A Comparison of Morphology among Four Termite Species with Different Moisture Requirements
Abstract
:1. Introduction
2. Materials and Methods
2.1. Termite Collection
2.2. Sectioning, Staining, and Slide Preparation
2.3. Data Analysis
3. Results
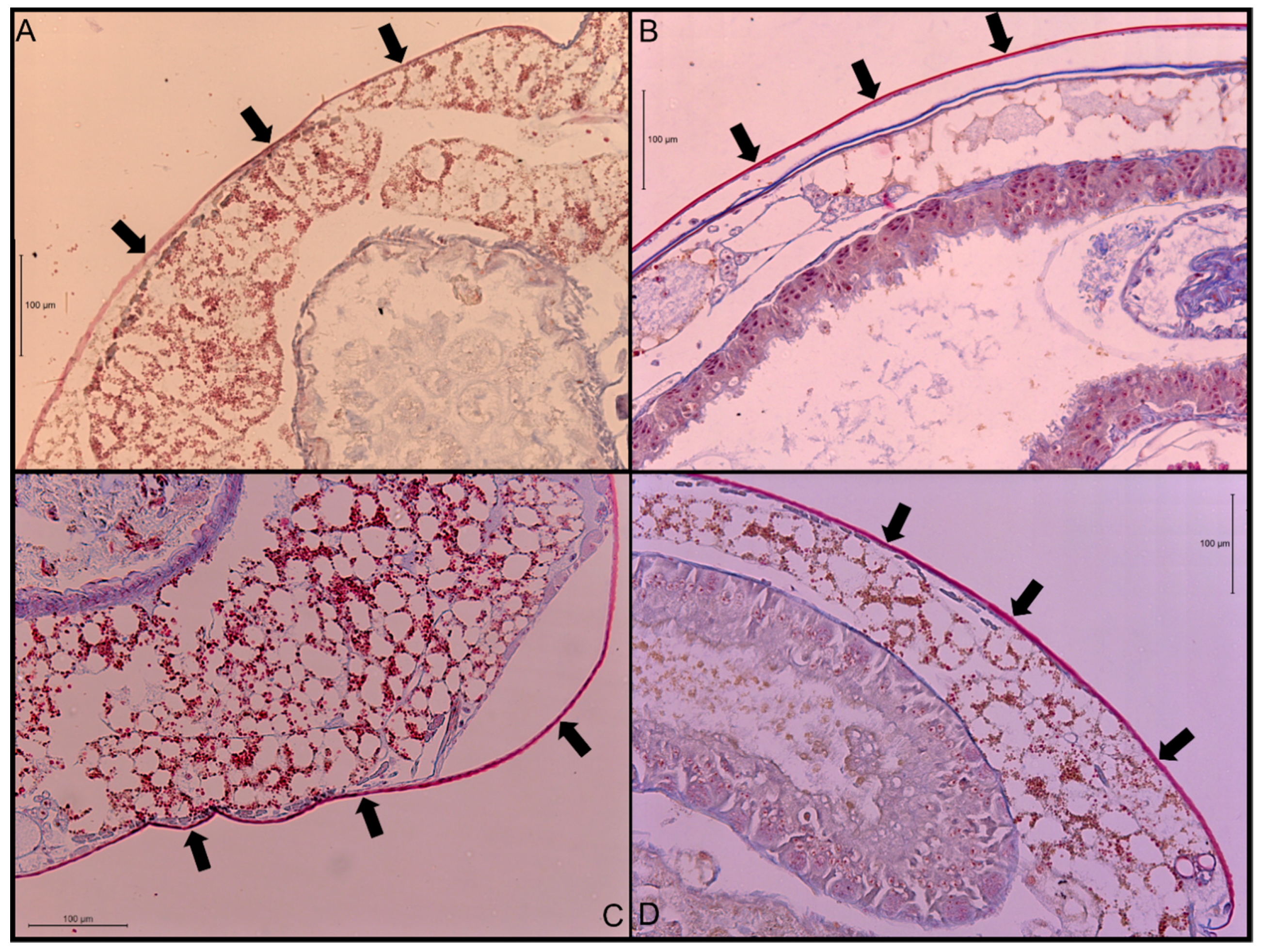
3.1. Cuticle Thickness
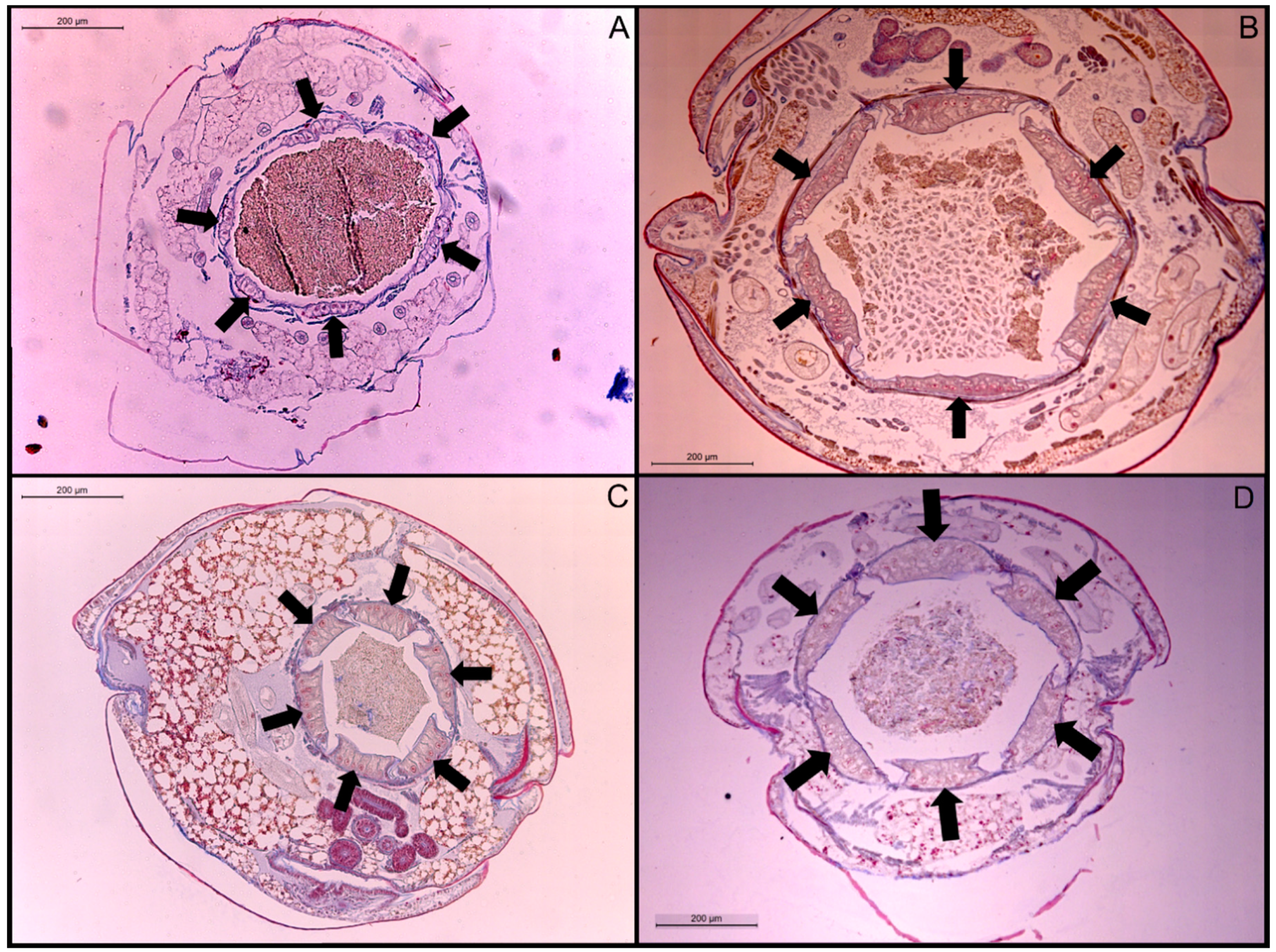
3.2. Rectal Pad Width
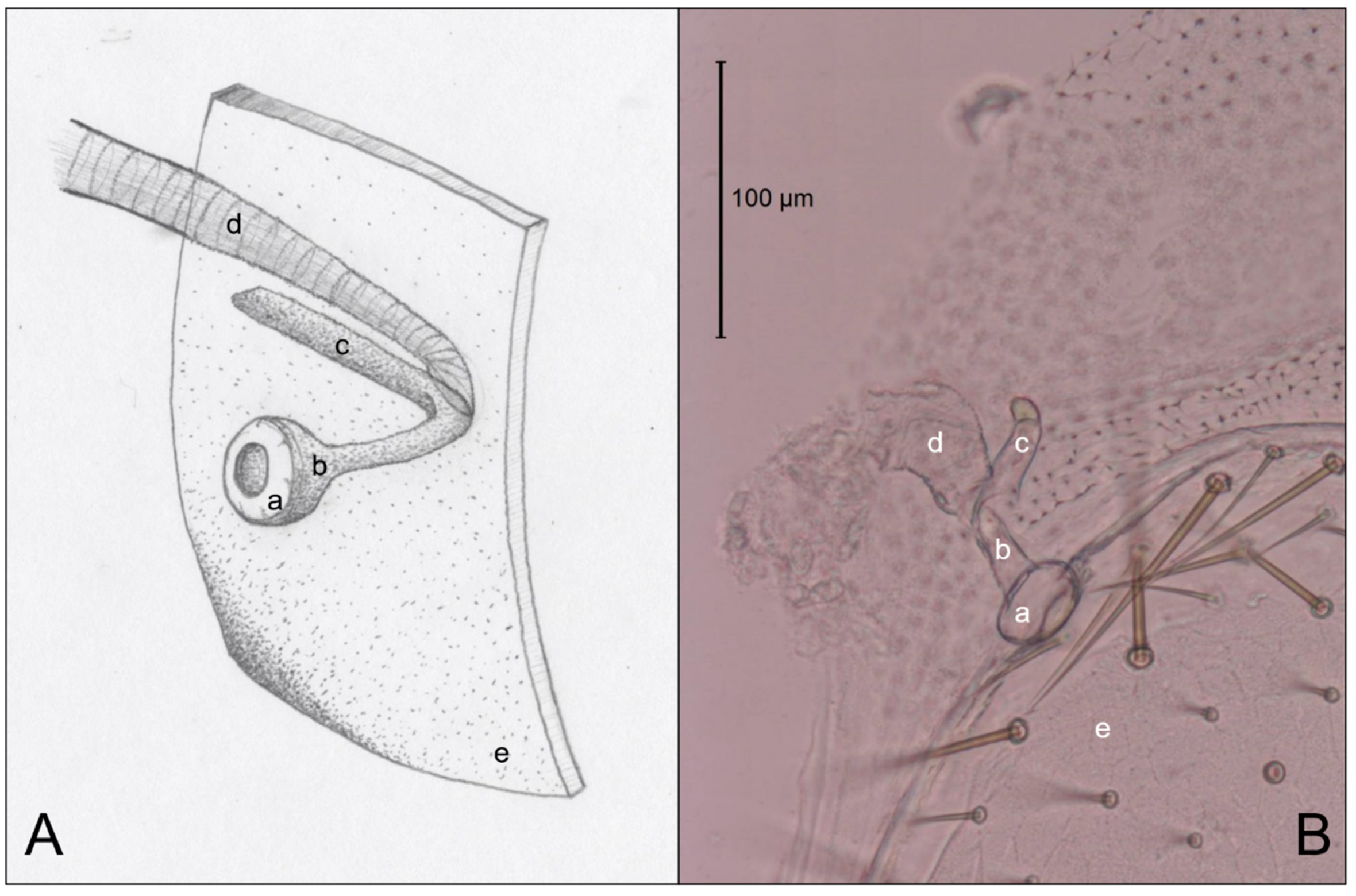
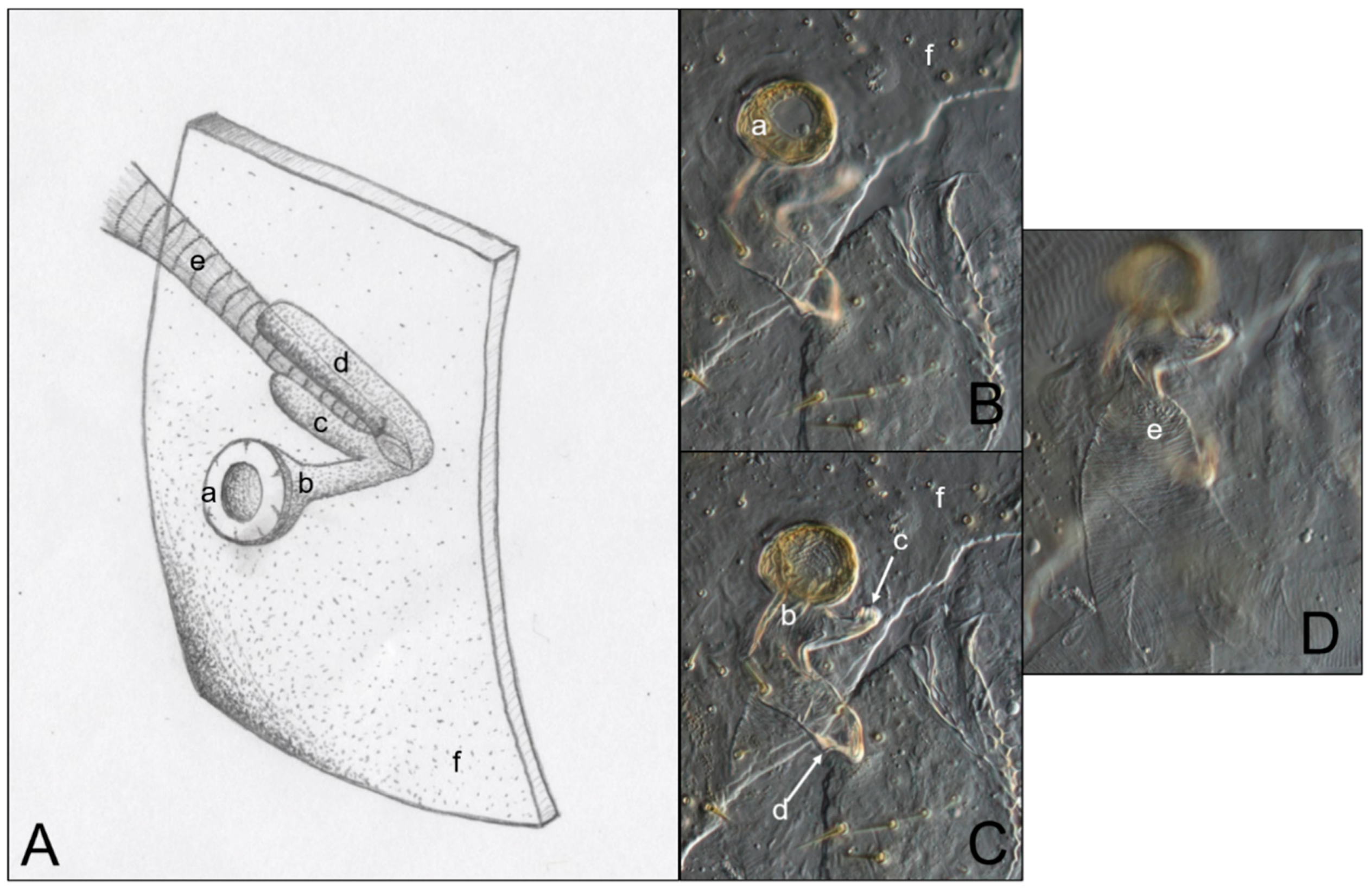
3.3. Abdominal Spiracles
4. Discussion
4.1. Cuticle Thickness
4.2. Rectal Pad Width
4.3. Abdominal Spiracles
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Edney, E.B. Zoophysiology and Ecology Series: Water Balance in Land Arthropods; Springer: Berlin/Heidelberg, Germany, 1977; Volume 9, p. 284. [Google Scholar]
- Hadley, N.F. Water Relations of Terrestrial Arthropods; Academic Press: New York, NY, USA, 1994; p. 356. [Google Scholar]
- Noirot, C.; Noirot-Timothée, C. Fine structure of the rectum in termites (Isoptera): A comparative study. Tissue Cell 1977, 9, 693–710. [Google Scholar] [CrossRef]
- Su, N.-Y.; Scheffrahn, R.H. A method to access, trap, and monitor field populations of the Formosan termite (Isoptera: Rhinotermitidae) in the urban environment. Sociobiology 1986, 12, 299–304. [Google Scholar]
- Martoja, R.; Martoja-Pierson, M. Initiation Aux Technique de 1′ Histologie Animale; Masson: Paris, France, 1967; p. 345. [Google Scholar]
- Mayer, R.T.; Meola, S.M.; Coppage, D.L.; Deloach, J.R. The pupal instar of Stomoxys calcitrans cuticle deposition and chitin synthesis. J. Insect Physiol. 1979, 25, 677–683. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.-Y.; Robert, A. Cellular encapsulation in the eastern subterranean termite, Reticulitermes flavipes (Isoptera), against infection by the entomopathogenic fungus Metarhizium anisopliae. J. Invertebr. Pathol. 2009, 101, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Wigglesworth, V.B. Transpiration through the cuticle of insects. J. Exp. Biol. 1945, 21, 97–114. [Google Scholar]
- Appel, A.G.; Reierson, D.A.; Rust, M.K. Comparative water relations and temperature sensitivity of cockroaches. Comp. Biochem. Physiol. 1983, 74A, 357–361. [Google Scholar] [CrossRef]
- Appel, A.G.; Reierson, A.G.; Rust, M.K. Cuticular water loss in the Smokybrown cockroach, Periplaneta fuliginosa. J. Insect Physiol. 1986, 32, 623–628. [Google Scholar] [CrossRef]
- Hadley, N.F. Epicuticular lipids of the desert tenebrionid beetle, Eleodes armata: Seasonal and acclamatory effects on chemical composition. Insect Biochem. 1977, 7, 277–283. [Google Scholar] [CrossRef]
- Hadley, N.F. Cuticular permeability of desert tenebrionid beetles: Correlations with epicuticular hydrocarbon composition. Insect Biochem. 1978, 8, 17–22. [Google Scholar] [CrossRef]
- Monzer, M.A.; Srour, H.A. Desiccation intolerance of the red palm weevil, Rhynchophorus ferrugineus (Oliv) adults in relation to their cuticular hydrocarbons. Egypt. Acad. J. Biol. Sci. 2009, 2, 47–53. [Google Scholar]
- Weissling, T.J.; Giblin-Davis, R.M. Water loss dynamics and humidity preference of Rhynchophorus cruentatus (Coleoptera: Curculionidae) adults. Environ. Entomol. 1983, 22, 93–98. [Google Scholar] [CrossRef]
- Locke, M. Permeability of the insect cuticle to water and lipids. Science 1965, 147, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Beament, J.W.L. The cuticular lipoids of insects. J. Exp. Biol. 1945, 21, 115–131. [Google Scholar]
- Ebeling, W.; Wagner, R.E. Rapid desiccation of drywood termites with inert sorptive dusts and other substances. J. Econ. Entomol. 1959, 52, 190–207. [Google Scholar] [CrossRef]
- Collins, M. Water relations in termites. In Biology of Termites; Krishna, K., Weesner, F.M., Eds.; Academic Press: New York, NY, USA, 1969; Volume 1, pp. 433–458. [Google Scholar]
- Sponsler, R.C.; Appel, A.G. Aspects of the water relations of the Formosan and Eastern subterranean termites (Isoptera: Rhinotermitidae). Environ. Entomol. 1990, 19, 15–20. [Google Scholar] [CrossRef]
- Beament, J.W.L. The water relations of insect cuticle. Biol. Rev. 1961, 36, 281–320. [Google Scholar] [CrossRef] [PubMed]
- Zukowski, J.; Su, N.-Y. Survival of termites (Isoptera) exposed to various levels of relative humidity (RH) and water availability, and their RH preferences. Fla. Entomol. 2017, 100, 532–538. [Google Scholar] [CrossRef] [Green Version]
- University of Florida, Featured Creatures: Florida Dampwood Termites. Available online: http://entomology.ifas.ufl.edu/creatures/urban/termites/neotermes.html (accessed on 31 October 2015).
- Snodgrass, R.E. Principles of Insect Morphology; Cornell University Press: Ithaca, NY, USA, 1993; p. 667. [Google Scholar]


| Species | Cuticle Thickness (Microns) * | Observations |
|---|---|---|
| C. brevis | 2.28 ± 0.03a | 180 |
| C. cavifrons | 2.10 ± 0.03b | 222 |
| Co. formosanus | 1.81 ± 0.03c | 196 |
| N. jouteli | 2.51 ± 0.05d | 339 |
| Species | Rectal Pad Widths (Microns) * | Observations |
|---|---|---|
| C. brevis | 160.5 ± 4.2a | 66 |
| C. cavifrons | 142.6 ± 3.7b | 59 |
| Co. formosanus | 101.8 ± 4.8c | 62 |
| N. jouteli | 171.6 ± 4.8a | 55 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zukowski, J.; Su, N.-Y. A Comparison of Morphology among Four Termite Species with Different Moisture Requirements. Insects 2020, 11, 262. https://doi.org/10.3390/insects11050262
Zukowski J, Su N-Y. A Comparison of Morphology among Four Termite Species with Different Moisture Requirements. Insects. 2020; 11(5):262. https://doi.org/10.3390/insects11050262
Chicago/Turabian StyleZukowski, John, and Nan-Yao Su. 2020. "A Comparison of Morphology among Four Termite Species with Different Moisture Requirements" Insects 11, no. 5: 262. https://doi.org/10.3390/insects11050262
APA StyleZukowski, J., & Su, N.-Y. (2020). A Comparison of Morphology among Four Termite Species with Different Moisture Requirements. Insects, 11(5), 262. https://doi.org/10.3390/insects11050262





