Chromosome Unipolar Division and Low Expression of Tws May Cause Parthenogenesis of Rice Water Weevil (Lissorhoptrus oryzophilus Kuschel)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Rice Water Weevil and Fly Stocks
2.2. Immunostaining
2.3. Total RNA Extraction and Reverse Transcription
2.4. Gene Cloning and Q-PCR
2.5. Statistical Analysis
3. Results
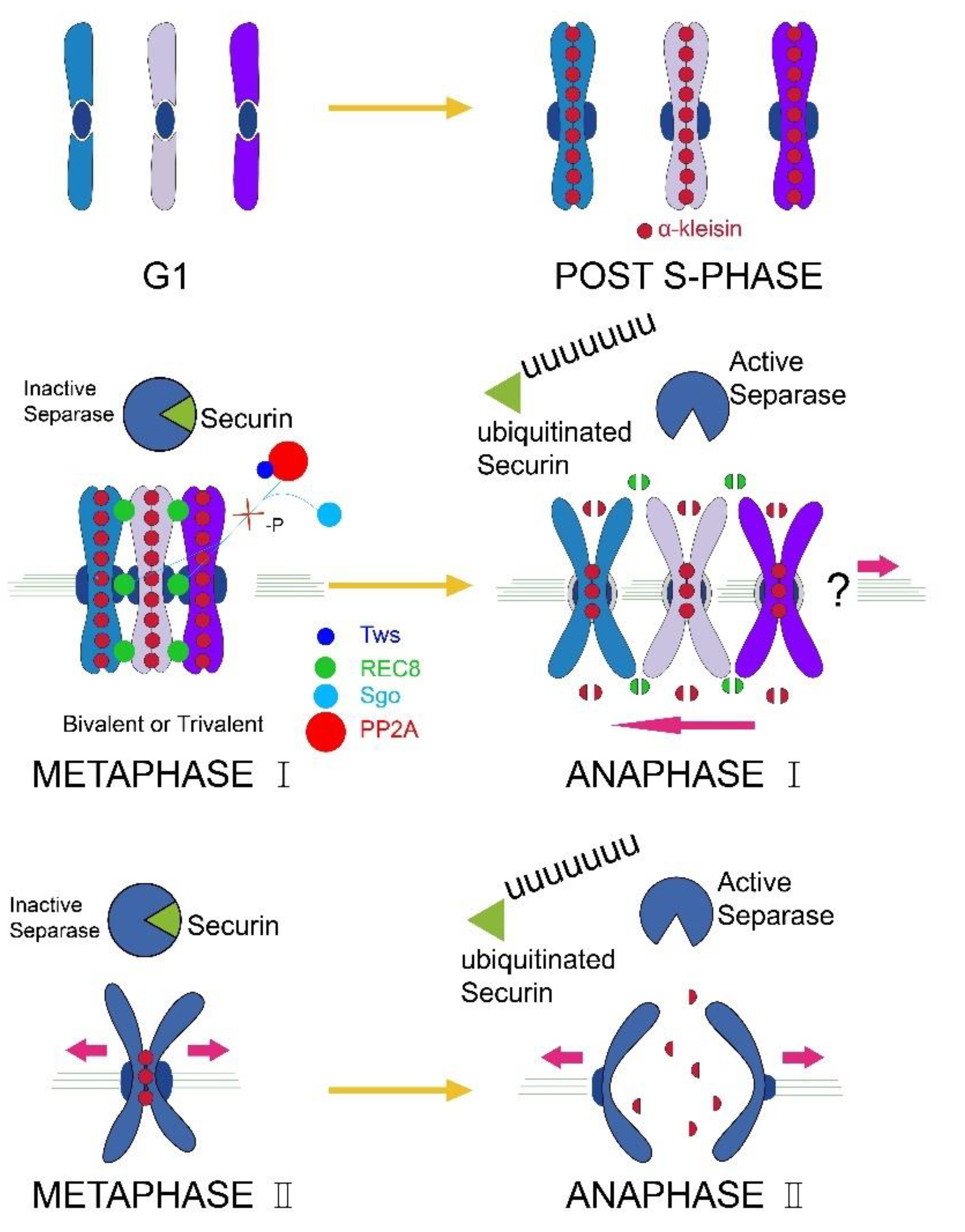
3.1. The Chromosomes of Parthenogenetic RWW Segregate at One Pole of the Meiotic Spindle during the Meiosis I Anaphase
3.2. Tws and REC8 Expression Levels
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kearney, M. Hybridization, glaciation and geographical parthenogenesis. Trends Ecol. Evol. 2005, 20, 495–502. [Google Scholar] [CrossRef]
- Vershinina, A.O.; Kuznetsova, V.G. Parthenogenesis in Hexapoda: Entognatha and non-holometabolous insects. J. Zool. Syst. Evol. Res. 2016, 54, 257–268. [Google Scholar] [CrossRef]
- Gokhman, V.E.; Kuznetsova, V.G. Parthenogenesis in Hexapoda: Holometabolous insects. J. Zool. Syst. Evol. Res. 2017, 56, 23–34. [Google Scholar] [CrossRef]
- Bowling, C.C. Rice Water Weevil Resistance to Aldrin in Texas. J. Econ. Èntomol. 1968, 61, 1027–1030. [Google Scholar] [CrossRef]
- Lange, W.; Grigarick, A. Rice water weevil: Beetle pest in rice growing areas of southern states discovered in California. Calif. Agric. 1959, 13, 10–11. [Google Scholar]
- Takenouchi, Y. A chromosome study of the parthenogenetic rice water weevil, Lissorhoptrus oryzophilus Kuschel (Coleoptera: Curculionidae), in Japan. Cell. Mol. Life Sci. 1978, 34, 444–445. [Google Scholar] [CrossRef]
- Huang, Y.; Ao, Y.; Jiang, M. Reproductive Plasticity of an Invasive Insect Pest, Rice Water Weevil (Coleoptera: Curculionidae). J. Econ. Èntomol. 2017, 110, 2381–2387. [Google Scholar] [CrossRef] [PubMed]
- Giantsis, I.A.; Sierra, J.C.; Chaskopoulou, A. The distribution of the invasive pest, rice water weevil Lissorhoptrus oryzophilus Kuschel (Coleoptera: Curculionidae), is expanding in Europe: First record in the Balkans, confirmed by CO1 DNA barcoding. Phytoparasitica 2017, 45, 147–149. [Google Scholar] [CrossRef]
- Saito, T.; Hirai, K.; Way, M.O. The rice water weevil, Lissorhoptrus oryzophilus Kuschel (Coleoptera: Curculionidae). Appl. Èntomol. Zool. 2005, 40, 31–39. [Google Scholar] [CrossRef]
- Jiang, C. Taxonomy of Mecinini and Bagoini (Coleoptera: Curculionidae) and Spread of Rice Water Weevil in China. Ph.D. Thesis, University of Chinese Academy of Sciences, Beijing China, 2018. [Google Scholar]
- Yang, P. Geographical Parthenogenesis of Rice Water Weevil. Ph.D. Thesis, Zhejiang University, Hangzhou, China, 2008. [Google Scholar]
- Kirchheimer, B.; Wessely, J.; Gattringer, A.; Hülber, K.; Moser, D.; Schinkel, C.C.F.; Appelhans, M.; Klatt, S.; Caccianiga, M.; Dellinger, A.; et al. Reconstructing geographical parthenogenesis: Effects of niche differentiation and reproductive mode on Holocene range expansion of an alpine plant. Ecol. Lett. 2018, 21, 392–401. [Google Scholar] [CrossRef]
- Lee, C.-C.; Hsu, S.-F.; Yang, C.; Lin, C.-C. Thelytokous parthenogenesis in the exotic dacetine ant Strumigenys rogeri (Hymenoptera: Formicidae) in Taiwan. Èntomol. Sci. 2018, 21, 28–33. [Google Scholar] [CrossRef]
- Galis, F.; Van Alphen, J.J.M. Parthenogenesis and developmental constraints. Evol. Dev. 2020, 22, 205–217. [Google Scholar] [CrossRef]
- Stenberg, P.; Saura, A. Cytology of Asexual Animals. In Lost Sex; Schön, I., Martens, K., Dijk, P., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 63–74. [Google Scholar] [CrossRef]
- Adachi-Hagimori, T.; Miura, K.; Stouthamer, R. A new cytogenetic mechanism for bacterial endosymbiont-induced parthenogenesis in Hymenoptera. Proc. R. Soc. B Biol. Sci. 2008, 275, 2667–2673. [Google Scholar] [CrossRef]
- Lynch, M.; Seyfert, A.; Eads, B.; Williams, E. Localization of the Genetic Determinants of Meiosis Suppression in Daphnia pulex. Genetics 2008, 180, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Innes, D.J.; Hebert, P.D.N. The Origin and Genetic Basis of Obligate Parthenogenesis in Daphnia pulex. Evolution 1988, 42, 1024. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Daimon, T. First molecular genetic evidence for automictic parthenogenesis in cockroaches. Insect Sci. 2019, 26, 649–655. [Google Scholar] [CrossRef]
- Eisman, R.C.; Kaufman, T.C. Cytological investigation of the mechanism of parthenogenesis in Drosophila mercatorum. Fly 2007, 1, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, K.; Fujimoto, M.; Goka, K. Sexual and asexual colony foundation and the mechanism of facultative parthenogenesis in the termite Reticulitermes speratus (Isoptera, Rhinotermitidae). Insectes Sociaux 2004, 51, 325–332. [Google Scholar] [CrossRef]
- Alavi, Y.; Van Rooyen, A.; Elgar, M.A.; Jones, T.M.; Weeks, A.R. Novel microsatellite markers suggest the mechanism of parthenogenesis in Extatosoma tiaratum is automixis with terminal fusion. Insect Sci. 2018, 25, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-C.; Ting, C.-T.; Chang, C.-H.; Fang, S.; Chang, H.-Y. The Persistence of Facultative Parthenogenesis in Drosophila albomicans. PLoS ONE 2014, 9, e113275. [Google Scholar] [CrossRef] [PubMed]
- Legay, J.M. Morphogenesis of the silkworm egg. J. Embryol. Exp. Morphol. 1976, 36, 13–18. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Zhang, H.; Gao, Y.-J.; Yan, L.-L.; Yu, X.-Y.; Yang, Y.-H.; Xu, W.-Y.; Pu, C.-X.; Sun, Y. Protein Phosphatase 2A B’α and B’β Protect Centromeric Cohesion during Meiosis I. Plant Physiol. 2019, 179, 1556–1568. [Google Scholar] [CrossRef] [PubMed]
- Petronczki, M.; Siomos, M.F.; Nasmyth, K. Un Ménage à Quatre: The Molecular Biology of Chromosome Segregation in Meiosis. Cell 2003, 112, 423–440. [Google Scholar] [CrossRef]
- Urban, E.; Nagarkar-Jaiswal, S.; Lehner, C.F.; Heidmann, S.K. The Cohesin Subunit Rad21 Is Required for Synaptonemal Complex Maintenance, but Not Sister Chromatid Cohesion, during Drosophila Female Meiosis. PLoS Genet. 2014, 10, e1004540. [Google Scholar] [CrossRef] [PubMed]
- Rieder, C.L.; Cole, R. Chromatid cohesion during mitosis: Lessons from meiosis. J. Cell Sci. 1999, 112, 2607–2613. [Google Scholar]
- Watanabe, Y.; Nurse, P. Cohesin Rec8 is required for reductional chromosome segregation at meiosis. Nature 1999, 400, 461–464. [Google Scholar] [CrossRef]
- Schleiffer, A.; Kaitna, S.; Maurer-Stroh, S.; Glotzer, M.; Nasmyth, K.; Eisenhaber, F. Kleisins: A Superfamily of Bacterial and Eukaryotic SMC Protein Partners. Mol. Cell 2003, 11, 571–575. [Google Scholar] [CrossRef]
- Buonomo, S.B.; Clyne, R.K.; Fuchs, J.; Loidl, J.; Uhlmann, F.; Nasmyth, K. Disjunction of Homologous Chromosomes in Meiosis I Depends on Proteolytic Cleavage of the Meiotic Cohesin Rec8 by Separin. Cell 2000, 103, 387–398. [Google Scholar] [CrossRef]
- Kim, L.-H.; Hong, S.-T.; Choi, K.-W. Protein phosphatase 2A interacts with Verthandi/Rad21 to regulate mitosis and organ development in Drosophila. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Kitajima, T.S.; Sakuno, T.; Ishiguro, K.-I.; Iemura, S.-I.; Natsume, T.; Kawashima, S.A.; Watanabe, Y. Shugoshin collaborates with protein phosphatase 2A to protect cohesin. Nat. Cell Biol. 2006, 441, 46–52. [Google Scholar] [CrossRef]
- Riedel, C.G.; Katis, V.L.; Katou, Y.; Mori, S.; Itoh, T.; Helmhart, W.; Gálová, M.; Petronczki, M.; Gregan, J.; Cetin, B.; et al. Protein phosphatase 2A protects centromeric sister chromatid cohesion during meiosis I. Nat. Cell Biol. 2006, 441, 53–61. [Google Scholar] [CrossRef]
- Horner, V.L.; Wolfner, M.F. Mechanical stimulation by osmotic and hydrostatic pressure activates Drosophila oocytes in vitro in a calcium-dependent manner. Dev. Biol. 2008, 316, 100–109. [Google Scholar] [CrossRef]
- Loidl, J. Meiotic chromosome pairing in triploid and tetraploid Saccharomyces cerevisiae. Genetics 1995, 139, 1511–1520. [Google Scholar] [CrossRef]
- Köhler, C.; Scheid, O.M.; Erilova, A. The impact of the triploid block on the origin and evolution of polyploid plants. Trends Genet. 2010, 26, 142–148. [Google Scholar] [CrossRef]
- Khanday, I.; Skinner, D.; Yang, B.; Mercier, R.; Sundaresan, V. A male-expressed rice embryogenic trigger redirected for asexual propagation through seeds. Nat. Cell Biol. 2019, 565, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Eads, B.D.; Tsuchiya, D.; Andrews, J.; Lynch, M.; Zolan, M.E. The spread of a transposon insertion in Rec8 is associated with obligate asexuality in Daphnia. Proc. Natl. Acad. Sci. USA 2012, 109, 858–863. [Google Scholar] [CrossRef]
- Yagound, B.; Dogantzis, K.A.; Zayed, A.; Lim, J.; Broekhuyse, P.; Remnant, E.J.; Beekman, M.; Allsopp, M.H.; Aamidor, S.E.; Dim, O.; et al. A Single Gene Causes Thelytokous Parthenogenesis, the Defining Feature of the Cape Honeybee Apis mellifera capensis. Curr. Biol. 2020, 30, 2248–2259.e6. [Google Scholar] [CrossRef]
- Matsuo, M.; Onuma, T.A.; Omotezako, T.; Nishida, H. Protein phosphatase 2A is essential to maintain meiotic arrest, and to prevent Ca2+ burst at spawning and eventual parthenogenesis in the larvacean Oikopleura dioica. Dev. Biol. 2020, 460, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Khetani, R.S.; Bickel, S.E. Regulation of meiotic cohesion and chromosome core morphogenesis during pachytene in Drosophila oocytes. J. Cell Sci. 2007, 120, 3123–3137. [Google Scholar] [CrossRef] [PubMed]
- Demarco, R.S.; Eikenes, Å.H.; Haglund, K.; Jones, D.L. Investigating spermatogenesis in Drosophila melanogaster. Methods 2014, 68, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, M.; Matsuzaki, M. Ovarian structure and oogenesis in the rice water weevil, Lissorphoptrus oryzophilus (Coleoptera, Curculionidae). Jpn. J. Enotomol. 1989, 57, 666–680. [Google Scholar]
- Büning, J. The trophic tissue of telotrophic ovarioles in polyphage coleoptera. Zoomorphology 1979, 93, 33–50. [Google Scholar] [CrossRef]
- Huebner, E.; Anderson, E. A cytological study of the ovary ofRhodnius prolixus. II. Oocyte differentiation. J. Morphol. 1972, 137, 385–415. [Google Scholar] [CrossRef] [PubMed]
- Blackman, R. Early development of the parthenogenetic egg in three species of aphids (homoptera: Aphididae). Int. J. Insect Morphol. Embryol. 1978, 7, 33–44. [Google Scholar] [CrossRef]
- Wilson, A.C.C.; Sunnucks, P.; Hales, D.F. SHORT PAPER Random loss of X chromosome at male determination in an aphid, Sitobion near fragariae, detected using an X-linked polymorphic microsatellite marker. Genet. Res. 1997, 69, 233–236. [Google Scholar] [CrossRef]
- Stevens, N.M. A study of the germ cells of Aphis rosæ and Aphis œnotheræ. J. Exp. Zool. 1905, 2, 313–333. [Google Scholar] [CrossRef][Green Version]
- Stevens, N.M. Studies on the Germ Cell of Aphids (No. 51); Carnegie: Washington, DC, USA, 1906. [Google Scholar] [CrossRef]
- Morgan, T.H. A biological and cytological study of sex determination in phylloxerans and aphids. Dev. Genes Evol. 1910, 29, 201–203. [Google Scholar] [CrossRef]
- Morgan, T.H. Sex determination and parthenogenesis in phylloxerans and aphids. Science 1909, 29, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, D.G.; Abdelhady, A.; Stern, D.L. Gene Expression Analysis of Parthenogenetic Embryonic Development of the Pea Aphid, Acyrthosiphon pisum, Suggests That Aphid Parthenogenesis Evolved from Meiotic Oogenesis. PLoS ONE 2014, 9, e115099. [Google Scholar] [CrossRef]
- Astaurov, B.L. Artificial parthenogenesis and experimental polyploidy in silkworm. J. Sericult. Sci. Jpn. 1967, 36, 277–285. [Google Scholar] [CrossRef]
- Astaurov, B.L. Experimental alterations of the developmental cytogenetic mechanisms in mulberry silkworms: Artificial parthenogenesis, polyploidy, gynogenesis, and androgenesis. In Advances in Morphogenesis; Abercrombie, M., Brachet, J., Eds.; Elsevier: Amsterdam, The Netherlands, 1967; Volume 6, pp. 199–257. [Google Scholar] [CrossRef]
- Grosmaire, M.; Launay, C.; Siegwald, M.; Brugière, T.; Estrada-Virrueta, L.; Berger, D.; Burny, C.; Modolo, L.; Blaxter, M.; Meister, P.; et al. Males as somatic investment in a parthenogenetic nematode. Science 2019, 363, 1210–1213. [Google Scholar] [CrossRef] [PubMed]
- Nokkala, C.; Kuznetsova, V.G.; Nokkala, S. Rare diploid females coexist with rare males: A novel finding in triploid parthenogenetic populations in the psyllid Cacopsylla myrtilli (W. Wagner, 1947) (Hemiptera, Psylloidea) in northern Europe. Genetic 2015, 143, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Nokkala, C.; Kuznetsova, V.G.; Nokkala, S. Meiosis in rare males in parthenogenetic Cacopsylla myrtilli (Wagner, 1947) (Hemiptera, Psyllidae) populations from northern Europe. Comp. Cytogenet. 2013, 7, 241–251. [Google Scholar] [CrossRef][Green Version]
- Nokkala, S.; Kuznetsova, V.G.; Nokkala, C. Characteristics of parthenogenesis in Cacopsylla ledi (Flor, 1861) (Hemiptera, Sternorryncha, Psylloidea): Cytological and molecular approaches. Comp. Cytogenet. 2017, 11, 807–817. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Peters, N.C.; Berg, C.A. In Vitro Culturing and Live Imaging of Drosophila Egg Chambers: A History and Adaptable Method. Breast Cancer 2016, 1457, 35–68. [Google Scholar] [CrossRef]
- Leonard, A.C.; Grimwade, J.E. Chromosome Replication and Segregation. Bacteriol Rev. 2019, 30, 3–32. [Google Scholar] [CrossRef]
- Nicholson, J.M.; Cimini, D. How Mitotic Errors Contribute to Karyotypic Diversity in Cancer. Adv. Cancer Res. 2011, 112, 43–75. [Google Scholar] [CrossRef]
- Cho, U.S.; Xu, W. Crystal structure of a protein phosphatase 2A heterotrimeric holoenzyme. Nat. Cell Biol. 2006, 445, 53–57. [Google Scholar] [CrossRef]
- Mayer-Jaekel, R.E.; Ohkura, H.; Gomes, R.; Sunkel, C.E.; Baumgärtner, S.; Hemmings, B.A.; Glover, D.M. The 55 kd regulatory subunit of Drosophila protein phosphatase 2A is required for anaphase. Cell 1993, 72, 621–633. [Google Scholar] [CrossRef]
- Snaith, H.A.; Armstrong, C.G.; Guo, Y.; Kaiser, K.; Cohen, P.T. Deficiency of protein phosphatase 2A uncouples the nucle-ar and centrosome cycles and prevents attachment of microtubules to the kinetochore in Drosophila microtubule star (mts) embryos. J. Cell Sci. 1996, 109, 3001–3012. [Google Scholar]
- Mehsen, H.; Boudreau, V.; Garrido, D.; Bourouh, M.; Larouche, M.; Maddox, P.S.; Swan, A.; Archambault, V. PP2A-B55 promotes nuclear envelope reformation after mitosis in Drosophila. J. Cell Biol. 2018, 217, 4106–4123. [Google Scholar] [CrossRef]
- Uemura, T.; Shiomi, K.; Togashi, S.; Takeichi, M. Mutation of twins encoding a regulator of protein phosphatase 2A leads to pattern duplication in Drosophila imaginal discs. Genes Dev. 1993, 7, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, R.; Makhijani, K.; Rao, P.R.; Shashidhara, L.S. Drosophila Twins regulates Armadillo levels in response to Wg/Wnt signal. Development 2004, 131, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, C.W.; Klebba, J.E.; Buster, D.W.; Rogers, G.C. The Protein Phosphatase 2A regulatory subunit Twins stabilizes Plk4 to induce centriole amplification. J. Cell Biol. 2011, 195, 231–243. [Google Scholar] [CrossRef] [PubMed]
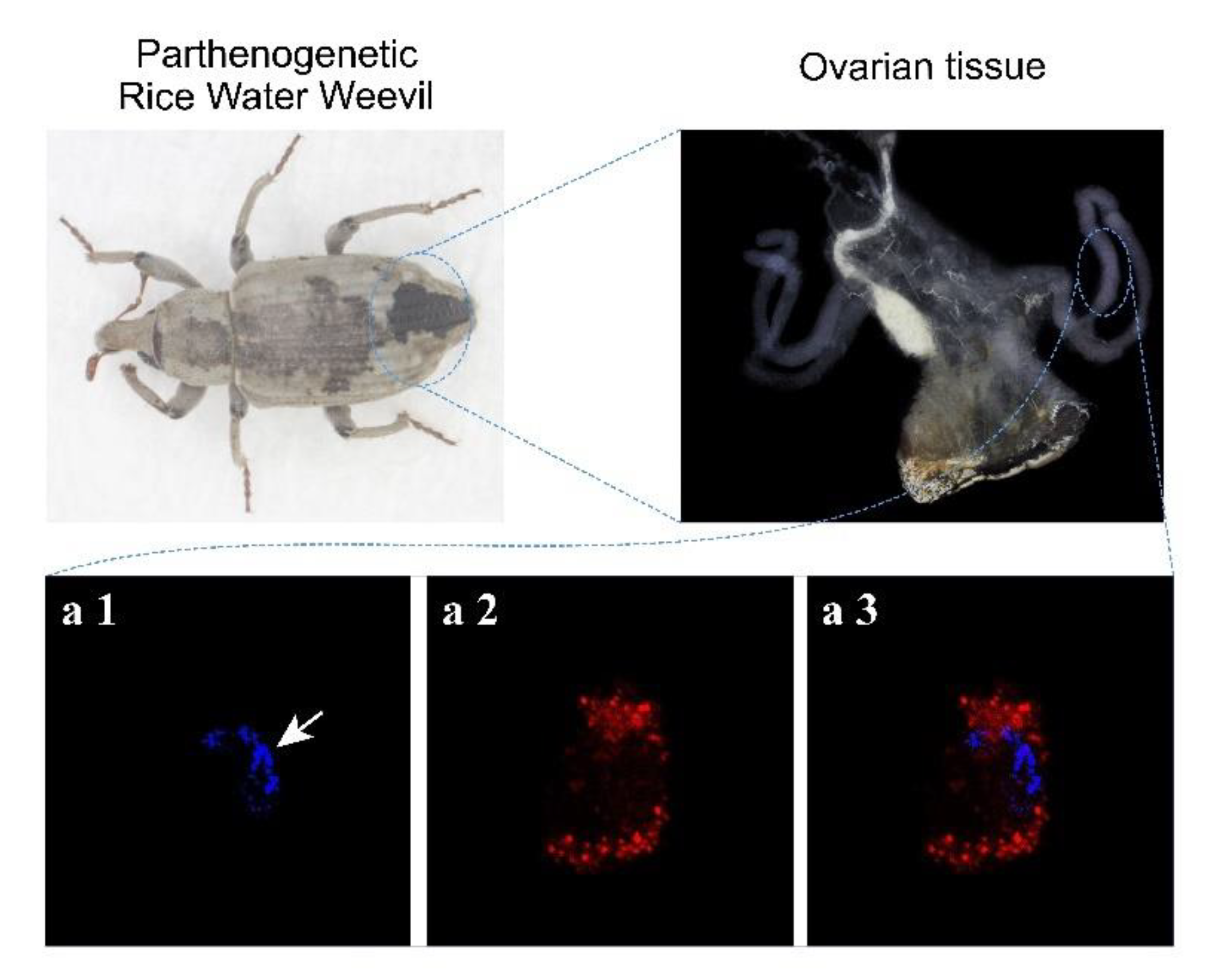
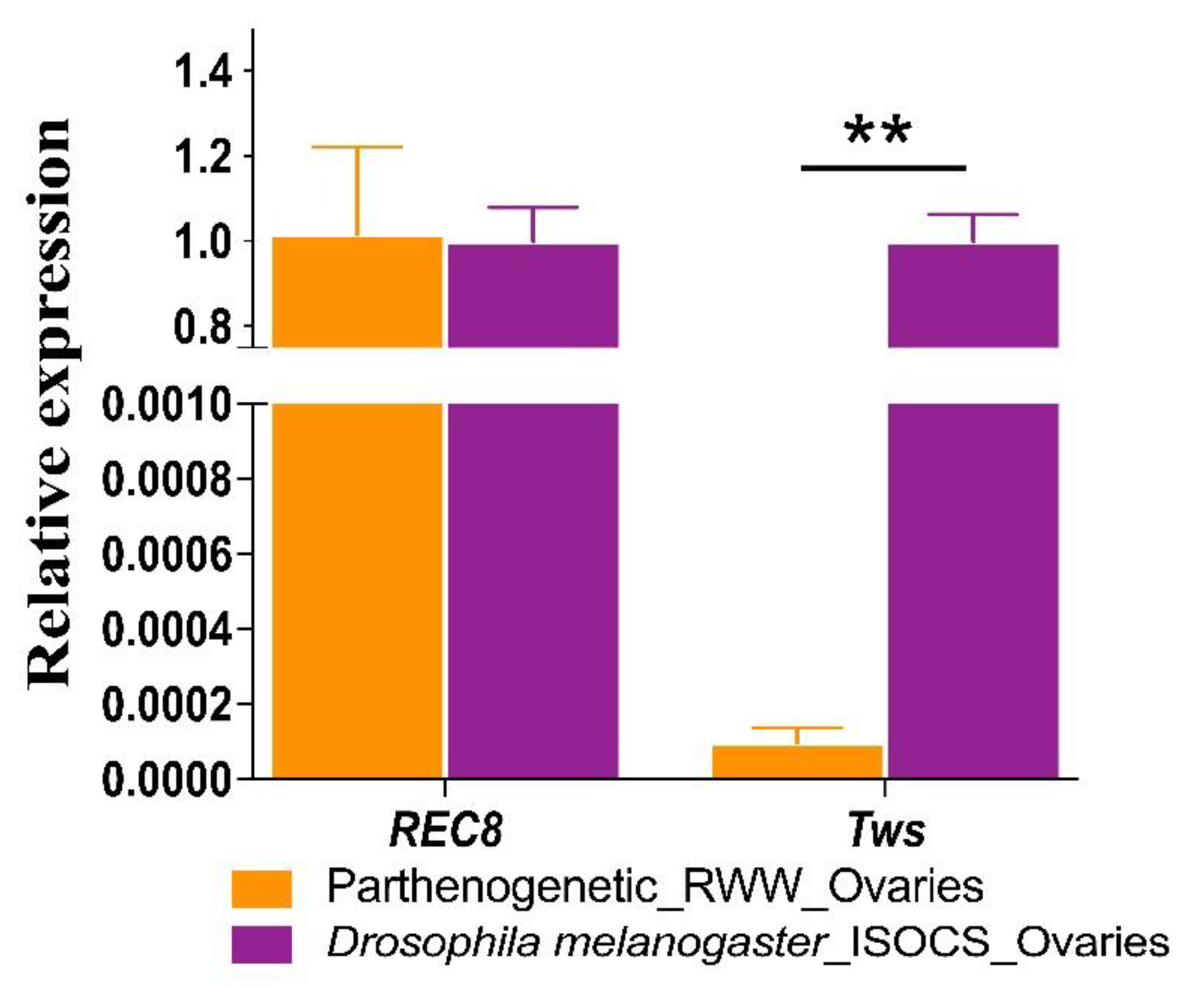
| Species | Relative Expression | |
|---|---|---|
| REC8 | Tws | |
| Lissorhoptrus oryzophilus-P 1 | 1.02 ± 0.20 | 9.70 × 10−5 ± 0.4 × 10−5 |
| Drosophila melanogaster | 1.00 ± 0.08 | 1.00 ± 0.0627 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, P.; Yang, F.; Ma, Z.; Zhang, R. Chromosome Unipolar Division and Low Expression of Tws May Cause Parthenogenesis of Rice Water Weevil (Lissorhoptrus oryzophilus Kuschel). Insects 2021, 12, 278. https://doi.org/10.3390/insects12040278
Wang P, Yang F, Ma Z, Zhang R. Chromosome Unipolar Division and Low Expression of Tws May Cause Parthenogenesis of Rice Water Weevil (Lissorhoptrus oryzophilus Kuschel). Insects. 2021; 12(4):278. https://doi.org/10.3390/insects12040278
Chicago/Turabian StyleWang, Pengcheng, Fangyuan Yang, Zhuo Ma, and Runzhi Zhang. 2021. "Chromosome Unipolar Division and Low Expression of Tws May Cause Parthenogenesis of Rice Water Weevil (Lissorhoptrus oryzophilus Kuschel)" Insects 12, no. 4: 278. https://doi.org/10.3390/insects12040278
APA StyleWang, P., Yang, F., Ma, Z., & Zhang, R. (2021). Chromosome Unipolar Division and Low Expression of Tws May Cause Parthenogenesis of Rice Water Weevil (Lissorhoptrus oryzophilus Kuschel). Insects, 12(4), 278. https://doi.org/10.3390/insects12040278








