Body Size and Behavioural Plasticity Interact to Influence the Performance of Free-Foraging Bumble Bee Colonies
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
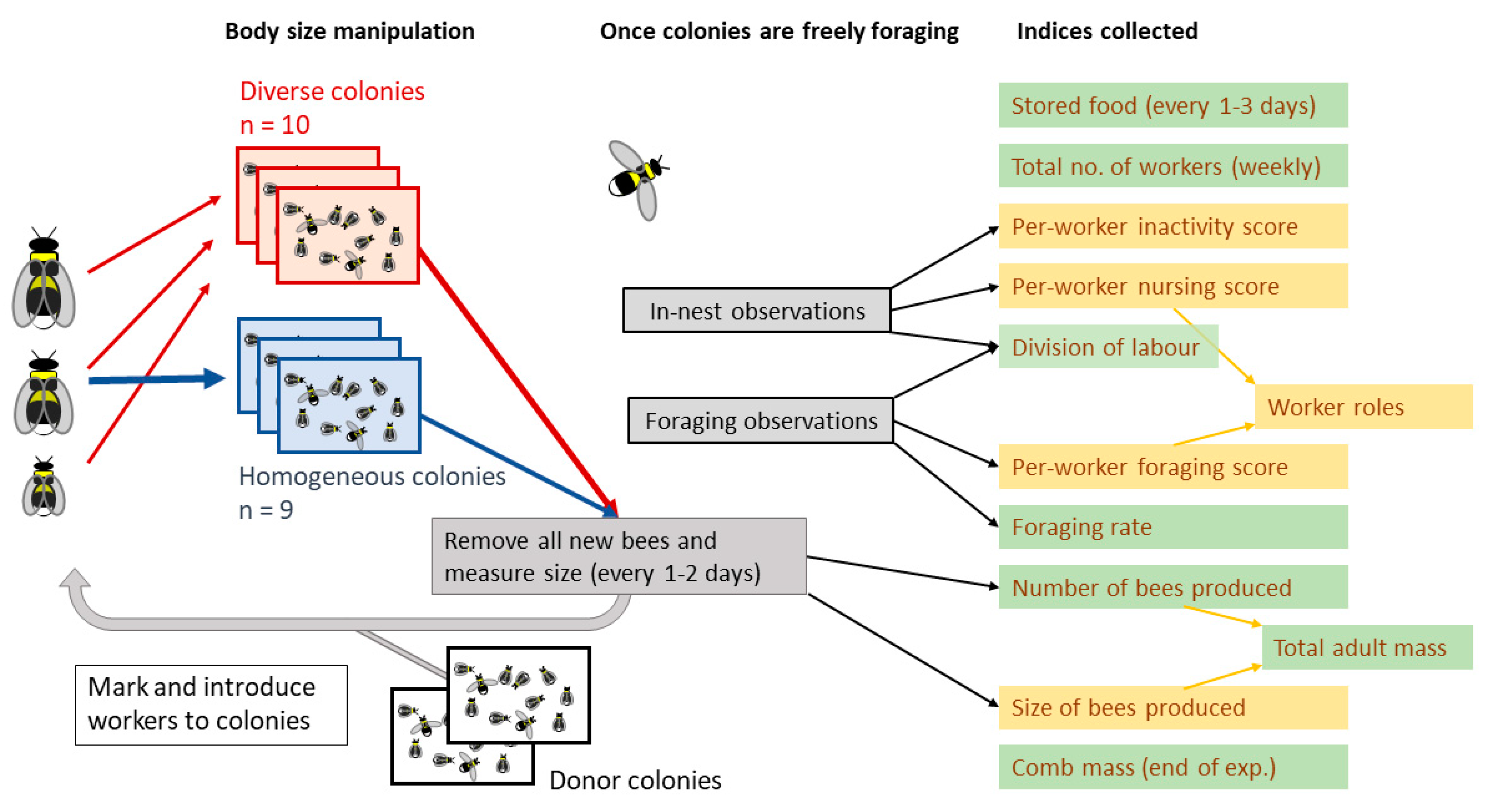
2.1. Colony Maintenance and Treatment
2.2. Behavioural Observations
2.3. Measuring Colony Performance
2.4. Statistical Analyses and Hypothesis Testing
2.4.1. Comparing Colony Performance
2.4.2. Comparing Responses to Reduced Size Distribution
2.4.3. Comparing across Trials
3. Results
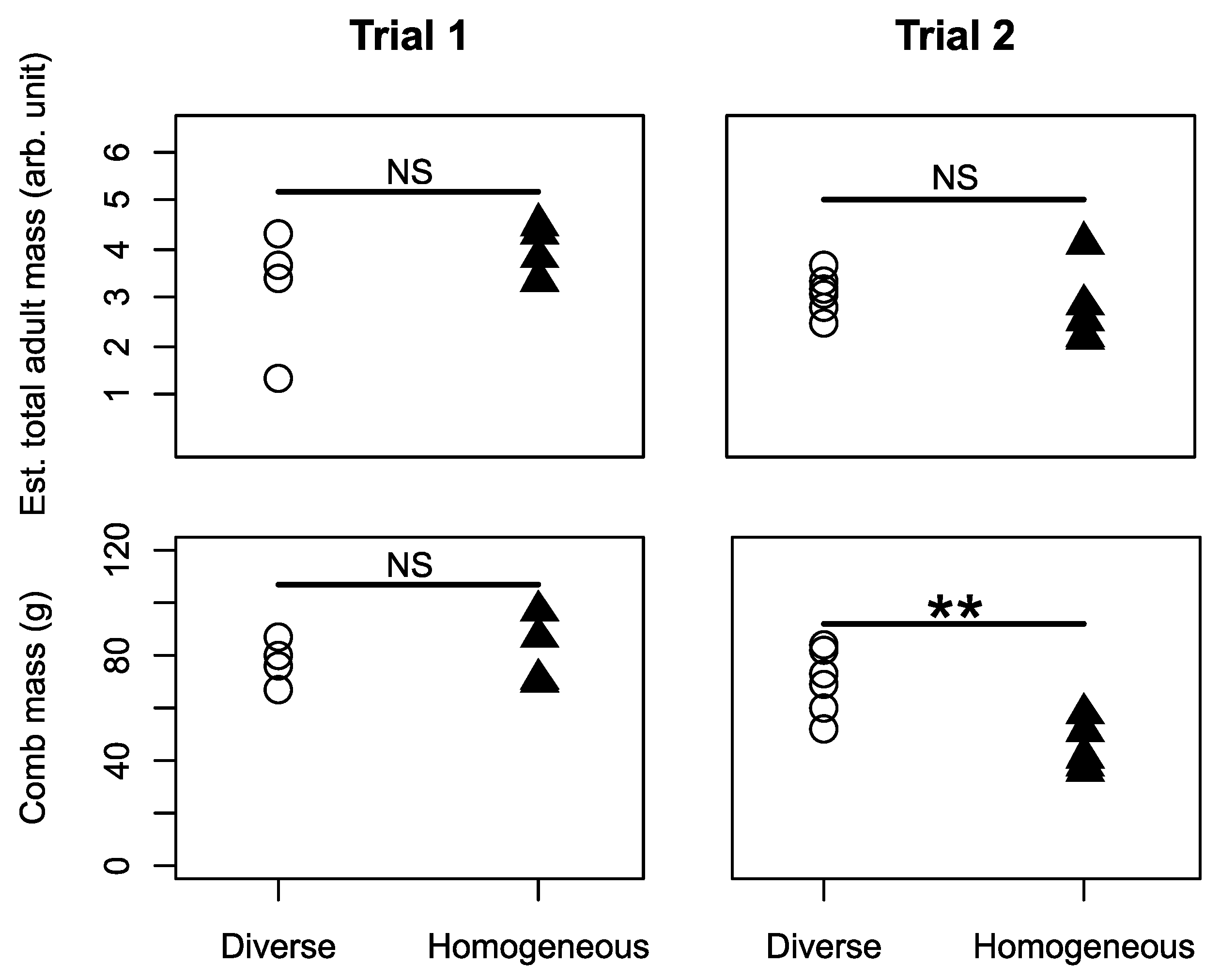
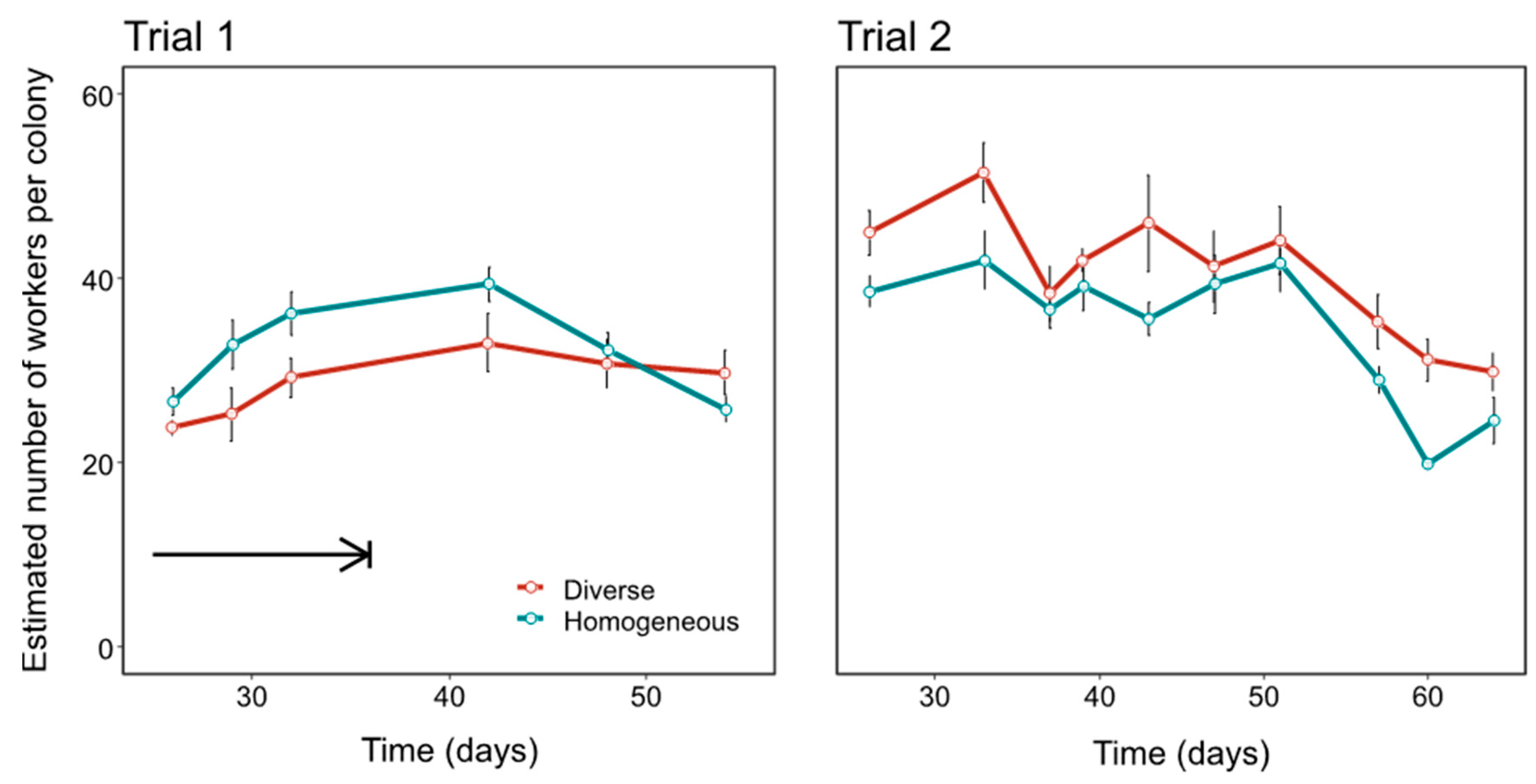
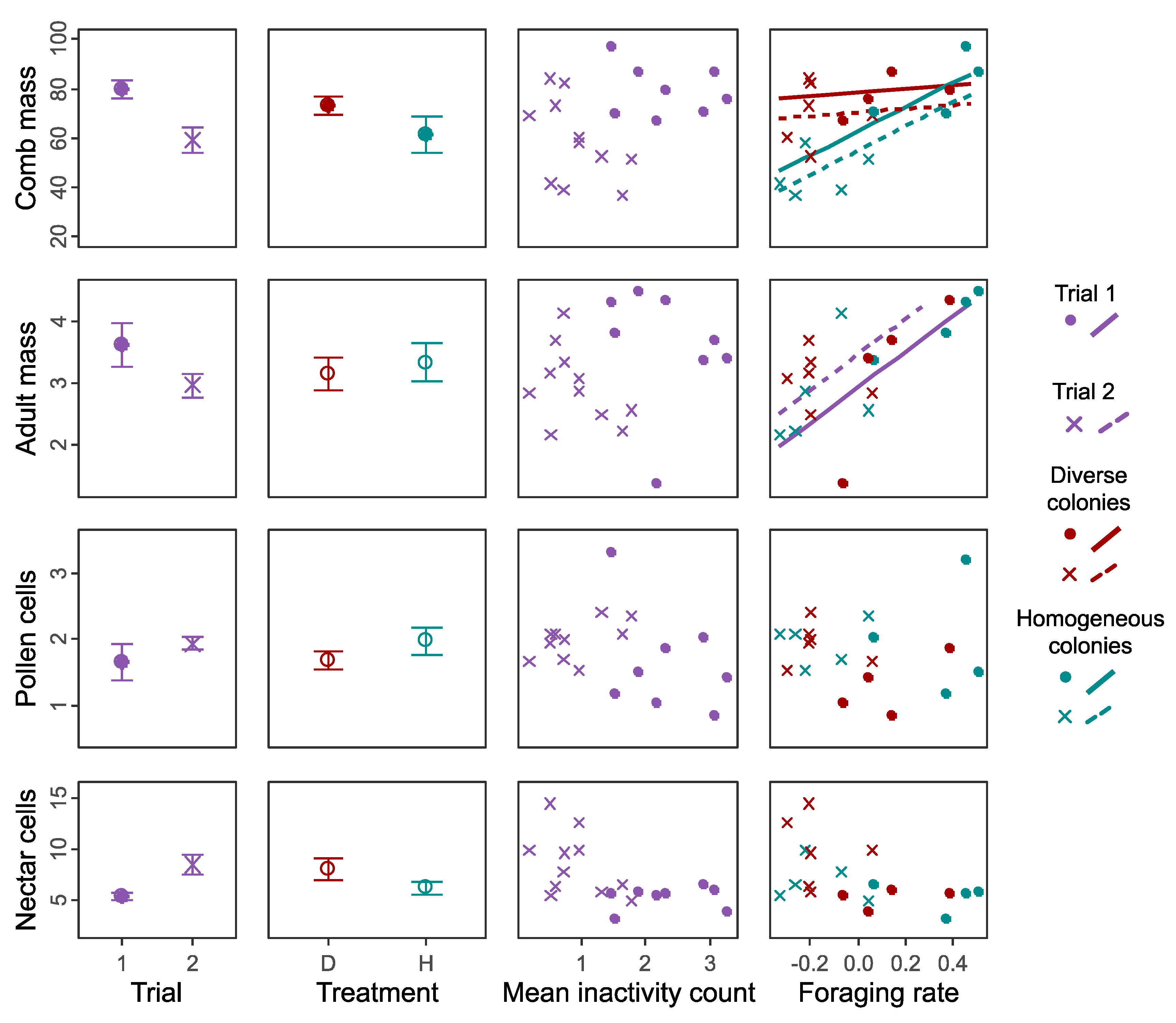
3.1. Colony Performance
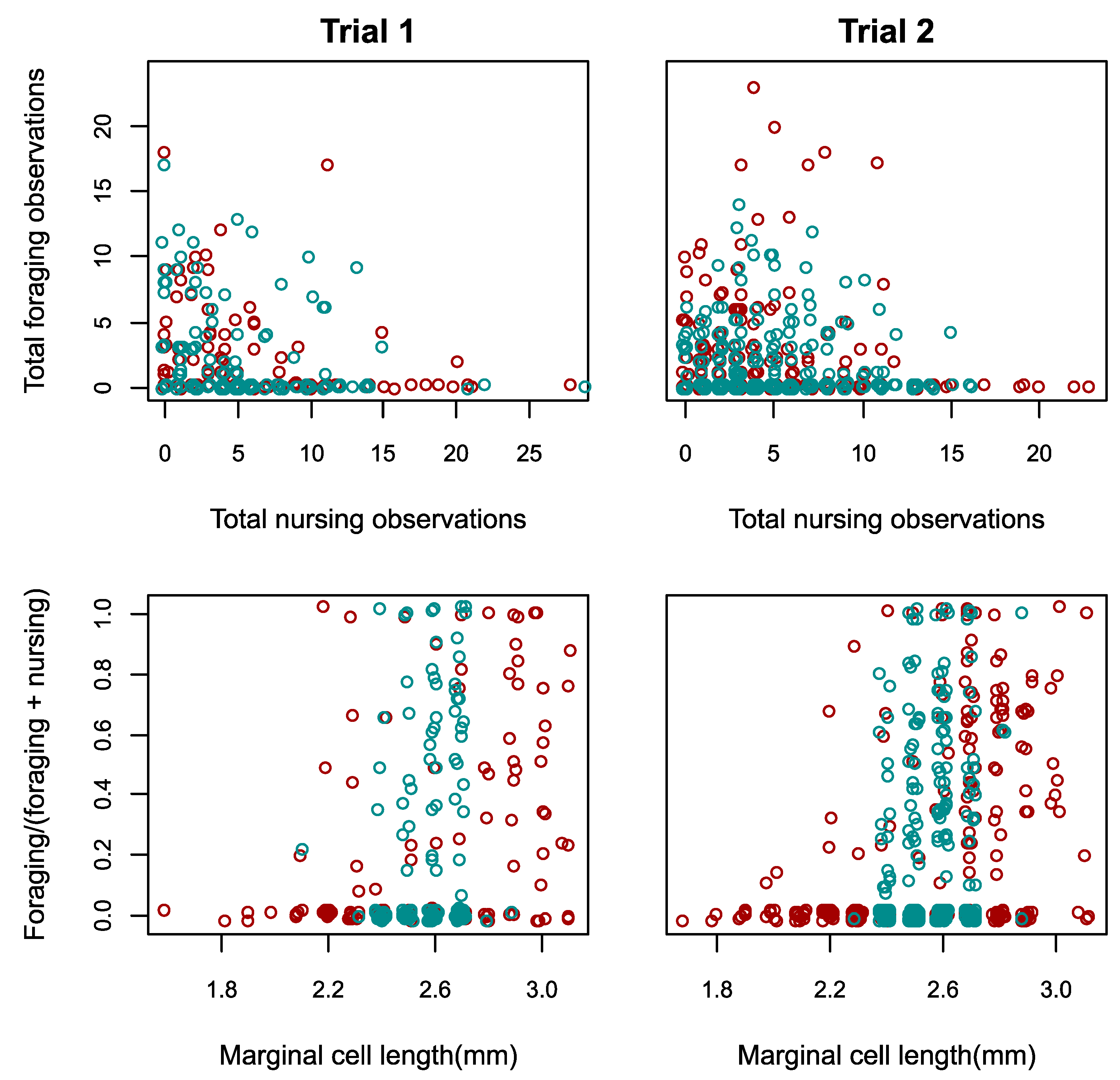
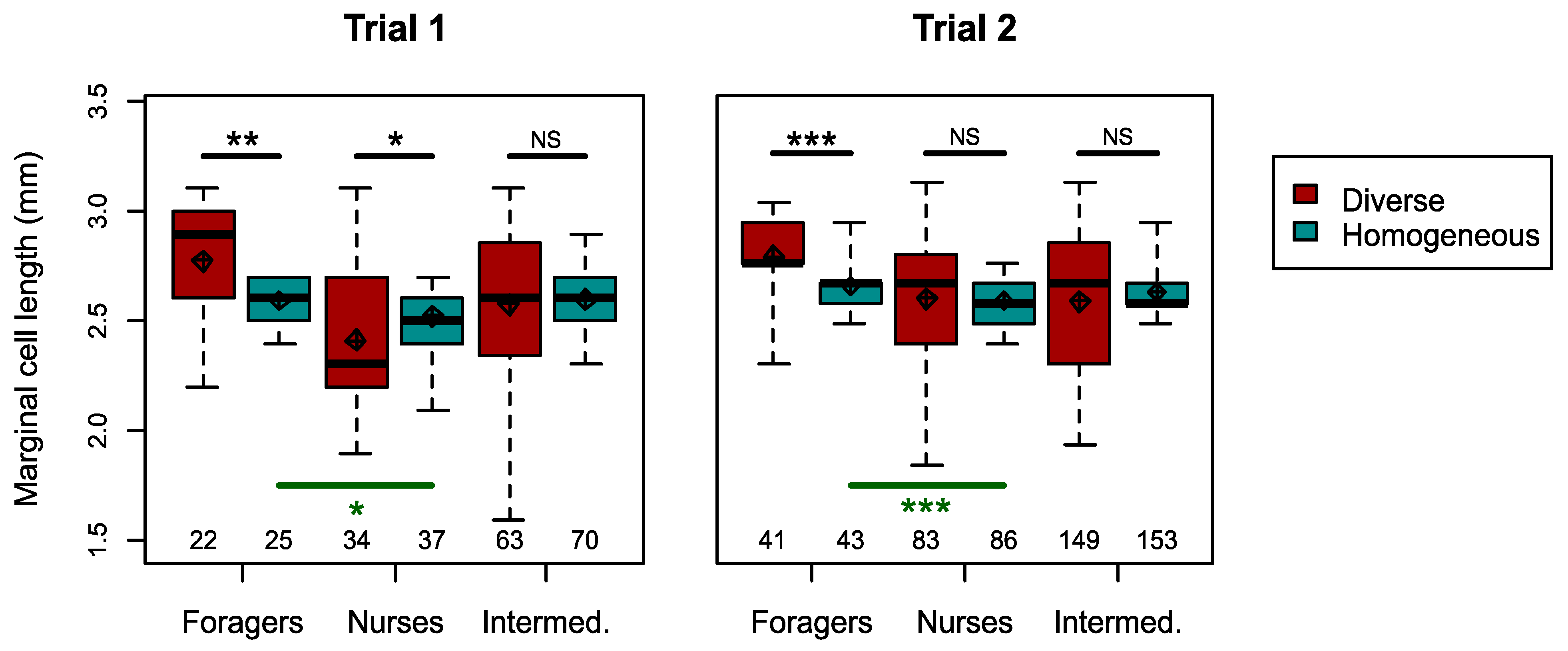
3.2. Responses to Reduced Size Distribution
3.3. Overall Effects on Performance across Both Trials
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beshers, S.N.; Fewell, J.H. Models of division of labor in social insects. Annu. Rev. Entomol. 2001, 46, 413–440. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.O. The Insect Societies; Belknap Press of Harvard University Press: Cambridge, MA, USA, 1971. [Google Scholar]
- Anderson, C.; McShea, D.W. Individual versus social complexity, with particular reference to ant colonies. Biol. Rev. 2001, 76, 211–237. [Google Scholar] [CrossRef] [PubMed]
- Goldsby, H.J.; Dornhaus, A.; Kerr, B.; Ofria, C. Task-switching costs promote the evolution of division of labor and shifts in individuality. Proc. Natl. Acad. Sci. USA 2012, 109, 13686–13691. [Google Scholar] [CrossRef]
- Oster, G.F.; Wilson, E.O. Caste and Ecology in the Social Insects; Princeton University Press: Princeton, NJ, USA, 1978; p. 371. [Google Scholar]
- West, S.A.; Fisher, R.M.; Gardner, A.; Kiers, E.T. Major evolutionary transitions in individuality. Proc. Natl. Acad. Sci. USA 2015, 112, 10112–10119. [Google Scholar] [CrossRef]
- Arnold, K.E.; Owens, I.P.F.; Goldizen, A.W. Division of labour within cooperatively breeding groups. Behaviour 2005, 142, 1577–1590. [Google Scholar] [CrossRef]
- Bennett, N.C.; Faulkes, C.G. African Mole Rats: Ecology and Eusociality; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Maynard Smith, J.; Szathmáry, E. The Major Transitions in Evolution; Oxford University Press: Oxford, UK, 1995; p. 346. [Google Scholar]
- Zhang, Z.R.; Claessen, D.; Rozen, D.E. Understanding microbial divisions of labor. Front. Microbiol. 2016, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, S.; Diner, D.; Holland, J.G.; Bloch, G.; Porfiri, M.; Nov, O. The influence of social information and self-expertise on emergent task allocation in virtual groups. Front. Ecol. Evol. 2018, 6, 16. [Google Scholar] [CrossRef]
- Smith, A. An Inquiry into the Nature and Causes of the Wealth of Nations; W. Strahan and T. Cadell: London, UK, 1776. [Google Scholar]
- Kennedy, P.; Baron, G.; Qiu, B.T.; Freitak, D.; Helantera, H.; Hunt, E.R.; Manfredini, F.; O’Shea-Wheller, T.; Patalano, S.; Pull, C.D.; et al. Deconstructing superorganisms and societies to address big questions in biology. Trends Ecol. Evol. 2017, 32, 861–872. [Google Scholar] [CrossRef]
- Ferguson-Gow, H.; Sumner, S.; Bourke, A.F.G.; Jones, K.E. Colony size predicts division of labour in attine ants. Proc. R. Soc. B Biol. Sci. 2014, 281. [Google Scholar] [CrossRef] [PubMed]
- Korb, J.; Hartfelder, K. Life history and development—A framework for understanding developmental plasticity in lower termites. Biol. Rev. 2008, 83, 295–313. [Google Scholar] [CrossRef]
- Johnson, B.R. Organization of work in the honeybee: A compromise between division of labour and behavioural flexibility. Proc. R. Soc. B Biol. Sci. 2003, 270, 147–152. [Google Scholar] [CrossRef]
- Moglich, M.; Holldobler, B. Social carrying behavior and division of labor during nest moving in ants. Psyche 1974, 81, 219–236. [Google Scholar] [CrossRef]
- Robinson, E.J.H.; Feinerman, O.; Franks, N.R. Flexible task allocation and the organization of work in ants. Proc. R. Soc. B Biol. Sci. 2009, 276, 4373–4380. [Google Scholar] [CrossRef]
- Tofts, C.; Franks, N.R. Doing the right thing—Ants, honeybees and naked mole-rats. Trends Ecol. Evol. 1992, 7, 346–349. [Google Scholar] [CrossRef]
- Bourke, A.F.G. Colony size, social complexity and reproductive conflict in social insects. J. Evol. Biol. 1999, 12, 245–257. [Google Scholar] [CrossRef]
- Page, R.E.; Erber, J.; Fondrk, M.K. The effect of genotype on response thresholds to sucrose and foraging behavior of honey bees (Apis mellifera L.). J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 1998, 182, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Weidenmüller, A. The control of nest climate in bumblebee (Bombus terrestris) colonies: Interindividual variability and self reinforcement in fanning response. Behav. Ecol. 2004, 15, 120–128. [Google Scholar] [CrossRef]
- Jeanson, R.; Weidenmüller, A. Interindividual variability in social insects—Proximate causes and ultimate consequences. Biol. Rev. 2014, 89, 671–687. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Vaamonde, C.; Koning, J.W.; Brown, R.M.; Jordan, W.C.; Bourke, A.F.G. Social parasitism by male-producing reproductive workers in a eusocial insect. Nature 2004, 430, 557–560. [Google Scholar] [CrossRef]
- Gill, R.J.; Ramos-Rodriguez, O.; Raine, N.E. Combined pesticide exposure severely affects individual- and colony-level traits in bees. Nature 2012, 491, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Shpigler, H.Y.; Siegel, A.J.; Huang, Z.Y.; Bloch, G. No effect of juvenile hormone on task performance in a bumblebee (Bombus terrestris) supports an evolutionary link between endocrine signaling and social complexity. Horm. Behav. 2016, 85, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Yerushalmi, S.; Bodenhaimer, S.; Bloch, G. Developmentally determined attenuation in circadian rhythms links chronobiology to social organization in bees. J. Exp. Biol. 2006, 209, 1044–1051. [Google Scholar] [CrossRef]
- Alford, D.V. Bumblebees; Davis-Poynter: London, UK, 1975. [Google Scholar]
- Brian, A.D. Division of labour and foraging in bombus agrorum fabricius. J. Anim. Ecol. 1952, 21, 223–240. [Google Scholar] [CrossRef]
- Cumber, R.A. The biology of humble-bees, with special reference to the production of the worker caste. Trans. R. Entomol. Soc. Lond. 1949, 100, 1–45. [Google Scholar] [CrossRef]
- Free, J.B. The division of labour within bumblebee colonies. Insectes Sociaux 1955, 2, 195–212. [Google Scholar] [CrossRef]
- Gardner, K.E.; Foster, R.L.; O’Donnell, S. Experimental analysis of worker division of labor in bumblebee nest thermoregulation (Bombus huntii, hymenoptera: Apidae). Behav. Ecol. Sociobiol. 2007, 61, 783–792. [Google Scholar] [CrossRef]
- Goulson, D.; Peat, J.; Stout, J.C.; Tucker, J.; Darvill, B.; Derwent, L.C.; Hughes, W.O.H. Can alloethism in workers of the bumblebee, Bombus terrestris, be explained in terms of foraging efficiency? Anim. Behav. 2002, 64, 123–130. [Google Scholar] [CrossRef]
- Chole, H.; Woodard, S.H.; Bloch, G. Body size variation in bees: Regulation, mechanisms, and relationship to social organization. Curr. Opin. Insect Sci. 2019, 35, 77–87. [Google Scholar] [CrossRef]
- Klein, S.; Pasquaretta, C.; Barron, A.B.; Devaud, J.-M.; Lihoreau, M. Inter-individual variability in the foraging behaviour of traplining bumblebees. Sci. Rep. 2017, 7, 4561. [Google Scholar] [CrossRef]
- Spaethe, J.; Weidenmüller, A. Size variation and foraging rate in bumblebees (Bombus terrestris). Insectes Sociaux 2002, 49, 142–146. [Google Scholar] [CrossRef]
- Kapustjanskij, A.; Streinzer, M.; Paulus, H.F.; Spaethe, J. Bigger is better: Implications of body size for flight ability under different light conditions and the evolution of alloethism in bumblebees. Funct. Ecol. 2007, 21, 1130–1136. [Google Scholar] [CrossRef]
- Spaethe, J.; Chittka, L. Interindividual variation of eye optics and single object resolution in bumblebees. J. Exp. Biol. 2003, 206, 3447–3453. [Google Scholar] [CrossRef] [PubMed]
- Mares, S.; Ash, L.; Gronenberg, W. Brain allometry in bumblebee and honey bee workers. Brain Behav. Evol. 2005, 66, 50–61. [Google Scholar] [CrossRef]
- Riveros, A.J.; Gronenberg, W. Brain allometry and neural plasticity in the bumblebee Bombus occidentalis. Brain Behav. Evol. 2010, 75, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Spaethe, J.; Brockmann, A.; Halbig, C.; Tautz, J. Size determines antennal sensitivity and behavioral threshold to odors in bumblebee workers. Naturwissenschaften 2007, 94, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.; Dov, A.; Fahrbach, S.E.; Bloch, G. Body size-related variation in pigment dispersing factor-immunoreactivity in the brain of the bumblebee Bombus terrestris (hymenoptera, apidae). J. Insect Physiol. 2009, 55, 479–487. [Google Scholar] [CrossRef]
- Merling, M.; Eisenmann, S.; Bloch, G. Body size but not age influences phototaxis in bumble bee (Bombus terrestris, L.) workers. Apidologie 2020, 51, 763–776. [Google Scholar] [CrossRef]
- Couvillon, M.J.; Dornhaus, A. Small worker bumble bees (Bombus impatiens) are hardier against starvation than their larger sisters. Insectes Sociaux 2010, 57, 193–197. [Google Scholar] [CrossRef]
- Shpigler, H.; Tamarkin, M.; Gruber, Y.; Poleg, M.; Siegel, A.J.; Bloch, G. Social influences on body size and developmental time in the bumblebee Bombus terrestris. Behav. Ecol. Sociobiol. 2013, 67, 1601–1612. [Google Scholar] [CrossRef]
- Holland, J.G.; Bloch, G. The complexity of social complexity: A quantitative multidimensional approach for studies of social organization. Am. Nat. 2020, 196, 525–540. [Google Scholar] [CrossRef]
- Goulson, D. Bumblebees: Behaviour, Ecology and Evolution, 2010 ed.; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Michener, C. The Social Behavior of the Bees; Belknap Press of Harvard University Press: Cambridge, MA, USA, 1974; p. 404. [Google Scholar]
- Jandt, J.M.; Dornhaus, A. Bumblebee response thresholds and body size: Does worker diversity increase colony performance? Anim. Behav. 2014, 87, 97–106. [Google Scholar] [CrossRef]
- Wills, B.D.; Powell, S.; Rivera, M.D.; Suarez, A.V. Correlates and consequences of worker polymorphism in ants. In Annual Review of Entomology; Berenbaum, M.R., Ed.; Annual Reviews; Palo Alto: Santa Clara, CA, USA, 2018; Volume 63, pp. 575–598. [Google Scholar]
- Billick, I. The relationship between the distribution of worker sizes and new worker production in the ant Formica neorufibarbis. Oecologia 2002, 132, 244–249. [Google Scholar] [CrossRef]
- Billick, I.; Carter, C. Testing the importance of the distribution of worker sizes to colony performance in the ant species Formica obscuripes Forel. Insectes Sociaux 2007, 54, 113–117. [Google Scholar] [CrossRef]
- Kelemen, E.P.; Davidowitz, G.; Dornhaus, A. Size variation does not act as insurance in bumble bees; instead, workers add weight in an unpredictable environment. Anim. Behav. 2020, 170, 99–109. [Google Scholar] [CrossRef]
- Elbgami, T.; Kunin, W.E.; Hughes, W.O.H.; Biesmeijer, J.C. The effect of proximity to a honeybee apiary on bumblebee colony fitness, development, and performance. Apidologie 2014, 45, 504–513. [Google Scholar] [CrossRef]
- Medler, J.T. Morphometric studies on bumble bees. Ann. Entomol. Soc. Am. 1962, 55, 212–218. [Google Scholar] [CrossRef]
- Porath, H.T.; Hazan, E.; Shpigler, H.; Cohen, M.; Band, M.; Ben-Shahar, Y.; Levanon, E.Y.; Eisenberg, E.; Bloch, G. Rna editing is abundant and correlates with task performance in a social bumblebee. Nat. Commun. 2019, 10, 14. [Google Scholar] [CrossRef] [PubMed]
- Holland, J.G.; Li, J.; Sovik, E.; Ben-Shahar, Y.; Hughes, M.E.; Bloch, G. Transcriptomic signatures of circadian plasticity in the social bumblebee Bombus terrestris. Unpublished.
- Shpigler, H.Y.; Herb, B.; Avidan-King, M.; Alkalay, S.; Cohen, M.; Zadeh, J.D.; Band, M.; Robinson, G.E.; Bloch, G. Task performance and queen presence influence brain gene expression in bumble bee workers. Unpublished.
- Gorelick, R.; Bertram, S.M. Quantifying division of labor: Borrowing tools from sociology, sociobiology, information theory, landscape ecology, and biogeography. Insectes Sociaux 2007, 54, 105–112. [Google Scholar] [CrossRef]
- Gorelick, R.; Bertram, S.M.; Killeen, P.R.; Fewell, J.H. Normalized mutual entropy in biology: Quantifying division of labor. Am. Nat. 2004, 164, 677–682. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Symonds, M.R.E.; Moussalli, A. A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using akaike’s information criterion. Behav. Ecol. Sociobiol. 2011, 65, 13–21. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing, 4.0.3; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Stoffel, M.A.; Nakagawa, S.; Schielzeth, H. Rptr: Repeatability estimation and variance decomposition by generalized linear mixed-effects models. Methods Ecol. Evol. 2017, 8, 1639–1644. [Google Scholar] [CrossRef]
- Chouvenc, T.; Basille, M.; Su, N.-Y. The production of soldiers and the maintenance of caste proportions delay the growth of termite incipient colonies. Insectes Sociaux 2015, 62, 23–29. [Google Scholar] [CrossRef]
- Passera, L.; Roncin, E.; Kaufmann, B.; Keller, L. Increased soldier production in ant colonies exposed to intraspecific competition. Nature 1996, 379, 630–631. [Google Scholar] [CrossRef]
- Charbonneau, D.; Sasaki, T.; Dornhaus, A. Who needs ‘lazy’ workers? Inactive workers act as a ‘reserve’ labor force replacing active workers, but inactive workers are not replaced when they are removed. PLoS ONE 2017, 12, e0184074. [Google Scholar] [CrossRef]
- Hasegawa, E.; Ishii, Y.; Tada, K.; Kobayashi, K.; Yoshimura, J. Lazy workers are necessary for long-term sustainability in insect societies. Sci. Rep. 2016, 6, 20846. [Google Scholar] [CrossRef]
- Crall, J.D.; Gravish, N.; Mountcastle, A.M.; Kocher, S.D.; Oppenheimer, R.L.; Pierce, N.E.; Combes, S.A. Spatial fidelity of workers predicts collective response to disturbance in a social insect. Nat. Commun. 2018, 9, 1201. [Google Scholar] [CrossRef]
- Pendrel, B.A.; Plowright, R.C. Larval feeding by adult bumble bee workers (hymenoptera: Apidae). Behav. Ecol. Sociobiol. 1981, 8, 71–76. [Google Scholar] [CrossRef]
- Couvillon, M.J.; Fitzpatrick, G.; Dornhaus, A. Ambient air temperature does not predict whether small or large workers forage in bumble bees (Bombus impatiens). Psyche 2010, 2010, 8. [Google Scholar] [CrossRef] [PubMed]
- Peat, J.; Darvill, B.; Ellis, J.; Goulson, D. Effects of climate on intra- and interspecific size variation in bumble-bees. Funct. Ecol. 2005, 19, 145–151. [Google Scholar] [CrossRef]
- Kerr, N.Z.; Crone, E.E.; Williams, N.M. Integrating vital rates explains optimal worker size for resource return by bumblebee workers. Funct. Ecol. 2019, 33, 467–478. [Google Scholar] [CrossRef]
- Heinrich, B. Bumblebee Economics; Harvard University Press: Cambridge, UK, 1979. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holland, J.G.; Nakayama, S.; Porfiri, M.; Nov, O.; Bloch, G. Body Size and Behavioural Plasticity Interact to Influence the Performance of Free-Foraging Bumble Bee Colonies. Insects 2021, 12, 236. https://doi.org/10.3390/insects12030236
Holland JG, Nakayama S, Porfiri M, Nov O, Bloch G. Body Size and Behavioural Plasticity Interact to Influence the Performance of Free-Foraging Bumble Bee Colonies. Insects. 2021; 12(3):236. https://doi.org/10.3390/insects12030236
Chicago/Turabian StyleHolland, Jacob G., Shinnosuke Nakayama, Maurizio Porfiri, Oded Nov, and Guy Bloch. 2021. "Body Size and Behavioural Plasticity Interact to Influence the Performance of Free-Foraging Bumble Bee Colonies" Insects 12, no. 3: 236. https://doi.org/10.3390/insects12030236
APA StyleHolland, J. G., Nakayama, S., Porfiri, M., Nov, O., & Bloch, G. (2021). Body Size and Behavioural Plasticity Interact to Influence the Performance of Free-Foraging Bumble Bee Colonies. Insects, 12(3), 236. https://doi.org/10.3390/insects12030236






