs of external body, or whole body. eDNA can then be used to reconstruct trophic interactions using NGS techniques such as DNA metabarcoding e.g., [12].
- HTS—High-throughput sequencing techniques using NGS to power the rapid processing of thousands of samples simultaneously to explore biodiversity or trophic structure in agricultural systems.
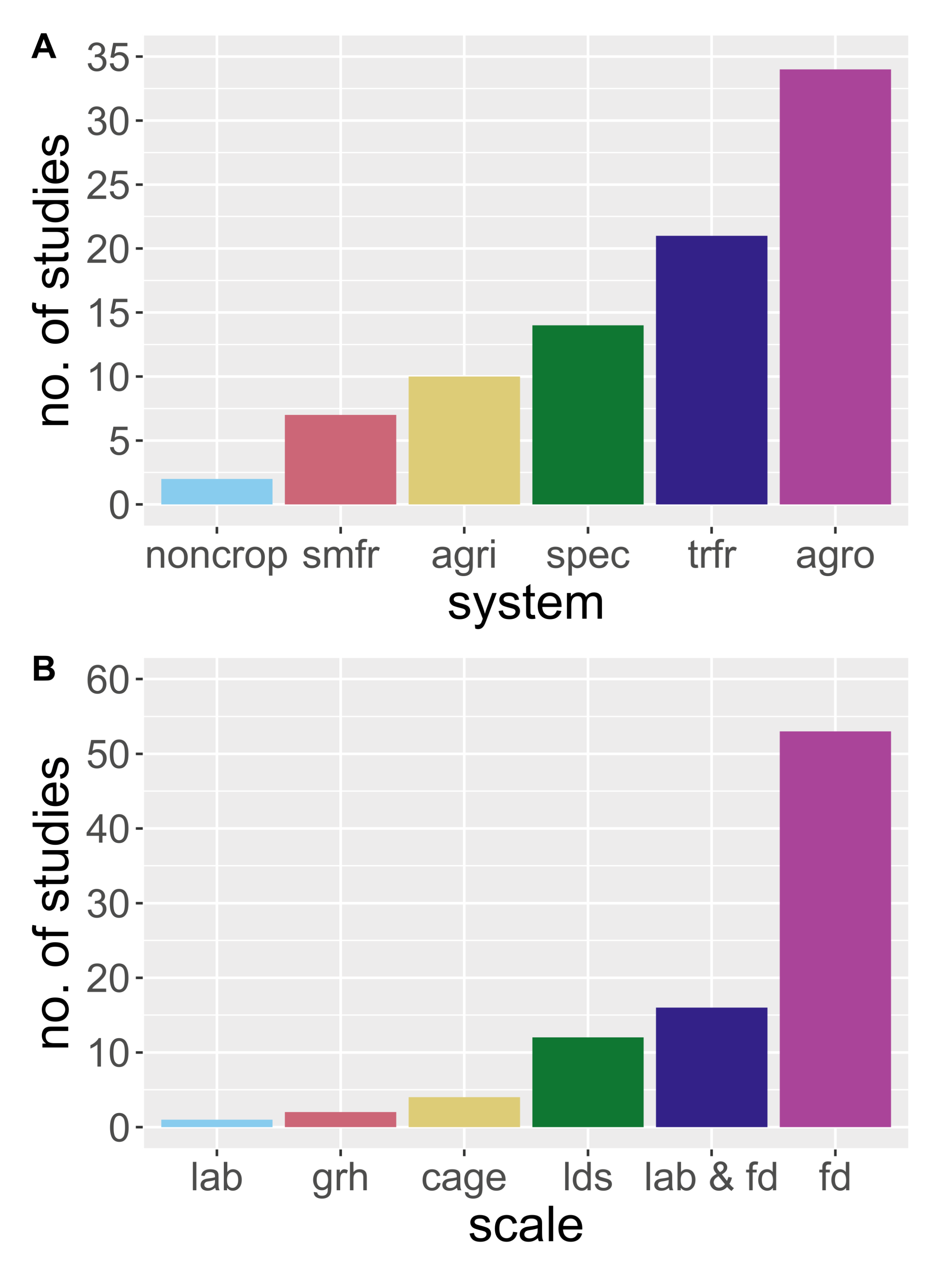
2. Methods: The Review Process and Scope
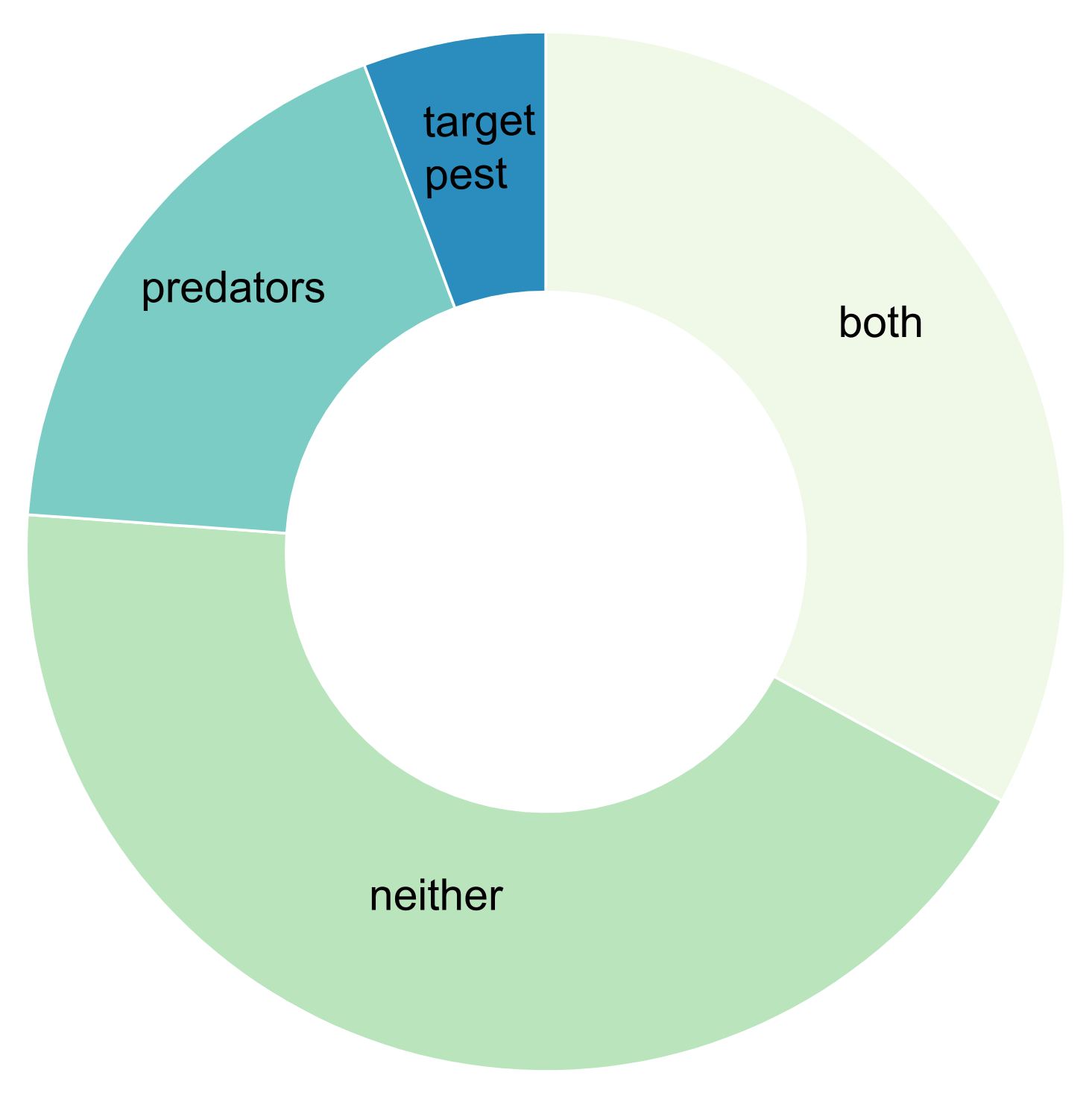
3. Results
3.1. MGCA in Agronomic Cropping Systems
3.2. Current Application of MGCA to Understand Tree Fruit and Tree Nut Systems
3.3. MGCA to Understand Predation in Specialty Row Crops
3.4. Applying MGCA to Agricultural Landscapes
3.5. Using MGCA to Expose Intraguild Predation
3.6. Roles of Vertebrates in Biocontrol Determined by MGCA
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gurr, G.M.; You, M.S. Conservation Biological Control of Pests in the Molecular Era: New Opportunities to Address Old Constraints. Front. Plant Sci. 2016, 6, 1255. [Google Scholar] [CrossRef]
- Losey, J.E.; Vaughan, M. The economic value of ecological services provided by insects. Bioscience 2006, 56, 311–323. [Google Scholar] [CrossRef]
- Gurr, G.M.; Lu, Z.X.; Zheng, X.S.; Xu, H.X.; Zhu, P.Y.; Chen, G.H.; Yao, X.M.; Cheng, J.; Zhu, Z.R.; Catindig, J.L.; et al. Multi-country evidence that crop diversification promotes ecological intensification of agriculture. Nat. Plants 2016, 2, 16014. [Google Scholar] [CrossRef]
- Heimpel, G.E.; Mills, N.J. Biologica Control: Ecology and Applications; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- Lundgren, J.G.; Fausti, S.W. Trading biodiversity for pest problems. Sci. Adv. 2015, 1, e1500558. [Google Scholar] [CrossRef]
- Perovic, D.J.; Gamez-Virues, S.; Landis, D.A.; Wackers, F.; Gurr, G.M.; Wratten, S.D.; You, M.S.; Desneux, N. Managing biological control services through multi-trophic trait interactions: Review and guidelines for implementation at local and landscape scales. Biol. Rev. 2018, 93, 306–321. [Google Scholar] [CrossRef] [PubMed]
- Symondson, W.O.C.; Sunderland, K.D.; Greenstone, M.H. Can generalist predators be effective biocontrol agents? Annu. Rev. Entomol. 2002, 47, 561–594. [Google Scholar] [CrossRef]
- Hooper, D.U.; Chapin, F.S.; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Symondson, W.O.C. The molecular revolution: Using polymerase chain reaction based methods to explore the role of predators in terrestrial food webs. In Biodiersity and Insect Pests: Key Issues for Sustainable Management; Gurr, G.M., Wratten, S., Snyder, W.E., Read, M.Y., Eds.; Wiley-Blackwell: West Sussex, UK, 2012; pp. 166–184. [Google Scholar]
- Gariepy, T.D.; Bruin, A.; Konopka, J.; Scott-Dupree, C.; Fraser, H.; Bon, M.C.; Talamas, E. A modified DNA barcode approach to define trophic interactions between native and exotic pentatomids and their parasitoids. Mol. Ecol. 2019, 28, 456–470. [Google Scholar] [CrossRef] [PubMed]
- Kitson, J.J.N.; Hahn, C.; Sands, R.J.; Straw, N.A.; Evans, D.M.; Lunt, D.H. Detecting host-parasitoid interactions in an invasive Lepidopteran using nested tagging DNA metabarcoding. Mol. Ecol. 2019, 28, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, P.F.; Sigsgaard, E.E. Environmental DNA metabarcoding of wild flowers reveals diverse communities of terrestrial arthropods. Ecol. Evol. 2019, 9, 1665–1679. [Google Scholar] [CrossRef] [PubMed]
- Clare, E.L. Molecular detection of trophic interactions: Emerging trends, distinct advantages, significant considerations and conservation applications. Evol. Appl. 2014, 7, 1144–1157. [Google Scholar] [CrossRef]
- González-Chang, M.; Wratten, S.D.; Lefort, M.-C.; Boyer, S. Food webs and biological control: A review of molecular tools used to reveal trophic interactions in agricultural systems. Food Webs 2016, 9, 4–11. [Google Scholar] [CrossRef]
- Sheppard, S.K.; Harwood, J.D. Advances in molecular ecology: Tracking trophic links through predator-prey food-webs. Funct. Ecol. 2005, 19, 751–762. [Google Scholar] [CrossRef]
- Deagle, B.E.; Thomas, A.C.; McInnes, J.C.; Clarke, L.J.; Vesterinen, E.J.; Clare, E.L.; Kartzinel, T.R.; Eveson, J.P. Counting with DNA in metabarcoding studies: How should we convert sequence reads to dietary data. Mol. Ecol. 2019, 28, 391–406. [Google Scholar] [CrossRef]
- Staniczenko, P.P.A.; Lewis, O.T.; Tylianakis, J.M.; Albrecht, M.; Coudrain, V.; Klein, A.M.; Reed-Tsochas, F. Predicting the effect of habitat modification on networks of interacting species. Nat. Commun. 2017, 8, 792. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Polo, P.; Alomar, O.; Castane, C.; Lundgren, J.G.; Pinol, J.; Agusti, N. Molecular assessment of predation by hoverflies (Diptera: Syrphidae) in Mediterranean lettuce crops. Pest Manag. Sci. 2015, 71, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Furlong, M.J. Knowing your enemies: Integrating molecular and ecological methods to assess the impact of arthropod predators on crop pests. Insect Sci. 2015, 22, 6–19. [Google Scholar] [CrossRef]
- King, R.A.; Symondson, W.O.C.; Thomas, R.J. Molecular analysis of faecal samples from birds to identify potential crop pests and useful biocontrol agents in natural areas. Bull. Entomol. Res. 2015, 105, 261–272. [Google Scholar] [CrossRef]
- Tillman, P.G.; Greenstone, M.H.; Hu, J.S. Predation of stink bugs (Hemiptera: Pentatomidae) by a complex of predators in cotton and adjoining soybean habitats in Georgia, USA. Fla. Entomol. 2015, 98, 1114–1126. [Google Scholar] [CrossRef]
- Bowers, C.; Toews, M.; Liu, Y.; Schmidt, J.M. Cover crops improve early season natural enemy recruitment and pest management in cotton production. Biol. Control 2020, 141, 104149. [Google Scholar] [CrossRef]
- Choate, B.A.; Lundgren, J.G. Invertebrate communities in spring wheat and the identification of cereal aphid predators through molecular gut content analysis. Crop Prot. 2015, 77, 110–118. [Google Scholar] [CrossRef]
- Li, J.H.; Yang, F.; Wang, Q.; Pan, H.S.; Yuan, H.B.; Lu, Y.H. Predation by generalist arthropod predators on Apolygus lucorum (Hemiptera: Miridae): Molecular gut-content analysis and field-cage assessment. Pest Manag. Sci. 2017, 73, 628–635. [Google Scholar] [CrossRef]
- Roubinet, E.; Birkhofer, K.; Malsher, G.; Staudacher, K.; Ekbom, B.; Traugott, M.; Jonsson, M. Diet of generalist predators reflects effects of cropping period and farming system on extra—And intraguild prey. Ecol. Appl. 2017, 27, 1167–1177. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Bao, W.; Yang, F.; Yang, Y.; Lu, Y. A PCR-based analysis of plant DNA reveals the feeding preferences of Apolygus lucorum (Heteroptera: Miridae). Arthropod-Plant Interact. 2018, 12, 567–574. [Google Scholar] [CrossRef]
- Wang, Q.; Bao, W.F.; Yang, F.; Xu, B.; Yang, Y.Z. The specific host plant DNA detection suggests a potential migration of Apolygus lucorum from cotton to mungbean fields. PLoS ONE 2017, 12, e0177789. [Google Scholar] [CrossRef] [PubMed]
- Ju, Q.; Ouyang, F.; Gu, S.M.; Qiao, F.; Yang, Q.F.; Qu, M.J.; Ge, F. Strip intercropping peanut with maize for peanut aphid biological control and yield enhancement. Agric. Ecosyst. Environ. 2019, 286, 8. [Google Scholar] [CrossRef]
- Li, J.H.; Liu, B.; Pan, H.S.; Luo, S.P.; Wyckhuys, K.A.G.; Yuan, H.B.; Lu, Y.H. Buckwheat strip crops increase parasitism of Apolygus lucorum in cotton. Biocontrol 2019, 64, 645–654. [Google Scholar] [CrossRef]
- Peterson, J.A.; Burkness, E.C.; Harwood, J.D.; Hutchison, W.D. Molecular gut-content analysis reveals high frequency of Helicoverpa zea (Lepidoptera: Noctuidae) consumption by Orius insidiosus (Hemiptera: Anthocoridae) in sweet corn. Biol. Control 2018, 121, 1–7. [Google Scholar] [CrossRef]
- Kheirodin, A.; Sharanowski, B.J.; Carcamo, H.A.; Costamagna, A.C. Consumption of cereal leaf beetle, Oulema melanopus, by generalist predators in wheat fields detected by molecular analysis. Entomol. Exp. Appl. 2020, 168, 59–69. [Google Scholar] [CrossRef]
- Athey, K.J.; Sitvarin, M.I.; Harwood, J.D. Laboratory and Field Investigation of Biological Control for Brown Marmorated Stink Bug (Halyomorpha halys (Stal) (Hemiptera: Pentatomidae)). J. Kans. Entomol. Soc. 2017, 90, 341–352. [Google Scholar] [CrossRef]
- Roubinet, E.; Jonsson, T.; Malsher, G.; Staudacher, K.; Traugott, M.; Ekbom, B.; Jonsson, M. High Redundancy as well as Complementary Prey Choice Characterize Generalist Predator Food Webs in Agroecosystems. Sci. Rep. 2018, 8, 8054. [Google Scholar] [CrossRef] [PubMed]
- Roubinet, E.; Straub, C.; Jonsson, T.; Staudacher, K.; Traugott, M.; Ekbom, B.; Jonsson, M. Additive effects of predator diversity on pest control caused by few interactions among predator species. Ecol. Entomol. 2015, 40, 362–371. [Google Scholar] [CrossRef]
- Staudacher, K.; Rubbmark, O.R.; Birkhofer, K.; Malsher, G.; Sint, D.; Jonsson, M.; Traugott, M. Habitat heterogeneity induces rapid changes in the feeding behaviour of generalist arthropod predators. Funct. Ecol. 2018, 32, 809–819. [Google Scholar] [CrossRef]
- Wang, Q.; Bao, W.; Zhang, Q.; Fu, X.; Yang, Y.; Lu, Y. Host plant use of a polyphagous mirid, Apolygus lucorum: Molecular evidence from migratory individuals. Ecol. Evol. 2019, 9, 11518–11528. [Google Scholar] [CrossRef] [PubMed]
- Hagler, J.R. It’s Gut Check Time! A Universal Food Immunomarking Technique for Studying Arthropod Feeding Activities. Ann. Entomol. Soc. Am. 2019, 112, 211–219. [Google Scholar] [CrossRef]
- Irvin, N.A.; Hagler, J.R.; Hoddle, M.S. Measuring natural enemy dispersal from cover crops in a California vineyard. Biol. Control 2018, 126, 15–25. [Google Scholar] [CrossRef]
- Hagler, J.R.; Mostafa, A.M. A Gut Analysis Technique for Pinpointing Egg-Specific Predation Events. J. Insect Sci. 2019, 19. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Ouyang, G.C.; Liu, H.; Hou, B.H.; Huang, S.S.; Guo, M.F. Molecular screening and predation evaluation of the key predators of Conopomorpha sinensis Bradley (Lepidoptera: Gracilariidae) in litchi orchards. Bull. Entomol. Res. 2014, 104, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.M.; Szendrei, Z.; Grieshop, M. Elucidating the Common Generalist Predators of Conotrachelus nenuphar (Herbst) (Coleoptera: Curculionidae) in an Organic Apple Orchard Using Molecular Gut-Content Analysis. Insects 2016, 7, 29. [Google Scholar] [CrossRef]
- Gutierrez-Coarite, R.; Yoneishi, N.M.; Pulakkatu-Thodi, I.; Mollinedo, J.; Calla, B.; Wright, M.G.; Geib, S.M. PCR-Based Gut Content Analysis to Detect Predation of Eriococcus ironsidei (Hemiptera: Eriococcidae) by Coccinellidae Species in Macadamia Nut Orchards in Hawaii. J. Econ. Entomol. 2018, 111, 885–891. [Google Scholar] [CrossRef]
- Bouvet, J.P.R.; Urbaneja, A.; Perez-Hedo, M.; Monzo, C. Contribution of predation to the biological control of a key herbivorous pest in citrus agroecosystems. J. Anim. Ecol. 2019, 88, 915–926. [Google Scholar] [CrossRef]
- de Roince, C.B.; Lavigne, C.; Ricard, J.M.; Franck, P.; Bouvier, J.C.; Garcin, A.; Symondson, W.O.C. Predation by generalist predators on the codling moth versus a closely-related emerging pest the oriental fruit moth: A molecular analysis. Agric. For. Entomol. 2012, 14, 260–269. [Google Scholar] [CrossRef]
- Heimoana, V.; Pilkington, L.J.; Raman, A.; Mitchell, A.; Nicol, H.I.; Johnson, A.C.; Gurr, G.M. Integrating spatially explicit molecular and ecological methods to explore the significance of non-crop vegetation to predators of brassica pests. Agric. Ecosyst. Environ. 2017, 239, 12–19. [Google Scholar] [CrossRef]
- Mabin, M.D.; Welty, C.; Gardiner, M.M. Predator richness predicts pest suppression within organic and conventional summer squash (Cucurbita pepo L. Cucurbitales: Cucurbitaceae). Agric. Ecosyst. Environ. 2020, 287, 106689. [Google Scholar] [CrossRef]
- Gomez-Polo, P.; Alomar, O.; Castane, C.; Aznar-Fernandez, T.; Lundgren, J.G.; Pinol, J.; Agusti, N. Understanding trophic interactions of Orius spp. (Hemiptera: Anthocoridae) in lettuce crops by molecular methods. Pest Manag. Sci. 2016, 72, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Rusch, A.; Chaplin-Kramer, R.; Gardiner, M.M.; Hawro, V.; Holland, J.; Landis, D.; Thies, C.; Tscharntke, T.; Weisser, W.W.; Winqvist, C.; et al. Agricultural landscape simplification reduces natural pest control: A quantitative synthesis. Agric. Ecosyst. Environ. 2016, 221, 198–204. [Google Scholar] [CrossRef]
- Gardiner, M.M.; Landis, D.A.; Gratton, C.; DiFonzo, C.D.; O’Neal, M.; Chacon, J.M.; Wayo, M.T.; Schmidt, N.P.; Mueller, E.E.; Heimpel, G.E. Landscape diversity enhances biological control of an introduced crop pest in the north-central USA. Ecol. Appl. 2009, 19, 143–154. [Google Scholar] [CrossRef]
- Grab, H.; Danforth, B.; Poveda, K.; Loeb, G. Landscape simplification reduces classical biological control and crop yield. Ecol. Appl. 2018, 28, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Martínez, S.A.; Lavandero, B. The effect of landscape context on the biological control of Sitobion avenae: Temporal partitioning response of natural enemy guilds. J. Pest Sci. 2018, 91, 41–53. [Google Scholar] [CrossRef]
- Bowers, C.L.; Toews, M.D.; Schmidt, J.M. Beyond soil health: The trophic effects of cover crops shape predator communities. bioRxiv 2020. [Google Scholar] [CrossRef]
- Ortiz-Martínez, S.; Staudacher, K.; Baumgartner, V.; Traugott, M.; Lavandero, B. Intraguild predation is independent of landscape context and does not affect the temporal dynamics of aphids in cereal fields. J. Pest Sci. 2020, 93, 235–249. [Google Scholar] [CrossRef]
- Tscharntke, T.; Tylianakis, J.M.; Rand, T.A.; Didham, R.K.; Fahrig, L.; Batary, P.; Bengtsson, J.; Clough, Y.; Crist, T.O.; Dormann, C.F.; et al. Landscape moderation of biodiversity patterns and processes—Eight hypotheses. Biol. Rev. 2012, 87, 661–685. [Google Scholar] [CrossRef] [PubMed]
- Ingrao, A.J.; Schmidtl, J.; Jubenville, J.; Grode, A.; Komondy, L.; VanderZee, D.; Szendrei, Z. Biocontrol on the edge: Field margin habitats in asparagus fields influence natural enemy-pest interactions. Agric. Ecosyst. Environ. 2017, 243, 47–54. [Google Scholar] [CrossRef]
- Morrison, W.R.; Gibson, G.A.P.; Szendrei, Z. The Parasitoids of the Asparagus Miner (Diptera: Agromyzidae): Field Parasitism and the Influence of Food Resources on Life History. Environ. Entomol. 2014, 43, 1526–1534. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.M.; Whitehouse, T.S.; Green, K.; Krehenwinkel, H.; Schmidt-Jeffris, R.; Sial, A.A. Local and landscape-scale heterogeneity shape spotted wing drosophila (Drosophila suzukii) activity and natural enemy abundance: Implications for trophic interactions. Agric. Ecosyst. Environ. 2019, 272, 86–94. [Google Scholar] [CrossRef]
- Brown, P.M.J.; Ingels, B.; Wheatley, A.; Rhule, E.L.; de Clercq, P.; van Leeuwen, T.; Thomas, A. Intraguild predation by Harmonia axyridis (Coleoptera: Coccinellidae) on native insects in Europe: Molecular detection from field samples. Entomol. Sci. 2015, 18, 130–133. [Google Scholar] [CrossRef]
- Rondoni, G.; Fenjan, S.; Bertoldi, V.; Ielo, F.; Djelouah, K.; Moretti, C.; Buonaurio, R.; Ricci, C.; Conti, E. Molecular detection of field predation among larvae of two ladybird beetles is partially predicted from laboratory experiments. Sci. Rep. 2018, 8, 2594. [Google Scholar] [CrossRef] [PubMed]
- Petrakova, L.; Michalko, R.; Loverre, P.; Sentenska, L.; Korenko, S.; Pekar, S. Intraguild predation among spiders and their effect on the pear psylla during winter. Agric. Ecosyst. Environ. 2016, 233, 67–74. [Google Scholar] [CrossRef]
- Masonick, P.; Hernandez, M.; Weirauch, C. No guts, no glory: Gut content metabarcoding unveils the diet of a flower-associated coastal sage scrub predator. Ecosphere 2019, 10, e02712. [Google Scholar] [CrossRef]
- Russo, D.; Bosso, L.; Ancillotto, L. Novel perspectives on bat insectivory highlight the value of this ecosystem service in farmland: Research frontiers and management implications. Agric. Ecosyst. Environ. 2018, 266, 31–38. [Google Scholar] [CrossRef]
- de Torrez, E.C.B.; Brown, V.A.; McCracken, G.F.; Kunz, T.H. Sympatric Bat Species Prey Opportunistically on a Major Moth Pest of Pecans. Sustainability 2019, 11, 6365. [Google Scholar] [CrossRef]
- Mangan, A.M.; Piaggio, A.J.; Hopken, M.W.; Werner, S.J.; Pejchar, L. A molecular analysis to assess codling moth Cydia pomonella L. (Lepidoptera: Tortricidae) predation by orchard birds. Ecol. Indic. 2018, 93, 1222–1225. [Google Scholar] [CrossRef]
- Crisol-Martinez, E.; Moreno-Moyano, L.T.; Wormington, K.R.; Brown, P.H.; Stanley, D. Using Next-Generation Sequencing to Contrast the Diet and Explore Pest-Reduction Services of Sympatric Bird Species in Macadamia Orchards in Australia. PLoS ONE 2016, 11, e0150159. [Google Scholar] [CrossRef]
- Garfinkel, M.B.; Minor, E.S.; Whelan, C.J. Birds suppress pests in corn but release them in soybean crops within a mixed prairie/agriculture system. Condor 2020, 122, duaa009. [Google Scholar] [CrossRef] [PubMed]
- Kemp, J.; Lopez-Baucells, A.; Rocha, R.; Wangensteen, O.S.; Andriatafika, Z.; Nair, A.; Cabeza, M. Bats as potential suppressors of multiple agricultural pests: A case study from Madagascar. Agric. Ecosyst. Environ. 2019, 269, 88–96. [Google Scholar] [CrossRef]
- Aizpurua, O.; Budinski, I.; Georgiakakis, P.; Gopalakrishnan, S.; Ibanez, C.; Mata, V.; Rebelo, H.; Russo, D.; Szodoray-Paradi, F.; Zhelyazkova, V.; et al. Agriculture shapes the trophic niche of a bat preying on multiple pest arthropods across Europe: Evidence from DNA metabarcoding. Mol. Ecol. 2018, 27, 815–825. [Google Scholar] [CrossRef]
- Baroja, U.; Garin, I.; Aihartza, J.; Arrizabalaga-Escudero, A.; Vallejo, N.; Aldasoro, M.; Goiti, U. Pest consumption in a vineyard system by the lesser horseshoe bat (Rhinolophus hipposideros). PLoS ONE 2019, 14, e0219265. [Google Scholar] [CrossRef]
- Cohen, Y.; Bar-David, S.; Nielsen, M.; Bohmann, K.; Korine, C. An appetite for pests: Synanthropic insectivorous bats exploit cotton pest irruptions and consume various deleterious arthropods. Mol. Ecol. 2020, 29, 1185–1198. [Google Scholar] [CrossRef]
- Jedlicka, J.A.; Vo, A.-T.E.; Almeida, R.P.P. Molecular scatology and high-throughput sequencing reveal predominately herbivorous insects in the diets of adult and nestling Western Bluebirds (Sialia mexicana) in California vineyards. Auk 2016, 134, 116–127. [Google Scholar] [CrossRef]
- Weier, S.M.; Moodley, Y.; Fraser, M.F.; Linden, V.M.G.; Grass, I.; Tscharntke, T.; Taylor, P.J. Insect pest consumption by bats in macadamia orchards established by molecular diet analyses. Glob. Ecol. Conserv. 2019, 18, e00626. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Garnett, T. Food security and sustainable intensification. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369. [Google Scholar] [CrossRef] [PubMed]
- Pompanon, F.; Deagle, B.E.; Symondson, W.O.C.; Brown, D.S.; Jarman, S.N.; Taberlet, P. Who is eating what: Diet assessment using next generation sequencing. Mol. Ecol. 2012, 21, 1931–1950. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, M.L.; Mondal, S. Sustainable Management of Key Lepidopteran Insect Pests of Vegetables. In I International Symposium on Sustainable Vegetable Production in Southeast Asia; DeNeve, S., Boehme, M., Everaarts, A., Neeteson, J., Eds.; ISHS: Leuven, Belgium, 2012; Volume 958, pp. 147–153. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, J.M.; Acebes-Doria, A.; Blaauw, B.; Kheirodin, A.; Pandey, S.; Lennon, K.; Kaldor, A.D.; Toledo, P.F.S.; Grabarczyk, E.E. Identifying Molecular-Based Trophic Interactions as a Resource for Advanced Integrated Pest Management. Insects 2021, 12, 358. https://doi.org/10.3390/insects12040358
Schmidt JM, Acebes-Doria A, Blaauw B, Kheirodin A, Pandey S, Lennon K, Kaldor AD, Toledo PFS, Grabarczyk EE. Identifying Molecular-Based Trophic Interactions as a Resource for Advanced Integrated Pest Management. Insects. 2021; 12(4):358. https://doi.org/10.3390/insects12040358
Chicago/Turabian StyleSchmidt, Jason M., Angelita Acebes-Doria, Brett Blaauw, Arash Kheirodin, Swikriti Pandey, Kylie Lennon, Amos D. Kaldor, Pedro F. S. Toledo, and Erin E. Grabarczyk. 2021. "Identifying Molecular-Based Trophic Interactions as a Resource for Advanced Integrated Pest Management" Insects 12, no. 4: 358. https://doi.org/10.3390/insects12040358
APA StyleSchmidt, J. M., Acebes-Doria, A., Blaauw, B., Kheirodin, A., Pandey, S., Lennon, K., Kaldor, A. D., Toledo, P. F. S., & Grabarczyk, E. E. (2021). Identifying Molecular-Based Trophic Interactions as a Resource for Advanced Integrated Pest Management. Insects, 12(4), 358. https://doi.org/10.3390/insects12040358





