Organic Control Strategies for Use in IPM of Invertebrate Pests in Apple and Pear Orchards
Abstract
:Simple Summary
Abstract
1. Introduction
2. Cultural Control
2.1. Soil Health and Properties
2.2. Cover Crops
2.3. Variety
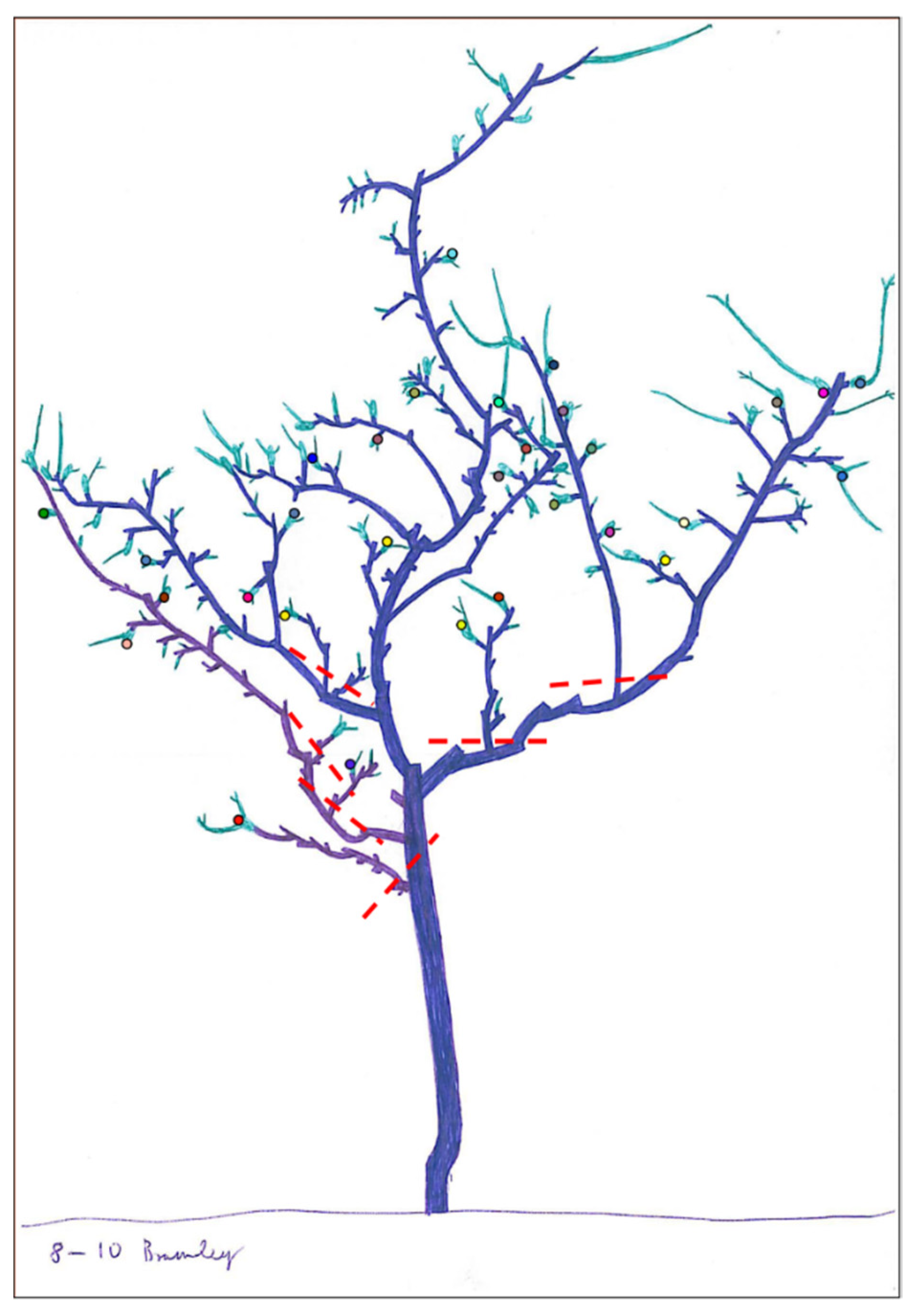
2.4. Canopy Maintenance
3. Biological Control
3.1. Natural Enemies
3.2. Introduced/Augmented Biological Control
3.3. Avoiding Practices Harmful to Beneficial Insects and Conservation Biological Control
3.4. Viruses
3.5. Entomopathogenic Fungi and Nematodes
3.6. Parasitoids
3.7. Semiochemicals
4. Physical Control
4.1. Netting and Barriers
4.2. Waste Removal
4.3. Particle Films
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Basha, M.B.; Mason, C.; Shamsudin, M.F.; Hussain, H.I.; Salem, M.A. Consumers attitude towards organic food. Procedia Econ. Financ. 2015, 31, 444–452. [Google Scholar] [CrossRef] [Green Version]
- Lesur-Dumoulin, C.; Malézieux, E.; Ben-Ari, T.; Langlais, C.; Makowski, D. Lower average yields but similar yield variability in organic versus conventional horticulture. A meta-analysis. Agron. Sustain. Dev. 2017, 37, 45. [Google Scholar] [CrossRef] [Green Version]
- Samnegård, U.; Alins, G.; Boreux, V.; Bosch, J.; García, D.; Happe, A.K.; Klein, A.M.; Miñarro, M.; Mody, K.; Porcel, M.; et al. Management trade-offs on ecosystem services in apple orchards across Europe: Direct and indirect effects of organic production. J. Appl. Ecol. 2018, 56, 802–811. [Google Scholar] [CrossRef] [Green Version]
- De Ponti, T.; Rijk, B.; Van Ittersum, M.K. The crop yield gap between organic and conventional agriculture. Agric. Syst. 2012, 108, 1–9. [Google Scholar] [CrossRef]
- Connor, D.J. Organically grown crops do not a cropping system make and nor can organic agriculture nearly feed the world. Field Crop. Res. 2013, 144, 145–147. [Google Scholar] [CrossRef]
- Raviv, M. Is organic horticulture sustainable? Chron. Hortic. 2010, 50, 7–14. [Google Scholar]
- Reganold, J.P.; Wachter, J.M. Organic agriculture in the twenty-first century. Nat. Plants 2016, 2, 15221. [Google Scholar] [CrossRef] [PubMed]
- Dib, H.; Sauphanor, B.; Capowiez, Y. Effect of management strategies on arthropod communities in the colonies of rosy apple aphid, Dysaphis plantaginea Passerini (Hemiptera: Aphididae) in south-eastern France. Agric. Ecosyst. Environ. 2016, 216, 203–206. [Google Scholar] [CrossRef]
- Wyss, E.; Pfiffner, L. Biodiversity in organic horticulture- an indicator for sustainability and a tool for pest management. In XXVII International Horticultural Congress-IHC2006: International Symposium on Sustainability through Integrated and Organic Production; ISHS: Seoul, Korea, 2006; pp. 75–80. [Google Scholar]
- Connor, D.J.; Mínguez, M.I. Evolution not revolution of farming systems will best feed and green the world. Glob. Food Secur. 2012, 1, 106–113. [Google Scholar] [CrossRef] [Green Version]
- Willer, H.; Lernoud, J. The World of Organic Agriculture. Statistics and Emerging Trends; Research Institute of Organic Agriculture Switzerland (FrBL): Frick, Switzerland, 2019. [Google Scholar]
- Cross, J.V.; Solomon, M.G.; Chandler, D.; Jarrett, P.; Richardson, P.N.; Winstanley, D.; Bathon, H.; Huber, J.; Keller, B.; Langenbruch, G.A.; et al. Biocontrol of pests of apples and pears in Northern and Central Europe: 1. Microbial Agents and Nematodes. Biocontrol Sci. Technol. 1999, 9, 125–149. [Google Scholar] [CrossRef]
- Powell, G. The biology and control of an emerging shield bug pest, Pentatoma rufipes (L.)(Hemiptera: Pentatomidae). Agric. For. Entomol. 2020, 22, 298–308. [Google Scholar] [CrossRef]
- Powell, G.; Barclay, M.V.L.; Couch, Y.; Evans, K.A. Current invasion status and potential for UK establishment of the Brown Marmorated Stink Bug, Halyomorpha halys (Hemiptera: Pentatomidae). Br. J. Entomol. Nat. Hist. 2021, 34, 9–21. [Google Scholar]
- Hillocks, R.J. Farming with fewer pesticides: EU pesticide review and resulting challenges for UK agriculture. Crop Prot. 2012, 31, 85–93. [Google Scholar] [CrossRef]
- Culliney, T.W. Crop losses to arthropods. In Integrated Pest Management Reviews; Pimentel, D., Peshin, R., Eds.; Springer Science & Business Media: Dordrecht, The Netherlands, 2014; pp. 201–225. [Google Scholar]
- Peshin, R.; Bandral, R.S.; Zhang, W.; Wilson, L.; Dhawan, A.K. Integrated pest management: A global overview of history, programs and adoption. In Integrated Pest Management: Innovation-Development Process; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1–49. [Google Scholar]
- Zhang, Q.; Wei, Q.; Liu, H.; Jiang, R.; Liu, X.; Wang, X. Multivariate analysis and optimum proposals of the relationship between soil nutrients and fruit qualities in apple orchard. Sci. Agric. Sin. 2011, 44, 1654–1661. [Google Scholar]
- Zehnder, G.; Gurr, G.M.; Kühne, S.; Wade, M.R.; Wratten, S.D.; Wyss, E. Arthropod pest management in organic crops. Annu. Rev. Entomol. 2007, 52, 57–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altieri, M.A.; Nicholls, C.I. Soil fertility management and insect pests: Harmonizing soil and plant health in agroecosystems. Soil Tillage Res. 2003, 72, 203–211. [Google Scholar] [CrossRef]
- Daugherty, M.P.; Briggs, C.J.; Welter, S. Bottom-up and top-down control of pear psylla (Cacopsylla pyricola): Fertilization, plant quality, and the efficacy of the predator Anthocoris nemoralis. Biol. Control 2007, 43, 257–264. [Google Scholar] [CrossRef]
- Altieri, M.; Ponti, L.; Nicholls, C. Enhanced pest management through soil health: Toward a belowground habitat management strategy. Biodynamics 2005, 253, 33. [Google Scholar]
- Simberloff, D.; Martin, J.L.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; Garcia-Berthou, E.; Pascal, M.; et al. Impacts of biological invasions: What’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [Green Version]
- Bugg, R.L.; Waddington, C. Using cover crops to manage arthropod pests of orchards: A review. Agric. Ecosyst. Environ. 1994, 50, 11–28. [Google Scholar] [CrossRef]
- Lim, K.; Kim, W.; Choi, H.; Lee, I.; Cho, W.; Koo, H.; Lee, Y. Nutrient contribution of the cover crops and fruit quality of pear trees as affected by ground cover treatments. J. Korean Soc. Int. Agric. 2011, 23, 297–301. [Google Scholar]
- Oh, Y.-J.; Kang, S.-B.; Song, Y.-I.; Choi, J.-H.; Paik, W.-K. Effects of cover plants on soil microbial community in organic apple orchards. Korean J. Soil Sci. Fertil. 2012, 45, 822–828. [Google Scholar] [CrossRef] [Green Version]
- Hart, J.; Righetti, T.; Stevens, B.; Stebbins, B.; Lombard, P.; Burkhart, D.; Van Buskirk, P. Pears: Fertilizer Guide; U.S. Department of Agriculture and Oregon State University Extension Service: Corvallis, OR, USA, 1997. [Google Scholar]
- Sánchez, E.E.; Giayetto, A.; Cichón, L.; Fernández, D.; Aruani, M.C.; Curetti, M. Cover crops influence soil properties and tree performance in an organic apple (Malus domestica Borkh) orchard in northern Patagonia. Plant Soil 2007, 292, 193–203. [Google Scholar] [CrossRef]
- Ferris, H.; Bongers, T. Nematode indicators of organic enrichment. J. Nematol. 2006, 38, 3. [Google Scholar] [PubMed]
- Altierr, M.; Schmidt, L. Cover crops affect insect and spider populations in apple orchards. Calif. Agric. 1986, 40, 15–17. [Google Scholar]
- Horton, D.R.; Jones, V.P.; Unruh, T.R. Use of a new immunomarking method to assess movement by generalist predators between a cover crop and tree canopy in a pear orchard. Am. Entomol. 2009, 55, 49–56. [Google Scholar] [CrossRef] [Green Version]
- Arnaoudov, V.; Kutinkova, H. Susceptibility of some apple cultivars to infestation by the rosy apple aphid (Dysaphis plantaginea Pass., Homoptera: Aphididae). J. Fruit Ornam. Plant Res. 2006, 14, 137. [Google Scholar]
- Kutinkova, H.; Dzhuvinov, V. Apple breeding for resistance to the aphids Dysaphis plantaginea and Aphis pomi in Bulgaria. Acta Hortic. 2016, 1127, 103–108. [Google Scholar] [CrossRef]
- Berrueta, L.A.; Sasía-Arriba, A.; Miñarro, M.; Antón, M.J.; Alonso-Salces, R.M.; Micheletti, D.; Gallo, B.; Dapena, E. Relationship between hydroxycinnamic acids and the resistance of apple cultivars to rosy apple aphid. Talanta 2018, 187, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Dapena, E.; Miñarro, M.; Blázquez, M.D. Evaluation of the resistance to the rosy apple aphid using a genetic marker. In XII EUCARPIA Symposium on Fruit Breeding and Genetics 814; ISHS: Zaragoza, Spain, 2007; pp. 787–790. [Google Scholar]
- Stoeckli, S.; Mody, K.; Gessler, C.; Patocchi, A.; Jermini, M.; Dorn, S. QTL analysis for aphid resistance and growth traits in apple. Tree Genet. Genomes 2008, 4, 833–847. [Google Scholar] [CrossRef] [Green Version]
- Roche, P.; Alston, F.H.; Maliepaard, C.; Evans, K.M.; Vrielink, R.; Dunemann, L.M.; Markussen, T.; Tartarini, S.; Brown, L.M.; Ryder, C.; et al. RFLP and RAPD markers linked to the rosy leaf curling aphid reistance gene (Sd1) in apple. Theor. Appl. Genet. 1997, 94, 528–533. [Google Scholar] [CrossRef]
- Sandanayaka, W.R.M.; Bus, V.G.M.; Connolly, P. Mechanisms of woolly aphid [Eriosoma lanigerum (Hausm.)] resistance in apple. J. Appl. Entomol. 2005, 129, 534–541. [Google Scholar] [CrossRef]
- Berrada, S.; Nguyen, T.X.; Lemoine, J.; Vanpoucke, J.; Fournier, D. Thirteen Pear Species and Cultivars Evaluated for Resistance to Cacopsylla pyri (Homoptera: Psyllidae). Environ. Entomol. 1995, 24, 1604–1607. [Google Scholar] [CrossRef]
- Puterka, G.J.; Bell, R.L.; Jones, S.K. Ovipositional Preference of Pear Psylla (Homoptera: Psyllidae) for Resistant and Susceptible Pear. J. Econ. Entomol. 1993, 86, 1297–1302. [Google Scholar] [CrossRef]
- Fotiric Aksic, M.M.; Dabic, D.C.; Gasic, U.M.; Zec, G.N.; Vulic, T.B.; Tesic, Z.; Natic, M.M. Polyphenolic profile of pear leaves with different resistance to pear psylla (Cacopsylla pyri). J. Agric. Food Chem. 2015, 63, 7476–7486. [Google Scholar] [CrossRef] [PubMed]
- Evans, K.M.; Govan, C.L.; Fernández-Fernández, F. A new gene for resistance to Dysaphis pyri in pear and identification of flanking microsatellite markers. Genome 2008, 51, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Brewer, L.; Shaw, P.; Wallis, R.; Alspach, P.; Aldworth, M.; Orellana-Torrejon, C.; Chagné, D.; Bus, V.G.M. Genetic mapping of pear sawfly (Caliroa cerasi) and pear blister mite (Eriophyes pyri) resistance in an interspecific pear family. Tree Genet. Genomes 2018, 14, 38. [Google Scholar] [CrossRef]
- Cummins, J.N.; Aldwinckle, H.S. Breeding Apple Rootstocks. In Plant Breeding Reviews; Janick, J., Ed.; Springer: Boston, MA, USA, 1983; Volume 1, pp. 294–394. [Google Scholar]
- Orpet, R.J.; Jones, V.P.; Reganold, J.P.; Crowder, D.W. Effects of restricting movement between root and canopy populations of woolly apple aphid. PLoS ONE 2019, 14, e0216424. [Google Scholar] [CrossRef]
- Bell, R.L. Genetics, genomics, and breeding for fire blight resistance in pear. In The Pear Genome; Springer: Berlin/Heidelberg, Germany, 2019; pp. 243–264. [Google Scholar]
- Brewer, L.; Volz, R. Genetics and Breeding of Pear. In The Pear Genome; Springer: Berlin/Heidelberg, Germany, 2019; pp. 63–101. [Google Scholar]
- Howard, N.; Albach, D.; Luby, J. The identification of apple pedigree information on a large diverse set of apple germplasm and its application in apple breeding using new genetic tools. In Proceedings of the 18th International Conference on Organic Fruit-Growing, Hohenheim, Germany, 19–21 February 2018; pp. 19–21. [Google Scholar]
- Laurens, F.; Aranzana, M.J.; Arus, P.; Bassi, D.; Bink, M.; Bonany, J.; Caprera, A.; Corelli-Grappadelli, L.; Costes, E.; Durel, C.E.; et al. An integrated approach for increasing breeding efficiency in apple and peach in Europe. Hortic. Res. 2018, 5, 11. [Google Scholar] [CrossRef] [Green Version]
- Briggs, J.B.; Alston, F.H. Pest avoidance by late-flowering apple varieties. East Malling Res. Stn. Annu. Rep. 1966, 1, 170–171. [Google Scholar]
- Minarro, M.; Dapena, E. Tolerance of some scab-resistant apple cultivars to the rosy apple aphid, Dysaphis plantaginea. Crop Prot. 2008, 27, 391–395. [Google Scholar] [CrossRef]
- Stuart, L.C.; Butt, B.A.; Bell, R.L. Effect of Host Phenology on Ovipositional Preferences of Winter Form Pear Psylla (Homoptera: Psyllidae. J. Entomol. Soc. Br. Columbia 1989, 86, 34–38. [Google Scholar]
- Nath, V.; Marboh, E.S.; Gupta, A.K.; Lal, N. Canopy management for sustainable fruit production. Int. J. Innov. Hortic. 2019, 8, 115–126. [Google Scholar] [CrossRef]
- Baldi, P.; Wolters, P.J.; Komjanc, M.; Viola, R.; Velasco, R.; Salvi, S. Genetic and physical characterisation of the locus controlling columnar habit in apple (Malus× domestica Borkh.). Mol. Breed. 2013, 31, 429–440. [Google Scholar] [CrossRef]
- Franck, P.; Reyes, M.; Olivares, J.; Sauphanor, B. Genetic architecture in codling moth populations: Comparison between microsatellite and insecticide resistance markers. Mol. Ecol. 2007, 16, 3554–3564. [Google Scholar] [CrossRef] [PubMed]
- Simon, S.; Lauri, P.; Brun, L.; Defrance, H.; Sauphanor, B. Does manipulation of fruit-tree architecture affect the development of pests and pathogens? A case study in an organic apple orchard. J. Hortic. Sci. Biotechnol. 2006, 81, 765–773. [Google Scholar] [CrossRef]
- Simon, S.; Sauphanor, B.; Lauri, P.-E. Control of fruit tree pests through manipulation of tree architecture. Pest Technol. 2007, 1, 33–37. [Google Scholar]
- Massimino Cocuzza, G.E. Insect pests of fruit; aphids. In Integrated Management of Diseases and Insect Pests of Fruit Trees, 1st ed.; Xu, X., Fountain, M.T., Eds.; Burleigh Dodds Science Publishing: Cambridge, UK, 2019. [Google Scholar]
- Yao, I. Costs and constraints in aphid-ant mutualism. Ecol. Res. 2014, 29, 383–391. [Google Scholar] [CrossRef] [Green Version]
- Wünsche, J.N.; Lakso, A.N. Apple tree physiology: Implications for orchard and tree management. Compact Fruit Tree 2000, 33, 82–88. [Google Scholar]
- Corelli, L.; Sansavini, S. Light interception and photosynthesis related to planting density and canopy management in apple. In IV International Symposium on Research and Development on Orchard and Plantation Systems 243; ISHS: Leuven, Belgium, 1989; pp. 159–174. [Google Scholar]
- Solomon, M.E.; Glen, D.M.; Kendall, D.A.; Milsom, N.F. Predation of overwintering larvae of codling moth (Cydia pomonella (L.)) by birds. J. Appl. Ecol. 1976, 13, 341–352. [Google Scholar] [CrossRef]
- García, D.; Miñarro, M.; Martínez-Sastre, R. Birds as suppliers of pest control in cider apple orchards: Avian biodiversity drivers and insectivory effect. Agric. Ecosyst. Environ. 2018, 254, 233–243. [Google Scholar] [CrossRef]
- Kirk, D.A.; Evenden, M.D.; Mineau, P. Past and current attempts to evaluate the role of birds as predators of insect pests in temperate agriculture. In Current Ornithology; Springer: Berlin/Heidelberg, Germany, 1996; pp. 175–269. [Google Scholar]
- Peisley, R.K.; Saunders, M.E.; Luck, G.W. Cost-benefit trade-offs of bird activity in apple orchards. PeerJ 2016, 4, e2179. [Google Scholar] [CrossRef] [Green Version]
- Mols, C.M.; Visser, M.E. Great tits can reduce caterpillar damage in apple orchards. J. Appl. Ecol. 2002, 39, 888–899. [Google Scholar] [CrossRef]
- Mols, C.M.; Visser, M.E. Great tits (Parus major) reduce caterpillar damage in commercial apple orchards. PLoS ONE 2007, 2, e202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawson, D.; Bull, P. A questionnaire survey of bird damage to fruit. N. Z. J. Agric. Res. 1970, 13, 362–371. [Google Scholar] [CrossRef] [Green Version]
- Cross, J.; Walklate, P.; Murray, R.; Richardson, G. Spray deposits and losses in different sized apple trees from an axial fan orchard sprayer: 3. Effects of air volumetric flow rate. Crop Prot. 2003, 22, 381–394. [Google Scholar] [CrossRef]
- Xu, X.; Wu, P.; Thorbek, P.; Hyder, K. Variability in initial spray deposit in apple trees in space and time. Pest Manag. Sci. 2006, 62, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Cross, J.; Fountain, M.; MarkÓ, V.; Nagy, C. Arthropod ecosystem services in apple orchards and their economic benefits. Ecol. Entomol. 2015, 40, 82–96. [Google Scholar] [CrossRef]
- McKerchar, M.; Potts, S.; Fountain, M.; Garratt, M.P.; Westbury, D.B. The potential for wildflower interventions to enhance natural enemies and pollinators in commercial apple orchards is limited by other management practices. Agric. Ecosyst. Environ. 2020, 301, 107034. [Google Scholar] [CrossRef]
- Markó, V.; Elek, Z.; Kovács-Hostyánszki, A.; Kőrösi, Á.; Somay, L.; Földesi, R.; Varga, Á.; Iván, Á.; Báldi, A. Landscapes, orchards, pesticides–Abundance of beetles (Coleoptera) in apple orchards along pesticide toxicity and landscape complexity gradients. Agric. Ecosyst. Environ. 2017, 247, 246–254. [Google Scholar] [CrossRef] [Green Version]
- Hardman, J.M.; Franklin, J.L.; Jensen, K.I.; Moreau, D.L. Effects of pesticides on mite predators (Acari: Phytoseiidae) and colonization of apple trees by Tetranychus urticae. Phytoparasitica 2006, 34, 449–462. [Google Scholar] [CrossRef]
- Cross, J.V.; Solomon, M.G.; Babandreier, D.; Blommers, L.; Easterbrook, M.A.; Jay, C.N.; Jenser, G.; Jolly, R.L.; Kuhlmann, U.; Lilley, R.; et al. Biocontrol of pests of apples and pears in Northern and Central Europe: 2. Parasitoids. Biocontrol Sci. Technol. 1999, 9, 277–314. [Google Scholar] [CrossRef]
- Fountain, M.; Nagy, C.; Harris, A.; Cross, J. Importance of naturally occurring predators for pear sucker control. IOBC-WPRS Bull. 2013, 91, 117–125. [Google Scholar]
- Vranken, K.; Trekels, H.; Thys, T.; Demaeght, P.; Van Leeuwen, T.; Clercq, D. Importance of alternative predators of pear suckers (Cacopsylla pyri) in organic versus IPM pear orchards. IOBC-WPRS Bull. 2014, 103, 37–40. [Google Scholar]
- Solomon, M.G.; Cross, J.V.; Fitzgerald, J.D.; Campbell, C.A.M.; Jolly, R.L.; Olszak, R.W.; Niemczyk, E.; Vogt, H. Biocontrol of pests of apples and pears in Northern and Central Europe-3. Predators. Biocontrol Sci. Technol. 2000, 10, 91–128. [Google Scholar] [CrossRef]
- Solomon, M.G.; Cranham, J.E.; Easterbrook, M.A.; Fitzgerald, J.D. Control of the pear psyllid, Cacopsylla pyricola, in South East England by predators and pesticides. Crop Prot. 1989, 8, 197–205. [Google Scholar] [CrossRef]
- Sigsgaard, L. Oviposition preference of Anthocoris nemorum and A. nemoralis for apple and pear. Entomol. Exp. Et Appl. 2004, 111, 215–223. [Google Scholar] [CrossRef]
- Sigsgaard, L. Habitat and prey preferences of the two predatory bugs Anthocoris nemorum (L.) and A. nemoralis (Fabricius) (Anthocoridae: Hemiptera-Heteroptera). Biol. Control 2010, 53, 46–54. [Google Scholar] [CrossRef] [Green Version]
- Sigsgaard, L.; Kollmann, J. Beneficial effects of hedgerow plants for insect predators in adjacent orchards–the value of pollen and nectar to Anthocoris nemorum (L.). In Proceedings of the International Workshop on Arthropod Pest Problems in Pome Fruit Production, Lleida, Spain, 4–6 September 2007. [Google Scholar]
- Happe, A.; Beni, L.R.; Bosch, J.; Alins, G.; Mody, K. Earwigs and aphids in apple orchards–influence of agri-environmental measures and landscape factors. In Proceedings of the 18th International Conference on Organic Fruit-Growing, Hohenheim, Germany, 19–21 February 2018; pp. 243–244. [Google Scholar]
- Cross, J.; Nagy, C.; Batki, M.; Linka, J. Conservation biocontrol of pear psyllids. Mitt. Klosterneubg. Rebe Wein Obstbau Früchteverwertung 2010, 60, 403–412. [Google Scholar]
- Wolton, R.; Pollard, K.; Goodwin, A.; Norton, L. Regulatory Services Delivered by Hedges: The Evidence Base. 2014. Available online: http://randd.defra.gov.uk (accessed on 30 November 2021).
- Garratt, M.P.; Senapathi, D.; Coston, D.J.; Mortimer, S.R.; Potts, S.G. The benefits of hedgerows for pollinators and natural enemies depends on hedge quality and landscape context. Agric. Ecosyst. Environ. 2017, 247, 363–370. [Google Scholar] [CrossRef] [Green Version]
- Debras, J.F.; Cousin, M.; Rieux, R. The conception of hedgerows for use in the control of pear psyllid. Phytoma 2000, 525, 44–48. [Google Scholar]
- Staerkle, M.; Kölliker, M. Maternal food regurgitation to nymphs in earwigs (Forficula auricularia). Ethology 2008, 114, 844–850. [Google Scholar] [CrossRef]
- Orpet, R.J.; Crowder, D.W.; Jones, V.P. Biology and management of European earwig in orchards and vineyards. J. Integr. Pest Manag. 2019, 10, 21. [Google Scholar] [CrossRef] [Green Version]
- Logan, D.P.; Maher, B.J.; Rowe, C.A. Predation of diaspidid scale insects on kiwifruit vines by European earwigs, Forficula auricularia, and steel-blue ladybirds, Halmus chalybeus. BioControl 2017, 62, 469–479. [Google Scholar] [CrossRef]
- Mueller, T.F.; Blommers, L.H.M.; Mols, P.J.M. Earwig (Forficula auricularia) predation on the woolly apple aphid, Eriosoma lanigerum. Entomol. Exp. Et Appl. 1988, 47, 145–152. [Google Scholar] [CrossRef]
- Vogt, H.; Just, J.; Grutzmacher, A. Impact of four insecticides on the European earwig, Forficula auricularia L., in an apple orchard. In Proceedings of the 7th International Conference on Integrated Fruit Production, Avignon, France, 27–30 October 2008; pp. 112–116. [Google Scholar]
- Fountain, M.T.; Harris, A.L. Non-target consequences of insecticides used in apple and pear orchards on Forficula auricularia L.(Dermaptera: Forficulidae). Biol. Control 2015, 91, 27–33. [Google Scholar] [CrossRef]
- Orpet, R.J.; Goldberger, J.R.; Crowder, D.W.; Jones, V.P. Field evidence and grower perceptions on the roles of an omnivore, European earwig, in apple orchards. Biol. Control 2019, 132, 189–198. [Google Scholar] [CrossRef]
- Suckling, D.M.; Burnip, G.M.; Hackett, J.; Daly, J.C. Frass sampling and baiting indicate European earwig (Forficula auricularia) foraging in orchards. J. Appl. Entomol. 2006, 130, 263–267. [Google Scholar] [CrossRef]
- Helsen, H.; Trapman, M.; Polfliet, M.; Simonse, J. Presence of the common earwig Forficula auricularia L. in apple orchards and its impact on the woolly apple aphid Eriosoma lanigerum (Haussmann). IOBC-WPRS Bull. 2007, 30, 31. [Google Scholar]
- Carroll, D.P.; Hoyt, S.C. Augmentation of European earwigs (Dermaptera: Forficulidae) for biological control of apple aphid (Homoptera: Aphididae) in an apple orchard. J. Econ. Entomol. 1984, 77, 738–740. [Google Scholar] [CrossRef]
- Rotheray, E.L.; MacGowan, I.; Rotheray, G.E.; Sears, J.; Elliott, A. The conservation requirements of an endangered hoverfly, Hammerschmidtia ferruginea (Diptera, Syrphidae) in the British Isles. J. Insect Conserv. 2008, 13, 569. [Google Scholar] [CrossRef]
- Garratt, M.; Breeze, T.; Boreux, V.; Fountain, M.; Mckerchar, M.; Webber, S.; Coston, D.; Jenner, N.; Dean, R.; Westbury, D. Apple pollination: Demand depends on variety and supply depends on pollinator identity. PLoS ONE 2016, 11, e0153889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossi, J.; Gamba, U.; Pinna, M.; Spagnolo, S.; Visentin, C.; Alberto, A. Hoverflies in organic apple orchards in north-western Italy. Bull. Insectology 2006, 59, 111–114. [Google Scholar]
- Almohamad, R.; Verheggen, F. Francis, F.; Haubruge, E. Impact of aphid colony size and associated induced plant volatiles on searching and oviposition behaviour of a predatory hoverfly. Belg. J. Entomol. 2008, 10, 17–26. [Google Scholar]
- Gontijo, L.M.; Beers, E.H.; Snyder, W.E. Flowers promote aphid suppression in apple orchards. Biol. Control 2013, 66, 8–15. [Google Scholar] [CrossRef]
- Hogg, B.N.; Nelson, E.H.; Mills, N.J.; Daane, K.M. Floral resources enhance aphid suppression by a hoverfly. Entomol. Exp. Et Appl. 2011, 141, 138–144. [Google Scholar] [CrossRef]
- Hodgkiss, D.; Brown, M.J.F.; Fountain, M.T. Syrphine hoverflies are effective pollinators of commercial strawberry. J. Pollinat. Ecol. 2018, 22, 55–66. [Google Scholar] [CrossRef]
- Hindayana, D.; Meyhofer, R.; Scholz, D.; Poehling, H.M. Intraguild Predation among the Hoverfly Episyrphus balteatus de Geer (Diptera: Syrphidae) and Other Aphidophagous Predators. Biol. Control 2001, 20, 236–246. [Google Scholar] [CrossRef] [Green Version]
- Dib, H.; Jamont, M.; Sauphanor, B.; Capowiez, Y. Predation potency and intraguild interactions between generalist (Forficula auricularia) and specialist (Episyrphus balteatus) predators of the rosy apple aphid (Dysaphis plantaginea). Biol. Control 2011, 59, 90–97. [Google Scholar] [CrossRef]
- Wyss, E.; Villiger, M.; Müller-Schärer, H. The potential of three native insect predators to control the rosy apple aphid, Dysaphis plantaginea. BioControl 1999, 44, 171–182. [Google Scholar] [CrossRef]
- Perrin, R. The role of the perennial stinging nettle, Urtica dioica, as a reservoir of beneficial natural enemies. Ann. Appl. Biol. 1975, 81, 289–297. [Google Scholar] [CrossRef]
- Baverstock, J.; Porcel, M.; Clark, S.; Copeland, J.; Pell, J. Potential value of the fibre nettle Urtica dioica as a resource for the nettle aphid Microlophium carnosum and its insect and fungal natural enemies. BioControl 2011, 56, 215–223. [Google Scholar] [CrossRef]
- Wyss, E.; Villiger, M.; Hemptinne, J.-L.; Müller-schärer, H. Effects of augmentative releases of eggs and larvae of the ladybird beetle, Adalia bipunctata, on the abundance of the rosy apple aphid, Dysaphis plantaginea, in organic apple orchards. Entomol. Exp. Et Appl. 1999, 90, 167–173. [Google Scholar] [CrossRef] [Green Version]
- Nagy, C.; Cross, J.; Markó, V. Sugar feeding of the common black ant, Lasius niger (L.), as a possible indirect method for reducing aphid populations on apple by disturbing ant-aphid mutualism. Biol. Control 2013, 65, 24–36. [Google Scholar] [CrossRef]
- Nagy, C.; Cross, J.; Markó, V. Can artificial nectaries outcompete aphids in ant-aphid mutualism? Applying artificial sugar sources for ants to support better biological control of rosy apple aphid, Dysaphis plantaginea Passerini in apple orchards. Crop Prot. 2015, 77, 127–138. [Google Scholar] [CrossRef]
- Offenberg, J. Balancing between mutualism and exploitation: The symbiotic interaction between Lasius ants and aphids. Behav. Ecol. Sociobiol. 2001, 49, 304–310. [Google Scholar] [CrossRef]
- Jay, C.; Cross, J. Increasing Hoverly Populations in Apple Orchards for Control of Apple Aphids; NIAB EMR: West Malling, UK, 2016. [Google Scholar]
- Rural Payments Agency and Natural England. BN11: Planting New Hedges. Available online: https://www.gov.uk/countryside-stewardship-grants/planting-new-hedges-bn11 (accessed on 30 November 2021).
- Amano, H.; Chant, D.A. Mating behaviour and reproductive mechanisms of two species of predacious mites, Phytoseiulus persimilis Athias-Henriot and Amblyseius andersoni (Chant) (Acarina: Phytoseiidae). Acarologia 1979, 20, 196–213. [Google Scholar]
- Fitzgerald, J.D.; Pepper, N.; Solomon, M.G. Interactions among predators and phytophagous mites on apple; possible impact on biocontrol of Panonychus ulmi by Typhlodromus pyri in orchards. Biocontrol Sci. Technol. 2007, 17, 1009–1019. [Google Scholar] [CrossRef]
- Strapazzon, A.; Montà, L.D. Role and distribution of Amblyseius andersoni (Chant) and Zetzellia mali (Ewing) in apple orchards infested by Aculus schlechtendali (Nalepa). Redia 1988, 71, 39–54. [Google Scholar]
- Easterbrook, M.A. The life-history of the eriophyid mite Aculus schlechtendali on apple in South-east England. Ann. Appl. Biol. 1979, 91, 287–296. [Google Scholar] [CrossRef]
- Skirvin, D.J.; Fenlon, J.S. The effect of temperature on the functional response of Phytoseiulus persimilis (Acari: Phytoseiidae). Exp. Appl. Acarol. 2003, 31, 37. [Google Scholar] [CrossRef] [PubMed]
- Laing, J.E. Life history and life table of Phytoseiulus persimilis. Acarologia 1968, 10, 578–588. [Google Scholar] [PubMed]
- AgroSciences, B. Anderline Amblyseius andersoni Product Sheet. Available online: https://www.biolineagrosciences.com/products/anderline/ (accessed on 25 February 2020).
- Amano, H.; Chant, D.A. Life history and reproduction of two species of predacious mites, Phytoseiulus persimilis Athias-Henriot and Amblyseius andersoni (Chant) (Acarina: Phytoseiidae). Can. J. Zool. 1977, 55, 1978–1983. [Google Scholar] [CrossRef]
- Llusià, J.; Peñuelas, J. Emission of volatile organic compounds by apple trees under spider mite attack and attraction of predatory mites. Exp. Appl. Acarol. 2001, 25, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Momen, F.; Fahim, S.; Barghout, M. Mass production of predatory mites and their efficacy for controlling pests. In Cottage Industry of Biocontrol Agents and Their Applications; Springer: Berlin/Heidelberg, Germany, 2020; pp. 157–200. [Google Scholar]
- Szabo, A.; Penzes, B. A new method for the release of Amblyseius andersoni (Acari: Phytoseiidae) in young apple orchards. Eur. J. Entomol. 2013, 110, 477–482. [Google Scholar] [CrossRef] [Green Version]
- Solomon, M.G. The colonization of an apple orchard by predators of the fruit tree red spider mite. Ann. Appl. Biol. 1975, 80, 119–122. [Google Scholar] [CrossRef]
- Solomon, M.; Fitzgerald, J. The role of resistant Typhlodromus pyri in apple orchards. In Proceedings of the British Crop Protection Conference. Pests and Diseases, Brighton Metropole, UK, 19–22 November 1984; pp. 1113–1116. [Google Scholar]
- Sekrecka, M.; Niemczyk, E. Introducing Typhlodromus pyri (Phytoseiidae) into apple orchards in Poland. J. Fruit Ornam. Plant Res. 2006, 14, 203–207. [Google Scholar]
- Horton, D.R. Phenology of emergence from artificial overwintering shelters by some predatory arthropods common in pear orchards of the Parcific Northwest. J. Entomol. Soc. Br. Columbia 2004, 101, 101–108. [Google Scholar]
- Horton, D.R.; Broers, D.A.; Hinojosa, T.; Lewis, T.M.; Miliczky, E.R.; Lewis, R.R. Diversity and phenology of predatory arthropods overwintering in cardboard bands placed in pear and apple orchards of central Washington State. Ann. Entomol. Soc. Am. 2002, 95, 469–480. [Google Scholar] [CrossRef]
- Costes, E.; Sinoquet, H.; Kelner, J.J.; Godin, C. Exploring within-tree architectural development of two apple tree cultivars over 6 years. Ann. Bot. 2003, 91, 91–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Begg, G.S.; Cook, S.M.; Dye, R.; Ferrante, M.; Franck, P.; Lavigne, C.; Lövei, G.L.; Mansion-Vaquie, A.; Pell, J.K.; Petit, S. A functional overview of conservation biological control. Crop Prot. 2017, 97, 145–158. [Google Scholar] [CrossRef]
- Kölliker, M. Benefits and costs of earwig (Forficula auricularia) family life. Behav. Ecol. Sociobiol. 2007, 61, 1489–1497. [Google Scholar] [CrossRef] [Green Version]
- Helsen, H.; Vaal, F.; Blommers, L. Phenology of the common earwig Forficula auricularia L. (Dermaptera: Forficulidae) in an apple orchard. Int. J. Pest Manag. 1998, 44, 75–79. [Google Scholar] [CrossRef]
- Kölliker, M.; Vancassel, M. Maternal attendance and the maintenance of family groups in common earwigs (Forficula auricularia): A field experiment. Ecol. Entomol. 2007, 32, 24–27. [Google Scholar] [CrossRef]
- Lamb, R. Parental behaviour in the Dermaptera with special reference to Forficular auricularia (Dermaptera Forficulidae). Can. Entomol. 1976, 108, 609–619. [Google Scholar] [CrossRef]
- Lys, J.-A.; Nentwig, W. Surface activity of carabid beetles inhabiting cereal fields: Seasonal phenology and the influence of farming operations on five abundant species. Pedobiologia 1991, 35, 129–138. [Google Scholar]
- Shearin, A.F.; Reberg-Horton, S.C.; Gallandt, E.R. Direct effects of tillage on the activity density of ground beetle (Coleoptera: Carabidae) weed seed predators. Environ. Entomol. 2014, 36, 1140–1146. [Google Scholar] [CrossRef]
- Boivin, G.; Hance, T. Ground beetle assemblages in cultivated organic soil and adjacent habitats: Temporal dynamics of microspatial changes. Pedobiologia 2003, 47, 193–202. [Google Scholar] [CrossRef]
- Kromp, B. Carabid beetles in sustainable agriculture: A review on pest control efficacy, cultivation impacts and enhancement. Agric. Ecosyst. Environ. 1999, 74, 187–228. [Google Scholar] [CrossRef]
- Miñarro, M.; Espadaler, X.; Melero, V.; Suárez-Álvarez, V. Organic versus conventional management in an apple orchard: Effects of fertilization and tree-row management on ground-dwelling predaceous arthropods. Agric. For. Entomol. 2009, 11, 133–142. [Google Scholar] [CrossRef]
- Brown, M.; Tworkoski, T. Pest management benefits of compost mulch in apple orchards. Agric. Ecosyst. Environ. 2004, 103, 465–472. [Google Scholar] [CrossRef] [Green Version]
- Ladurner, E.; Bosch, J.; Kemp, W.P.; Maini, S. Foraging and Nesting Behavior of Osmia lignaria (Hymenoptera: Megachilidae) in the Presence of Fungicides: Cage Studies. J. Econ. Entomol. 2008, 101, 647–653. [Google Scholar] [CrossRef]
- Artz, D.R.; Pitts-Singer, T.L. Effects of Fungicide and Adjuvant Sprays on Nesting Behavior in Two Managed Solitary Bees, Osmia lignaria and Megachile rotundata. PLoS ONE 2015, 10, e0135688. [Google Scholar] [CrossRef] [PubMed]
- Gebhardt, M.M.; Eberle, K.E.; Radtke, P.; Jehle, J.A. Baculovirus resistance in codling moth is virus isolate-dependent and the consequence of a mutation in viral gene pe38. Proc. Natl. Acad. Sci. USA 2014, 111, 15711–15716. [Google Scholar] [CrossRef] [Green Version]
- Glen, D.M.; Wiltshire, C.W.; Milsom, N.F.; Brain, P. Codling moth granulosis virus: Effects of its use on some other orchard arthropods. Ann. Appl. Biol. 1984, 104, 99–106. [Google Scholar] [CrossRef]
- Bouvier, J.-C.; Buès, R.; Boivin, T.; Boudinhon, L.; Beslay, D.; Sauphanor, B. Deltamethrin resistance in the codling moth (Lepidoptera: Tortricidae): Inheritance and number of genes involved. Heredity 2001, 87, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Charmillot, P.; Blanc, G.; Pasquier, D. First case of summer fruit tortrix moth (Adoxophyes orana) resistance to insecticides in Switzerland. Rev. Suisse De Vitic. Arboric. Hortic. 2006, 38, 87–93. [Google Scholar]
- Kadoic Balasko, M.; Bazok, R.; Mikac, K.M.; Lemic, D.; Pajac Zivkovic, I. Pest management challenges and control practices in codling moth: A review. Insects 2020, 11, 38. [Google Scholar] [CrossRef] [Green Version]
- Salamin, C.; Charmillot, P.; Pasquier, D. New Case of Summer Fruit Tortrix (Adoxophyes orana) Resistance to Insecticides; Revue Suisse de Viticulture, Arboriculture, Horticulture: Lausanne, Switzerland, 2007. [Google Scholar]
- Arthurs, S.P.; Lacey, L.A. Field evaluation of commercial formulations of the codling moth granulovirus: Persistence of activity and success of seasonal applications against natural infestations of codling moth in Pacific Northwest apple orchards. Biol. Control 2004, 31, 388–397. [Google Scholar] [CrossRef]
- Lacey, L.A.; Thomson, D.; Vincent, C.; Arthurs, S.P. Codling moth granulovirus: A comprehensive review. Biocontrol Sci. Technol. 2008, 18, 639–663. [Google Scholar] [CrossRef]
- Arthurs, S.P.; Lacey, L.A.; Fritts, J.R. Optimizing use of codling moth granulovirus: Effects of application rate and spraying frequency on control of codling moth larvae in pacific northwest apple orchards. J. Econ. Entomol. 2005, 95, 1459–1468. [Google Scholar] [CrossRef]
- Sekita, N.; Kawashima, K.; Aizu, H.; Shiraski, S.; Yamada, M. A short term control of Adoxophyes orana fasciata Walsingham (Lepidoptera:Tortricidae) by a granulosis virus in apple orchards. Appl. Entomol. Zool. 1984, 19, 498–508. [Google Scholar] [CrossRef]
- Peters, D.; Wiebenga, J.; van Maanen, H.J.; Blommers, L. Control of the summer fruit tortrix moth, Adoxophyes orana, with a nuclear polyhedrosis virus in orchards. In Proceedings of the VIIth Symposium on Integrated Plant Protection in Orchards, Wageningen, The Netherlands, 26–29 August 1985. [Google Scholar]
- Wallhead, M.; Zhu, H. Decision support systems for plant disease and insect management in commercial nurseries in the Midwest: A perspective review. J. Environ. Hortic. 2017, 35, 84–92. [Google Scholar] [CrossRef]
- Trapman, M. Validation of the RIMpro decision support system for apple sawfly (Hoplocampa testudinea) with field observations in The Netherlands, Belgium, Denmark and Austria 2010–2015. In Proceedings of the 17th International Conference on Organic Fruit-Growing, Stuttgart, Germany, 15–17 February 2016; pp. 69–76. [Google Scholar]
- Trapman, M.; Helsen, H.; Polfliet, M. Development of a dynamic population model as a decision support system for Codling Moth (Cydia pomonella L) management. In Proceedings of the Ecofruit-13th International Conference on Cultivation Technique and Phytopathological Problems in Organic Fruit-Growing, Weinsberg, Germany, 18–20 February 2008; pp. 247–251. [Google Scholar]
- Berling, M.; Blachere-Lopez, C.; Soubabere, O.; Lery, X.; Bonhomme, A.; Sauphanor, B.; Lopez-Ferber, M. Cydia pomonella granulovirus genotypes overcome virus resistance in the codling moth and improve virus efficiency by selection against resistant hosts. Appl. Environ. Microbiol. 2009, 75, 925–930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lacey, L.A.; Arthurs, S.P.; Knight, A.L.; Huber, J. Microbial control of lepidopteran pests of apple orchards. In Field Manual of Techniques in Invertebrate Pathology: Application and Evaluation of Pathogens for Control of Insects and Other Invertebrate Pests; Lacey, L.A., Kaya, H.K., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 528–546. [Google Scholar]
- Kaya, H.K.; Gaugler, R. Entomopathogenic nematodes. Annu. Rev. Entomol. 1993, 38, 181–206. [Google Scholar] [CrossRef]
- Bedding, R.; Miller, L. Use of a nematode, Heterorhabditis heliothidis, to control black vine weevil, Otiorhynchus sulcatus, in potted plants. Ann. Appl. Biol. 1981, 99, 211–216. [Google Scholar] [CrossRef]
- Grewal, P.S.; Ehlers, R.-U.; Shapiro-Ilan, D.I. Nematodes as Biocontrol Agents; CABI: Wallingford, UK, 2005. [Google Scholar]
- Vincent, C.; Babendreier, D.; Świergiel, W.; Helsen, H.; Blommers, L.H. A review of the apple sawfly, Hoplocampa testudinea (Hymenoptera Tenthredinidae). Bull. Insectology 2019, 72, 35–54. [Google Scholar]
- Divya, K.; Sankar, M. Entomopathogenic nematodes in pest management. Indian J. Sci. Technol. 2009, 2, 53–60. [Google Scholar] [CrossRef]
- Labaude, S.; Griffin, C.T. Transmission success of entomopathogenic nematodes used in pest control. Insects 2018, 9, 72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vincent, C.; Belair, G. Biocontrol of the apple sawfly, Hoplocampus testudinea, with entomogenous nematodes. Entomophaga 1992, 37, 575–582. [Google Scholar] [CrossRef]
- Wright, D.; Peters, A.; Schroer, S.; Fife, J. Application Technology. In Nematodes as Biocontrol Agents; Grewal, P.S., Ehlers, R.-U., Shapiro-Ilan, D.I., Eds.; CABI Publishing: Wallingford, UK, 2005. [Google Scholar]
- Curto, G.; Boselli, M.; Vergnani, S.; Reggiani, A. Effectiveness of entomopathogenic nematodes in the control of sawfly (Hoplocampa brevis) in pear orchards. IOBC-WPRS Bull. 2007, 30, 13–17. [Google Scholar]
- Fornaciari, M.; Vergnani, S. Organic and integrated pear production: Towards a common strategy? Riv. Di Fruttic. E Di Ortofloric. 2006, 68, 60–63. [Google Scholar]
- Lacey, L.A.; Arthurs, S.P.; Unruh, T.R.; Headrick, H.; Fritts, R. Entomopathogenic nematodes for control of codling moth (Lepidoptera: Tortricidae) in apple and pear orchards: Effect of nematode species and seasonal temperatures, adjuvants, application equipment, and post-application irrigation. Biol. Control 2006, 37, 214–223. [Google Scholar] [CrossRef]
- Brown, M.; Jaegers, J.; Pye, A.; Schmitt, J. Control of edaphic populations of woolly apple aphid using entomopathogenic nematodes and a systemic aphicide. J. Entomol. Sci. 1992, 27, 224–232. [Google Scholar] [CrossRef]
- Mantzoukas, S.; Eliopoulos, P.A. Endophytic entomopathogenic fungi: A valuable biological control tool against plant pests. Appl. Sci. 2020, 10, 360. [Google Scholar] [CrossRef] [Green Version]
- Sookar, P.; Bhagwant, S.; Awuor Ouna, E. Isolation of entomopathogenic fungi from the soil and their pathogenicity to two fruit fly species (Diptera: Tephritidae). J. Appl. Entomol. 2008, 132, 778–788. [Google Scholar] [CrossRef]
- Litwin, A.; Nowak, M.; Różalska, S. Entomopathogenic fungi: Unconventional applications. Rev. Environ. Sci. Bio/Technol. 2020, 19, 23–42. [Google Scholar] [CrossRef] [Green Version]
- Shah, P.; Pell, J. Entomopathogenic fungi as biological control agents. Appl. Microbiol. Biotechnol. 2003, 61, 413–423. [Google Scholar] [CrossRef]
- Tkaczuk, C.; Tipping, C.; Majchrowska-Safaryan, A.; Krol, A.; Wyrzykowska, M.; Wegensteiner, R. The occurrence of entomopathogenic fungi in soils from apple orchards protected in different farming systems. Fresenius Environ. Bull. 2019, 28, 7906–7914. [Google Scholar]
- Clifton, E.H.; Jaronski, S.T.; Hodgson, E.W.; Gassmann, A.J. Abundance of soil-borne entomopathogenic fungi in organic and conventional fields in the Midwestern USA with an emphasis on the effect of herbicides and fungicides on fungal persistence. PLoS ONE 2015, 10, e0133613. [Google Scholar] [CrossRef]
- Easterbrook, M. The life-history and bionomics of Epitrimerus piri (Acarina: Eriophyidae) on pear. Ann. Appl. Biol. 1978, 88, 13–22. [Google Scholar] [CrossRef]
- Robles-Acosta, I.N.; Chacón-Hernández, J.C.; Torres-Acosta, R.I.; Landeros-Flores, J.; Vanoye-Eligio, V.; Arredondo-Valdés, R. Entomopathogenic Fungi as Biological Control Agents of Phyllocoptruta oleivora (Prostigmata: Eriophyidae) under Greenhouse Conditions. Fla. Entomol. 2019, 102, 303–308. [Google Scholar] [CrossRef]
- Demirci, F.; Denizhan, E. Paecilomyces lilacinus, a potential biocontrol agent on apple rust mite Aculus schlechtendali and interactions with some fungicides in vitro. Phytoparasitica 2010, 38, 125–132. [Google Scholar] [CrossRef]
- Yagimuma, K. Seasonal occurrence of entomopathogenic fungi in apple orchard not sprayed with insecticides. Jpn. J. Appl. Entomol. Zool. 2007, 51, 213–220. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.; Kostromytska, O.S.; Goble, T.; Hajek, A.E.; Koppenhöfer, A.M. Compatibility of a microsclerotial granular formulation of the entomopathogenic fungus Metarhizium brunneum with fungicides. BioControl 2020, 65, 113–123. [Google Scholar] [CrossRef]
- Wenzel Rodrigues, I.M.; Batista Filho, A.; Giordano, I.B.; Denadae, B.E.; Fernandes, J.B.; Forim, M.R. Compatibility of polymers to fungi Beauveria bassiana and Metarhizium anisopliae and their formulated products stability. Acta Scientiarum. Agron. 2017, 39, 457–464. [Google Scholar] [CrossRef] [Green Version]
- Godfray, H. Parasitoids: Behavioral and Evolutionary Ecology; Princeton University Press: Princeton, NJ, USA, 1994; Volume 67. [Google Scholar]
- Cross, J.; Jay, C. Exploiting the parasitoids Lathrolestes ensator and Platygaster demades for control of apple sawfly and apple leaf midge in IPM in apple orchards. IOBC-WPRS Bull. 2001, 24, 161–166. [Google Scholar]
- Mates, S.G.; Perfecto, I.; Badgley, C. Parasitoid wasp diversity in apple orchards along a pest-management gradient. Agric. Ecosyst. Environ. 2012, 156, 82–88. [Google Scholar] [CrossRef] [Green Version]
- Zijp, J.; Blommers, L. Lathrolestes ensator, a parasitoid of the apple sawfly. Proc. Sect. Exp. Appl. Entomol. Neth. Entomol. Soc. 1993, 4, 237–242. [Google Scholar]
- Zijp, J.; Blommers, L. Apple sawfly Hoplocampa testudinea (Hym., Tenthredinidae) and its parasitoid Lathrolestes ensator in Dutch apple orchards (Hym., Ichneumonidae, Ctenopelmatinae). J. Appl. Entomol. 2002, 126, 265–274. [Google Scholar] [CrossRef]
- Mols, P.; Boers, J. Comparison of a Canadian and a Dutch strain of the parasitoid Aphelinus mali (Hald)(Hym., Aphelinidae) for control of woolly apple aphid Eriosoma lanigerum (Haussmann)(Hom., Aphididae) in the Netherlands: A simulation approach. J. Appl. Entomol. 2001, 125, 255–262. [Google Scholar] [CrossRef]
- Quarrell, S.R.; Corkrey, R.; Allen, G.R. Predictive thresholds for forecasting the compatibility of Forficula auricularia and Aphelinus mali as biological control agents against woolly apple aphid in apple orchards. BioControl 2017, 62, 243–256. [Google Scholar] [CrossRef]
- Morris, M.; Mendel, H.; Barclay, R.; Booth, R.; Cannon, M.F.; Conroy, C.E.; Csokay, L.K.; Fisher, C.; Fountain, M.T.; Jay, C.N. Anthonomus spilotus Redtenbacher, 1847 (Curculionidae) new to Britain, a pest in pear orchards in Southern England. Coleopterist 2017, 26, 117–122. [Google Scholar]
- Zijp, J.; Blommers, L.H.M. Biology of Centistes delusorius, a parasitoid of adult apple blossom weevil. Agric. For. Entomol. 2002, 4, 275–282. [Google Scholar] [CrossRef]
- Bianchi, F.J.; Wäckers, F.L. Effects of flower attractiveness and nectar availability in field margins on biological control by parasitoids. Biol. Control 2008, 46, 400–408. [Google Scholar] [CrossRef]
- Balázs, K. The importance of parasitoids in apple orchards. Biol. Agric. Hortic. 1997, 15, 123–129. [Google Scholar] [CrossRef]
- Sjöberg, P.; Rämert, B.; Thierfelder, T.; Hillbur, Y. Ban of a broad-spectrum insecticide in apple orchards: Effects on tortricid populations, management strategies, and fruit damage. J. Pest Sci. 2015, 88, 767–775. [Google Scholar] [CrossRef]
- Davis, V.A.; Sandanayaka, W.M.; Charles, J.G. Parasitoids associated with codling moth (Cydia pomonella) in apple-growing regions in New Zealand. N. Z. Plant Prot. 2018, 71, 352. [Google Scholar] [CrossRef]
- Tingley, R.; García-Díaz, P.; Arantes, C.R.R.; Cassey, P. Integrating transport pressure data and species distribution models to estimate invasion risk for alien stowaways. Ecography 2018, 41, 635–646. [Google Scholar] [CrossRef] [Green Version]
- Zhu, G.; Bu, W.; Gao, Y.; Liu, G. Potential geographic distribution of brown marmorated stink bug invasion (Halyomorpha halys). PLoS ONE 2012, 7, e31246. [Google Scholar] [CrossRef] [PubMed]
- Charles, J.; Avila, G.; Hoelmer, K.A.; Hunt, S.; Gardner-Gee, R.; MacDonald, F.; Davis, V. Experimental assessment of the biosafety of Trissolcus japonicus in New Zealand, prior to the anticipated arrival of the invasive pest Halyomorpha halys. Biocontrol 2019, 64, 367–379. [Google Scholar] [CrossRef]
- Avila, G.; Charles, J. Modelling the potential geographic distribution of Trissolcus japonicus: A biological control agent of the brown marmorated stink bug, Halyomorpha halys. BioControl 2018, 63, 505–518. [Google Scholar] [CrossRef] [Green Version]
- Norin, T. Semiochemicals for insect pest management. Pure Appl. Chem. 2007, 79, 2129–2136. [Google Scholar] [CrossRef]
- Witzgall, P.; Kirsch, P.; Cork, A. Sex pheromones and their impact on pest management. J. Chem. Ecol. 2010, 36, 80–100. [Google Scholar] [CrossRef] [PubMed]
- Cork, A.; Beevor, P.; Gough, A.; Hall, D. Gas chromatography linked to electroantennography: A versatile technique for identifying insect semiochemicals. In Chromatography and Isolation of Insect Hormones and Pheromones; Springer: Berlin/Heidelberg, Germany, 1990; pp. 271–279. [Google Scholar]
- Reddy, G.V.; Guerrero, A. New pheromones and insect control strategies. In Vitamins & Hormones; Elsevier: Amsterdam, The Netherlands, 2010; Volume 83, pp. 493–519. [Google Scholar]
- Hall, D.R.; Amarawardana, L.; Cross, J.V.; Francke, W.; Boddum, T.; Hillbur, Y. The chemical ecology of cecidomyiid midges (Diptera: Cecidomyiidae). J. Chem. Ecol. 2012, 38, 2–22. [Google Scholar] [CrossRef] [PubMed]
- Leskey, T.C.; Short, B.D.; Butler, B.R.; Wright, S.E. Impact of the invasive brown marmorated stink bug, Halyomorpha halys (Stål), in mid-Atlantic tree fruit orchards in the United States: Case studies of commercial management. Psyche 2012, 2012, 535062. [Google Scholar] [CrossRef] [Green Version]
- Rice, K.B.; Bergh, C.J.; Bergmann, E.J.; Biddinger, D.J.; Dieckhoff, C.; Dively, G.; Fraser, H.; Gariepy, T.; Hamilton, G.; Haye, T. Biology, ecology, and management of brown marmorated stink bug (Hemiptera: Pentatomidae). J. Integr. Pest Manag. 2014, 5, A1–A13. [Google Scholar] [CrossRef]
- Vandervoet, T.F.; Bellamy, D.E.; Anderson, D.; MacLellan, R. Trapping for early detection of the brown marmorated stink bug, Halyomorpha halys, in New Zealand. N. Z. Plant Prot. 2019, 72, 36–43. [Google Scholar] [CrossRef] [Green Version]
- Short, B.D.; Khrimian, A.; Leskey, T.C. Pheromone-based decision support tools for management of Halyomorpha halys in apple orchards: Development of a trap-based treatment threshold. J. Pest Sci. 2017, 90, 1191–1204. [Google Scholar] [CrossRef]
- Cross, J.; Innocenzi, P.; Hall, D. Investigations of pheromones of soft fruit pests. IOBC-WPRS Bull. 2000, 23, 67–72. [Google Scholar]
- Fountain, M.; Jåstad, G.; Hall, D.; Douglas, P.; Farman, D.; Cross, J. Further studies on sex pheromones of female Lygus and related bugs: Development of effective lures and investigation of species-specificity. J. Chem. Ecol. 2014, 40, 71–83. [Google Scholar] [CrossRef]
- Baroffio, C.; Sigsgaard, L.; Ahrenfeldt, E.J.; Borg-Karlson, A.-K.; Bruun, S.; Cross, J.; Fountain, M.; Hall, D.; Mozuraitis, R.; Ralle, B. Combining plant volatiles and pheromones to catch two insect pests in the same trap: Examples from two berry crops. Crop Prot. 2018, 109, 1–8. [Google Scholar] [CrossRef]
- Wibe, A.; Cross, J.V.; Borg-Karlson, A.-K.; Hall, D.R.; Trandem, N.; Sigsgaard, L.; Baroffio, C.; Ralle, B.; Fountain, M.T. Softpest Multitrap-management of strawberry blossom weevil and European tarnished plant bug in organic strawberry and raspberry using semiochemical traps. Build. Org. Bridges 2014, 3, 883–886. [Google Scholar]
- Woodford, J.; Birch, A.; Gordon, S.; Griffiths, D.; McNicol, J.; Robertson, G. Controlling raspberry beetle without insecticides. IOBC-WPRS Bull. 2003, 26, 87–92. [Google Scholar]
- Pajač Živković, I.; Benitez, H.A.; Barić, B.; Drmić, Z.; Kadoić Balaško, M.; Lemic, D.; Dominguez Davila, J.H.; Mikac, K.M.; Bažok, R. Codling moth wing morphology changes due to insecticide resistance. Insects 2019, 10, 310. [Google Scholar] [CrossRef] [Green Version]
- Miller, J.R.; McGhee, P.S.; Siegert, P.Y.; Adams, C.G.; Huang, J.; Grieshop, M.J.; Gut, L.J. General principles of attraction and competitive attraction as revealed by large-cage studies of moths responding to sex pheromone. Proc. Natl. Acad. Sci. USA 2010, 107, 22–27. [Google Scholar] [CrossRef] [Green Version]
- Rothschild, G.H.L. Mating Disruption of Lepidopterous Pests: Current Status and Future Prospects. In Management of Insect Pests with Semiochemicals: Concepts and Practice; Mitchell, E.R., Ed.; Springer: Boston, MA, USA, 1981; pp. 207–228. [Google Scholar]
- Knight, A.L. Codling moth areawide integrated pest management. In Areawide Pest Management: Theory and Implementation; Publications from USDA-ARD/UNL Faculty: Lincoln, NE, USA, 2008; pp. 159–190. [Google Scholar]
- Cardé, R.T.; Minks, A.K. Control of moth pests by mating disruption: Successes and constraints. Annu. Rev. Entomol. 1995, 40, 559–585. [Google Scholar] [CrossRef]
- Suckling, D.M. Issues affecting the use of pheromones and other semiochemicals in orchards. Crop Prot. 2000, 19, 677–683. [Google Scholar] [CrossRef]
- Lance, D.R.; Leonard, D.S.; Mastro, V.C.; Walters, M.L. Mating disruption as a suppression tactic in programs targeting regulated Lepidopteran pests in US. J. Chem. Ecol. 2016, 42, 590–605. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Lucchi, A.; Thomson, D.; Ioriatti, C. Sex pheromone aerosol devices for mating disruption: Challenges for a brighter future. Insects 2019, 10, 308. [Google Scholar] [CrossRef] [Green Version]
- Kong, W.N.; Li, J.; Fan, R.J.; Li, S.C.; Ma, R.Y. Sex-pheromone-mediated mating disruption technology for the oriental fruit moth, Grapholita molesta (Busck)(Lepidoptera: Tortricidae): Overview and prospects. Psyche A J. Entomol. 2014, 2014, 253924. [Google Scholar] [CrossRef] [Green Version]
- McGhee, P.S.; Gut, L.J.; Miller, J.R. Aerosol emitters disrupt codling moth, Cydia pomonella, competitively. Pest Manag. Sci. 2014, 70, 1859–1862. [Google Scholar] [CrossRef] [PubMed]
- Knight, A.L.; Judd, G.J.; Gilligan, T.; Fuentes-Contreras, E.; Walker, W.B. Integrated management of tortricid pests of tree fruit. In Integrated Management of Disease and Insect Pests of Tree Fruit, 1st ed.; Xu, X., Fountain, M.T., Eds.; Burleigh Dodds Serise in Agricultural Science; Burleigh Dodds Sciences Publishing: Cambridge, UK, 2019. [Google Scholar]
- Verheggen, F.J.; Arnaud, L.; Bartram, S.; Gohy, M.; Haubruge, E. Aphid and plant volatiles induce oviposition in an aphidophagous hoverfly. J. Chem. Ecol. 2008, 34, 301–307. [Google Scholar] [CrossRef] [Green Version]
- James, D.G. Field evaluation of herbivore-induced plant volatiles as attractants for beneficial insects: Methyl salicylate and the green lacewing, Chrysopa nigricornis. J. Chem. Ecol. 2003, 29, 1601–1609. [Google Scholar] [CrossRef]
- James, D.G. Further field evaluation of synthetic herbivore-induced plan volatiles as attractants for beneficial insects. J. Chem. Ecol. 2005, 31, 481–495. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Ghany, N.M. Semiochemicals for controlling insect pests. J. Plant Prot. Res. 2019, 59, 1–11. [Google Scholar]
- Groot, A.T.; Drijfhout, F.P.; Heijboer, A.; Van Beek, T.A.; Visser, J.H. Disruption of sexual communication in the mirid bug Lygocoris pabulinus by hexyl butanoate. Agric. For. Entomol. 2001, 3, 49–55. [Google Scholar] [CrossRef] [Green Version]
- Song, B.; Jiao, H.; Tang, G.; Yao, Y. Combining repellent and attractive aromatic plants to enhance biological control of three tortricid species (Lepidoptera: Tortricidae) in an apple orchard. Fla. Entomol. 2014, 97, 1679–1689. [Google Scholar] [CrossRef]
- Souza, I.L.; Marucci, R.C.; Silveira, L.C.P.; de Paulo, N.C.P.; Lee, J.C. Effects of marigold on the behavior, survival and nutrient reserves of Aphidius Platensis. BioControl 2018, 63, 543–553. [Google Scholar] [CrossRef]
- Hodgkiss, D.; Brown, M.J.F.; Fountain, M.T. The effect of within-crop floral resources on pollination, aphid control and fruit quality in commercial strawberry. Agric. Ecosyst. Environ. 2019, 275, 112–122. [Google Scholar] [CrossRef]
- Agriculture and Horticulture Development Board. Apple Best Practise Guide; Agriculture and Horticulture Development Board: Warwickshire, UK, 2021. [Google Scholar]
- Collatz, J.; Dorn, S. A host-plant-derived volatile blend to attract the apple blossom weevil Anthonomus pomorum–the essential volatiles include a repellent constituent. Pest Manag. Sci. 2013, 69, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Dickens, J.C. Green leaf volatiles enhance aggregation pheromone of boll weevil, Anthonomus grandis. Entomol. Exp. Appl. 1989, 52, 191–203. [Google Scholar] [CrossRef]
- Eller, F.J.; Bartelt, R.J.; Shasha, B.S.; Schuster, D.J.; Riley, D.G.; Stansly, P.A.; Mueller, T.F.; Shuler, K.D.; Johnson, B.; Davis, J.H. Aggregation pheromone for the pepper weevil, Anthonomus eugenii Cano (Coleoptera: Curculionidae): Identification and field activity. J. Chem. Ecol. 1994, 20, 1537–1555. [Google Scholar] [CrossRef] [PubMed]
- Innocenzi, P.; Hall, D.; Cross, J. Components of male aggregation pheromone of strawberry blossom weevil, Anthonomus rubi Herbst.(Coleoptera: Curculionidae). J. Chem. Ecol. 2001, 27, 1203–1218. [Google Scholar] [CrossRef]
- Xu, Q.; Hatt, S.; Lopes, T.; Zhang, Y.; Bodson, B.; Chen, J.; Francis, F. A push–pull strategy to control aphids combines intercropping with semiochemical releases. J. Pest Sci. 2018, 91, 93–103. [Google Scholar] [CrossRef]
- Pickett, J.A.; Woodcock, C.M.; Midega, C.A.; Khan, Z.R. Push–pull farming systems. Curr. Opin. Biotechnol. 2014, 26, 125–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The use of push-pull strategies in integrated pest management. Annu. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef] [Green Version]
- Mazzi, D.; Bravin, E.; Meraner, M.; Finger, R.; Kuske, S. Economic impact of the introduction and establishment of Drosophila suzukii on sweet cherry production in Switzerland. Insects 2017, 8, 18. [Google Scholar] [CrossRef] [Green Version]
- Mupambi, G.; Anthony, B.M.; Layne, D.R.; Musacchi, S.; Serra, S.; Schmidt, T.; Kalcsits, L.A. The influence of protective netting on tree physiology and fruit quality of apple: A review. Sci. Hortic. 2018, 236, 60–72. [Google Scholar] [CrossRef]
- Alaphilippe, A.; Capowiez, Y.; Severac, G.; Simon, S.; Saudreau, M.; Caruso, S.; Vergnani, S. Codling moth exclusion netting: An overview of French and Italian experiences. IOBC-WPRS Bull. 2016, 112, 31–35. [Google Scholar]
- Sauphanor, B.; Severac, G.; Maugin, S.; Toubon, J.F.; Capowiez, Y. Exclusion netting may alter reproduction of the codling moth (Cydia pomonella) and prevent associated fruit damage to apple orchards. Entomol. Exp. Appl. 2012, 145, 134–142. [Google Scholar] [CrossRef]
- Candian, V.; Pansa, M.G.; Santoro, K.; Spadaro, D.; Tavella, L.; Tedeschi, R. Photoselective exclusion netting in apple orchards: Effectiveness against pests and impact on beneficial arthropods, fungal diseases and fruit quality. Pest Manag. Sci. 2020, 76, 179–187. [Google Scholar] [CrossRef]
- Bal, H.K.; Adams, C.; Grieshop, M. Evaluation of off-season potential breeding sources for spotted wing Drosophila (Drosophila suzukii Matsumura) in Michigan. J. Econ. Entomol. 2017, 110, 2466–2470. [Google Scholar] [CrossRef] [PubMed]
- Noble, R.; Dobrovin-Pennington, A.; Shaw, B.; Buss, D.S.; Cross, J.V.; Fountain, M.T. Fermentation for disinfesting fruit waste from Drosophila species (Diptera: Drosophilidae). Environ. Entomol. 2017, 46, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Landi, S.; Pennacchio, F.; Papini, R.; D’errico, G.; Torrini, G.; Strangi, A.; Barabaschi, D.; Roversi, P.F. Evaluation of sheep grazing effects on nematode community, insect infestation and soil fertility in sweet chestnut orchards: A case of study. Redia 2017, 99, 117. [Google Scholar]
- Buehrer, K.A.; Grieshop, M.J. Postharvest grazing of hogs in organic fruit orchards for weed, fruit, and insect pest management. Org. Agric. 2014, 4, 223–232. [Google Scholar] [CrossRef]
- Nunn, L.; Embree, C.; Hebb, D.; Bishop, S.; Nichols, D. Rotationally grazing hogs for orchard floor management in organic apple orchards. Acta Hortic. 2007, 737, 71. [Google Scholar] [CrossRef] [Green Version]
- Grieshop, M.J. Cultural control of arthropod pests in temperate tree fruit. In Integrated Managment of Disease and Insect Pests of Tree Fruit, 1st ed.; Xu, X., Fountain, M.T., Eds.; Burleigh Dodds Science Publishing: Cambridge, UK, 2019; pp. 499–525. [Google Scholar]
- Glenn, D.M.; Puterka, G.J. Particle films: A new technology for agriculture. Hortic. Rev. 2005, 31, 1–44. [Google Scholar]
- Saour, G.; Makee, H. A kaolin-based particle film for suppression of the olive fruit fly Bactrocera oleae Gmelin (Dip., Tephritidae) in olive groves. J. Appl. Entomol. 2004, 128, 28–31. [Google Scholar] [CrossRef]
- Showler, A. Effects of kaolin-based particle film application on boll weevil (Coleoptera: Curculionidae) injury to cotton. J. Econ. Entomol. 2002, 95, 754–762. [Google Scholar] [CrossRef]
- Saour, G.; Ismail, H.; Hashem, A. Impact of kaolin particle film, spirodiclofen acaricide, harpin protein, and an organic biostimulant on pear psylla Cacopsylla pyri (Hemiptera: Psyllidae). Int. J. Pest Manag. 2010, 56, 75–79. [Google Scholar] [CrossRef]
- Thomas, A.L.; Muller, M.E.; Dodson, B.R.; Ellersieck, M.R.; Kaps, M. A kaolin-based particle film suppresses certain insect and fungal pests while reducing heat stress in apples. J. Am. Pomol. Soc. 2004, 58, 42. [Google Scholar]
- Marko, V.; Blommers, L.; Bogya, S.; Helsen, H. Kaolin particle films suppress many apple pests, disrupt natural enemies and promote woolly apple aphid. J. Appl. Entomol. 2008, 132, 26–35. [Google Scholar] [CrossRef]

| 2020 Provisional Data | |||
|---|---|---|---|
| Area Grown (Hectares) | Yield (Thousand Tonnes) | Value (£ Million) | |
| Dessert Apples | 6372 | 200.7 | 158.1 |
| Culinary Apples | 2473 | 92.2 | 81.2 |
| Cider Apples and Perry Pears | 6700 | 154.1 | 27.6 |
| Dessert Pears | 1470 | 25 | 19.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaw, B.; Nagy, C.; Fountain, M.T. Organic Control Strategies for Use in IPM of Invertebrate Pests in Apple and Pear Orchards. Insects 2021, 12, 1106. https://doi.org/10.3390/insects12121106
Shaw B, Nagy C, Fountain MT. Organic Control Strategies for Use in IPM of Invertebrate Pests in Apple and Pear Orchards. Insects. 2021; 12(12):1106. https://doi.org/10.3390/insects12121106
Chicago/Turabian StyleShaw, Bethan, Csaba Nagy, and Michelle T. Fountain. 2021. "Organic Control Strategies for Use in IPM of Invertebrate Pests in Apple and Pear Orchards" Insects 12, no. 12: 1106. https://doi.org/10.3390/insects12121106
APA StyleShaw, B., Nagy, C., & Fountain, M. T. (2021). Organic Control Strategies for Use in IPM of Invertebrate Pests in Apple and Pear Orchards. Insects, 12(12), 1106. https://doi.org/10.3390/insects12121106







